Abstract
Background:
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have emerged as potential neuroprotective agents for Parkinson’s disease (PD). However, evidence regarding their efficacy remains inconclusive.
Objective:
To assess the therapeutic effects and safety profile of GLP-1 RAs in patients with mild-to-moderate PD. We aim to conduct an updated systematic review to evaluate the effects of GLP-1 RAs in patients with mild-to-moderate PD.
Design:
Systematic review and meta-analysis of randomized controlled trials (RCTs) with trial sequential analysis (TSA) and Grading of Recommendations, Assessment, Development, and Evaluations certainty assessment.
Data sources:
PubMed, Embase, and the Cochrane Library were searched through April 14, 2025.
Methods:
We conducted a systematic review and meta-analysis of RCTs with TSA comparing GLP-1 RAs to placebo in patients with mild-to-moderate PD. The primary outcome was change in the Movement Disorder Society-unified Parkinson’s Disease Rating Scale (MDS-UPDRS) scores.
Results:
Five RCTs involving a total of 708 nondiabetic patients with mild-to-moderate PD were included. GLP-1 RAs significantly attenuated motor symptom progression, as evidenced by a mean difference in MDS-UPDRS Part III (off medication) of −2.06 (95% confidence interval (CI): −4.09; −0.03; I2 = 56%), with conclusive evidence supported by TSA. No statistically significant improvements were observed in other MDS-UPDRS domains, levodopa equivalent daily dose reduction, or functional scales (Parkinson’s Disease Questionnaire-39, Non-Motor Symptoms Scale for Parkinson’s Disease, UDysRS). A nonsignificant trend toward increased serious adverse events or treatment discontinuation was observed (odds ratio = 1.52; 95% CI: 0.66; 3.50), with low heterogeneity. TSA for secondary outcomes indicated that additional trials are required.
Conclusion:
GLP-1 RAs may provide a modest benefit in slowing motor progression in PD. However, their effects on nonmotor symptoms, medication use, and long-term safety remain uncertain due to the limited number of available trials. Further large-scale, long-duration trials are warranted.
Trial registration:
INPLASY2024110119.
Plain language summary
Parkinson’s disease is a brain disorder that worsens over time, causing problems with movement, balance, and daily functioning. Current treatments can reduce symptoms, but they do not slow the progression of the disease. Recently, drugs originally designed for diabetes—called GLP-1 receptor agonists—have shown promise in protecting brain cells. In this study, we analyzed data from five clinical trials involving 708 people with early to moderate Parkinson’s disease who did not have diabetes. We compared patients who received GLP-1 drugs with those who received a placebo. Our results showed that people taking GLP-1 drugs had slightly slower worsening of movement symptoms. However, the effect on other symptoms and medication needs was unclear. There were also some concerns about side effects, especially with higher doses. These findings suggest that GLP-1 drugs may offer a new way to help manage Parkinson’s disease. Larger, long-term studies are needed to confirm these benefits and better understand safety risks.
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder presented with motor and nonmotor symptoms. The incidence increases with advancing age. 1 While current treatment protocols primarily rely on dopamine-related medications to alleviate symptoms, disease progression often necessitates higher dosages over time. 2 This escalation in medication presents two significant challenges: a narrowing therapeutic window and increased side effects. 3 Despite alternative interventions such as deep brain stimulation and exercise therapy, optimizing levodopa concentrations in the brain continues to be the preferred therapeutic approach. 4
Recently, glucagon-like peptide-1 receptor agonists (GLP-1 RAs), originally developed for diabetes mellitus (DM), 5 have emerged as potential therapeutic candidates in PD. Evidence suggests that GLP-1 may exert neuroprotective, anti-inflammatory effects, reducing mitochondrial fragmentation and oxidative stress which potentially mitigates disease progression and alleviating symptoms.6–10 However, current randomized controlled trials (RCTs) findings are inconsistent, with some studies demonstrating significant benefits while others report minimal or no effects.11–14 Ongoing research seeks to evaluate the efficacy and safety of these agents in this context.15,16
Two systematic reviews have highlighted the potential benefits of GLP-1 RAs in PD. A Cochrane review reported low-certainty evidence for improvements in motor function.17,18 In contrast, a more recent systematic review demonstrated potential benefits of GLP-1 RAs in slowing motor deterioration, concluding high-certainty evidence with minimal risk of bias. However, one of the included studies in this review were unpublished and lacked peer review, potentially inflating the perceived robustness of findings. Notably, this review also did not assess the certainty of evidence using a standardized framework such as Grading of Recommendations, Assessment, Development, and Evaluations (GRADE), limiting the ability to fully interpret the certainty of the reported conclusions. 18
Given these conflicting quality assessments and persistent uncertainty, the robustness of current evidence remains inadequate for definitive conclusions regarding the efficacy of GLP-1 RAs. Therefore, to address these critical methodological limitations, our study aims to conduct a comprehensive and updated systematic review incorporating recently published trials and employs trial sequential analysis (TSA) along with GRADE assessment to rigorously evaluate the therapeutic potential and reliability of GLP-1 RAs in modifying PD progression.
