Abstract
Background:
Two principal methods for detecting anti-John Cunningham virus (JCV) antibodies are currently utilized in clinical practice: STRATIFY JCV™, an ELISA developed by Biogen, and IMMUNOWELL™, a solid-phase ELISA assay by Polpharma Biologics/GenBio.
Objective:
We aimed to evaluate the concordance between STRATIFY and IMMUNOWELL in detecting anti-JCV antibodies in a real-world population of patients with relapsing-remitting multiple sclerosis (RRMS) undergoing natalizumab therapy.
Design:
This monocentric observational study screened all patients treated with natalizumab for at least 6 months, referring to the MS Center of the University Hospital of Catania.
Methods:
Each patient’s serum was tested simultaneously using STRATIFY-2 (STRATIFY JCV DxSelect) and IMMUNOWELL assays. The qualitative results (positive/negative) were compared, and the index values were analyzed using Pearson’s correlation and Bland–Altman plots. Inter-method agreement was calculated using Cohen’s kappa coefficient.
Results:
Among the 120 patients tested, 82 were positive and 31 negative with both STRATIFY and IMMUNOWELL. Four cases were STRATIFY-negative but IMMUNOWELL-positive, and three were the opposite. Overall concordance was 94.2%, with a Cohen’s Kappa of 0.86, indicating strong agreement. The index values showed strong correlation (Pearson r = 0.79, p < 0.001) and the coefficient of determination (r2) was 0.62.
Conclusion:
STRATIFY and IMMUNOWELL demonstrate a high level of agreement in the detection of anti-JCV antibodies in patients with RRMS receiving natalizumab. IMMUNOWELL may serve as a reliable complementary method, especially in cases where borderline serostatus could influence therapeutic strategy. Regular and accurate monitoring of JCV status remains essential for guiding long-term treatment safety and optimizing individual patient outcomes.
Plain language summary
Natalizumab is a medication used to treat relapsing-remitting multiple sclerosis (RRMS), but it can increase the risk of a rare brain infection called progressive multifocal leukoencephalopathy (PML), caused by the JC virus (JCV). To help assess this risk, doctors test patients for antibodies against JCV in their blood. Two main tests are available to detect these antibodies: STRATIFY JCV™ and IMMUNOWELL™. This study compared how well these two tests agree in real-world patients receiving natalizumab. Researchers tested blood samples from 120 patients using both tests at the same time. They found that the results matched in over 94% of cases, showing strong agreement. The few differences happened mostly in borderline cases, where the antibody levels were close to the cutoff point between negative and positive. This suggests that IMMUNOWELL™ is a reliable alternative to the standard STRATIFY JCV™ test. Using either test can help doctors monitor patients’ JCV status and make safer treatment decisions.
Introduction
Natalizumab is a highly effective therapy for relapsing-remitting multiple sclerosis (RRMS), but its use is associated with a risk of progressive multifocal leukoencephalopathy (PML), a rare opportunistic brain infection caused by the John Cunningham virus (JCV).1,2 The presence of anti-JCV antibodies in a patient’s blood is a well-established risk factor for PML development in those receiving natalizumab.1,3 Serological testing for anti-JCV antibodies has therefore become a cornerstone of PML risk stratification and guides treatment decisions for natalizumab therapy. 4 Patients who test negative for anti-JCV antibodies (seronegative) have a very low risk of PML, on the order of <0.1 per 1000 (<0.01%). 5 In contrast, patients who are anti-JCV antibody positive (seropositive) face a higher risk that increases with cumulative natalizumab exposure and prior immunosuppressant use. 5 For example, in seropositive patients with no history of immunosuppression, the PML risk is approximately 0.56 per 1000 during the first 2 years of natalizumab therapy, rising to ~4.6 per 1000 after 2 years. These risk estimates lead to widespread adoption of JCV serological testing to stratify PML risk in clinical practice.1,6,7 Subsequently, it was shown that not only the presence but also the level of anti-JCV antibody (reported as an index value) correlates with PML risk. This prompted refined algorithms incorporating antibody index thresholds (e.g., 0.9 or 1.5) to further differentiate risk among seropositive patients. 8
