Abstract
Background:
Detection of atrial fibrillation (AF) after ischemic stroke (IS) is crucial for starting anticoagulant therapy to prevent IS recurrence. Strategies to identify patients who might benefit the most from extended electrocardiography (ECG) monitoring would be highly desirable.
Objectives:
We aimed to investigate the role of brain natriuretic peptide (BNP) and echocardiographic parameters as biomarkers for predicting new AF detection in a large cohort of patients with acute IS.
Design:
We retrospectively analyzed data of 2411 consecutive patients admitted for IS to a single Stroke Center from January 1, 2018, to May 31, 2023.
Methods:
BNP levels were measured within 48 h of onset. Clinical and echocardiographic variables were evaluated. Single or multiple 12-lead ECG and continuous monitoring with external or implantable devices were used to detect new AF. The outcome measure was new AF detection at 3 months.
Results:
Of 2337 included patients, 1907 (81.6%) had not previously known AF, and new AF was detected in 422 (22%) patients. In the multivariate analysis, older age (odds ratio (OR) 1.03, 95% confidence interval (CI) 1.02–1.05, p < 0.001), higher National Institutes of Health Stroke Scale score (OR 1.05, 95% CI 1.03–1.07, p < 0.001), BNP ⩾150 pg/ml (OR 4.10, 95% CI 2.95–5.69, p < 0.001), and left atrium (LA) enlargement (OR 2.88, 95% CI 1.99–4.18, p < 0.001) were independently associated with new AF detection. The combination of both BNP ⩾150 pg/ml and LA enlargement showed a strong association (adjusted OR 4.74, 95% CI 3.47–6.48, p < 0.001) and a good discriminative performance for predicting new AF detection at 3 months (area under the receiver operating characteristic curve 0.74, 95% CI 0.71–0.77, sensitivity 0.70 (95% CI 0.65–0.74), specificity 0.78 (95% CI 0.76–0.80)).
Conclusion:
BNP ⩾150 pg/ml within 48 h of IS onset and LA enlargement, especially if combined, may be valuable biomarkers for predicting new AF detection and identifying patients who might benefit the most from extended ECG monitoring.
Introduction
Cardioembolic stroke accounts for nearly 30% of ischemic stroke (IS) and atrial fibrillation (AF) is the most common cause of cardioembolism. 1 Early detection of AF after acute IS is crucial for starting an effective secondary prevention strategy based on anticoagulation treatment. Recent guidelines recommend extended electrocardiography (ECG) monitoring for AF detection after IS, but there is uncertainty on timing, duration, and patient selection. 2 Studies on extended ECG monitoring with implantable loop recorders in patients with cryptogenic IS have shown that AF is detected in about 20%–30% of cases after 2 years.3–5 However, routine use of these devices in clinical practice presents several logistic and resource issues, and selection strategies to identify patients who might benefit the most from such interventions would be highly desirable. Previous studies have identified predictors of AF detection after IS, including age, vascular risk factors, and stroke occlusion pattern (embolic pattern, with cortical branch occlusion and multiterritorial involvement).5–8 Among the most promising are blood and echocardiographic biomarkers. Blood biomarkers previously associated with heart failure, such as brain natriuretic peptide (BNP) and N-terminal pro-BNP (NT-proBNP), have also been related to an increased risk of AF detection after IS.9–14 However, most studies were limited by small sample sizes, heterogeneous populations, and lack of a complete cardiologic assessment.
We aimed to investigate the role of BNP and echocardiographic parameters as biomarkers for predicting new AF detection in a large and homogenously studied cohort of patients with acute IS.
Materials and methods
Study population and data collection
We retrospectively analyzed data of 2411 consecutive patients admitted for IS to a single Stroke Center (Department of Neurology and Stroke Center, Maggiore Hospital, Bologna, Italy) from January 1, 2018, to May 31, 2023.
Inclusion criteria were acute IS admitted within 48 h of symptom onset, availability of BNP plasma levels within 48 h of symptom onset, and complete data on AF history. There were no specific exclusion criteria.
