Abstract
Background:
Non-ischemic cerebral enhancing (NICE) lesions are a rare complication following endovascular therapy (EVT) for cerebral aneurysms. Although first described in 2008, data on long-term outcome and treatment response remain limited.
Objectives:
In this study, we investigated the long-term follow-up of patients with NICE lesions, including magnetic resonance imaging (MRI) findings, clinical course, and treatment.
Design:
For this single-center ambispective observational study, we enrolled nine patients with NICE lesions after EVT for cerebral aneurysms.
Methods:
We analyzed patients diagnosed with NICE lesions following EVT between 2008 and 2024 at the University Hospital of Augsburg. Data collection included patients’ and procedural characteristics, clinical course, MRI findings, and response to immunotherapies.
Results:
We present the long-term follow-up of five patients already published and four additional cases. Nine female patients (mean age at diagnosis 50.67 ± 11.82 (± standard deviation, SD) years) were identified and analyzed with a mean follow-up of 1659.44 ± 1426.87 (SD) days, ranging from 328 to 5223 days (cumulative follow-up of 40.92 patient-years). In total, 112 MRIs were available for evaluation. Eight patients developed symptoms at a mean of 11 ± 13.41 (SD) days post-EVT, one patient remained asymptomatic. New NICE lesions during follow-up were detected in six patients, five patients developed new or increasing symptoms. All patients received glucocorticosteroids with variable duration, six patients required additional immunotherapies. At final follow-up, all patients had a favorable outcome (modified Rankin Scale 0–1), though residual symptoms persisted in four of them.
Conclusion:
Hitherto, this study presents the longest follow-up period of patients developing NICE lesions after EVT. NICE lesions may have a highly variable course regarding radiological and clinical characteristics, with potential for both clinical and radiological recurrence years after initial presentation. While immunosuppressive therapy appears effective, optimal treatment regimens and duration have yet to be determined. Our findings underline the importance of regular clinical and MRI controls for individual patient care in this rare condition.
Introduction
Endovascular therapy (EVT) has emerged as the predominant treatment modality for cerebral aneurysms in recent decades. 1 While major complications such as procedural aneurysm perforations and thromboembolic events are well documented, delayed non-ischemic cerebral enhancing (NICE) lesions have been identified as a rare but significant complication following EVT.2–19 NICE lesions are characterized by punctate, nodular, or annular foci of leptomeningeal, cortical, and subcortical enhancements with perilesional edema, predominantly distributed in the territory of the endovascular access route. 2 Reported incidence rates vary between 0.05% and 2.3%, with most studies suggesting rates below 1%.2–5,7,10,14,20,21
The pathogenesis of NICE lesions has been attributed primarily to foreign body emboli, particularly hydrophilic polymer coating emboli leading to granulomatous reactions, as confirmed through biopsy-proven cases.2,6,8,10,11,22,23 Additional mechanisms have been proposed, including cerebral metallic hypersensitivity and nickel allergy. However, these mechanisms have since been refuted.2,4
Clinical manifestations of NICE lesions are heterogeneous, encompassing asymptomatic cases to mild and severe neurological presentations, including headache, epileptic seizures, and focal neurological deficits such as hemiparesis.3–5
Regarding treatment of this rare disorder, several immunotherapies have been reported, mostly glucocorticosteroids (GCS) and various steroid-sparing immunosuppressants like mycophenolate mofetil (MMF), azathioprine (AZA), or methotrexate (MTX),23–25 with heterogeneous results. Up to now, comprehensive data on long-term outcomes and treatment efficacy remain limited.2–7,10,14,16,21,25 Due to the highly variable course of the disease and the asymptomatic cases, there are no recommendations, on which patients should be selected for treatment and how long treatment should be continued.
Current literature on NICE lesions is predominantly based on case reports and small case series with limited follow-up periods. In our recent comprehensive analysis 5 of 746 patients undergoing EVT at the University Hospital of Augsburg, delayed NICE lesions emerged as a rare but significant complication with an incidence rate of 0.67%. The therapeutic response to various immunotherapies during a follow-up interval of up to 132 months (60.17 ± 48.74 months; mean ± SD) showed marked heterogeneity, with GCS, tocilizumab (TCZ), and MMF showing favorable outcomes in select cases, while rituximab (RTX) and AZA failed to achieve disease stabilization. The present study aims to reveal further long-term data and includes new cases focusing on lesion development in magnetic resonance imaging (MRI), clinical course, and response to immunotherapies
Methods
We conducted a prospective and retrospective single-center study to analyze the long-term follow-up of patients with NICE lesions. All patients aged 18 years and older diagnosed with NICE lesions at the University Hospital of Augsburg after EVT for symptomatic or asymptomatic cerebral aneurysms between May 1, 2008 and December 20, 2024 were included. No exclusion criteria were set. Data were collected retrospectively and prospectively up to the data cut-off in December 2024. Cases 1–5 have been reported previously, 5 here, we provide further follow-up data; additionally, four newly diagnosed cases (cases 6–9) are included. NICE lesions were defined as symptomatic or asymptomatic punctate, circular or nodular contrast enhancement of the brain parenchyma or leptomeninges (on Gadolinium-enhanced T1-weighted imaging) with or without perilesional edema (on T2 fluid-attenuated inversion recovery (FLAIR) weighted imaging) occurring after an endovascular procedure and mainly located in the vascular territory of the endovascular procedure. 2
All endovascular interventions incorporated a standardized digital subtraction angiography (DSA) of the brain-supplying vessels, comprising evaluation of bilateral vertebral arteries and internal carotid arteries in four standard planes, supplemented by three-dimensional rotational angiography of the aneurysm-hosting vessel. Treatment decisions and device selection were determined based on angiographic findings and encompassed multiple devices, including various coils, stent- or balloon-assisted coiling, and both intra- and extra-aneurysmal flow diversion devices. The standardized MRI protocol for post-endovascular aneurysm treatment surveillance consisted of diffusion-weighted imaging (DWI), high-resolution T2-weighted sequences over the treated aneurysm in axial and coronal planes, MR angiography with and without intravenous contrast administration, and T1-weighted sequences of the entire brain following intravenous contrast administration. 5 Susceptibility-weighted imaging (SWI) sequences were not available for most of our patients, as they are not included in our follow-up MRI protocol; therefore, an analysis of SWI sequences was not possible in our cohort.
