Abstract
Background:
The novel criteria for the diagnosis of chronic inflammatory demyelinating polyneuropathy (CIDP) have established imaging with nerve ultrasound (NUS) and magnetic resonance neurography (MRN) as complementary methods for CIDP diagnosis.
Objectives:
Our goal was to investigate the role of MRN and NUS for CIDP monitoring.
Methods and design:
We longitudinally examined 12 CIDP patients from 2016 to 2022 using NUS, MRN, nerve conduction studies (NCS), and clinical parameters (inflammatory neuropathy cause and treatment (INCAT)/overall disability sum score (ODSS)). NUS evaluated the cross-sectional area (CSA) of the median, ulnar, radial, tibial, fibular, and sural nerve as well as the intranerve CSA variability (INVcsa) of the tibial, fibular, ulnar, and median nerve, whereas MRN evaluated T2-weighted sequences of the fibular and tibial nerve at the popliteal fossa.
Results:
Five patients showed clinical improvement/stability with corresponding improved or stable NCS/NUS parameters (number of nerves with increased CSA and INVCSA). Seven deteriorating patients showed deteriorating NCS and either increasing or decreasing NUS markers possibly indicating inflammatory activity or degenerative CSA reduction. The difference ΔINCAT/ODSS2022–2016 correlated positively with NUS ΔINVCSA2022–2016 (p = 0.007, r = 0.731, n = 12) and with NUS ΔCSA2022–2016 of the tibial nerve (p = 0.0005, r = 0.865, n = 12). Further, NUS-CSA of the tibial nerve in the popliteal fossa in 2016 correlated inversely with the difference of the INCAT/ODSS score (ΔINCAT/ODSS2022–2016; r = −0.653; p = 0.033; n = 11). Finally, the Bland–Altman analyses for the tibial and fibular nerve showed a bias of −1.903 and 2.195 mm2 (bias = NUS-CSA − MRN-CSA) accordingly revealing a difference between MRN and NUS measurements for deeper nerves.
Conclusion:
CSA and INVCSA of the tibial and fibular nerve can be used for monitoring in CIDP, and increased CSA of the tibial nerve is a good prognostic marker. MRN is more reliable for evaluating inflammation in proximal leg nerve segments.
Keywords
Introduction
The diagnostic criteria of the most common treatable autoimmune disease of the peripheral nervous system, the chronic inflammatory demyelinating polyneuropathy (CIDP), have been extensively revised in 2021 (European Academy of Nerve Society/Peripheral Nerve Society, EAN/PNS criteria) to include peripheral nerve imaging with both nerve ultrasound (NUS) and magnetic resonance neurography (MRN) as supportive criteria. 1 Together with the nerve conduction studies (NCS), the goal is to achieve an earlier diagnosis and treatment.
Numerous markers have been developed based on these imaging methods to visualize nerve pathology, which mostly correspond to features of inflammation. NUS examination evaluates cross-sectional area (CSA) of all peripheral nerves apart from the lumbosacral plexus. Longitudinal studies have shown that the intranerve CSA variability (INVCSA = maximal CSA/minimal CSA) of the tibial and fibular nerve is increasing over time for patients with progressive disease. Also, nerve echogenicity has been reported to change from hypoechogenic to hyperechogenic nerves over the years correlating with nerve degeneration.2–6
While ultrasound allows the assessment of superficial nerves, deeper nerves and the lumbosacral plexus may be visualized better with MRN. Our group has already published that apart from the imaging of nerve morphology by classical T2-weighted sequences quantitative MRN techniques such as T2 relaxometry and diffusion tensor imaging (DTI) allow the assessment of biomarkers in the context of inflammation and neurodegeneration.7–10
In this study, we proceed to answer the question of the value of NUS and MRN for disease monitoring and prognosis evaluation. Building on the previously published study by Pitarokoili et al. 9 in 2016, we examined the course of a cohort of 12 CIDP patients longitudinally between 2016 and 2022 by applying and correlating clinical, electrophysiological, and imaging markers.