The standard method for detecting anti-JCV antibodies in natalizumab-treated patients has been the STRATIFY-2 JCV™ DxSelect two-step ELISA assay developed by Biogen® Cambridge, Massachusetts, United States.9,10 This assay uses JCV virus-like particles as antigen and classifies patients as seropositive or seronegative based on a calibrated index value. It includes a confirmatory inhibition step for borderline cases to ensure high specificity. In clinical trials and post-marketing studies, the STRATIFY assay has demonstrated reliable performance, with a reported seroprevalence approximately of 50%–70% in multiple sclerosis (MS) patients and a low analytical false-negative rate (about 2%–3%).5,11 Detection of anti-JCV antibodies by this method has proven effective in stratifying PML risk, as seronegative status corresponds to the lowest risk category. 5 However, a small fraction of patients can have latent JCV infection without detectable antibodies (“false negative” serology).6,12
In August 2023, the European Medicines Agency approved the natalizumab biosimilar (Tyruko®), the first biosimilar of natalizumab, for the treatment of RRMS. 13 Tyruko was granted marketing authorization based on comprehensive biosimilarity data demonstrating equivalent pharmacokinetics, efficacy, safety, and immunogenicity compared to the reference product (Tysabri®).14,15 As with the originator, the risk of PML remains a central concern in natalizumab biosimilar-treated patients, and anti-JCV antibody testing continues to play a critical role in risk stratification. For this reason, a new anti-JCV antibody ELISA kit, IMMUNOWELL™ Anti-JCV (Polpharma Biologics/GenBio) Gdańsk, Poland, has emerged as a potential alternative to the Biogen STRATIFY assay.16,17 The IMMUNOWELL JCV test is an analytically and clinically validated two-step ELISA, available as a diagnostic kit or centralized service, designed to similarly detect anti-JCV IgG antibodies in human serum. According to the manufacturer’s technical documentation, this assay uses a screening ELISA with defined cutoff ranges (index >0.50 considered positive, <0.20 negative, and 0.20–0.50 as an indeterminate zone requiring confirmation). A confirmatory step (competitive inhibition test) is built-in for samples with borderline index values, mirroring the approach of the reference Biogen assay. Initial validation studies reported a high positive agreement of ~97% with the Biogen STRATIFY test, along with a negative agreement around ~74%. 16 Given that natalizumab and its biosimilars may use different antibody tests, it is clinically important to know whether these two assays yield comparable results.
In this study, we aimed to evaluate the concordance between STRATIFY-2 JCV DxSelect and IMMUNOWELL anti-JCV antibody assays in a real-world cohort of RRMS patients on natalizumab therapy.
Materials and methods
Study design and patients
This was a single-center, observational comparative study conducted at the University of Catania MS Center in the period between August 2024 and March 2025. We screened all patients with relapsing-remitting MS according to the Lublin criteria, 18 who were receiving natalizumab treatment. All patients had been on natalizumab for at least 6 months at the time of sampling, ensuring a stable treatment exposure. Patient inclusion criteria were confirmed RRMS diagnosis, current natalizumab therapy (⩾6 infusions), and age ⩾18 years. We excluded patients with prior diagnosis of PML, history of hematologic malignancies or primary immunodeficiency, or insufficient clinical or laboratory data for anti-JCV testing. Patients with any immunosuppressive treatments (mitoxantrone, methotrexate, azathioprine, cyclophosphamide, S1P receptors modulators, etc.) prior to natalizumab were noted, as this is a known PML risk factor. Blood samples were collected during routine monitoring visits.
Ethical approval was obtained from the local institutional review board, and all participants provided informed consent.