According to our institution’s standard follow-up protocol for EVT of intracerebral aneurysms, all patients underwent a cerebral MRI follow-up 3 or 6 months after EVT; if new or worsening clinical symptoms were detected, MRI was performed immediately. All patients with NICE lesions, symptomatic or asymptomatic, were included in our analysis and received further MRI and clinical follow-up, with the frequency depending on their clinical course, treatment, and lesion development. MRIs were analyzed by at least one senior neuroradiologist.
In our study, we also employed the LST-AI method for the automated segmentation of NICE lesions using T1-weighted and FLAIR MRI scans. 26 LST-AI is an advanced, deep learning-based extension of the Lesion Segmentation Tool (LST), 27 specifically designed for the accurate delineation of brain white matter lesions (Supplemental Figure 1). Details on automated lesion segmentation are summarized in Supplemental Figure 1. 28
The following data at diagnosis and during follow-up were collected: patient characteristics (including age, gender, allergies, comorbidities), aneurysm characteristics (i.e., number, location, maximum diameter, ruptured, symptomatic or asymptomatic), procedure-related data (i.e., intervention duration, materials), laboratory findings (where available including cerebrospinal fluid (CSF)), dermatological testing, clinical course and treatment, cognitive testing (self-reported and where available test results performed by a qualified neuropsychologist), electroencephalography, duplex sonography and DSA. Moreover, we analyzed MRI characteristics (i.e., number of lesions, perilesional edema, presence of lesions beyond the primarily targeted vascular territory at onset and dynamics of lesions and perilesional edema during follow-up). The modified Rankin Scale (mRS) was used as overall parameter for clinical outcome. Further outcomes of interest included new or worsening symptoms (including headache, epileptic seizures, cognitive deficits, focal neurological deficits), as well as MRI outcome parameters (presence or absence of new NICE lesions and perilesional edema).
This study followed the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statements. 29
Statistical analysis
Statistical analyses were performed with SPSS version 29.0 (IBM Corporation, New York, NY, USA). Continuous variables are presented as mean ± standard deviation (SD). For categorial variables, data are reported as counts or frequencies. Pearson correlation test was used to analyze the correlation between two variables. p Value below 0.05 was considered statistically significant.
Results
Patients’ and procedural characteristics
Nine patients were diagnosed with NICE lesions between May 1, 2008 and December 20, 2024, at the University Hospital of Augsburg. In this period, a total of 1759 aneurysms were treated, of which 838 were unruptured, this corresponds to an incidence of 0.51% for NICE lesions.
All nine patients were female with a mean age of 50.67 (±11.82 SD) years at diagnosis. Two out of nine patients were treated for symptomatic aneurysms (case 3 presented with stroke and case 8 with an oculomotor nerve palsy and subarachnoid hemorrhage (Hunt and Hess Scale grade 2), the remaining seven patients had asymptomatic aneurysms (Table 1).
EVT characteristics.

For case 1–5 data according to Bayas et al. 5 For all patients guide wire Terumo 0.035″ and sheath Terumo 6F or 7F were used.
AcomA, anterior communicating artery; EVT, endovascular treatment; F, female; ICA, internal carotid artery; M, male; MCA, middle cerebral artery; MPD, multipurpose D; NA, not available; PcaA, pericallosal artery; PcomA, posterior communicating artery; SACE, stent-assisted coil embolization; WEB, Woven EndoBridge.
All treated intracranial aneurysms (n = 18) were situated in characteristic locations within the Circle of Willis or at the middle cerebral artery bifurcation. The aneurysms demonstrated small to medium dimensions, with maximum diameters not exceeding 12 mm. No patients with inflammatory aneurysms developed NICE lesions.