Methods
Patients
A total of 12 patients from the neuropathy out-patient clinic of St Josef-Hospital Bochum diagnosed with typical CIDP fulfilled the European Federation of Neurological Societies/Peripheral Nerve Society (EFNS/PNS) criteria 2010, 11 were included in the INHIBIT register. They received annual follow-up examinations, and their disease progression was recorded using NCS, ultrasound examination, and the following clinical scores: inflammatory neuropathy cause and treatment (INCAT), overall disability sum score (ODSS), and the Medical Research Council Score (MRCSS). Additionally, they underwent MR-Neurography at Heidelberg University Hospital approved by the local ethics committee of the Heidelberg University in the year 2016 and the year 2022 (study duration 6 years; No. S-146/2021). Detailed correlation analyses of multiple MRN parameters of this cohort with clinical and electrophysiological parameters have been published separately. 10 The reporting of this study conforms to STROBE guidelines. 12
Nerve conduction studies
All electrophysiological studies were performed by the examiners (B.L. and D.C.). We used Dantec™ Keypoint Focus EMG device (Natus Medical GmbH, Planegg, Germany). Each examination includes the measurement of motor and sensory nerves. On the upper limb, the median and ulnar nerves were measured for the motor function and the median, ulnar, and radial nerves for sensory function. On the lower limb, we performed bilateral tibial and fibular nerves motor measurements and fibular and sural nerves sensory measurements. The electrophysiological examination provided compound motor action potential (cMAP), sensory nerve action potential (sNAP), nerve conduction velocity (NCV), and F-waves-latency. We used the reference values proposed by Stöhr and Pfister 13 and defined a deterioration of cMAP or sNAP <−3 µV/mV in at least one nerve.
Ultrasound examination
NUS examinations were performed on the same day as the NCS by the examiners (A.A. and E.S.). For each examination, we used an Affinity® 70G ultrasound system (Philips, Hamburg, Germany). For better visualization of superficial and deeper nerves, we used an 18-MHz linear array transducer for the fibular nerve at the fibular head, tibial nerve at the ankle, sural nerve, and tibial and fibular nerve at the popliteal fossa. The transducer was always placed perpendicular to the nerves to avoid anisotropy with no additional force to prevent artificial deformation of the nerve. The patients’ extremities were kept in neutral position. The CSA measurements were performed as described previously. 9
Additionally, we calculated the INV CSA variability for the median, ulnar, tibial, and fibular nerve calculated by dividing the maximum CSA by the minimum CSA, as described by Fisse et al. 3
The maximum CSA of all peripheral nerves and the brachial plexus were measured bilaterally in all CIDP patients at the following sites as described before 9 :
median nerve at the entrance to the carpal tunnel (retinaculum flexorum), forearm (approximately 15 cm proximal to the retinaculum flexorum), and upper arm (around the middle of the distance between the medial epicondyle and axillary fossa);
ulnar nerve at Guyon’s canal, forearm (approximately 15 cm proximal to Guyon’s canal), elbow (between medial epicondyle and olecranon), upper arm (around the middle of the distance between the medial epicondyle and axillary fossa);
radial nerve in the spiral groove;
tibial nerve in the popliteal fossa and at the ankle;
fibular nerve at the fibular head and in the popliteal fossa;
sural nerve (between the lateral and medial head of the gastrocnemius muscle).
MRN technique, image interpretation, and MRN outcome measures
MRN was performed by F.P. and M.K. on all participants using a 3.0 Tesla MR-scanner (Magnetom Prismafit; Siemens Healthineers, Forchheim, Germany).
In this study, we analyze correlation of NUS parameters with CSA measurements in the following peripheral nerves based on a fat-saturated T2-weighted 3D sequence of the lumbosacral plexus and a fat-saturated high-resolution T2-weighted turbo spin-echo sequence for visualization of nerve morphology. 10
The following peripheral nerves were measured with MRN:
maximum CSA of the median nerve in the upper arm between 8 and 15 cm proximally to the middle of the elbow;
maximum CSA of the ulnar nerve in the upper arm between 8 and 15 cm proximally to the middle of the elbow (line crossing horizontally through the elbow joint);
maximum CSA of the tibial nerve at the thigh (tibial portion of the sciatic nerve), which was between 10 and 20 cm proximally to the popliteal fossa as well as at the site of the maximal CSA at the lower leg, which was 8–12 cm distally to the popliteal fossa;
maximum fibular nerve CSA at the thigh, which was between 10 and 20 cm proximal to the popliteal fossa (fibular portion of the sciatic nerve).
All examiners were blinded for the results of all further examinations.
