Abstract
Background:
Early use of high-efficacy therapies (EHT) in multiple sclerosis (MS) is a promising but novel treatment strategy. Its adoption in Germany’s MS care warrants further research.
Objectives:
This study assessed treatment initiation patterns among newly diagnosed MS patients in Germany (2017–2022).
Design:
This is a retrospective observational study.
Methods:
Claims data from 4.5 million individuals insured by German statutory health insurance included 1448 newly diagnosed MS patients from 2017 to 2022. Patients were identified by International Statistical Classification of Diseases and Related Health Problems, 10th revision, German modification (ICD-10-GM) code G35 – in two different quarters of the same year, and a disease-modifying therapy (DMT) prescription, with no prior MS diagnosis or DMT prescription in the preceding 2 years. DMTs were categorized according to German Society of Neurology S2k guidelines: category 1 (low-efficacy), category 2 (moderate-efficacy) and category 3 (high-efficacy).
Results:
Of patients initiating treatment, 77.1% started with category 1, 8.1% with category 2 and 14.8% with category 3 DMTs. From 2017 to 2022, category 1 initiations declined by 7.6% points (pp), while categories 2 and 3 initiations increased by 2.8 and 4.8 pp, respectively. Escalation to category 3 occurred in 10.5% of category 1/2 starters, with 31.5% taking over 2 years. De-escalation to category 1/2 occurred in 3.3% of category 3 starters.
Conclusion:
Real-world data highlight a shift towards EHT in MS care.
Keywords
Introduction
Disease-modifying therapies (DMTs) are central to multiple sclerosis (MS) management and consist of a range of immune-modulating drugs available in Germany, classified by their efficacy as low, moderate or high based on their effects on annual relapse rates. 1 Traditionally, the treatment paradigm has followed a ‘treat-to-target’ model, initiating therapy with low-efficacy medications and escalating to higher-efficacy options only if necessary. However, this approach is now being re-evaluated as emerging evidence highlights the benefits of early intervention with high-efficacy therapies (EHT).2,3
The primary goal of DMT treatment is to mitigate disease activity and prevent disability accumulation, which can lead to a secondary progressive course.4–6 Initiating an effective treatment early-on in the disease may reduce relapse rates and the underlying inflammatory process could delay irreversible neurological damage and conversion to secondary progressive multiple sclerosis (SPMS),7,8 particularly during the critical ‘window of opportunity’, when these therapies are believed to be most effective. 9 Although the results of ongoing randomized controlled trials, such as DELIVER-MS and TREAT-MS, are awaited to provide direct comparisons between the stepwise escalation approach and EHT, retrospective analyses and real-world studies have shown that early EHT is associated with improved outcomes, including reduced disability progression and a lower risk of transitioning to SPMS.10–15
Current national guidelines, such as the German S2k guideline as of 2023, recognize the potential of EHT while calling for more prospective randomized studies to validate its efficacy. 1 Recent real-world data from Germany suggest a trend towards initiating high-efficacy treatment for newly diagnosed MS patients.16,17 However, the current evidence does not differentiate between different MS subtypes. MS is categorized into three primary phenotypes, namely relapsing-remitting multiple sclerosis (RRMS), SPMS and primary progressive multiple sclerosis (PPMS), with most DMTs being approved for RRMS. 1 This study aims to extend the evidence of the recent shift towards the EHT approach by analysing statutory health insurance (SHI) claims data from Germany spanning the years 2017–2022 and providing new insights into the application of the EHT strategy across different disease subtypes.
Methods
Dataset and data processing
This analysis utilized longitudinal routine data from German statutory health insurers, encompassing both corporate health insurers and regular insurers. The dataset covers approximately 4.5 million insured individuals. The data consist of anonymized and aggregated patient-level information, including disease diagnoses based on the International Statistical Classification of Diseases and Related Health Problems, 10th revision, German modification (ICD-10-GM), Version 23 codes sourced from inpatient and outpatient claims, sociodemographic characteristics as well as reimbursements for treatments and procedures.