Multiple aneurysms were treated in cases 1–3 and case 5, while the other patients underwent EVT for a single aneurysm. Endovascular treatment consisted of standalone coiling in case 8, whereas the other aneurysms were treated with either Woven EndoBridge (n = 2), stent-assisted coiling (n = 12), or flow-diverter (n = 2). Aneurysm characteristics, endovascular devices and materials used, and procedural duration are provided in Table 1. Of note, case 3 underwent three endovascular interventions during the follow-up period.
During follow-up, DSA was performed in cases 1, 3, 5, 6, 8, and 9; case 1 underwent DSA several years post-EVT due to amaurosis fugax without an explanatory finding. Notably, in case 6, NICE lesions manifested 223 days after EVT, but only 7 days following the routine follow-up DSA. Cases 3 and 5 demonstrated subtle irregularities of peripheral arteries within the affected vascular territory, potentially suggestive of vasculitis. 5
CSF analysis was performed in seven patients, with pleocytosis detected in two patients, showing leucocyte counts of 37 and 56/µl, respectively.
Clinical course and MRI follow-up
The mean interval between the first EVT and the detection of NICE lesions was 69.78 days (±63.03, SD, range 16–223 days), whereby in case 6 NICE lesions did not occur until 223 days after EVT but 7 days after DSA. Table 2 shows patients’ characteristics, as well as symptoms, MRI and CSF results at the time of diagnosis. Figure 1 summarizes the clinical course, MRI activity, and treatment.
Patients characteristics.

For case 1–5 data according to Bayas et al. 5
Because of intermediate-type (fet al/adult) PCA both sides, NICE lesions in the PCA territory cannot be reliably assigned to the anterior or posterior circulation.
Initially, the condition was not reported by the patient but detected through cognitive testing.
CSF, cerebrospinal fluid; DSA, diagnostic angiography; EVT, endovascular treatment; F, female; M, male; MRI, magnetic resonance imaging; NA, not available; NICE, non-ischemic cerebral enhancing; PCA, posterior cerebral arteries.

Clinical course.
Symptoms developed in eight out of nine patients 11 ± 13.41 days (mean ± SD; range 1–40 days) after EVT, while one patient (case 6) remained asymptomatic. The presenting symptoms comprised headache (n = 8), focal neurological deficits (n = 8; including (hemi-)paresis (n = 5), aphasia or dysarthria (n = 2), sensory disturbance (n = 3), hemianopsia or visual disturbance (n = 3), neglect (n = 1), dizziness (n = 2)), epileptic seizures (n = 1), and cognitive impairment (n = 6; Table 2). At onset, symptomatic patients were mildly to moderately affected, with a mRS of 1 in one patient, 2 in six patients, and mRS 3 in two patients.
The follow-up period varied from 328 to 5223 days (mean ± SD, 1659.44 ± 1426.87 days) and covered 40.92 patient-years in total (Table 3). During the follow-up period, new or increasing symptoms were documented in five patients (cases 1–3, 5, and 9). Case 2 presented with isolated headache, while the remaining four patients exhibited new or progressive seizure activity, focal neurological deficits, and/or cognitive deterioration. In case 3, deterioration was observed following each subsequent EVT (second and third EVT). The onset of new or worsening symptoms ranged from 36 to 2014 days post-EVT (case 1: 2014 days; case 2: 115 days; case 3: 1673 days after second EVT (3761 days after initial EVT); case 5: 1156 days; case 9: 36 days post-EVT). Case 6 remained asymptomatic throughout the observation period. Cases 4, 7, and 8 demonstrated clinical stability or improvement, with no evidence of symptom recurrence or progression.
Follow-up times and immunotherapy.

GCS also for dermatological indications.
Died due to coexisting aggressive B-cell lymphoma.
AZA, azathioprine; CYC, cyclophosphamide; EVT, endovascular treatment; GCS, glucocorticosteroids; MMF, mycophenolate mofetil; MRI, magnetic resonance imaging; MTX, methotrexate; RTX, rituximab; TCZ, tocilizumab.
MRI demonstrated characteristic punctate or patchy contrast enhancement patterns. In one case (case 3), linear to cortical enhancement was observed, likely associated with concurrent changes from aneurysm-related infarction of a partially thrombosed symptomatic aneurysm. 5 NICE lesions were predominantly localized in the territory of the treated aneurysm(s). All vascular territories exhibiting NICE lesions had undergone aneurysm treatment or DSA. Initial MRI characteristics occasionally included extensive perifocal edema on T2-weighted sequences, without significant mass effect, and T2-shine through on DWI. No patients exhibited midline shift or hemorrhagic components.
Long-term MRI follow-up ranged from 328 to 5222 days (mean ± SD: 1639.11 ± 1435.38 days; equivalent to 4 years, 5 months, and 25 days), representing a cumulative follow-up of 40.42 patient-years (Table 3). During this period, the cohort (n = 9) underwent a total of 112 MRI examinations (including baseline diagnostic scans), with individual patients receiving between 5 and 23 MRIs.