Statistics
Statistical analysis was conducted using Prism 10 (GraphPad Software, La Jolla, CA, USA). All values are shown as mean ± standard deviation (SD) unless stated otherwise; p < 0.05 was regarded as statistically significant. The D’Agostino and Pearson, Anderson–Darling, Shapiro–Wilk, and Kolmogorov–Smirnov test were applied to test the distribution of NUS and NCS data from 2016 and 2022. We applied the nonlinear Spearman’s rank correlation coefficient for correlations with NCV, cMAP, F-wave latency, INCAT/ODSS, and MRCSS with NUS and additionally with the difference between 2016 and 2022. The Pearson correlation coefficient r was reported for all correlation analyses unless stated otherwise. To assess agreement between NUS and MRN-CSA measurements, a Bland–Altman plot was used. Mean bias (bias = NUS-CSA − MRN-CSA) and 95% limits of agreement (±1.96 SD of the bias) were depicted as dotted lines in the plot.
Results
Patients and clinical characteristics
The cohort comprises six male and six female patients with typical CIDP based on EFNS/PNS criteria 2010 11 with an average age of 66.17 years (range 54–73). All patients were screened negative for paranodal antibodies. In 2022, mean disease duration after initial diagnosis was 11.5 ± 5.6 years. Mean INCAT/ODSS was 3.83 ± 1.34 (range 1–7; respective cohort in 2016: 3.0 ± 1.41) and mean MRCSS was 53.96 ± 5.25 (range, 46–60; respective cohort in 2016: 57.08 ± 3.5).
Of the 12 patients and based on INCAT/ODSS, two remained stable, seven deteriorated (1–3 points), and three improved (1–2 points) between 2016 and 2022. Of the seven patients that deteriorated in INCAT/ODSS five of them showed also a reduction of the MRCSS (1.5–13 points) whereas two of them remained stable in MRCSS. One patient deteriorated in the MRCSS (10 points) but remained stable in INCAT/ODSS (Table 1).
Epidemiological, clinical and ultrasound characteristics of the cohort according to INCAT/ODSS in to INCAT/ODSS P1, P2, P5 (improved), P4, P8 (stable) and P3, P6, P7, P9, P10, P11, P12 (deteriorated).

INCAT, inflammatory neuropathy cause and treatment; INVCSA, intranerve cross-sectional area; IVIg, intravenous immunoglobulins; MRCSS, Medical Research Council sum score (an increase is indicating clinical improvement); ODSS, overall disability sum score (a decrease is indicating clinical improvement over the observed timespan).
NCS between 2016 and 2022
NCS for the years 2016 and 2022 are presented in Table 1 and Supplemental Table 1(A) for the upper extremities and Table 1 and Supplemental Table 1(B) for the lower extremities.
Two of the three patients (P1, P2) that
The two patients that remained
For all patients that
NUS parameters: CSA and intranerve CSA variability
Ultrasound images of an exemplary patient are presented in Figure 1 showing a comparison of 2016 and 2022 with improving CSA of the ulnar nerve in the left upper arm and the tibial nerve in the left popliteal fossa.

Representative ultrasound pictures of patient 5 (Table 1) showing comparative CSA of the ulnar nerve measured at the upper arm and the tibial nerve measured at the popliteal fossa between 2016 and 2022. (a) Shows the left ulnar nerve 2016 with a CSA of 9.6 mm2, and comparatively, (b) shows the left ulnar nerve 2022 with a CSA of 7.56 mm2. In addition, the left tibial nerve 2016 has a CSA of 27.2 mm2 (c), compared to the left tibial nerve 2022 with a CSA of 19.7 mm2 (d). In this exemplary patient P5, there was a reduction in the CSA showing in the decreasing CSA and thus an improvement. This improvement was also reflected in the clinical scores, such as INCAT/ODSS and MRCSS. INCAT/ODSS improved from 5 to 3 and MRCSS increased from 48 to 53. The patient received 90 g IVIg every 4 weeks.
We next proceeded to evaluate markers of progression in ultrasound. As reported in the 2021 EAN/PNS guidelines nerve
We proceeded to evaluate further markers for disease activity in NUS and used
INCAT/ODSS showed a negative correlation with INVCSA variability of the right fibular nerve (p = 0.039, r = −0.599, n = 12) in 2022, meaning that ultrasound inflammatory activity with high INVCSA >2 implies a low clinical severity in 2022, which could be a sign of positive anti-inflammatory disease response. On the other hand, ΔINCAT/ODSS2022–2016 correlated positively with ΔINVCSA variability of the right tibial nerve (p = 0.007, r = 0.731, n = 12), meaning that the tibial INVCSA >2 implies a clinical deterioration over the years as a possible sign of increase of the inflammatory activity despite treatment (Table 1 and Supplemental Table 4).