The data were stored in the Permea platform (Temedica GmbH, Munich, Germany) in a General Data Protection Regulation compliant manner and no personal information, that might allow identification of individuals, was revealed. Data for specific subgroups were only available if the subgroup comprised a minimum of five individuals.
Cohort definitions
The cohort comprised continuously insured patients who received a MS diagnosis based on ICD-10-GM codes G35.0 (initial manifestation of MS), G35.1- (RRMS), G35.2- (PPMS), G35.3- (SPMS) and/ or G35.9 (MS, unspecified) in at least two quarters in the same calendar year (M2Q criterion) between January 2017 and December 2022. The M2Q criterion was used to ensure the inclusion of patients with a confirmed MS diagnosis. 18 To approximate newly diagnosed patients, patients must not have had a recorded G35 – diagnosis or relevant DMT prescription in the 2 years prior to their initial MS diagnosis. The initial diagnosis was used to categorize the disease type of newly diagnosed patients. Since claims data are only available on a quarterly basis, all MS diagnoses recorded within a patient’s first diagnostic quarter were considered initial diagnoses. If a patient received multiple MS diagnoses with different subtypes in the same quarter, they were assigned to each relevant subtype. While each patient was counted only once in the overall cohort, they could be included in multiple disease subtype groups due to this classification approach. As the main focus was on therapy initiation, only patients who received a prescription of a relevant DMT after initial diagnosis were included.
Prescriptions for relevant DMTs after the initial MS diagnosis were identified by the corresponding Anatomical Therapeutic Chemical code and further categorized into medication categories 1, 2 and 3 based on DGN guidelines regarding effectiveness. 1 If medications were approved by the European Medicines Agency between 2017 and 2022, the approval dates are noted. Medication category 1 (mild efficacy): Dimethyl fumarate, diroximel fumarate (2021 19 ), glatiramer acetate, interferon beta-1a, interferon beta-1b, peginterferon beta-1a and teriflunomide. Medication category 2 (moderate efficacy): cladribine (2017 20 ), fingolimod, ozanimod (2020 21 ), siponimod (2020 22 ) and ponesimod (2021 23 ). Medication category 3 (high efficacy): alemtuzumab, natalizumab, ocrelizumab (2018 24 ), ofatumumab (2021 25 ) and rituximab (off-label). Untreated patients did not receive such prescriptions in the observation period.
Patients meeting the inclusion criteria could contribute to each disease type and – in the case of treated patients – DMT cohort only once for the whole observation period and per calendar year but could contribute to multiple different cohorts.
Variables and statistical methods
Therapy initiation was analysed regarding DMT, medication category, disease type, age groups, sex and by calendar year in which the initial diagnosis of the patients took place through absolute and relative frequencies. Analyses by disease type were restricted to clinically distinct MS subtypes (RRMS, SPMS and PPMS). Switches between medication categories were analysed through absolute and relative frequencies including time to switch as quarters from first prescription of a relevant DMT to a second prescription of a relevant DMT of another category.
Data processing and frequency calculations were performed with Python 3.9. Group differences were assessed using two-sided Fisher’s exact test via GraphPad Prism 10. RRMS was used as the reference population for cross-disease type comparisons, as it constitutes the largest MS subtype population. Figures were generated via GraphPad Prism 10.
Results
Cohort characteristics
The dataset comprised 1448 newly diagnosed MS patients who received a first prescription for a relevant MS medication within the observation period (Table 1). A total of 60.3% of patients initially presented with RRMS, 7.7% with PPMS and 2.9% with SPMS. The remaining patients presented with either initial manifestation of MS or unspecified MS. A total of 64.4% of patients were females and 28.8% were over 45 years old. The age and sex distribution of RRMS patients was similar to the overall cohort. PPMS and SPMS patients were more likely to be over 45 years than RRMS patients (57.7% and 40.5%–50.0%, respectively), and fewer SPMS (54.8%) were female than RRMS and PPMS patients.