During follow-up, new NICE lesions were detected in six out of nine patients (cases 1–3, 5, 7, and 8), while lesions with new or increasing perilesional edema were observed in five out of nine patients (cases 2, 3, 5, 7, and 8). In case 3, the radiological worsening occurred exclusively in temporal association with subsequent EVTs, and not independently. New or enlarging NICE lesions were detected up to 2396 days post-EVT (case 1: 1163 days, case 2: 2396 days, case 5: 1472 days; case 7: 369 days; case 8: 989 days; mean ± SD: 1277.80 ± 664.92 days), while new or increasing perifocal edema was observed up to 1044 days post-EVT (case 2: 544 days, case 5: 1044 days, case 7: 369 days, case 8: 774 days; mean ± SD: 682.75 ± 253.24 days). Figure 2 illustrates the dynamics of MRI manifestation and clinical symptoms for all cases. The data suggests that with increasing time interval from EVT, recurrent NICE lesions are less frequently accompanied by perifocal edema.

MRI and clinical follow-up for cases 1–9.
New lesions occurred with or without perilesional edema or clinical symptoms, respectively. For instance, Figure 3 shows the disappearance and partial recurrence of NICE lesions, accompanied by perilesional edema to varying degrees in case 5.
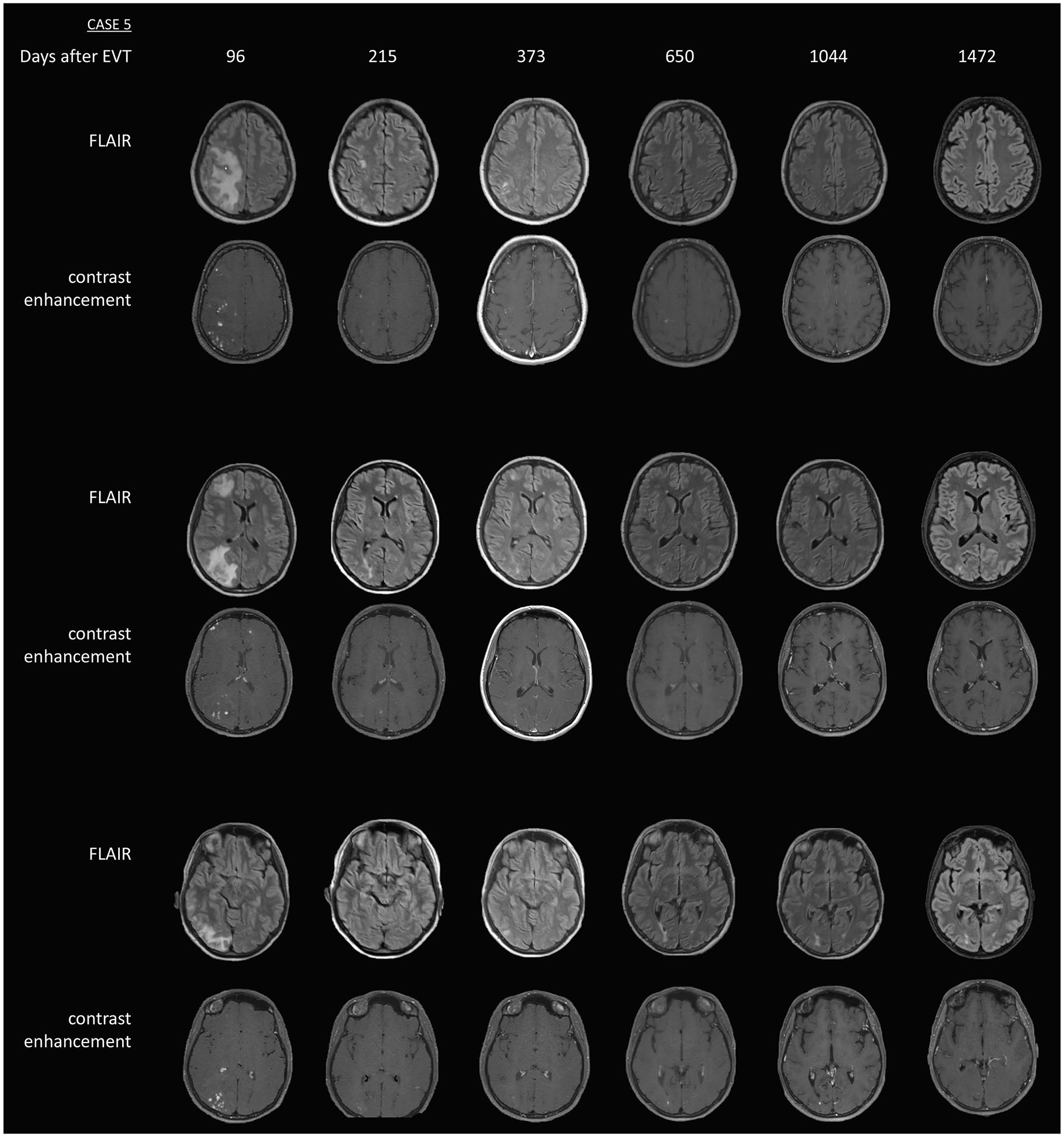
MRI at diagnosis of NICE lesions and during follow-up (select time points) in case 5.