Looking closer to the INVCSA of individual patients, we partially confirmed the correlation findings reported above:
The three patients that
Patient P4
Among the patients that
Comparison between ultrasound and MR-neurography measurements
A basic question is whether NUS and MRN can both reliably and reproducibly depict the CSA of the same nerve segments, particularly for deeper nerves such as the tibial and fibular nerves in the popliteal fossa.
As depicted in Figure 2, we performed a Bland–Altman analysis to compare the reliability of MRN and NUS measurements. For both the tibial and fibular nerves, the CSA was measured using MRN and NUS in the popliteal fossa. The Bland–Altman analysis for the tibial nerve showed a positive mean bias (bias = NUS-CSA − MRN-CSA) of 2.195 (SD of the bias 8.111), while the 95% limits of agreement (±1.96 SD of the bias) ranged between −13.70 and 18.09. These results indicate that nerve CSA measured by MRN is frequently lower compared to NUS measurements for the tibial nerve in the popliteal fossa.

Comparison between ultrasound and MR-Neurography evaluation of tibial nerve (a) and the fibular nerve (b) both measured in popliteal fossa shown by Bland–Altman plots. The Bland–Altman plot for the tibial nerve (a) shows a positive mean bias (bias = NUS-CSA − MRN-CSA) between −13.70 and 18.09. The Bland–Altman plot for the fibular nerve (b) shows a negative mean bias (bias = NUS-CSA − MRN-CSA) between −11.14 and 7.339.
The Bland–Altman analysis for the fibular nerve in the popliteal fossa showed a negative mean bias (bias = NUS-CSA − MRN-CSA) of −1.903 (SD of the bias 4.715) and the 95% limits of agreement (±1.96 SD of the bias) ranged between −11.14 and 7.339.
These results indicate that nerve CSA measured by MRN is frequently higher than the CSA measured by NUS for the fibular nerve in the popliteal fossa. A representative T2-weighted SPACE 3D sequence demonstrating hallmark features of CIDP (diffuse enlargement and T2w-hyperintense signal of the lumbosacral plexus nerves (arrows)) indicative of demyelination and inflammation is shown in Figure 3.

MRN of the lumbosacral plexus in CIDP. MIP of a T2-weighted SPACE 3D sequence demonstrates hallmark features of CIDP, including diffuse enlargement (hypertrophy) and T2w-hyperintense signal of the lumbosacral plexus nerves (arrows), indicative of demyelination and inflammation.
Correlation analyses between ultrasound, nerve conduction, and clinical markers
The correlations between ultrasound and nerve conduction markers are presented in Table 2 and confirm the theory that CSA increase is associated mostly with demyelinating inflammation.
Correlation analyses between ultrasound, clinical, and electrophysiological markers in 2022.