Patient population (2017–2022).

Number and age sex distribution of patients in each disease type cohort. As data are only available on a quarterly basis, all MS diagnoses made in the first diagnostic quarter counted as initial diagnoses. Patients were only counted once in the overall cohort but could contribute to several disease types.
MS, multiple sclerosis; PPMS, primary progressive MS; RRMS, relapsing-remitting MS; SPMS, secondary progressive MS.
Therapy initiation
Of all MS patients newly diagnosed between 2017 and 2022 who started treatment in the observation period (N = 1448), 77.1% initiated therapy with category 1, 8.1% with category 2 and 14.8% with category 3 DMTs (Figure 1(a)). The choice of initiation DMT depended on disease type: 13.3% of RRMS, 40.5% of PPMS (RRMS vs PPMS, p < 0.0001) and 23.8% of SPMS patients (RRMS vs SPMS, not significant (ns)) initiated treatment with category 3 DMTs. Overall, the most frequently used initiation DMTs within each category were glatiramer acetate for category 1 (27.1%), fingolimod for category 2 (3.2%) and ocrelizumab for category 3 (8.8%; Figure 1(b)). These three medications were the leading initiation DMTs across all disease types in their respective categories (data not shown). Alemtuzumab and siponimod were used by fewer than five patients each and were therefore grouped under ‘other’.
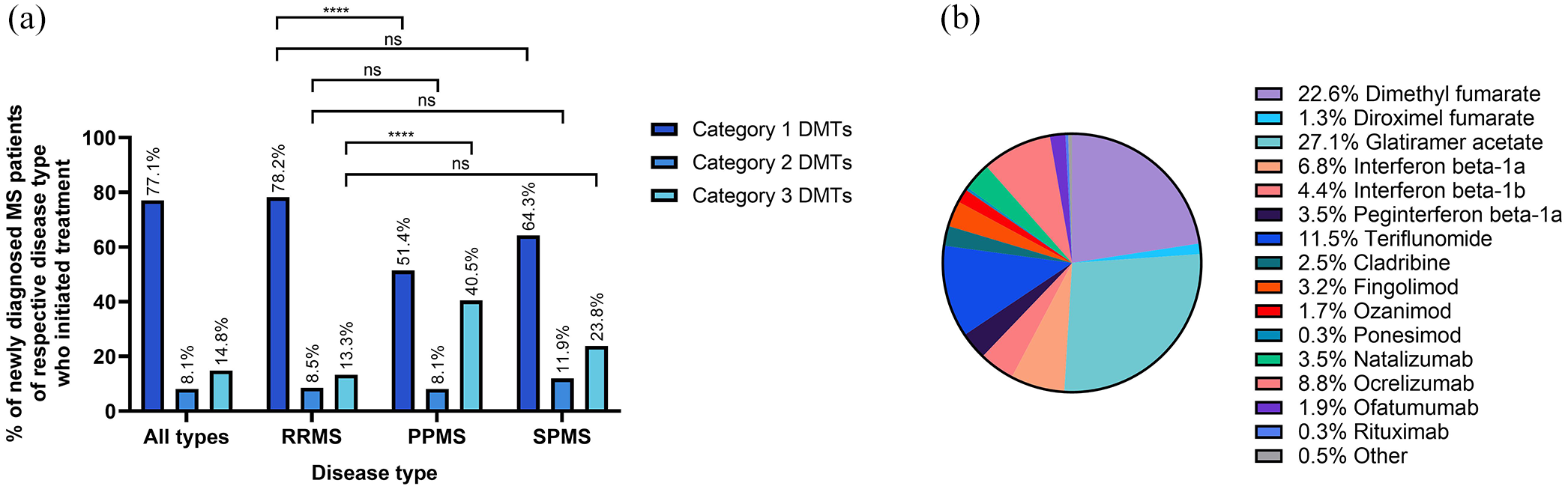
Distribution of DMT initiation among newly diagnosed MS patients (2017–2022). Proportion of newly diagnosed MS patients who initiated treatment with respective (a) medication category by disease type, (b) DMT, all MS types.