Clinical progression or improvement was observed in some cases without corresponding MRI changes. Four patients (cases 1, 2, 5, and 8) exhibited clinical changes accompanied by concurrent radiological progression (new lesions and/or edema) and regression (reduction in lesions and/or edema). The relationship between MRI abnormalities and clinical manifestations is illustrated in Figure 4 (bubble chart) and Figure 5 (bar chart). Correlation analysis showed that among symptoms (including headache, epileptic seizures, cognitive deficits, focal neurological deficits), headache (Pearson correlation coefficient (PCC) = 0.344, p = 0.0001) and focal neurological deficits (PCC = 0.365, p = 0.0001) had the strongest correlation with the recurrence of NICE lesions. The correlation between epileptic seizures (PCC = 0.181, p = 0.0657) and NICE lesion recurrence did not reach statistical significance, potentially related to the limited sample size of only one patient experiencing seizures. Considering any symptoms (headache, epileptic seizures, cognitive deficits, and/or focal neurological deficits), correlation analysis revealed a PCC of 0.368 (p = 0.0001).
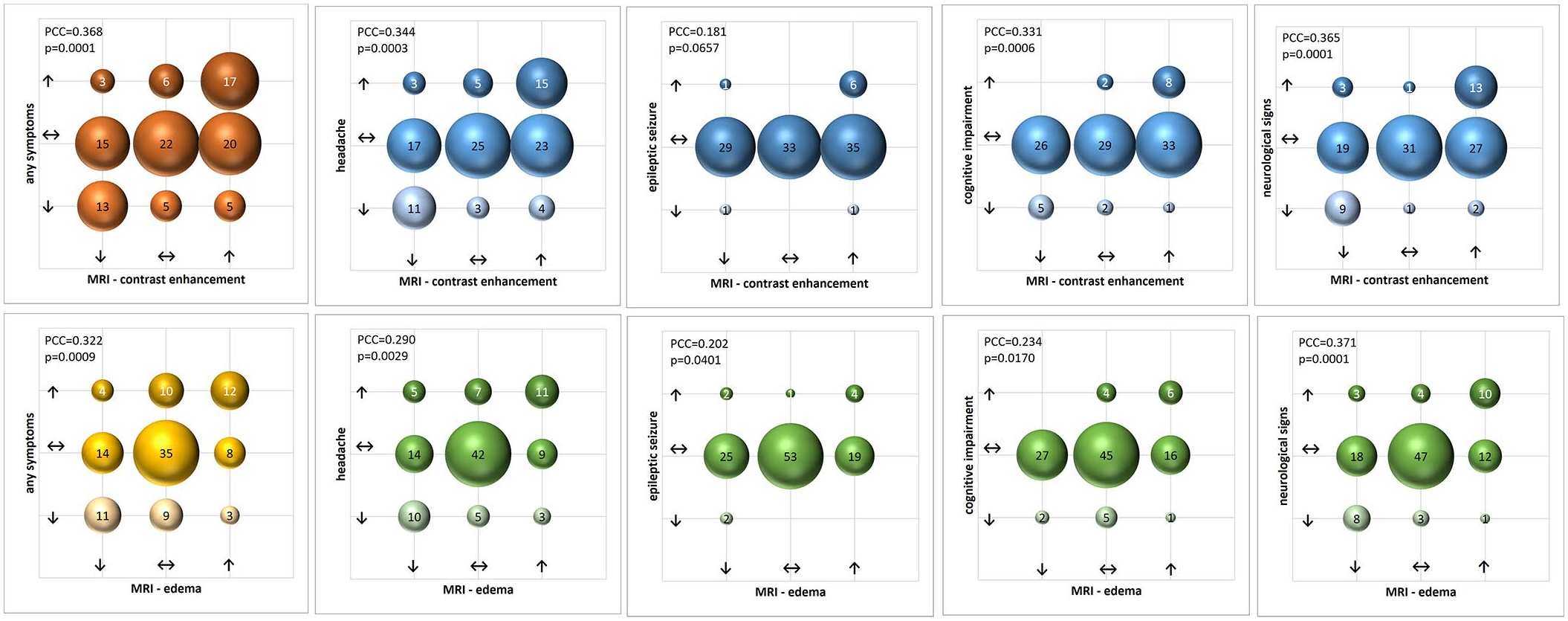
Correlation between MRI and clinical symptoms.

MRI changes visualized in relation to the change of clinical symptoms during follow-up.
New or enlarging NICE lesions were detected in 34 of the 112 MRIs, 13 out of these 34 MRIs showed increasing or new perilesional edema. Thirty-three of 34 MRI examinations were performed while patients were undergoing immunotherapy.
Treatment
Following NICE diagnosis, with symptomatic presentation in eight patients, the therapeutic regimen included GCS in all patients (n = 9), AZA in two patients, MMF in five patients, MTX in one patient, RTX in one patient, cyclophosphamide (CYC) in two patients, and TCZ in four patients. Three patients received GCS monotherapy (Figure 1 and Table 3). In symptomatic patients, immunotherapy was initiated at a mean of 73.25 days (±110.04 days, SD) after symptom onset. The patient without symptoms received GCS only for 16 days, also due to pre-existent type 1 diabetes mellitus. At final follow-up, five patients remained on immunotherapy, while treatment was discontinued in four patients with a mean duration of 8 months and 7 days (250.67 ± 300.39 days, mean ± SD). Significant adverse events occurred in two patients: cytomegalovirus colitis (case 5; while under GCS, MMF, TCZ) and depression requiring psychiatric hospitalization (case 7; under GCS therapy). Case 4 died due to coexisting aggressive B-cell lymphoma.