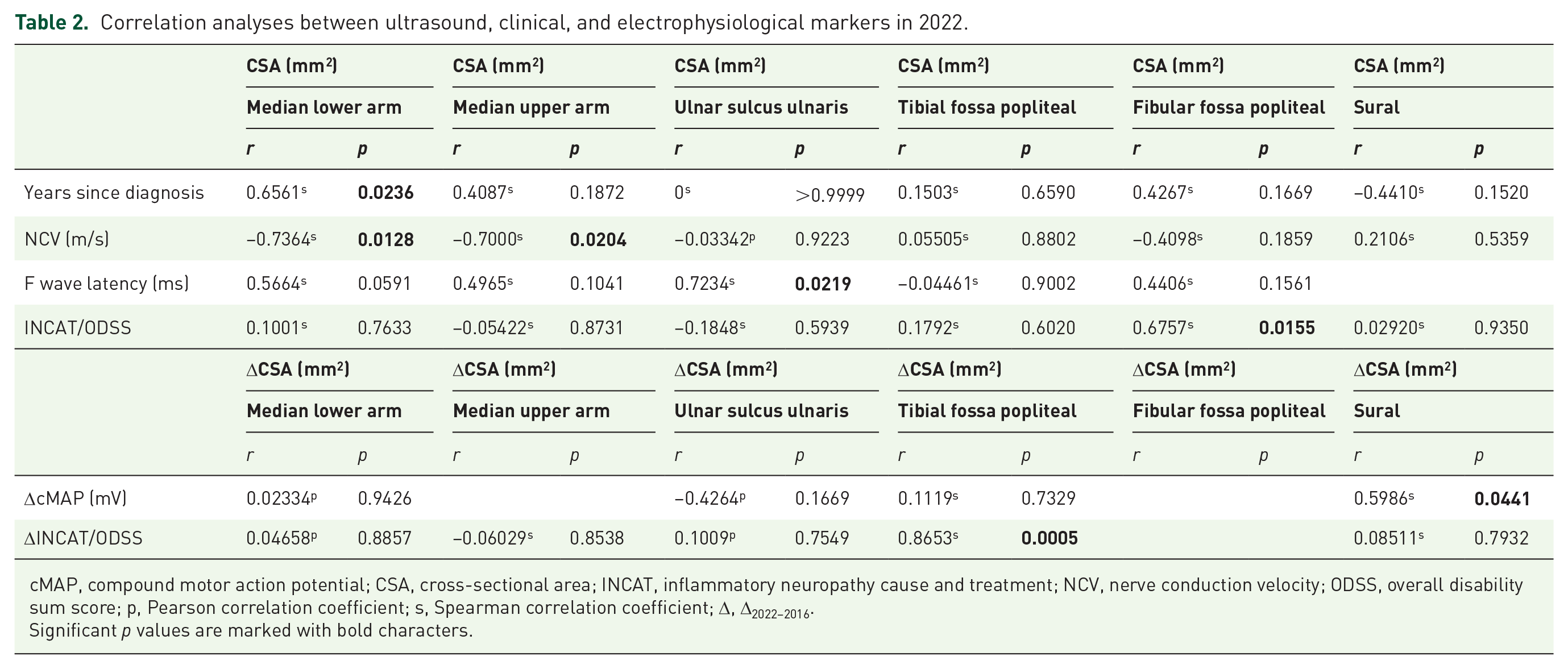
cMAP, compound motor action potential; CSA, cross-sectional area; INCAT, inflammatory neuropathy cause and treatment; NCV, nerve conduction velocity; ODSS, overall disability sum score; p, Pearson correlation coefficient; s, Spearman correlation coefficient; Δ, Δ2022–2016.
Significant p values are marked with bold characters.
The median nerve CSA at lower arm and at upper arm level showed a negative correlation with NCV of the median nerve (p = 0.013, r = −0.736, n = 11; p = 0.020; r = −0.700, n = 11).
The CSA of the ulnar nerve in the ulnar sulcus showed a positive correlation with the F-wave latency of the ulnar nerve (p = 0.022, r = 0.7234, n = 10).
In addition, the difference of the CSA between 2022 and 2016 (ΔCSA2022–2016) of the sural nerve showed a significant positive correlation with the difference of sNAP (ΔsNAP2022–2016; p = 0.044, r = 0.599, n = 12; Table 2).
Regarding the correlations with clinical markers, the analysis revealed the following interesting findings indicating that ultrasound inflammatory activity defines clinical severity.
The fibular nerve CSA measured in popliteal fossa and marking an increased inflammatory activity showed a positive correlation with INCAT/ODSS (p = 0.016, r = 0.676, n = 12) in 2022.
The difference of the CSA between 2022 and 2016 (ΔCSA2022–2016) of the tibial nerve measured in popliteal fossa showed a significant positive correlation with the difference of INCAT/ODSS (ΔINCAT/ODSS2022–2016; p = 0.0005, r = 0.865, n = 12).
Most importantly, the CSA of the tibial nerve in the popliteal fossa in 2016 correlated inversely with the difference of the INCAT/ODSS score (ΔINCAT/ODSS2022–2016; r = −0.653; p = 0.033; n = 11), indicating that a high tibial CSA (high demyelinating inflammatory activity) at the beginning of the monitoring period indicated a decrease of the INCAT/ODSS until 2022 (Figure 4 and Table 1 and Supplemental Table 5).

The CSA of the tibial nerve measured in 2016 showed a significant negative correlation with the ΔINCAT/ODSS (r = −0.653; p = 0.033; n = 11).
Discussion
The main goal of our current research project was to investigate the role of peripheral nerve imaging with NUS and MRN in a longitudinal observation study over 6 years for CIDP.