Materials and methods
Search strategy
We conducted this systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline. 19 We performed a comprehensive search for RCTs in PubMed, Embase, and the Cochrane Library, covering studies published from database inception through November 28, 2024, without restrictions on language or publication date. Additionally, we conducted an updated search for any newly published or relevant literature up to April 14, 2025, to ensure the inclusion of the most recent studies. The search strategy incorporated a combination of free text, Medical Subject Headings, and Emtree terms. Two independent reviewers (K.-H.L., W.-W.T.) screened titles and abstracts using predefined eligibility criteria, with the senior author (M.-T.H.) providing oversight and resolving discrepancies during both screening phases. Key search terms included GLP-1 RAs and related medications and PD. Comprehensive information on the systematic review, including eligibility criteria, search method, and the selection process, is provided in Supplemental Material. The systematic review and protocol were registered on the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY2024110119).
Eligibility criteria
We included all RCTs aligned with our predefined criteria, encapsulated within the framework of PICO (patient, intervention, comparison, and outcome). The population of interest consisted of patients with PD with or without DM under medications treatment. The intervention is using GLP-1 RAs and the comparison is placebo. The primary outcomes of interest encompassed a change of Movement Disorder Society-unified Parkinson’s Disease rating scale (MDS-UPDRS) scores. When both ON- and OFF-medication assessments were reported, we analyzed them separately. Secondary outcomes are change of levodopa equivalent daily dose (LEDD), risk of adverse event and functional outcomes by change in scales of the Parkinson’s Disease Questionnaire (PDQ-39), unified dyskinesia rating scale and Non-Motor Symptoms Scale for Parkinson’s Disease (NMSS). Nonrandomized studies, noninterventional trials, observational studies, meeting abstracts, and articles without completed peer review or preprints were excluded.
Data extraction
We extracted the following data from each included study: trial name, first author, year of publication, sample size, PD duration at baseline, GLP-1 RA agents used, dose and duration, control condition, primary and secondary outcomes, and adverse events. When available, we prioritized results reported in the OFF-medication state. For trials with multiple time points or treatment arms, we used data corresponding to the highest dose and longest follow-up, unless otherwise justified. Only primary outcomes from RCTs were included in the meta-analysis; secondary analyses, such as post hoc biomarker studies or long-term extensions, were synthesized narratively or used to support safety and mechanistic interpretations.
Risk of bias
The risk of bias in the included studies was evaluated using the Cochrane Risk of Bias 2.0 (RoB2) tool for randomized clinical trials. 20 RoB2 assessments were conducted independently by two reviewers (K.-H.L., W.-W.T.). In instances of disagreement between the two evaluators on the risk of bias assessment, a third reviewer (M.-T.H.) facilitated resolution and reached a consensus. The evaluators examined various domains including the randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selection of reported results. The studies were then classified as having low risk, some concerns, or high risk of bias based on the assessment outcomes.