IS was clinically diagnosed by stroke neurologists and confirmed by neuroimaging. Computer tomography (CT) and/or magnetic resonance imaging (MRI) were used. In cases where CT imaging yielded negative results during hospitalization, and MRI was not performed, IS was defined based on clinical criteria as one or more focal neurological deficits of presumed vascular origin lasting more than 24 h, and exclusion of stroke mimics.
Patients received acute reperfusion treatments (intravenous thrombolysis, thrombectomy, or both), as indicated by the most recent guidelines on stroke treatment at that time.
We recorded demographic data (age, sex), vascular risk factors (hypertension, hypercholesterolemia, and diabetes mellitus), and medical history related to AF (previous AF), previous cerebrovascular events, coronary artery disease (CAD), and congestive heart failure (CHF). Stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS) score at admission.
To evaluate stroke etiology, we conducted various examinations based on patient profile, test availability, and patient consent. These examinations always included neck and intracranial vessel studies (CT angiography and/or neck and transcranial echo color Doppler), and cardiologic assessment for heart rhythm (12-lead ECG, 24-h Holter-ECG, and prolonged ECG monitoring in selected cases) and cardiac morphology (transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) in selected cases). All patients performed routine blood exams, including complete blood cell count, glucose, hemoglobin glycated (HbA1c), prothrombin time, partial thromboplastin activated time, fibrinogen, lipid profile, renal function, C-reactive protein, and BNP within 48 h of symptom onset. Stroke subtypes were classified according to the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) criteria. 15
Measurement of BNP plasma level
Whole venous blood samples were collected upon admission, within 48 h from symptoms onset, using EDTA-containing plastic tubes. Plasma BNP concentrations were promptly measured after collection through a commercially available immunoassay (Triage BNP Test, Biosite Inc., San Diego, California, USA). This assay employs a two-site immunoenzymatic approach, comprising two antibodies: a mouse monoclonal anti-human BNP antibody, immobilized on the solid phase, which binds to plasma BNP, and a mouse anti-human BNP antibody-alkaline phosphatase conjugate that specifically reacts with the bound BNP. A chemiluminescent substrate (Lumi-Phos 530) is added to the reaction and the light generated is measured with a luminometer.
Cardiologic assessment
To detect AF, patients underwent single or multiple 12-lead ECG, continuous ECG monitoring, single or multiple 24-h Holter monitoring with 7-day dynamic monitoring in most patients, or implantable loop recorder as feasible. Most patients received TTE. Patients under 60 years old with an embolic stroke pattern of undetermined origin received TEE for a more detailed cardiac characterization and exclusion of other cardioembolic sources. The following TTE parameters were recorded: left atrium (LA) volume index (LAVI), left ventricle (LV) end-diastolic volume index (LVVI), left ventricular ejection fraction (LVEF) with Simpson method, mitral valve (MV) regurgitation, LV filling pattern, and estimated pulmonary artery systolic pressure (ePASP). LA enlargement was defined as LAVI >28 ml/m2. LV dilatation was defined as LVVI >75 ml/m2. LV dysfunction was defined as LVEF ⩽50%. Severe MV regurgitation was defined as the presence of at least one among these parameters: central jet mitral regurgitation >40% LA or holosystolic eccentric jet, vena contracta ⩾0.7 cm, regurgitant volume ⩾60 ml, regurgitant fraction ⩾50%, effective regurgitant orifice ⩾0.40 cm2. Restrictive LV filling pattern was defined as E/A wave ratio >2 or 1–2 with an E wave deceleration time ⩽140 ms. Pulmonary hypertension was defined as ePASP ⩾50 mmHg.
Outcome measure
The outcome measure was the detection of new AF at 3 months. New AF was defined as a dysrhythmia, lasting at least 30 continuous seconds, with irregular RR interval and no detectable P waves on ECG, in patients without previous AF.