Anti-JCV antibody testing
For each patient, a single blood draw provided serum that was divided into two aliquots. One aliquot was tested using the STRATIFY-2 JCV DxSelect ELISA (Biogen Inc.), and the other was tested using the IMMUNOWELL Anti-JCV ELISA (Polpharma Biologics, GenBio), in parallel, according to each manufacturer’s instructions. Both assays are two-step ELISAs for the qualitative detection of anti-JCV IgG. In the STRATIFY assay, JCV capsid antigen (virus-like particles of JCV VP1) coated on plates is used to capture anti-JCV antibodies from serum.1,10 Bound antibodies are detected via an enzyme-conjugated anti-human IgG, producing a colorimetric signal measured as an optical density that is converted to an index value relative to calibrators. The STRATIFY assay defines a predefined index cutoff for positivity; in prior publications this two-step method classifies any sample as antibody-positive if the index >0.50, negative if index <0.20, with an intermediate “indeterminate” range (~0.20–0.50) that triggers a confirmatory inhibition assay. Only if the confirmatory step is also positive (showing >45% inhibition of binding) is an indeterminate classified as final positive. This two-step algorithm is designed to maximize specificity by ruling out false positives in the borderline range.9,10 The IMMUNOWELL anti-JCV ELISA uses a very similar approach: serum is tested in a screening ELISA with JCV-like antigen, yielding an index value. According to current clinical stratification models, for IMMUNOWELL, the risk categories are defined as follows: index ⩽ 0.8 (low risk), 0.8 < index ⩽ 1.4 (intermediate risk), and index > 1.4 (high risk), as reported in recent studies.16,17 Borderline samples undergo a confirmatory neutralization test included in the kit, also using a 45% inhibition cutoff for positivity (i.e., if ⩾45% signal is inhibited by excess antigen, the sample is confirmed positive). 16
Statistical analysis
We compared the qualitative serostatus results of STRATIFY versus IMMUNOWELL for each patient. The primary outcome was the concordance rate between the two assays, calculated as the percentage of patients with the same result by both methods. A 2 × 2 contingency table was constructed (STRATIFY positive/negative vs IMMUNOWELL positive/negative), and from this we calculated Cohen’s kappa coefficient to assess inter-assay agreement beyond chance. Kappa values were interpreted using conventional benchmarks (e.g., κ > 0.80 = “almost perfect” agreement). We also computed the sensitivity and specificity of IMMUNOWELL relative to STRATIFY as the reference, although recognizing that there is no true gold standard in this context. Additionally, McNemar’s test was performed on the discordant pairs to evaluate if one assay tended to yield positive results significantly more often than the other (p < 0.05 considered significant).
For the quantitative index values, we assessed the linear correlation between the two assays. Pearson’s correlation coefficient (r) was calculated for the STRATIFY versus IMMUNOWELL index values across all patients. The correlation was tested for significance (with a two-tailed test, α = 0.05). To further examine the agreement in continuous values and identify any systematic bias, we performed a Bland–Altman analysis. For each patient, the difference between the IMMUNOWELL and STRATIFY index (IMMUNOWELL minus STRATIFY) was plotted against the average of the two assay indices. We calculated the mean difference (bias) and the 95% limits of agreement (mean difference ± 1.96 standard deviations).
All statistical analyses were conducted using STATA 18.0 StataCorp. Throughout the analysis, a p-value < 0.05 was considered statistically significant.
Results
Patient characteristics
The analysis included 120 RRMS patients on natalizumab. All patients had been exposed to natalizumab for at least 6 months (median therapy duration at sampling was not formally recorded, but all had ⩾6 infusions and 86 (71.7%) had >1 year on treatment). Demographic and clinical characteristics were illustrated in Table 1. No patient in this cohort had known PML, and none experienced PML during the study period.
Demographic and clinical characteristics of the cohort.