Statistical analysis
In this study, we performed random-effects meta-analysis using R software, version 4.3.1 (R Project for Statistical Computing) using the “meta” package. 21 We calculated the risk ratios (RRs) for binary variables and the mean differences for continuous variables, each accompanied by their respective 95% confidence intervals (CIs). Furthermore, we assessed the presence of heterogeneity in the data using Cochran’s Q and I2 tests. We considered a significance level of p < 0.05 (two-sided) for the Q statistic. The I2 values were interpreted as follows: 0%–40% might not be important; 30%–60% may represent moderate heterogeneity; 50%–90% may represent substantial heterogeneity; and 75%–100% indicate considerable heterogeneity. 22 To investigate the influence of each study on the overall effect-size estimate, we conducted sensitivity analysis using the leave-one-out method. If the number of included studies is fewer than 10, it will not be possible to conduct an Egger’s regression test to assess publication bias or perform a meta-regression analysis.23,24
To account for the risk of false-positive findings due to multiple testing, we performed a TSA using TSA Viewer version 0.9.5.10 Beta to evaluate the robustness of cumulative evidence for the primary outcome. The analysis was performed based on the pre-specified type I error of 5%, and type II error of 20% (power at 80%), examined the interaction between the cumulative Z-curve and the TSA boundary, incorporating calculations of trial sequential monitoring thresholds and the required information size. If the cumulative Z-curve crossed the TSA boundary, it indicated strong evidence supporting the intervention effect, reducing the need for further studies. In contrast, if the Z-curve did not intersect the boundary, it suggested that the evidence was insufficient for a conclusive interpretation. 25
Certainty of evidence
We assessed the certainty of evidence using the GRADE framework 26 evaluating factors including risk of bias, imprecision, inconsistency, indirectness, and publication bias. In alignment with GRADE methodology, we presented the findings within a minimally contextualized framework, assigning ratings to the strength of the observed effects. 27
Results
Study selection
An initial literature search identified a total of 946 study titles. The search process is illustrated in the flowchart provided in Figure 1. After removing duplicates, 120 studies were excluded. We applied a highly sensitive filter for RCTs to exclude 642 studies. Additionally, 177 studies were excluded based on titles and abstracts that did not meet our inclusion criteria. During the updated search, one study previously identified as a trial protocol had subsequently published its results and was therefore included in the full-text screening. 28 Consequently, eight studies11–14,28–31 underwent a full review. Among these, three were secondary analyses or long-term follow-up studies of previous RCTs, and thus were excluded from the meta-analysis but included in the qualitative synthesis.29–31

Identification and selection of studies for meta-analysis.
Study characteristics
Our analysis included five RCTs comprising 708 patients, of whom 67.09% (n = 475) were male. All RCTs were conducted in Europe, with four in the United Kingdom and one in France. One study was a single-center study, while the remaining four were multicenter studies. In these five RCTs, there were no diabetic patients, and the severity of PD was relatively mild to moderate (Hoehn and Yahr scale <3). Not all studies reported the race of the participants; however, given the locations of these studies, most participants were likely white. Additional individual study characteristics are provided in Table 1.
Basic characteristic of included trials.