Statistical analysis
Continuous variables were presented as median (interquartile range, IQR), while categorical variables were presented as frequencies (percentages, %). For comparison between groups, the Mann–Whitney U test was used for quantitative variables with skewed distribution, while the Chi-square test (χ2) and Fisher’s exact test were used for qualitative variables. Quantitative data were tested with the Kolmogorov–Smirnov test to assess whether they were normally distributed. We examined the association of the variables, including BNP levels, with our outcome measure (i.e., new AF detection at 3 months). Youden’s index was used to identify the BNP threshold with the best discriminative performance for new AF detection. Variables that showed a statistically significant association with the outcome measure were included in a logistic regression multivariate analysis to assess an independent association. Discrimination performance was assessed by calculation of the area under the receiver operating characteristic curve (AUC-ROC). The results are presented as odds ratios (OR) and 95% confidence intervals (CI). Complete case analysis was used for handling missing data. Data were analyzed using a commercially available software package (IBM SPSS, version 24, IBM, Armonk, New York, USA). We established statistical significance at a two-tailed 0.05 level (p < 0.05).
Results
Of 2411 consecutive patients with IS, we finally included 2337 patients, of which 430 (18.4%) had previously known AF and 1907 (81.6%) had not previously known AF (Figure 1).

Flowchart of patients’ enrollment.
The characteristics of the population are summarized in Table 1. The median age was 78 years (IQR, 66–85), and 1145 (49%) patients were females. In comparison to patients without known AF, patients with known AF were older, and more often, females had more frequently a history of hypertension, CAD, CHF, and previous cerebrovascular events, had a higher baseline NIHSS score, and were less frequently treated with intravenous thrombolysis and more frequently with endovascular thrombectomy. As regards stroke etiology, patients with known AF had most of the time a cardioembolic stroke, while patients without known AF had more frequently a stroke of undetermined origin. The median BNP level was 160 (IQR, 62–372) pg/ml with a right-skewed distribution. Patients with known AF had a higher median BNP level than patients without known AF. As for echocardiographic parameters, patients with known AF had more often LA enlargement, LV dilatation, LV dysfunction, severe MV regurgitation, and PH in comparison to patients without known AF.
Characteristics of the population.

AF, atrial fibrillation; BNP, brain natriuretic peptide; CAD, coronary artery disease; CHF, congestive heart failure; IQR, interquartile range; LA, left atrium; LAA, large artery atherosclerosis; LV, left ventricle; MV, mitral valve; NIHSS, National Institutes of Health Stroke Scale; TIA, transient ischemic attack; TOAST, Trial of ORG 10172 in Acute Stroke Treatment.
New AF was detected in 422 (22.1%) patients without previously known AF, in 304 cases before hospital discharge and 118 cases after hospital discharge, during the 3-month follow-up.
Median BNP levels were significantly higher in patients with new AF in comparison with patients without new AF (282 (IQR, 168–548) vs 95 (IQR, 42–213) pg/ml, p < 0.001; Figure 2). The maximum value of Youden’s index was 0.46 for a BNP cutoff ranging from 149 to 151 pg/ml. We, therefore, identified a threshold of BNP ⩾150 pg/ml with the best discriminative performance for new AF detection to include as a categorical variable in the multivariate logistic regression model.

Levels of BNP in patients with and without new AF detection at 3 months.
The echocardiographic parameters associated with new AF detection in the univariate analysis that entered the multivariate logistic regression model were LA enlargement, LV dysfunction, restrictive LV filling pattern, severe MV regurgitation, and pulmonary hypertension.
The other variables associated with new AF detection in the univariate analysis that entered the multivariate logistic regression model were older age, female sex, history of hypertension, and higher NIHSS score.