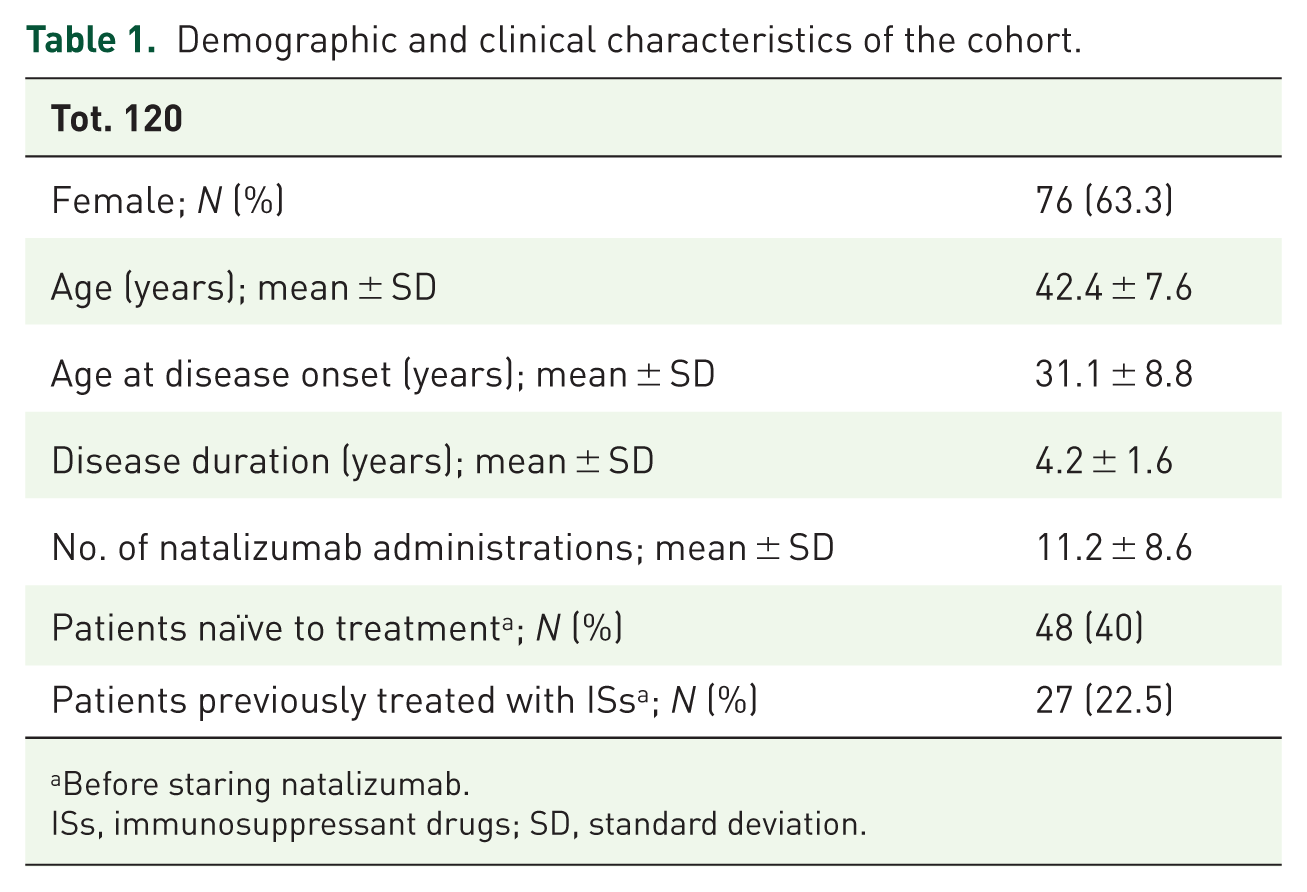
Before staring natalizumab.
ISs, immunosuppressant drugs; SD, standard deviation.
Qualitative concordance
The serostatus results obtained by the two assays showed a high level of agreement. Out of 120 patients, 113 had concordant results between STRATIFY and IMMUNOWELL. Specifically, 82 patients (68.3%) were JCV-antibody positive on both assays, and 31 patients (25.8%) were negative on both assays. Thus, 113/120 (94.2%) of samples gave the same qualitative interpretation with both methods. Only seven patients (5.8%) showed a discrepancy between the assays’ results (Table 2). Of the seven discordant cases, four patients were STRATIFY-negative but IMMUNOWELL-positive, and three patients were STRATIFY-positive but IMMUNOWELL-negative. The overall agreement rate between the assays was 94.2%. The Cohen’s κ coefficient was calculated to be κ = 0.86, with a 95% confidence interval approximately (0.75–0.97). We found no significant systematic bias in qualitative outcomes favoring one assay over the other—the discordant counts were nearly symmetric (4 vs 3, p = 0.72 by McNemar’s test, n.s.). The IMMUNOWELL assay’s positivity rate was 71.7% (86/120) compared to 70.8% (85/120) for STRATIFY.
Cross-tabulation of anti-JCV antibody results by assay.