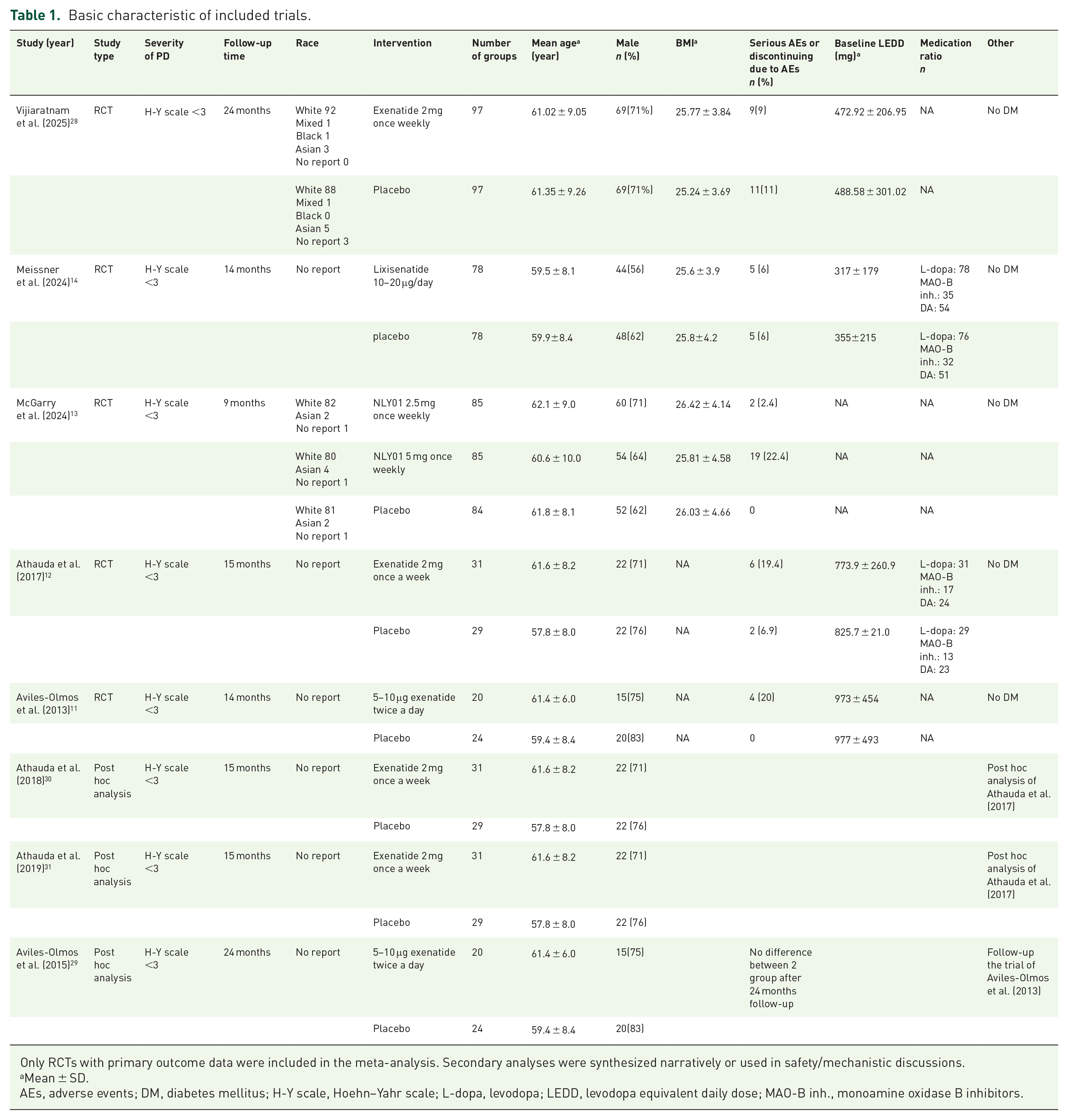
Only RCTs with primary outcome data were included in the meta-analysis. Secondary analyses were synthesized narratively or used in safety/mechanistic discussions.
Mean ± SD.
AEs, adverse events; DM, diabetes mellitus; H-Y scale, Hoehn–Yahr scale; L-dopa, levodopa; LEDD, levodopa equivalent daily dose; MAO-B inh., monoamine oxidase B inhibitors.
Risk of bias
A summary of the individual studies is provided in Figure S1. Most of the studies demonstrated a low risk of bias. One of the included studies was assessed as having a high risk in Domain 2 (bias due to deviations from intended interventions) because the investigators reported that participants might have detected their treatment allocation due to adverse events, including injection site reactions. Due to the limited number of studies included in each analysis (<10), we were unable to conduct an Egger regression test to assess publication bias or perform meta-regression.