At last follow-up, four patients remained on GCS therapy. One patient received combination therapy with MMF at 2 g/day, and another patient was maintained on TCZ monotherapy (8 mg/kg every 4 weeks; Figure 1 and Table 3). One patient required concurrent antiepileptic medication for seizure management (levetiracetam and lamotrigine). The last clinical assessment revealed favorable outcomes in all patients, with mRS of 0–1, indicating minimal or no disability. Complete remission of symptoms was noted in five of nine patients, while residual symptoms persisted in four patients, that is, headache (case 5), cognitive impairment (cases 1, 5, 7, and 9), and mild gait ataxia (case 1).
Automated segmentation
Despite utilizing state-of-the-art methodology, the lesion segmentation process did not achieve complete automation in this work (Supplemental Figure 2). Due to variations in study protocols and scanner specifications affecting the pre-trained LST-AI model’s performance, manual adjustments remain necessary to ensure accurate lesion localization.
Discussion
NICE lesions represent a rare complication after EVT for cerebral aneurysms. Here, we report a series of nine patients with NICE lesions after EVT with the to date most extensive long-term follow-up of 40.92 patient-years in total. Our study not only uncovers previously unreported temporal patterns of perilesional edema regression over time but also highlights significant correlations between radiological progression of NICE lesions and clinical manifestations, especially in relation to headache and focal neurological deficits.
The initial description of NICE lesions following EVT was first published in 2008, 8 followed by numerous case reports and series in the literature.2–4,6,7,10,13–15 The largest cohort to date was reported by Shotar et al. 3 based on retrospective data from the French National Registry analyzing 32 treated aneurysms in 31 patients. The radiological follow-up of 22 of these patients has been reported recently. 13
The mean follow-up period in our study was 1659.44 days (approximately 4.55 years), substantially exceeding previously reported long-term data. Prior studies reported a mean follow-up of 548.2 ± 467.0 days 4 and 25 ± 26 months 3 . The longer observation period allowed us to identify several important aspects of NICE lesions’ development and clinical courses.
Compared to our previous report, 5 our current analysis, covering a period of about 16.5 years, demonstrated a comparable incidence rate of NICE lesions following EVT. The observed incidence was 0.51% among 1759 treated aneurysms between May 2008 and December 2024, with all cases except one occurring in patients with unruptured aneurysms. This finding is consistent with our previously reported incidence rate of 0.67% for the period until December 2020. The rates observed fall within the range of previously reported incidences in the literature, which demonstrate considerable variability from 0.05% 3 to 2.3%. 14 Comparable incidence rates were documented by Shotar et al. 2 at 0.5% and Shapiro et al. 10 at 0.6%. However, other studies have reported substantially different figures, ranging from 0.05% 3 and 0.14% 21 up to 1% 4 and 2.3%. 14 The wide range of reported incidences may be attributed to different follow-up protocols, particularly regarding asymptomatic patients, and also follow-up durations.
Regarding the potential correlation between the type of endovascular devices used and the clinical course or treatment response, our cohort included six patients treated with bare platinum coils and six patients with LVIS EVO stents. However, due to the limited sample size, no meaningful conclusions can be drawn about the impact of specific devices, associated catheter materials, their combination, or the use of double platelet inhibition on the manifestation or progression of NICE lesions. Further investigations involving larger patient cohorts are required to better understand potential associations in this rare condition.
Our cohort consisted exclusively of female patients, which may to some extent reflect the known higher prevalence of intracranial aneurysms in females. 30 The mean age of 50.67 years aligns with previous reports, where mean ages ranged from 45 to 59.5 years.3,4,6,7,14 The female predominance in our series exceeds the high proportions reported in previous studies, which ranged from 53.3% to 85%.3,4,6,7 Notably, a case series by Nakagawa et al. also included only female patients. 14
Similar to previous reports,3,4,7,15,20 most aneurysms reported here were unruptured and located in the anterior circulation.
Our cohort exclusively comprises patients with NICE lesions following EVT for aneurysms. Recent evidence suggests that NICE lesions can also occur as a rare complication following mechanical thrombectomy (MT) for acute ischemic stroke. Forestier et al. 31 reported three cases of NICE lesions after MT through a retrospective French nationwide registry, with an overall incidence of 3 out of 34,824 MT procedures. The clinical and radiological presentation of post-MT NICE lesions was similar to those after endovascular aneurysm treatment. This finding highlights the importance of considering NICE lesions as a potential adverse event following various endovascular interventions and underscores the role of contrast-enhanced MRI in the diagnostic workup of unexplained symptoms or imaging findings during post-procedural follow-up.