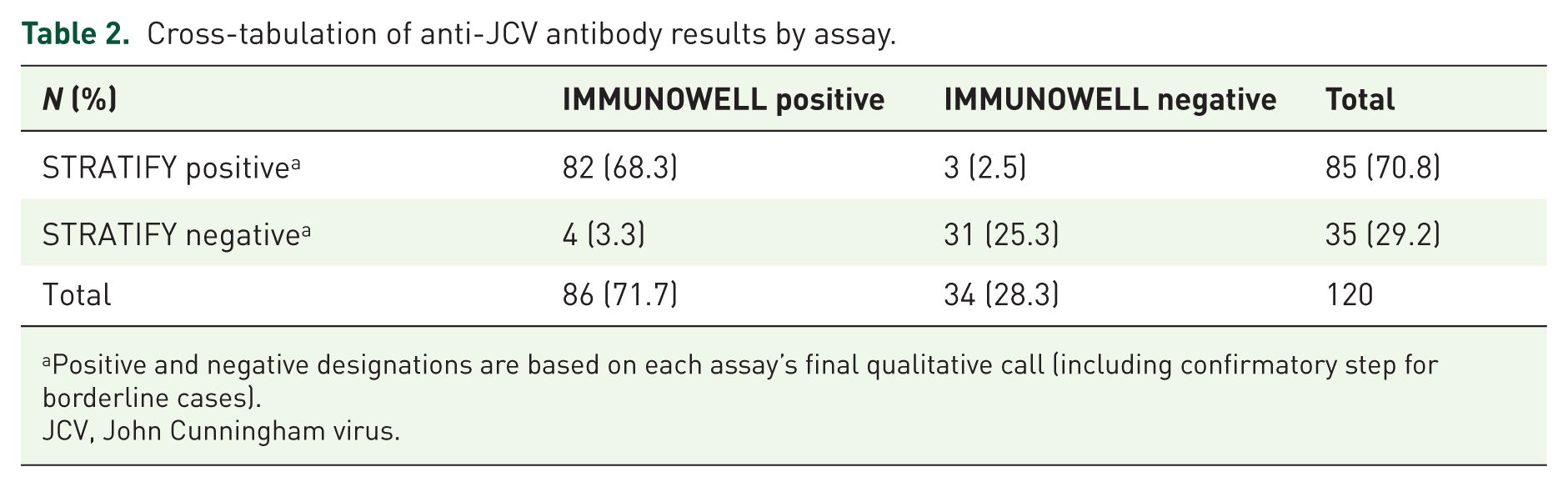
Positive and negative designations are based on each assay’s final qualitative call (including confirmatory step for borderline cases).
JCV, John Cunningham virus.
Notably, the discordant patients had antibody levels near the cut-off. In the four STRATIFY(−)/IMMUNOWELL(+) cases, the STRATIFY index values ranged from 0.30 to 0.49 (just below the typical 0.50 positive threshold), whereas their IMMUNOWELL indices ranged from ~0.55 to 0.65 (just above the threshold). All four of these had IMMUNOWELL indices in the low-positive range and presumably fell into the confirmatory testing zone; they were confirmed as true positives by IMMUNOWELL’s inhibition step, indicating low-level anti-JCV antibodies. Conversely, the three STRATIFY(+)/IMMUNOWELL(−) patients had STRATIFY indices between ~0.55 and 0.70 (borderline positive) while their IMMUNOWELL indices were slightly below 0.50 (in the 0.35–0.49 range). It appears that in those cases, the IMMUNOWELL assay did not register a high enough signal on initial screening to call positive, or perhaps the confirmatory step rendered them negative.
Quantitative correlation
Across the 120 paired measurements, there was a strong positive correlation between STRATIFY and IMMUNOWELL index values (Pearson r = 0.79, p < 0.001). The coefficient of determination (r2) of about 0.62 suggests that ~62% of the variance in one assay’s readings can be explained by the other’s, which is substantial given the biological and technical variability inherent in immunoassays (Figure 1).

Scatter plot of anti-JCV antibody index values measured by STRATIFY JCV™ versus IMMUNOWELL™ assays.