MDS-UPDRS change in different domains
The analysis comparing changes in the MDS-UPDRS across different domains between patients receiving GLP-1 RAs included 619 patients from all 5 studies11–14,28 for Parts I, II, and III (off medication), and 450 patients from 4 studies11,12,14,28 for Part III (on medication) and Part IV (Figure 2). The group receiving GLP-1RAs demonstrated no significant trend in MDS-UPDRS Part I compared to the control group (mean difference = −0.03, 95% CI (−0.65; 0.58)) with moderate heterogeneity (I2 = 41%), but there was no statistical significance. The group receiving GLP-1 RAs demonstrated a slightly slower progression in MDS-UPDRS Part II compared to the control group (MD = −0.69, 95% CI (−2.40; 1.01)) with considerable heterogeneity (I2 = 76%), but the difference was no significant. The group receiving GLP-1 RAs demonstrated a slightly slower progression in MDS-UPDRS Part III (on medication) compared to the control group (MD = −1.64, 95% CI (−4.30; 1.03)) with considerable heterogeneity (I2 = 68%), but the difference did not reach statistical significance. The progression of MDS-UPDRS part III (off medication) significantly reduced motor progression compared to the control group (MD = −2.06, 95% CI (−4.09; −0.03)) with moderate heterogeneity (I2 = 56%). The progression of MDS-UPDRS part IV was found to be slightly less pronounced compared to the control group (MD = −0.12, 95% CI (−0.50; 0.25)) with low heterogeneity (I2 = 26%), but there was no significant difference. The overall change of MDS-UPDRS revealed significantly better outcome in the group with GLP-1 RAs (MD = −0.76, 95% CI (−1.36; −0.15)) with substantial heterogeneity (I2 = 61%).

Forest plot for change of UPDRS.
To assess the robustness and reliability of the accumulated evidence, TSA was performed. For most domains of the MDS-UPDRS, including Parts I, II, III (on medication), and IV, the cumulative Z-curve did not cross the trial sequential monitoring boundary, suggesting that the current evidence is insufficient and further randomized trials are warranted to confirm these findings (Figure S4(A)). In contrast, for MDS-UPDRS Part III (off medication), the cumulative Z-curve crossed both the conventional significance boundary and the TSA monitoring boundary before reaching the required information size, indicating that the evidence supporting the treatment effect is statistically significant and robust (Figure 3).

Trial sequential analyses for change of UPDRS part III off medication.
Change from baseline in LEDD
In comparing changes in LEDD between patients receiving GLP-1 RAs and placebo included 450 patients from 4 studies11,12,14,28 (Figure 4). The LEDD was found to be reduced more in patients receiving GLP-1 RAs than in the control group (MD = −3.10, 95% CI (−31.77; 25.57)) with moderate heterogeneity (I2 = 44%), but there was no significant difference. In TSA, the Z-curve did not surpass the TSA boundary, suggesting that additional trials are needed to confirm these results (Figure S4(B)).

Forest plot for change from baseline in LEDD.
Adverse events
Adverse events were assessed with a specific focus on serious adverse events (SAEs) or treatment discontinuations attributed to adverse effects. Data from 5 RCTs involving 619 participants were included in this analysis (Figure 5).

Forest plot for serious AEs or discontinuing due to AEs.
A dose-dependent pattern of adverse events was observed in McGarry et al., 13 where the 5 mg once-weekly NLY01 group exhibited a markedly higher rate of SAEs or discontinuations (29 participants, 22.4%) compared to the 2.5 mg group (2 participants, 2.4%). Similarly, Athauda et al. 12 reported a higher rate of SAEs in the exenatide group (6 participants, 19.4%) than in the placebo group (2 participants, 6.9%). The majority of these adverse events were gastrointestinal in nature and led to participant withdrawal.
The pooled analysis from all five studies revealed no statistically significant increase in the risk of SAEs or discontinuations in the GLP-1 RAs group compared to placebo (odds ratio (OR) = 1.52; 95% CI (0.66; 3.50)), with low heterogeneity (I2 = 27%).
Assessment of GRADE
The certainty of evidence was assessed using the GRADE framework, as detailed in Table S1. Most outcomes were graded as low-to-moderate certainty, primarily due to imprecision and heterogeneity.