Older patients initiated treatment with category 3 DMTs more frequently than younger patients (26.3% of 56–65-year-olds vs 17.2% of 46–55-year-olds, p < 0.05; Figure 2(a)). Sample size for the 76+ years age group was too small for analysis. There was a trend that male patients (17.1%) initiated treatment with category 3 DMTs more frequently than female patients (13.5%; Figure 2(b)). Analysis by disease types was not possible due to insufficient sample sizes.
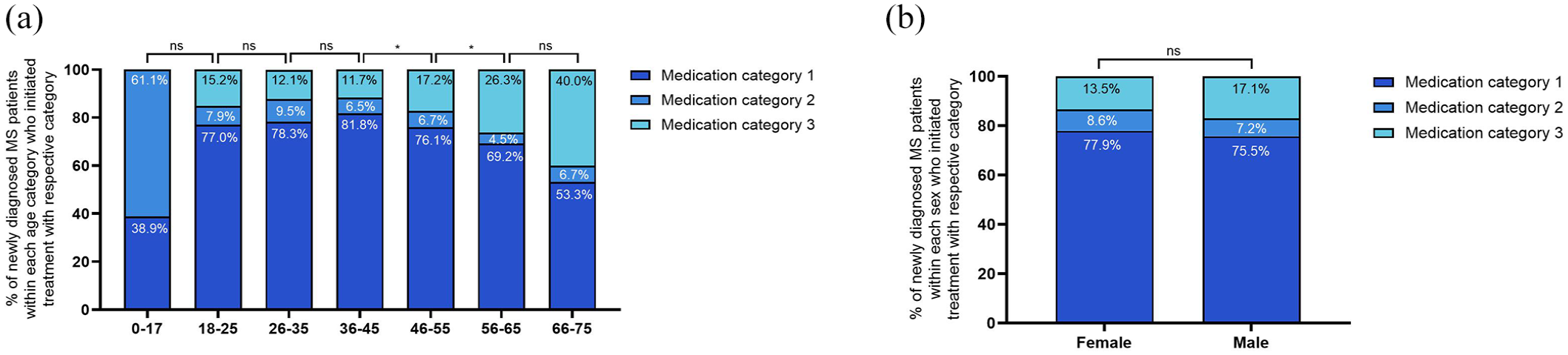
Distribution of medication category initiations within age and sex populations among newly diagnosed MS patients of all MS types (2017–2022). Proportion of newly diagnosed MS patients across all MS types who initiated treatment with respective medication category (a) within each age category, (b) within each sex population.
Therapy initiation shift over time
Among MS patients diagnosed between 2017 and 2022, the proportion of patients initiating treatment with category 1 DMTs decreased significantly from 84.1% to 76.5% (−7.6 percentage points (pp), p < 0.01; Figure 3(a)). In contrast, the proportion of patients initiating with category 3 DMTs increased from 10.5% to 15.3% (+4.8 pp, p < 0.05), while initiations with category 2 DMTs showed a rising trend from 5.4% to 8.2% (+2.8 pp, ns). Among individual DMTs, teriflunomide exhibited the largest increase as a first-line medication (7.8%–12.0%, +4.2 pp, p < 0.05), followed by an increasing trend for ocrelizumab (6.8%–9.3%, +2.6 pp, ns; Figure 3(b)). Conversely, the most notable declines were observed for interferon beta-1a (11.1%–6.5%, −4.7 pp, p < 0.05) and interferon beta-1b (8.1%–3.8%, −4.3 pp, p < 0.05). DMTs used by fewer than five patients in each calendar year are not shown.