In the multivariate analysis, older age (OR 1.03, 95% CI 1.02–1.05, p < 0.001), higher NIHSS score (OR 1.05, 95% CI 1.03–1.07, p < 0.001), BNP ⩾150 pg/ml (OR 4.10, 95% CI 2.95–5.69, p < 0.001), and LA enlargement (OR 2.88, 95% CI 1.99–4.18, p < 0.001) were the variables independently associated with a greater likelihood of new AF detection at 3 months (Table 2). BNP ⩾150 pg/ml had good discriminative performance for predicting new AF detection at 3 months, with high sensitivity and moderate specificity (AUC-ROC 0.73, 95% CI 0.70–0.76, sensitivity 0.80 (95% CI 0.76–0.84) specificity 0.66 (95% CI 0.63–0.68); Figure 3).
Univariate and multivariate analyses for new AF detection at 3 months.
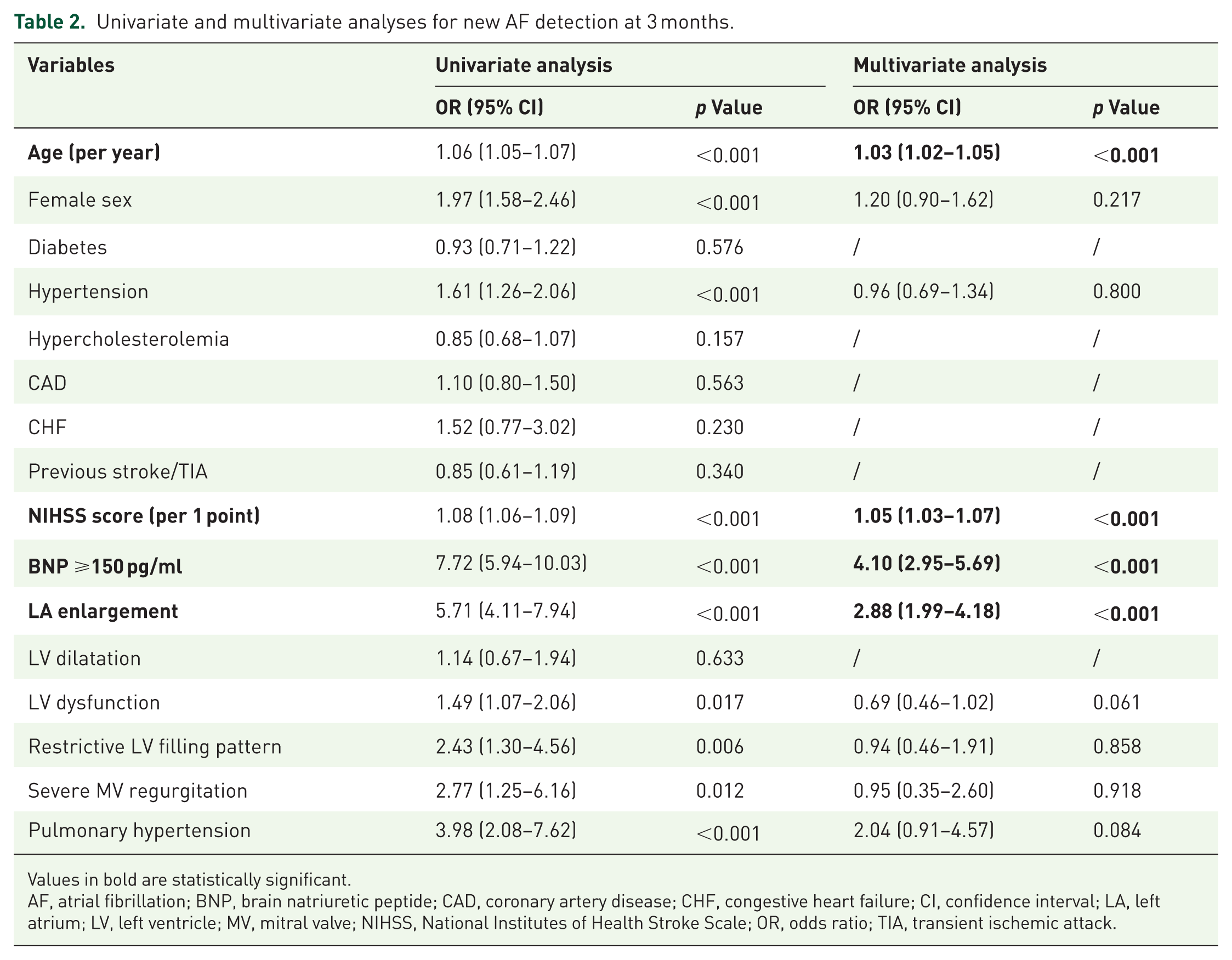
Values in bold are statistically significant.