Discussion
This meta-analysis evaluated the effectiveness of GLP-1 RAs in patients with mild-to-moderate PD without DM. Our findings demonstrated a significant reduction in symptom progression, with a mean decrease of 2.06 points in the MDS-UPDRS part III (off medication). This positive outcome remains robust in the TSA model, underscoring the conclusiveness of the results. Sensitivity analysis using a leave-one-out approach showed consistent effect estimates across all included studies, indicating that no single-study disproportionately influenced the overall result (Figure S2). However, in other domains of the MDS-UPDRS and in changes to the LEDD, no significant findings were observed. In addition, we also reviewed other functional outcomes, including the PDQ-39, the Unified Dyskinesia Rating Scale, and the NMSS. As shown in Figure S3(A)–(C), these forest plots revealed no significant differences between the GLP-1 RAs and placebo groups. TSA of these outcomes (Figure S4(C)–(E)) also indicated that the evidence remains inconclusive, and further studies are needed to clarify their effects. To address the limitations of prior systematic review, 18 our investigation adopted a more comprehensive framework. Specifically, we excluded articles lacking peer review, applied TSA to strengthen the conclusiveness of our findings, and employed enhanced critical appraisal methods to improve the rigor and reliability of our evidence quality assessment. This approach aims to provide more definitive insights into the therapeutic potential of GLP-1 RAs in PD management.
It is anticipated that MDS-UPDRS scores will worsen over time, given that PD is a slowly progressive disorder. We observed that GLP-1 RAs have a beneficial effect on motor symptoms, which may be attributed to their neuroprotective properties; however, most supporting evidence comes from animal studies. 8 The exact mechanisms underlying the neuroprotective potential of GLP-1 RAs remain to be fully elucidated. Previous studies have demonstrated their efficacy in neurodegenerative diseases such as Alzheimer’s disease (AD) and PD. 9 GLP-1 RAs enhance mitochondrial function by activating the cyclic adenosine monophosphate/protein kinase A pathway, leading to reduced mitochondrial fragmentation and oxidative stress, and restoration of mitochondrial membrane potential. 10 They also promote myelination and axonal regeneration, offering protection against demyelinating disorders like multiple sclerosis. 32 Additionally, these agents improve neuron-astrocyte metabolic coupling and support brain energy metabolism, thereby addressing proteopathic stress. 33 In the study by Athauda et al., 12 imaging demonstrated that patients using GLP-1 RAs exhibited increased dopamine signaling in the basal ganglia. However, in this meta-analysis, it is unclear whether the observed effect arises from a symptomatic benefit or a neuromodulatory mechanism.
Although there was no significant reduction in the LEDD compared to baseline, a downward trend in the use of dopamine medications was observed. A decrease in LEDD may have clinical implications, such as reducing dyskinesia and dopaminergic dysregulation syndrome. However, this meta-analysis included only mild-to-moderate PD, who had relatively low baseline LEDD and were followed for a short duration, potentially limiting the ability to detect these potential benefits. Nevertheless, the observed trend is promising and warrants further investigation in future research.
In evaluating functional outcomes for PD, GLP-1 RAs were associated with reductions in PDQ-39 scores and less severe changes in dyskinesia and NMSS scales compared to placebo (Supplemental Material). However, the lack of statistical significance, wide confidence intervals, and low heterogeneity highlight the uncertainty of these findings, potentially due to small sample sizes and study design variability. These results are consistent with prior research indicating the potential neuroprotective effects of GLP-1 RAs in PD. Notably, a systematic review and meta-analysis of preclinical rodent models demonstrated that GLP-1 RAs therapy confers neuroprotective benefits, 34 supporting its further investigation in clinical contexts.
It remains challenging to determine whether nonmotor symptoms directly influence motor function. Previous studies have shown that certain nonmotor symptoms can indeed have impact on motor function 35 ; however, it is difficult to determine whether this influence is driven by cognitive or emotional factors. Moreover, existing assessment scales often lack the precision to distinguish direct effects on motor function. In our study, we observed improvements in motor function, but it is challenging to ascertain whether these improvements were mediated by changes in emotional states.