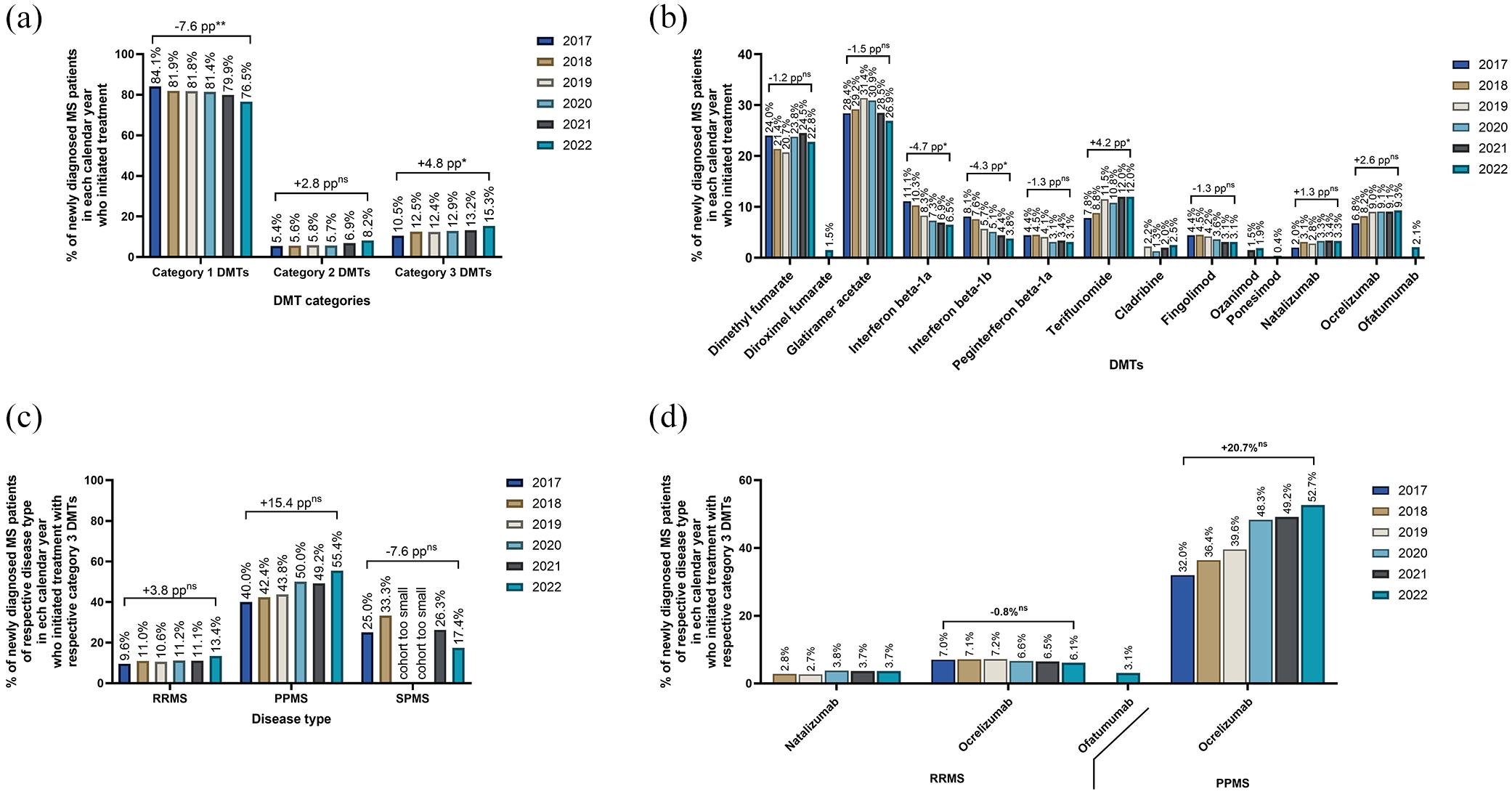
Trends in initiation by medication category and calendar year (2017–2022). Proportion of newly diagnosed MS patients in each calendar year who initiated treatment (a) with respective medication category across all MS types, (b) with respective DMT across all MS types, including only those DMTs used as initiation medications by five or more patients in each calendar year, (c) with category 3 DMTs by disease type, (d) with respective category 3 DMT by disease type, including only those DMTs used as initiation medications by five or more patients in each calendar year.
