Abstract
Renal artery aneurysm (RAA), a type of visceral aneurysm with atypical symptoms, is difficult to detect and is usually discovered incidentally by imaging examination. Hilar RAA (HRAA) represents a relatively rare subgroup of RAA that is located in the distal part of the renal artery, close to the renal parenchyma. We reported a 55-year-old woman with an HRAA measuring 19 mm × 20 mm × 20 mm. She underwent endovascular therapy with bare-metal stent implantation with nested coil embolization. She was discharged without complications. The uniqueness of this case is the aneurysm location, which was at the distal right renal artery, making it difficult to preserve the blood supply to the right kidney. The novelty of the minimally invasive technique was that this endovascular treatment not only eliminated the aneurysm, but also preserved the blood supply to the ipsilateral kidney. Endovascular therapy is effective in the management of HRAA.
Keywords
Introduction
Renal artery aneurysm (RAA) is a rare disease with an incidence of 0.1% in the general population.1,2 RAA is defined as the arterial diameter of the dilated segment double that of a normal segment. 3 Most patients are asymptomatic. Recently, owing to the popularization of computed tomography angiography (CTA), more RAA cases have been identified. The symptoms and signs of RAA are atypical, with some patients presenting with gross or microscopic hematuria. Additionally, persistent pain could be caused by infarction of the target kidney or compression of the surrounding organs by the dilated RAA. However, the sudden onset of severe abdominal pain should be taken seriously as this may indicate the threat of RAA rupture.
Women are more likely to suffer RAA than men because of the high incidence of fibromuscular dysplasia in women. 1 According to the morphology and anatomical location, RAAs can be categorized into four subgroups: saccular, fusiform, dissecting, and intrarenal.4,5 Depending on the lesion location and shape, another popular classification (Rundback’s classification) divides RAAs into three types: type I is a saccular aneurysm arising from the main renal artery or a large segment branch; type II comprises fusiform aneurysms involving renal artery bifurcations; and type III comprises intralobar artery aneurysms.3,5,6 Herein, we describe a rare case of type I RAA, which is also called a hilar RAA (HRAA).
Case presentation
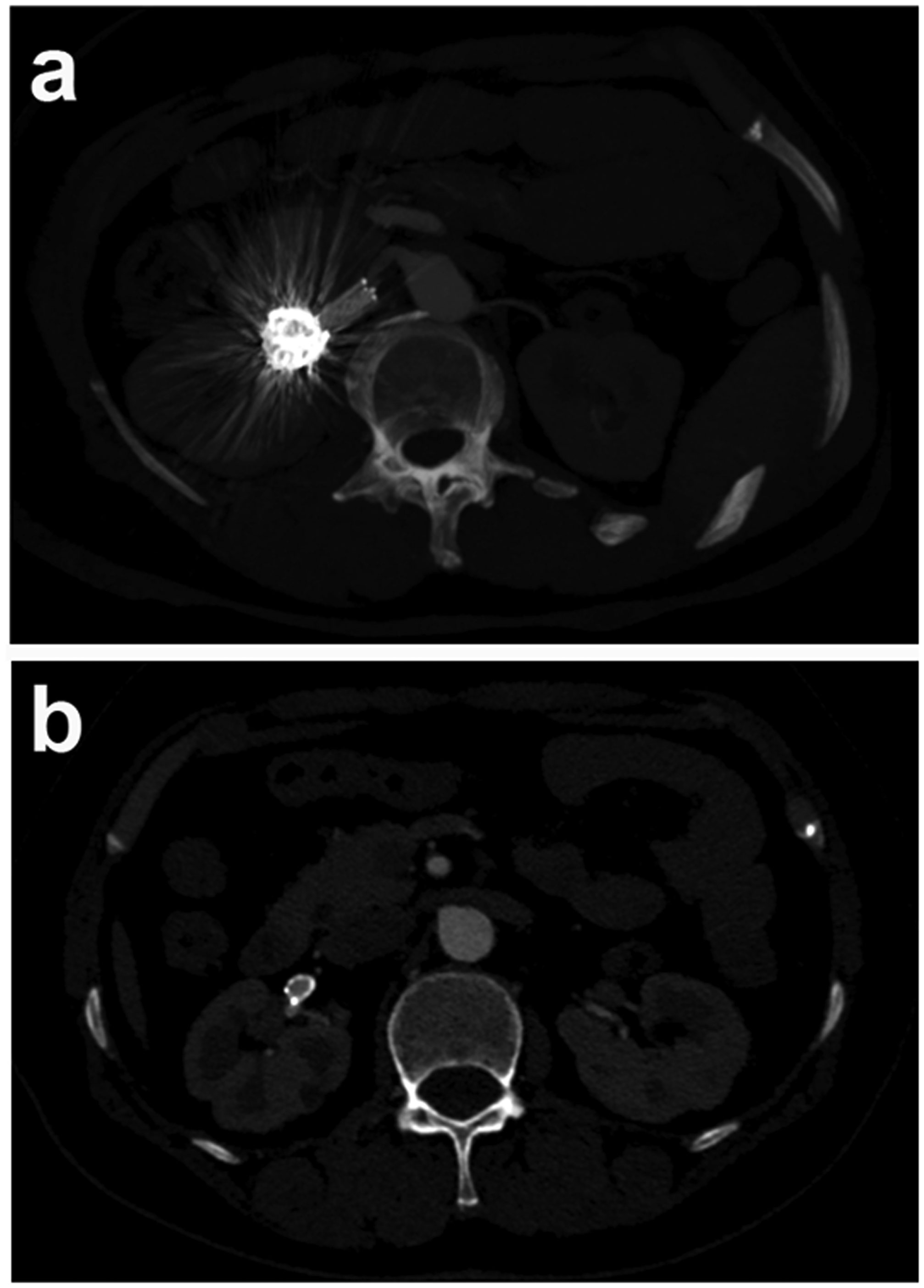
The reporting of this study conforms to the CARE guidelines. 7 A 55-year-old female patient complained of indigestion. Her only medical history of note was right ovarian cystectomy 14 years earlier. She denied any abdominal trauma. During routine abdominal CT, a right RAA was identified that measured 19 mm × 20 mm × 20 mm. On admission to the hospital, physical examination revealed negative percussion findings over the bilateral renal regions. Subsequently, CTA was performed to confirm the RAA, and she was diagnosed as having an HRAA (Figure 1a‒c).