The mean time to symptom onset in our cohort was on average 11 ± 13.41 days (mean ± SD) post-EVT, the mean time to detection on MRI 69.78 days (±63.03, SD, range 16–223 days), whereby in case 6, NICE lesions were not detectable until day 223 after EVT but 7 days after control DSA. The time interval to symptom onset or MRI findings in our cohort is notably shorter than previously reported. Shotar et al. 3 reported a mean interval to diagnosis of 5 ± 9 months, with 32% of cases diagnosed within 1 month, while in single cases longer intervals up to 36 months have been described. Richter et al. 4 reported MRI findings 65.1 days (mean) following EVT, while Ikemura et al. 7 observed a median period of 71.5 days for symptom onset or MRI lesions. The shorter interval in our cohort may reflect the heightened awareness of this complication and systematic post-procedural monitoring at our center. However, NICE lesions can occur over long intervals, ranging from days to several years after EVT. 24
The clinical correlate of NICE lesions is broad, ranging from incidental findings to symptoms of variable severity over years.3,13,14 The proportion of symptomatic patients at onset (eight out of nine patients) was higher than previously reported rates of 44%–74%,3,4,7 possibly reflecting increased awareness or referral bias. Symptoms in our cohort included headache (8/9 patients), focal neurological deficits (8/9 patients), and cognitive impairment (6/9 patients), broadly aligning with previous reports, though the overall symptom burden appeared to be higher than previously reported.3,4,7 Epileptic seizures were observed in one case, representing a lower incidence compared to previous studies, where seizures were reported in one third of patients by Shotar et al. 3 and in 2 out of 15 patients by Richter et al. 4 One of our cases was asymptomatic, in contrast Shotar et al. 3 reported eight asymptomatic patients in their cohort of 31 cases, and Ikemura et al. 7 nine asymptomatic cases among 16 patients.
The high rate of radiological recurrence (6/9 patients) and clinical progression (5/9 patients) in our cohort during follow-up suggests that NICE lesions may have a more dynamic long-term course than previously assumed. New lesions were detected up to 2396 days post-EVT, considerably longer than the timeframes reported in previous studies. Guetarni et al. 13 noted that new lesions occurred mainly within the first 200 weeks after the procedure in 73% of patients, while our findings suggest that the window for new lesion development may extend beyond this period. This observation is compatible with histological evidence showing granulomatous foreign body lesions in patients with NICE lesions up to 4 years post-procedure. 24 The recurrence rate of NICE lesions in our cohort (6/9 patients) is similar to the previously reported rate of Guetarni et al., 13 who found that among 22 patients 16 (72%) had new lesions during follow-up. Our close MRI monitoring revealed complex patterns of lesion evolution. The observation that new lesions can occur with or without perilesional edema, and that clinical progression may occur independently of radiological changes, aligns with recent findings by Guetarni et al., 13 showing that individual lesions can follow autonomous and discordant evolution patterns. In their study, manual lesion tracking through sequential imaging (median 3 MRI scans per patient, range 2–16) over a longitudinal observation period spanning 1–164 weeks was used, facilitating comprehensive monitoring of lesion-specific characteristics. This systematic tracking revealed that NICE lesions demonstrate significant heterogeneity in their imaging patterns, with each lesion exhibiting autonomous and discordant progression. Lesions showed variable temporal dynamics, including emergence, resolution, and in some cases, transient manifestation. The trajectory and progression of these lesions demonstrated no correlation with identifiable factors, suggesting patient-specific evolutionary patterns. 13
Despite utilizing state-of-the-art methodology, our aim of fully automated lesion segmentation for MRI evaluation has not been achieved in our study, making new research approaches necessary.
In our cohort, the declining occurrence of perilesional edema over time represents a novel observation. This may be related to immunotherapies applied in all our cases. Since data from untreated subjects are lacking, this warrants further investigation.
The correlation analysis revealed significant associations between radiological progression and clinical aspects, particularly headache (PCC = 0.344) and focal neurological deficits (PCC = 0.365). This finding adds to our understanding of the clinical–radiological relationship, which has been poorly characterized in previous studies due to limited sample sizes and follow-up durations.
Our treatment approach, incorporating both GCS and immunosuppressive agents in patients with ongoing disease activity, represents a more intense strategy than reported in the literature. While previous studies mainly focused on GCS monotherapy2–4,6,7,10,14,16,25 our experience with combination therapy, including various agents like RTX, CYC, AZA, MTX, TCZ, and MMF, provides new insights into potential options for refractory cases.
In our first report, 5 we could show that CYC was effective in reducing NICE lesions but it had to be discontinued due to long-term toxicity concerns. AZA, RTX, and (low dose) MTX were insufficient for disease control. MMF and TCZ resulted in heterogeneous treatment responses: some patients achieved clinical and radiological stabilization, while others experienced persistent MRI activity and symptoms. 5 Our long-term follow-up provides important further insights, especially regarding the efficacy of TCZ. Among four cases who received TCZ, due to insufficient response to GCS and RTX in case 1, and GCS and MMF in cases 2, 5, and 8, all experienced recurrent NICE lesions. However, MRI activity was associated with fewer and/or less severe clinical symptoms compared to the period before. Only two patients (cases 5 and 8) demonstrated NICE lesions with new or progressive edema during TCZ therapy. TCZ was discontinued in three out of four patients for being overall stable (with case 2 maintaining GCS monotherapy and case 5 continuing MMF and GCS). Among these three patients, one remained asymptomatic despite persistent NICE lesions on MRI, another showed no new NICE lesions but exhibited mild symptoms, and the third case experienced both clinical symptoms and radiological recurrence. While all five previously reported patients maintained immunotherapy at their last follow-up in 2020, 5 in the current long-term follow-up of nine patients, immunotherapy was discontinued in five cases. The longitudinal data suggest heterogeneous disease patterns, with some patients demonstrating short disease courses (e.g., cases 4, 6, and 9) and others showing recurrent NICE lesions, either symptomatic or asymptomatic (e.g., cases 1, 2, 5, and 8).