In this study, we observed a dose-dependent increase in adverse events among participants receiving higher doses of GLP-1 RAs, with gastrointestinal symptoms being the most prevalent. 36 This finding aligns with existing literature, which reports that GLP-1 RAs commonly induce gastrointestinal side effects such as nausea, vomiting, and diarrhea, particularly during the initiation and dose-escalation phases of treatment. These adverse effects are often transient and can be mitigated by gradual dose titration. However, in some cases, they may lead to treatment discontinuation. Notably, a systematic review highlighted that the incidence of these gastrointestinal events is dose-dependent, emphasizing the importance of careful dose management to balance therapeutic efficacy with tolerability. 37 Therefore, clinicians should monitor patients closely during the initiation and adjustment of GLP-1 RAs therapy to manage and minimize potential adverse effects effectively.
Strength and limitation
This study has several limitations. First, because we included only patients with mild-to-moderate PD, our conclusions cannot be extended to those with more severe PD. Second, we analyzed outcomes only with the 2.5 mg dose of NLY01, excluding the 5 mg dose used in the trials by McGarry et al., 13 to avoid double counting the placebo group and erroneously inflating the sample size. Our review of this trial revealed that both dosages had similar efficacy, but the 5 mg group experienced more adverse effects. Third, as PD is a neurodegenerative disorder characterized by slow progression, our analysis with only a 1 to 2-year follow-up period may not capture long-term effects; therefore, longer-term studies are necessary. Fourth, the effect observed in MDS-UPDRS Part III might be amplified due to the normal disease progression seen in the placebo group. 38 Fifth, only five RCTs were included in our meta-analysis. To mitigate the risk of false-positive findings due to multiple testing, we employed TSA. Sixth, since the patients included in our study did not have DM, we cannot extend our findings to PD patients who also have diabetes. Therefore, the efficacy of GLP-1 RAs in this population remains uncertain. Seventh, the RCTs we included utilized only subcutaneous GLP-1 receptor agonists. It remains uncertain whether oral GLP-1 RAs would exhibit similar efficacy. Seventh, establishing a direct influence of nonmotor symptoms on motor function is challenging and difficult to demonstrate in our meta-analyses.
Conclusion
GLP-1 RAs may help slow motor symptom progression in mild-to-moderate PD but show no benefits in other domains or levodopa dose reduction. Higher doses increase gastrointestinal adverse events. While these findings suggesting GLP-1 RAs have potential protective effect in motor symptoms progressive of PD, they should be considered as adjunctive options rather than replacements for existing PD treatment regimens. Further large-scale, long-term studies are needed to confirm their efficacy and safety.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251372747 – Supplemental material for GLP-1 receptor agonists in Parkinson’s disease progression: a meta-analysis with trial sequential analysis of randomized controlled trials
Supplemental material, sj-docx-1-tan-10.1177_17562864251372747 for GLP-1 receptor agonists in Parkinson’s disease progression: a meta-analysis with trial sequential analysis of randomized controlled trials by Wen-Wen Tsai, Kuan-Hsien Lu, Jheng-Yan Wu, Min-Hsiang Chuang, Jui-Yi Chen, Chih-Cheng Lai, Kuo Chuan Hung and Meng-Tsang Hsieh in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251372747 – Supplemental material for GLP-1 receptor agonists in Parkinson’s disease progression: a meta-analysis with trial sequential analysis of randomized controlled trials
Supplemental material, sj-docx-2-tan-10.1177_17562864251372747 for GLP-1 receptor agonists in Parkinson’s disease progression: a meta-analysis with trial sequential analysis of randomized controlled trials by Wen-Wen Tsai, Kuan-Hsien Lu, Jheng-Yan Wu, Min-Hsiang Chuang, Jui-Yi Chen, Chih-Cheng Lai, Kuo Chuan Hung and Meng-Tsang Hsieh in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We extend our gratitude to Chi Mei Medical Center for providing the academic environment and resources essential to the success of this study. Their invaluable support has significantly enhanced the quality and breadth of this work.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