Preoperative radiographic information. (a‒c) Before surgery, CTA revealed the hilar renal artery aneurysm in the distal segment of the right renal artery. The distal blood supply to the right kidney was normal. The white arrow indicates the aneurysm.
Initial laboratory tests showed normal creatine (59.3 μmol/L), blood urine nitrogen (4.96 mmol/L), hemoglobin (125 g/L), D-dimer (0.35 mg/L), and fibrinogen (FIB) (3.27 g/L) concentrations. We developed varied therapeutic plans for this patient. The key was to eliminate the aneurysm while preserving as much of the affected renal function as possible. Open surgery based on autologous blood vessel transplantation can preserve renal function but may induce marked trauma, slow recovery, and an increased risk of cardiovascular and cerebrovascular complications. Additionally, if an operation fails, the affected kidney may need to be removed. In contrast, endovascular repair is less invasive but may not preserve all the renal branch arteries. This patient’s HRAA affected at least two branches, as shown in Figure 1a and b. To maximize the blood supply to preserve as much of the right renal function as possible, the patient declined the simple stent graft plan. Owing to the extreme size of the aneurysm, a bare-metal stent could not thoroughly resolve this HRAA. Moreover, simple coil embolization could have induced unexpected renal infarction owing to the wide neck and turbulent flow in the HRAA. However, coil embolization with stent implantation could address the above disadvantages. After a detailed comparative analysis and adequate communication, and considering her history of right ovarian cystectomy, the patient consented to minimally invasive endovascular intervention instead of traditional renal arterial revascularization surgery. Briefly, after two successful Seldinger punctures of the right femoral artery, 5F and a 8F sheaths were inserted, and angiography was performed to confirm the 20.5-mm × 16.5-mm HRAA (Figure 2a). Next, a 6-mm × 40-mm ev3 bare-metal stent (Medtronic, Minneapolis, MN) was implanted to cover the aneurysm segment through a 5F Cobra catheter (Cook Medical, Bloomington, IN). As shown in Figure 2b, we then filled the gap between the stent and the HRAA with five Nester® MWCE-35-14-12 coils and four Nester® MWCE-35-14-10 coils (Cook Medical). We calculated the volume embolization ratio at approximately 91% (2934.44 ± 6.69 mm3 vs. 3233.45 ± 8.58 mm3; coil volume vs. total aneurysmal volume, respectively). Postoperative angiography showed that the HRAA was well embolized, and the blood