Bland–Altman agreement
The average index value for each sample was plotted against the difference between the IMMUNOWELL and STRATIFY values. The mean difference (IMMUNOWELL minus STRATIFY) was very close to zero: −0.03 in index units, meaning that on average the IMMUNOWELL index was 0.03 lower than the STRATIFY index. This difference was not statistically significant (paired t-test for mean bias ~0, p = 0.82). The 95% limits of agreement were approximately, 0.91 to +0.86 in index units. Thus, for 95% of samples, the IMMUNOWELL index fell within about ±0.9 of the STRATIFY index. There was no pronounced proportional bias observed: the mean difference remained near zero across the range of index magnitudes, and a linear regression of the difference versus the mean did not show a significant slope (no trend for differences to grow or shrink at higher antibody levels).
As shown in Figure 2, the differences between assays are tightly distributed around zero. The vast majority of samples had differences well below 0.5 in either direction. A few points fall near the upper LoA (+0.8) or lower LoA (−0.9); these are the cases where one assay reported a borderline positive and the other borderline negative. For instance, the highest difference observed was about +0.8, corresponding to a sample where STRATIFY gave an index ~0.4 and IMMUNOWELL ~1.2 (average 0.8, difference +0.8; this was a low-positive per IMMUNOWELL but negative by STRATIFY initially). Conversely, the largest negative difference (−0.8) was a case where STRATIFY index was about 1.4 versus IMMUNOWELL 0.6 (average ~1.0, difference −0.8; positive by STRATIFY, low-borderline by IMMUNOWELL). All these lie within the statistically expected limits.

Bland–Altman plot comparing the IMMUNOWELL and STRATIFY assays for anti-JCV index values.
Discussion
In this comparative study, we found that the IMMUNOWELL ELISA is largely equivalent to the established STRATIFY JCV™ assay for detecting anti-JCV antibodies in natalizumab-treated MS patients. The overall agreement of 94% and Cohen’s kappa of 0.86 reflect excellent concordance. This suggests that, in practice, both assays identify the same patients as JCV-seropositive or seronegative in the vast majority of cases. Such a high level of agreement is critical because JCV serostatus is used as a major criterion for PML risk stratification. 1
In our cohort, we observed a relatively high overall seroprevalence (68%), which lies at the upper end of previously reported ranges in MS populations. Indeed, a recent systematic review reported seropositivity rates between 40% and 69%, with a median of approximately 57%. 19 These variations are likely influenced by multiple factors, including assay sensitivity, demographic characteristics, and geographic distribution.
In our study, the strong correlation (r = 0.79) between the assays’ index values further supports their comparability. Minor quantitative differences are expected given differences in assay design details (e.g., antigen preparation, calibration curves, detection antibodies). However, the lack of any significant systematic bias (mean difference ~0) means neither assay consistently reads higher or lower than the other. In practical terms, a patient’s antibody level can be tracked over time even if the testing method changes, with only small adjustments for assay variability. This is reassuring for scenarios such as patients transitioning care or switching to a natalizumab biosimilar that uses a different assay platform.10,16 A recent analysis reported a slightly lower agreement (85.5%) and a moderate kappa value (κ = 0.6222) compared with our results, with a trend toward higher risk classification by IMMUNOWELL, particularly at intermediate and high-risk thresholds. 20 These differences may be attributed to variations in cohort size, patient characteristics, or methodological differences such as timing of sampling, laboratory procedures, and interpretation of borderline results.
Despite the overall high concordance, we observed discordant results in 5.8% of patients, specifically seven cases near the cutoff. This finding is not surprising as any two assays can yield conflicting results around the threshold of detection, where assay variability and biological noise have the greatest impact. Interestingly, the IMMUNOWELL assay tended to call slightly more patients positive than the STRATIFY assay (4 vs 3 in discordants, and 86 vs 85 overall). This pattern aligns with the notion that IMMUNOWELL might have a marginally higher analytical sensitivity.16,17 The manufacturer’s data for IMMUNOWELL reported a positive percent agreement of over 97% with the reference method, implying a small number of additional positives detected. In our real-world sample, those additional positives correspond to low-index positives that STRATIFY had missed (false negatives by STRATIFY’s criteria).16,17 From a clinical perspective, capturing these borderline cases could be seen as an advantage: a patient with a true low-level JCV antibody who was falsely labeled seronegative might be at some PML risk unbeknownst to the clinician. Furthermore, it has been noted that a proportion of JCV-seronegative results can occur in patients who actually have evidence of JCV infection (e.g., JCV DNA in urine).5,21
In addition, although differences between assays were generally minor, even small shifts in index values around clinically relevant thresholds (e.g., 0.8–0.9 or 1.4–1.5) may lead to different risk categorizations. For instance, a patient with an index of 0.85 would be considered low risk by STRATIFY (cutoff ⩽0.9), but intermediate risk by IMMUNOWELL (cutoff >0.8). These borderline cases may prompt different clinical decisions, such as whether to continue natalizumab beyond 24 months, intensify MRI monitoring, or initiate treatment switch discussions. 22 Thus, while the assays are highly concordant overall, clinicians should be aware that even subtle assay-dependent variations near threshold cutoffs can influence management, particularly when decisions hinge on crossing from low to intermediate risk categories.