Residual ultrasound inflammatory activity in stable/improving patients
Due to longitudinal design over a period of 6 years, our cohort is small but represents a typical cohort of the clinical routine. Half of the patients improved or remained stable over the years, and this was apparent in the clinical scores and NCS. For two of the patients who improved over the years, immunomodulatory treatment was stopped, which also represents a common consideration for stable/improving patients in every-day clinical practice. Notably and despite their improvement, these patients presented with an increased nerve CSA in 2016 at the beginning of the observation period as well as in 2022. These inflammatory imaging signs never normalized but showed a tendency to decrease (based on the number of nerves with INVcsa ⩾2 and the number of nerves with increased CSA) compared to the rest of the patients, who clinically deteriorated. This is a critical finding and confirms our previous publications, implying that imaging signs of inflammation in CIDP do not reside completely over the course of several years, even if the patient is improving or remains clinically stable. However, deescalating or stopping treatment for such patients is still a feasible option and must be evaluated by clinicians using clinical scores, NCS, and imaging NUS parameters.2,15 Interestingly, one of the patients who improved in INCAT/ODSS but deteriorated in the NCS and showed a high INVCSA of many nerves further received IVIg through the study period. Therefore, NCS deterioration and increasing/high number of nerves with INVCSA ⩾2 should be considered as markers of clinical activity (“red flags”) for clinicians to continue treatment.
Reliable ultrasound monitoring markers
Based on our current study, CSA and INVCSA of the tibial, fibular, and ulnar nerves should be predominantly considered as monitoring markers. The importance of the nerves of the lower extremities for the longitudinal follow-up was reported before from our group.2,15 On the other hand, the ulnar nerve has been reported to be affected early in the CIDP and is part of the majority of the ultrasound scores for CIDP evaluation.2,15,16
However, the most appropriate NUS marker for detecting inflammatory activity remains an open question. CSA increase alone is a marker of inflammation and contributes to disease diagnosis.6,16 A combination of different nerves mostly of the upper extremities distinguishing CIDP from other non-inflammatory neuropathies has been reported.17,18 CSA-based scores do not seem to be suitable as a marker for longitudinal studies probably due to the inhomogeneous distribution of the inflammation between nerves and the heterogeneous distribution of inflammation between patients. In our cohort, the number of nerves exceeding normal values neither in 2016 nor in 2022 was insufficient to predict deterioration. In contrast, patients with improvement or stable clinical outcomes generally exhibited a corresponding improvement or stability in the number of nerve segments with increased CSA.
To address this challenge in ultrasound-based disease monitoring, the marker of INVCSA was developed some years ago. 4 A high INVCSA is a marker of an inhomogeneous distribution of the CSA of each nerve and depicts the inhomogeneous inflammatory CSA increase in CIDP. In our cohort, two of the patients who deteriorated over the years showed an increasing number of nerves with a INVCSA ⩾2 in 2022. The remaining deteriorating patients, however, showed stable or reducing INVCSA ⩾2. A reducing number of nerves with INVCSA ⩾2 could also imply an increase in secondary axonal damage, also leading to clinical deterioration. This is supported by the nerve CSA findings, which remained stable or reduced in deteriorating patients, as previously reported by our group. 3 Concluding, on the role of NUS for CIDP, we propose its use as a supplementary tool alongside clinical scores and NCS for treatment monitoring, with INVCSA (mostly for the lower extremities and ulnar nerves) serving as a marker of inflammatory activity. However, clinical deterioration should primarily guide therapeutic decisions and an increase of nerves with INVCSA ⩾2 should only be regarded as a “red flag” for a possibly increasing inflammation. A stable number of nerves with INVCSA ⩾2, however, do not guarantee disease stability.
The role of MRN and NUS in disease monitoring
On the other hand, the role of MRN for CIDP monitoring is clearly influenced by two factors: the availability and cost of the method, along with the need for highly specialized radiologists as well as its additional advantage over NUS in evaluating inflammatory activity. Based on our previous publication but also on the present data (Bland–Altman analyses between US and MRN), we confirmed that NUS can detect the CSA of the superficial nerves with minimal bias, while the bias increases for deeper nerves, such as the fibular and tibial nerve in the popliteal fossa. 9
In this context, it is important to be aware that MRN can never capture a nerve in its exact orthogonal plane in every cross-sectional image due to the nerve’s varying course, influenced by factors such as changes in surrounding tissue characteristics and the nerve’s trajectory.