supply to the right kidney was intact (Figure 2c). Postinterventional laboratory tests showed normal creatine (63.9 μmol/L) and blood urine nitrogen (2.32 mmol/L) concentrations. Two days after the endovascular treatment, the patient was discharged without complications. Three months later, she underwent follow-up CTA (Figure 3), which showed that the proximal and distal segments of the bare-metal stent were patent (Figure 3a), and there was no infarction in the right kidney (Figure 3b). During the 6-month follow-up, the patient experienced no symptoms or complications, and her serum blood urea nitrogen and creatinine concentrations remained within their respective normal ranges.

Endovascular therapy for the hilar renal artery aneurysm. (a) Angiography confirmed the HRAA. (b) One 6-mm × 40-mm ev3 bare-metal stent (Medtronic, Minneapolis, MN) was inserted to cover the HRAA. (c) After delivery of five Nester® MWCE-35-14-12 coils and four Nester® MWCE-35-14-10 coils (Cook Medical, Bloomington, IN) into the aneurysmal space, the HRAA was cured, which was accompanied by intact blood flow to the right kidney. The white arrow indicates the aneurysmal area.
Three-month follow-up CTA findings. (a) Three months post-procedure, CTA revealed patent blood flow in the proximal segment of the bare-metal stent. (b) The aneurysm was embolized. There was no renal infarction, and the distal segment of the bare-metal stent was patent.
Discussion
We have described a successful endovascular intervention for an HRAA case. Although the natural course of HRAAs is undetermined, the surgical indications for the treatment of HRAA are an enlarged renal arterial diameter of >2 cm, female sex and childbearing age, and symptoms such as flank pain, hematuria, refractory hypertension, thromboembolism, dissection, and rupture.1,3,8 In the present case, the patient was cured by endovascular therapy.
RAAs can be treated by open surgery and endovascular intervention. For a long time, open surgery was the standard treatment for RAAs. The surgical strategies comprise nephrectomy, in situ HRAA reconstruction, ex vivo HRAA repair plus auto-renal transplantation, and robot-assisted laparoscopic repair. 9 Minimally invasive endovascular therapy is another option, which comprises stent graft implantation, stent-assisted coiling, and simple coil embolization. There is controversy regarding the best treatment. Currently, no reliable data are available favoring one option over another. 10 The optimal treatment depends on the affected anatomy, the operator’s skill and experience, and the availability of devices and materials. In addition, the patient’s wishes must be respected. HRAAs located in the distal artery and around the renal hilar area present a therapeutic challenge owing to the complex anatomy.