AF, atrial fibrillation; BNP, brain natriuretic peptide; CAD, coronary artery disease; CHF, congestive heart failure; CI, confidence interval; LA, left atrium; LV, left ventricle; MV, mitral valve; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio; TIA, transient ischemic attack.

ROC curves of BNP ⩾150 pg/ml (AUC 0.73, 95% CI 0.70–0.76, sensitivity 0.80 (95% CI 0.76–0.84), specificity 0.66 (95% CI 0.63–0.68)), and of the combination of BNP ⩾150 pg/ml and LA enlargement (AUC 0.74, 95% CI 0.71–0.77, sensitivity 0.70 (95% CI 0.65–0.74), specificity 0.78 (95% CI 0.76–0.80)) for predicting AF detection at 3 months.
A second logistic regression model was performed, entering the combination of BNP ⩾150 pg/ml and LA enlargement as a single variable and the other variables associated with new AF detection in the univariate analysis. In this analysis, the combination of BNP ⩾150 pg/ml and LA enlargement showed an adjusted OR of 4.74 (95% CI 3.47–6.48, p < 0.001; Table 3). The combination of BNP ⩾150 pg/ml and LA enlargement showed good discriminative performance for predicting new AF detection at 3 months, with less sensitivity but significantly increasing specificity compared to BNP ⩾150 pg/ml alone (AUC-ROC 0.74, 95% CI 0.71–0.77, sensitivity 0.70 (95% CI 0.65–0.74), specificity 0.78 (95% CI 0.76–0.80); Figure 3).
Univariate and multivariate analyses for new AF detection at 3 months, considering the combination of BNP ⩾150 pg/ml and LA enlargement as a single variable.

Values in bold are statistically significant.
AF, atrial fibrillation; BNP, brain natriuretic peptide; CI, confidence interval; LA, left atrium; LV, left ventricle; MV, mitral valve; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio.
Discussion
Our study showed that older age, higher NIHSS score, BNP ⩾150 pg/ml, and LA enlargement were independently associated with a greater likelihood of new AF detection at 3 months in patients with acute IS.
These results are in line with previous evidence showing that older age and stroke severity are predictors of AF and cardioembolism.6,16,17 Moreover, our study confirms previous evidence on the potential role of laboratory (BNP) and echocardiographic (LA enlargement) parameters as biomarkers strongly associated with new AF detection.
A BNP level ⩾150 pg/ml within the first 48 h of IS emerges as the strongest predictor, increasing the risk of new AF detection by up to fourfold (OR 4.10, 95% CI 2.95–5.69, p < 0.001). BNP is a peptide produced by cardiomyocytes in response to various stimuli and is an established biomarker of cardiac diseases. 18 Previous studies showed elevated BNP levels in both paroxysmal and permanent AF.19,20 Moreover, BNP elevation predicted the occurrence of AF in a cohort of apparently healthy individuals. 21 Other studies found an association between natriuretic peptides and the risk of AF in patients with IS.9–14,22–26 BNP was the only laboratory biomarker externally validated in two studies,9,10 and one small study showed that a baseline high BNP level reduced the number needed to screen with extended ECG monitoring. 13 However, previous studies on BNP and AF detection after IS had important limitations due to small sample sizes, heterogeneous populations, and lack of a complete cardiologic assessment.