Between 2017 and 2022, the proportion of RRMS and PPMS patients who initiated treatment with category 3 DMTs showed an increasing trend from 9.6% to 13.4% (+3.8 pp, ns) and from 40.0% to 55.4% (+15.4 pp, ns), respectively (Figure 3(c)). In contrast, the proportion of SPMS patients who initiated treatment with category 3 DMTs declined from 25.0% to 17.4% (−7.6 pp, ns). Ocrelizumab initiations among PPMS patients increased markedly from 2017 to 2022, rising from 32.0% to 52.7% (+20.7 pp, ns; Figure 3(d)). The proportion of RRMS patients treated with natalizumab or ocrelizumab remained stable.
Therapy switch between medication categories
In total, 89.5% of patients who initiated treatment on category 1 and 2 DMTs remained on category 1 and 2 DMTs and 96.7% of patients who initiated treatment with category 3 DMTs remained on category 3 medication. A total of 10.5% of patients who initiated treatment with category 1 or 2 DMTs escalated to category 3, whereas 3.3% of patients who initiated treatment with category 3 DMTs de-escalated to category 1 or 2 DMTs (Figure 4(a)).

Therapy journey. (a) Proportion of newly diagnosed MS patients across all MS types (2017–2022) who initiated treatment with respective medication category and who (did not) switch to another medication category. (b) Time to therapy escalation. Proportion of newly diagnosed MS patients across all MS types (2017–2022) who initiated treatment with category 1 or 2 DMTs and switched to category 3 DMTs by quarter since first prescription of category 1 or 2 DMT.
Among patients who escalated from a category 1 or 2 DMT to a category 3 DMT, 8.5% did so within the first 6 months of their initial prescription, while 31.5% took more than 2 years to escalate (Figure 4(b)).
Discussion
Early high-efficacy treatment (EHT) approach is a promising new strategy in the treatment of MS. This study aimed to provide evidence into the application of the EHT strategy across different MS disease subtypes in Germany based on SHI claims data from 2017 to 2022.
Our results showed that most (85.2%) newly diagnosed MS patients between 2017 and 2022 started treatment with mild- and moderate-efficacy category 1 or 2 DMTs, and 14.8% started treatment with high-efficacy category 3 DMTs. These results are in line with findings from a prescription sales data analysis from Germany 2020–2022 showing that among adult MS therapy beginners, 86.3% of patients redeemed their first prescription for category 1 or 2 medication, and 13.7% for category 3 medications. 16 We additionally showed that the initiation medication category depended on disease type with 13.3% of RRMS, 40.5% of PPMS patients and 23.8% of SPMS patients initiating treatment with category 3 DMTs.