We searched the literature for HRAA cases and summarized the evidence for the different therapeutic strategies to explore the best treatment (Table 1). To date, as shown in Table 1, we identified 54 patients and 55 aneurysms. Among the reported cases, four patients had a solitary kidney, two patients were pregnant, and two repaired kidneys were successfully transplanted to two men, respectively, which provided benefits to both donors and recipients.5,8,10,11,13–19 After analysis of the therapeutic strategies, we found that 47% (N = 26) of HRAAs were treated by ex vivo reconstruction (EVR), 44% (N = 24) were treated by in situ reconstruction (ISR), 5% (N = 3) were treated by endovascular therapy, and 4% (N = 2) were treated with nephrectomy. Laparoscopy was used to reduce surgical trauma owing to its lower invasiveness, and 17% (N = 9) of the operations were performed with the aid of laparoscopy. Most patients underwent reconstruction successfully, and their renal function returned to normal and was stable. However, three patients received an unplanned nephrectomy owing to EVR failure or thromboembolic events. Nevertheless, no nephrectomy was performed after ISR or endovascular treatment, and no deaths or ruptures were reported after therapy. Overall, the prognosis of HRAA was acceptable.
Summary of the findings in reported HRAA cases.

EVR, ex vivo reconstruction; Embo, embolization; F/U (M), follow-up (months); ISR, in situ reconstruction; UN, unplanned nephrectomy; NO., number of patients/number of aneurysms; NR, not reported; HTN, hypertension; Mor., mortality; Lap, laparoscopic; GSV, great saphenous vein; SD, standard deviation; GFR, glomerular filtration rate.
As shown in Table 1, EVR or ISR was the most commonly used method for HRAAs owing to the location of the aneurysms; the distal arterial segments were considered unsuitable for endovascular therapy. The difference between the decision to perform EVR vs. ISR is whether the affected kidney needs to be isolated. The extent of damage to the isolated kidney is associated with a longer operation time. Therefore, in one study, the kidney surface was cooled to minimize warm ischemic damage during preservation to prolong the preservation time to 3 hours. 8 Autologous grafting or prosthetic bypass was used for the replacement of the aneurysmal tract if a primary closure was not available. The great saphenous vein (GSV) was the most frequently used graft. As shown in Table 1, 24 patients received autologous grafts, and the GSV was used in 17 patients, the internal iliac artery was used in 6 patients, and the hypogastric artery was used in 1 patient.
Currently, conventional endovascular treatment combining coil embolization with stent implement for parenchymal aneurysms can be used to reconstruct the abnormal aneurysmal anatomical structure. 11 A single-center experiment conducted by Eldem et al. 12 demonstrated the efficacy and safety of endovascular treatment for true RAAs (TRAAs) with a high success rate and low morbidity. We reported this rare case of HRAA and presented the details of the case, including the preoperative and postoperative angiographic findings and the details of the endovascular procedure. This successful case may be a useful reference for the treatment of distal RAAs in the future. In this patient, endovascular intervention combining coil embolization and stent implantation was suitable for the HRAA. Nevertheless, for extremely distal renal aneurysms, open surgery or destructive embolism may be better options.
Conclusion
HRAA is a relatively rare subgroup of RAA. Most HRAAs are discovered incidentally on imaging examination for unrelated reasons. CTA is considered the first choice for evaluating HRAA. It is difficult to select the best treatment plan for HRAA. We must consider many factors, such as the patient’s age, underlying diseases, current renal function, surgical facilities, and the surgeon’s experience. Endovascular therapy is an effective choice for HRAA treatment. Additionally, open surgery, comprising laparoscopic nephrectomy with EVR, in situ HRAA reconstruction, and kidney transplantation, are possible endovascular salvage strategies. In our case, considering the patient’s normal renal function, our technical expertise, and the patient’s wishes, we chose endovascular treatment. The advantages were lower surgical trauma, lower complication rate, and shorter hospital stay.
Footnotes
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Union Hospital Wuhan, China. All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This research was supported by the National Natural Science Foundation of China (No. 81670512 to PL and No. 82000729 to CC).
Authors' contributions
HW and PY analyzed and interpreted the patient’s data and reviewed the manuscript. YC interpreted the patient’s data and reviewed the manuscript. HW and YL were the major contributors to writing the manuscript. CC and PL designed this study and interpreted the patient’s data. PY, CC, and PL revised the manuscript. All authors read and approved the final manuscript.