Noteworthily, according to current risk stratification algorithms, patients with a JCV antibody index ⩽0.9 are considered to be at low risk of developing PML, with an estimated risk of <0.1 per 1000 patient-years, which is only marginally higher than that of seronegative individuals.1,11,23 Therefore, whether a borderline case is labeled “negative” or “positive (low titer)” might not drastically change the objective risk estimation. However, we acknowledge that in clinical practice, any “positive” designation, even at low index values, may lead to heightened caution or to avoidance of natalizumab, particularly at treatment initiation or after prolonged exposure. This highlights the need for careful interpretation of borderline results within their full clinical context and the importance of consistent testing methodology over time for an individual patient. If a change in assay is unavoidable (for instance, transferring care to a center using a different test), clinicians should interpret a single discordant result with caution and possibly confirm it with repeat testing.
On the other hand, when both assays register a high antibody index (i.e., >1.5), they almost invariably agree as no case of one high-positive and the other negative were detected. This implies that both assays have robust performance in clearly positive or clearly negative cases, and the slight differences are confined to the low-titer realm.
Our study has some limitations. First, the generalizability of the results may be limited by the monocentric design and the moderate sample size but larger multicenter studies would further validate these findings across diverse populations and laboratory settings. Second, we did not conduct longitudinal testing to assess whether either assay is more prone to fluctuations in serostatus over time, such as seroconversion or seroreversion, which are known to occur in natalizumab-treated patients.24,25 Third, the absence of confirmed PML cases in our cohort limits our ability to evaluate the predictive value of either assay for clinical outcomes. Additionally, our analysis treated STRATIFY as the de facto reference method; however, without an independent gold standard, such as JCV DNA detection, clinical follow-up, or serial serological monitoring, we could not definitively determine which assay was more accurate in the seven discordant cases. In particular, four patients (≈11%) tested negative with STRATIFY but positive with IMMUNOWELL, with low index values confirmed by inhibition testing. Whether these represent true low-level seropositivity missed by STRATIFY or false positives remains uncertain. The lack of retesting or follow-up limits our ability to resolve these discrepancies. Future studies incorporating molecular assays (e.g., JCV DNA PCR in blood or urine) and clinical endpoints could help clarify the clinical relevance of such discordant results.
Conclusion
Our findings strongly suggest that IMMUNOWELL can serve as a reliable alternative to STRATIFY for anti-JCV antibody detection. This has implications for laboratories and clinics, for instance, the IMMUNOWELL kit could be used, for example, in regions where Biogen’s assay service is not readily accessible, or in contexts like the emerging biosimilar natalizumab therapies which are paired with this test. Clinicians should be aware that a patient’s JCV serostatus result may vary slightly depending on the assay used, especially if the value is near the threshold. As a precaution, if a patient who was previously antibody-negative by one method tests low-positive by another, it may be wise to repeat the test or seek confirmation (perhaps even using both assays if available) before making major therapeutic decisions. On the other hand, a consistently negative result across either test is highly reassuring, and a strongly positive result is unequivocal. Ultimately, accurate and timely JCV antibody monitoring remains essential for the safe long-term use of natalizumab. By improving our toolkit for PML risk stratification, whether through new assays like IMMUNOWELL or refined risk models, clinicians can better tailor therapies to individual patient risk profiles, maximizing treatment benefits while minimizing risks.