We observed a shift towards increased adoption of the EHT approach. Between 2017 and 2022, the proportion of patients initiating treatment with category 3 DMTs significantly increased by 4.8 pp, while the proportion of those starting on category 1 DMTs significantly decreased by 7.6 pp. Similar trends were seen in prescription sales data from Germany from 2020 to 2022, with a significant 14.5 pp increase in category 2 and/or 3 initiations and an equivalent decrease in category 1 initiations, as well as in US claims data during the same period. 17 Notably, in earlier claims data analyses covering the periods 2010–2017 and 2012–2019, this shift towards category 3 initiations was not yet apparent.26,27
These trends can be linked to shifts in DMT approvals based on MS types. Specifically, our data show an increasing trend of category 3 initiations from 2017 to 2022 among PPMS patients with a rise of 15.4 pp. This is likely due to the approval of ocrelizumab as the sole DMT for PPMS in January 2018. 24 While we observed an increase in category 3 initiations in PPMS, the proportion of RRMS patients remained stable, even with the availability of another category 3 DMT, ofatumumab, since 2021. 25
Older patients seem more likely to start treatment with category 3 DMTs compared to younger patients, aligning with findings from the 2020–2022 German prescription sales data analysis. 16 This trend is particularly notable, given that a meta-analysis from 2017 indicates that patients over 40 do not experience greater benefits from high-efficacy drugs compared to low-efficacy, 28 yet appear to frequently start on high-efficacy treatments in real-world settings. However, the conclusions of the meta-analysis may be limited given the comparatively smaller number of clinical trials for category 3 DMTs. Additionally, stage migration resulting from changes in the MS diagnostic criteria may influence the interpretation of treatment efficacy, as patients diagnosed earlier may have different disease trajectories and responses to therapy compared to those diagnosed under older criteria. Several factors may contribute to the real-world preference for category 3 DMTs in older patients. Known medication side effects like hepatic enzyme elevation in interferons, 29 and contraindications, such as coronary diseases in fingolimod 30 may influence treatment decisions. Additionally, the longer dosing intervals of category 3 DMTs, like ocrelizumab, can improve compliance, 31 especially for older patients with cognitive deficits who struggle with daily medication adherence. In Germany, treatment decisions are made collaboratively between the physician and the patient, with insurance companies playing no restrictive role in the choice of DMT. Physicians provide a rationale for treatment selection, but insurance companies do not impose limitations or require stepwise treatment escalation before accessing high-efficacy therapies. Therefore, the uptake (or lack thereof) of EHT is primarily driven by physician and patient preferences rather than reimbursement restrictions.
Furthermore, we found a trend that male patients initiated treatment with category 3 DMTs more often than female patients. These trends may reflect personalized MS treatment approaches, 32 influenced by age- and sex-based preferences among patients and/or physicians, as well as safety concerns related to DMT use during pregnancy and breastfeeding. Interferon-beta and glatiramer acetate are among the few DMTs deemed safe in the first trimester and while breastfeeding, leading many women to prefer these low-efficacy options over high-efficacy therapies, which could pose risks to fetal and neonatal health. 32
Our results show that most patients – around 90% – remained on their initially prescribed medication category without escalating or de-escalating their treatment. This finding is consistent with the 2020–2022 German prescription sales data analysis. 16 Notably, a higher proportion of patients who began with low- or moderate-efficacy therapies escalated to high-efficacy therapies (10.5%) compared to those who started on high-efficacy therapies and subsequently de-escalated to low- or moderate-efficacy options (3.3%). This trend is also in line with the findings from the 2020–2022 German data analysis. 16 Among patients who began on category 1 or 2 therapies and eventually escalated to category 3, approximately one-third of patients took longer than 2 years to make the switch. This suggests that escalation often occurs slowly, potentially missing the optimal window of opportunity for more effective intervention.
Limitations
Although the dataset is structural representative of the German SHI population in terms of age and sex, 33 and integrates treatment data from various sectors, this study has certain limitations. Claims data may not provide exact information on prescribed dosages, and filled prescriptions do not confirm actual medication intake by patients. Furthermore, the results are limited to SHI-insured individuals, and inconsistencies in coding quality could influence the accuracy of the data. Newly diagnosed patients were identified by considering a 2-year lookback period which may have led to some misclassification, as patients previously diagnosed with RRMS before this window could have been categorized as newly diagnosed SPMS patients. Furthermore, the use of DMTs was linked to the year of diagnosis rather than the year of prescription, which may lead to reporting the use of DMTs in years before their official approval dates.
Conclusion
Real-world data provide critical insights into the actual care practices for newly diagnosed MS patients and the adoption of novel treatment initiation strategies. This analysis has identified a trend towards earlier initiation of high-efficacy treatments, driven by the availability of newly approved high-efficacy DMTs for specific MS subtypes.