Safety and tolerability of treatments applied were generally favorable. However, two patients experienced significant adverse events: one patient (case 5) developed cytomegalovirus colitis while receiving combination therapy with GCS, MMF, and TCZ, necessitating antiviral intervention and hospitalization. The immunosuppressive regimen with GCS and MMF was maintained, TCZ was discontinued (also due to clinical stability), and the colitis resolved. Another patient (case 7) developed severe depression during GCS therapy, requiring psychiatric hospitalization. GCS were tapered and stopped, the patient exhibited transient new NICE lesions during further follow-up, cognitive function gradually improved over time. Our findings suggest that due to the highly variable disease course, individualized immunosuppressive treatment strategies and regular clinical and MRI controls, including awareness for possible adverse effects, are essential for optimal management.
The favorable outcomes observed in our cohort with all patients reaching mRS 0–1 align with the generally benign long-term prognosis reported in the literature.3,4 However, the persistence of residual symptoms in four of our nine patients suggests that complete remission may not always be achieved despite intensive immunotherapy.
The occurrence of new lesions during immunotherapy (33 of 34 MRIs showing progression during treatment) suggests that treatment strategies used do not completely suppress disease activity. This observation, together with the variable clinical–radiological correlation, makes treatment decisions challenging and highlights the need for individual approaches and more effective therapies.
The long-term persistence and recurrence of lesions in our cohort may support the hypothesis that NICE lesions possibly result from ongoing foreign body reactions to embolic material.8,10,23
Our study highlights the challenge in determining the optimal treatment duration. The observation that new lesions can develop even years after initial presentation, and that over half of the patients remained on immunotherapy at final follow-up, raises important questions about the appropriate length of immunotherapy. This uncertainty is reflected in the literature, where treatment approaches vary widely. 3
There are some limitations in our study. The small monocentric sample size (due to the rarity of the disease), yet comparable to most published series, limits statistical power and generalizability. All patients were treated with immunotherapies of varying intensity with favorable outcomes. However, without a control group, therapeutic effects cannot be assessed reliably. Additionally, the absence of histological confirmation through biopsy leaves some uncertainty about the exact pathophysiological mechanisms. Another limitation of our study is the lack of a formal power analysis for sample size calculation. Due to the rarity of NICE lesions, the sample size was determined by the number of patients diagnosed at our institution during the observation period. While our sample size is comparable to most published series, it still limits the statistical power and generalizability of our findings.
Future research should focus on developing standardized protocols for monitoring disease activity and treatment response. The identification of predictive factors for recurrence and treatment response would be particularly valuable. Our findings suggest that longer follow-up periods than previously considered may be necessary to better understand the pathophysiology of NICE lesions.
Conclusion
Our study contributes to our understanding of the long-term course of NICE lesions, demonstrating that disease activity can persist for years after initial presentation. Despite generally favorable outcomes, the potential for late recurrence and the variable treatment response highlight the need for individualized management approaches and extended follow-up. Our findings contribute to the evolving body of knowledge on this rare but relevant complication of EVT, offering valuable insights for clinical practice.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251345650 – Supplemental material for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment
Supplemental material, sj-docx-1-tan-10.1177_17562864251345650 for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment by Monika Ellssel, Ansgar Berlis, Markus Naumann, Muthuraman Muthuraman, Hao Ding, Sönke Schwarting, Felix Joachimski, Christoph J. Maurer and Antonios Bayas in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-2-tan-10.1177_17562864251345650 – Supplemental material for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment
Supplemental material, sj-pdf-2-tan-10.1177_17562864251345650 for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment by Monika Ellssel, Ansgar Berlis, Markus Naumann, Muthuraman Muthuraman, Hao Ding, Sönke Schwarting, Felix Joachimski, Christoph J. Maurer and Antonios Bayas in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-3-tan-10.1177_17562864251345650 – Supplemental material for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment
Supplemental material, sj-pdf-3-tan-10.1177_17562864251345650 for Long-term follow-up of patients with non-ischemic cerebral enhancing lesions following endovascular aneurysm treatment: magnetic resonance imaging findings, clinical course, and treatment by Monika Ellssel, Ansgar Berlis, Markus Naumann, Muthuraman Muthuraman, Hao Ding, Sönke Schwarting, Felix Joachimski, Christoph J. Maurer and Antonios Bayas in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
None.
Declarations
Availability of data and materials
All relevant data analyzed in this study are reported in the manuscript. Further patient information or materials cannot be shared for privacy reasons.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