Therefore, for CIDP treatment monitoring and diagnosis, we propose initially screening the peripheral nerves with NUS, evaluating INVCSA, and using the data in conjunction with NCS and clinical scores to influence prognostic evaluation and treatment decisions.
However, in cases of predominantly proximal distribution of nerve inflammation in CIDP (lumbosacral plexus or sciatic nerve), INVCSA ⩾2 is not applicable. In these instances, MRN would be the preferred additional imaging method to evaluate disease activity, as it detects CSA increases and active inflammation with Gadolinium enhancement. Furthermore, MRN clearly holds major potential for more advanced imaging compared to NUS through various quantitative techniques, such as T2 relaxometry and DTI, to more specifically define inflammatory and degenerative signs in CIDP. As reported by our group, the DTI parameter fractional anisotropy and magnetization transfer ratio correlate with markers of demyelination in NCS, which is not always the case for CSA in NUS.8,10
Reliable ultrasound prognostic markers
The next question is whether inflammatory imaging markers of the peripheral nerves could be used as a prognostic marker for anti-inflammatory treatment response.
Indeed, high tibial CSA in 2016 correlated with a low change in INCAT/ODSS over the years. Hence, patients with high (inflammatory) CSA of the tibial nerve in 2016 had a better prognosis on treatment as inflammatory imaging markers are indeed prognostic markers over the years. The same findings for the sciatic nerve and lumbosacral plexus were published by our group for MRN-CSA in the same CIDP cohort. 10 These findings highlight the importance of proximal nerve segments of the peripheral nerves for prognosis evaluation. Therefore, screening patients with NUS and MRN before treatment initiation provides valuable additional insight, as indicated by our study.
The major limitation of our study is the small number of patients and the lack of previous longitudinal studies on CIDP MRN and US evaluation, which did not allow us to perform power analyses on the number of the study cohort. However, we present a thorough evaluation of the patients over a long period of time, thereby pointing out clinically relevant aspects of disease activity in CIDP.
Conclusion
We have demonstrated in a small but representative cohort that peripheral nerve imaging can offer comprehensive inflammatory markers in CIDP. It should be used prior to treatment to evaluate inflammatory activity as a prognostic marker (e.g., CSA of tibial nerve in NUS or sciatic nerve and lumbosacral plexus in MRN) and should also be utilized for treatment monitoring as a marker for current inflammation (INVCSA of mostly the tibial and fibular nerve) but should influence treatment decisions only in combination with clinical scores and NCS.
Supplemental Material
sj-docx-2-tan-10.1177_17562864251342336 – Supplemental material for Comparison of imaging markers of nerve ultrasound and MR-neurography in a longitudinal course in chronic inflammatory demyelinating polyneuropathy
Supplemental material, sj-docx-2-tan-10.1177_17562864251342336 for Comparison of imaging markers of nerve ultrasound and MR-neurography in a longitudinal course in chronic inflammatory demyelinating polyneuropathy by Benjamin Lüling, Fabian Preisner, Jeremias Motte, Anna Lena Fisse, Thomas Grüter, Rafael Klimas, Emelie Schäfer, Annika Altenborg, Devrim Colak, Jörg Philipps, Tim Godel, Daniel Schwarz, Sabine Heiland, Min-Suk Yoon, Ralf Gold, Martin Bendszus, Moritz Kronlage and Kalliopi Pitarokoili in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-1-tan-10.1177_17562864251342336 – Supplemental material for Comparison of imaging markers of nerve ultrasound and MR-neurography in a longitudinal course in chronic inflammatory demyelinating polyneuropathy
Supplemental material, sj-pdf-1-tan-10.1177_17562864251342336 for Comparison of imaging markers of nerve ultrasound and MR-neurography in a longitudinal course in chronic inflammatory demyelinating polyneuropathy by Benjamin Lüling, Fabian Preisner, Jeremias Motte, Anna Lena Fisse, Thomas Grüter, Rafael Klimas, Emelie Schäfer, Annika Altenborg, Devrim Colak, Jörg Philipps, Tim Godel, Daniel Schwarz, Sabine Heiland, Min-Suk Yoon, Ralf Gold, Martin Bendszus, Moritz Kronlage and Kalliopi Pitarokoili in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