As regards echocardiographic parameters, multiple studies have detected an association between LA enlargement and the detection of AF.27–32 LA enlargement is the main echocardiographic biomarker of atrial cardiomyopathy, which is considered the common pathological feature of AF and an independent risk factor of IS. 33 LA remodeling, including changes in atrial tissue, size, cellular ultrastructure, and fibrosis, is believed to be the main cause of AF, and conversely, AF itself can lead to atrial remodeling.33,34 However, there is no single cardiac biomarker that is validated for AF screening after IS, and a recent international clinical consensus statement on atrial cardiomyopathy suggested, as a possible future direction, the use in combination of imaging and circulating biomarkers. 35
In this respect, the strength of our results is precisely the availability of both BNP and echocardiographic parameters in the largest population with acute IS ever studied to our knowledge. The combination of both BNP ⩾150 pg/ml and LA enlargement was strongly associated with new AF detection (adjusted OR 4.74, 95% CI 3.47–6.48, p < 0.001) and showed a good discriminative performance for predicting new AF detection within 3 months of IS (sensitivity 0.70 (95% CI 0.65–0.74), specificity 0.78 (95% CI 0.76–0.80)). Similar diagnostic accuracy was recently reported for NT-proBNP by a systematic review and meta-analysis on 2171 patients from 7 studies. 36 It is noteworthy that we included a larger sample size in a single-center study.
There is growing evidence supporting the use of prolonged ECG monitoring as an effective stroke prevention strategy in carefully selected patients. In particular, prolonged ECG monitoring is associated with a higher likelihood of AF detection and anticoagulant initiation to prevent stroke recurrence.37,38 Taken together, the results of our study may help to identify a subgroup of patients with IS who are at higher risk of developing AF. In particular, patients with both high BNP levels and LA enlargement should be carefully taken into consideration for more prolonged and costly ECG monitoring with implantable loop recorders. The practical use of BNP as a biomarker is highly feasible since the test is noninvasive, cost-effective, provides rapid results, and is widely available in hospital laboratories.
Our study has several limitations. First, it is characterized by the inherent weakness of any retrospective study, such as collection and entry bias and possible residual confounding. In particular, the duration and methods of cardiac monitoring were not the same for all patients, and variations in monitoring strategy may have influenced the likelihood of AF detection across patients. Nevertheless, this is a single-center study performed in a Comprehensive Stroke Center with standardized diagnostic protocols, and the vast majority of patients had at least a 7-day ECG monitoring during hospitalization or after discharge. Second, we did not collect information about several factors (i.e., pharmacological therapies and renal function) that may have an impact on BNP levels. Third, BNP rises early after stroke independently of AF, presumably due to BNP release from the brain, and samples taken in the first hours after stroke contain BNP not only from the heart but also from the damaged brain tissue. 22 Fourth, we did not collect data on thyroid disease, and therefore, we could not calculate the C2HEST score, 39 which was recently shown to be superior to other scores for AF detection in patients with cryptogenic stroke. 40 Fifth, there is a minority of patients with missing data on echocardiographic assessment. Sixth, the study population was relatively old, which may limit application to younger patients with cryptogenic or embolic strokes of undetermined source. Seventh, the study is not externally validated, and further evidence from other cohorts is warranted to assess generalizability.
Nevertheless, our study also has several strengths, mainly the considerably large sample size, the largest ever studied to our knowledge, with complete data on BNP levels. Other strengths are the inclusion of consecutive and homogeneously studied IS patients and the high percentage (about 75%) of patients with complete echocardiographic assessment.
Conclusion
Our study suggests that elevated BNP levels (i.e., ⩾150 pg/ml) within 48 h of acute IS onset and LA enlargement may serve as valuable biomarkers for identifying patients with a higher likelihood of new AF detection and who might benefit the most from extended ECG monitoring. In particular, the combination of both elevated BNP levels and LA enlargement showed a good discriminative performance for predicting the detection of new AF within 3 months of IS.
