Abstract
Background:
Serum neurofilament light chain (sNfL) is a biomarker for neuro-axonal injury.
Objectives:
To assess sNfL’s utility as a diagnostic marker for Lyme neuroborreliosis (LNB).
Methods:
We compared serum and CSF NfL levels in LNB patients and age-matched controls. Age-adjusted NfL values were used in receiver operating characteristic (ROC) analysis.
Design:
Retrospective cohort study.
Results:
Eighty-six patients (30 LNB, 29 with-, and 27 without neurological disorders) were included. Compared to individuals without neurological disease, LNB patients showed increased serum (median (interquartile range, IQR): 36.3 pg/ml (19.3–112.0) vs 20 pg/ml (12.9–37.3), p < 0.001) and CSF NfL levels (median (IQR): 1000.0 pg/ml (286.0–6471.0) vs 182 pg/ml (99.3–474.0), p < 0.001). NfL concentrations were similar in LNB and other neurological disorders. ROC analysis of age-adjusted sNfL and CSF NfL levels showed areas under the curve of 0.78 (95% confidence interval (CI): 0.66–0.89) and 0.83 (95% CI: 0.71–0.94), respectively.
Conclusion:
sNfL concentrations lack sufficient diagnostic capability for LNB diagnosis.
Introduction
Erythema migrans is the most common clinical manifestation of Lyme disease, but the infection can also affect organs beyond the skin, including the joints, heart, and nervous system. 1 In Europe, the most frequent presentation of Lyme neuroborreliosis (LNB) in adults is Garin–Bujadoux–Bannwarth’s syndrome, characterized by painful radiculoneuritis.2,3 Other symptoms of LNB include cranial nerve palsy, meningitis, and rarely encephalomyelitis.2,3 Since Garin and Bujadoux 4 first described tick paralysis in 1922, significant advancements have been made in the pathophysiological understanding and diagnosis of LNB. The diagnostic criteria for LNB are based on the combination of neurological symptoms and cerebrospinal fluid (CSF) findings, with lymphocytic pleocytosis, and evidence for intrathecal Borrelia burgdorferi specific antibody production. 5 In addition, CSF CXCL13, a B-cell chemoattractant, has been recognized as a valuable marker, particularly in early cases where there may not yet be detectable intrathecal antibody production specific to B. burgdorferi. 6 Neurofilaments are cytoskeletal proteins expressed exclusively in neurons of the peripheral and central nervous systems. 7 Neurofilament light chains (NfL) enter the CSF and blood as a result of axonal damage. 7 Recently, serum neurofilament light chain (sNfL) measurements have been adopted for monitoring the progression of central nervous system injury across several neurodegenerative disorders such as multiple sclerosis, Alzheimer’s disease, and amyotrophic lateral sclerosis. 8 Additionally, studies assessed the potential of sNfL measurements for quantifying infection induced neuro-axonal injury.9 –11
The objective of this pilot study was to assess the characteristics of sNfL as an additional diagnostic marker for assisting the diagnostic armamentarium for LNB.
Methods
Study design
We conducted a retrospective analysis of serum and CSF samples from all patients diagnosed with definite LNB according to the European Federation of Neurological Societies (EFNS) criteria. 5 We included patients treated at Inselspital (University Hospital of Bern, Switzerland) between January 1, 2018, and December 31, 2021, who provided consent for the reuse of their biological samples for research purposes. Patients without consent or without stored serum were excluded. We did not conduct a sample size calculation for this pilot study. We compared serum and CSF NfL levels in LNB patients with those in two age-matched (±5 years) control groups: one with neurological diseases and one without. Patients presenting with headache syndromes and transient neurologic disorders, such as paresthesia and dizziness, of unidentified etiology were categorized as not having a neurologic disease (controls). We employed age-matched controls, considering that sNfL levels naturally rise with age. 8 All patients consented to the reuse of biological samples for research purposes. We reported our results in accordance with the STARD 2015 guidelines for diagnostic accuracy studies. 12 The study was approved by the Ethics Commission of the Canton of Bern, Switzerland, Nr. 2021-00483.
Definitions
We applied the EFNS criteria for diagnosis of LNB. 5 Patients were classified to have definite LNB if they fulfilled all following criteria (i) neurological symptoms of LNB without other obvious reasons, (ii) CSF pleocytosis (>5 leukocytes/μl), and (iii) intrathecal B. burgdorferi antibody production by examination of simultaneously sampled CSF and serum.
We categorized LNB as early LNB if symptom duration was <6 months and as late LNB if symptom duration was ⩾6 months. 5
We implemented stringent criteria to exclude LNB in control patients. They were required to have a CSF CXCL13 concentration below the manufacturer’s specified threshold for LNB and to lack intrathecal B. burgdorferi antibody production. The NfL measurement results were not available when categorizing the patients into one of the three groups.
Laboratory analyses
We used frozen (−30°C) serum and CSF which was stored after performing the analyses for confirming or excluding LNB. To quantify serum and CSF NfL levels, we used an automated enzyme-linked immunosorbent assay (ELISA)-based microfluidic system (ELLA®; ProteinSimple, San Jose, CA, USA). The algorithm for diagnosis of B. burgdorferi infection, included a screening test (Serion ELISA classic B. burgdorferi IgG/IgM®; SERION Diagnostics, Würzburg, Germany) according to the manufacturer’s instructions followed by a confirmatory Westernblot (recomLine Borrelia®; Mikrogen, Neuried, Germany). The CSF/serum antibody index was determined using a commercially available kit (Serion ELISA classic B. burgdorferi IgG/IgM, considered positive if ⩾1.5). The individuals conducting the NfL measurements were not blinded to the categorization of the patient samples.
Statistics
Categorical variables are displayed as percentages, and continuous variables as medians with interquartile ranges (IQR). Differences in NfL levels among age-matched groups were analyzed using Wilcoxon signed-rank tests. For calculation of age-adjusted NfL levels, we used the method described by Harp et al. 13 In brief, a log-linear model was employed to standardize the sNfL and CSF NfL levels of patients without neurological disorders to a reference age of 18 years. The slope (sNfL: 0.0154206 log10 sNfL increase per year of age; CSF NfL: 0.0260784 log10 CSF NfL increase per year of age) of the regression line was used to calculate age-adjusted NfL levels: NfLadj = 10log10(NfL)−Slope(Age-18). 13
Nonparametric receiver operating characteristic (ROC) analysis was performed to assess the diagnostic performance of age-adjusted sNfL and CSF NfL levels as described by DeLong et al. 14 We used the Liu index to calculate the optimal serum and CSF NfL cut-offs for discriminating LNB patients and individuals without neurological disease. 15 Statistical significance was set at a p value < 0.05. Statistical analyses were performed using Stata software version 16.0 (Stata Corp., College Station, TX, USA).
Results
We obtained serum samples from 30 patients with definite LNB. Twenty-nine and 27 serum samples of age-matched individuals with non-LNB neurological diseases and without neurological diseases were included (Table 1). Sufficient CSF volume for NfL analysis was available for most patients (29/30 with LNB, 29/29 with non-LNB neurological disease, and 26/27 without neurological disease, Supplemental Figure 1).
Patient characteristics.
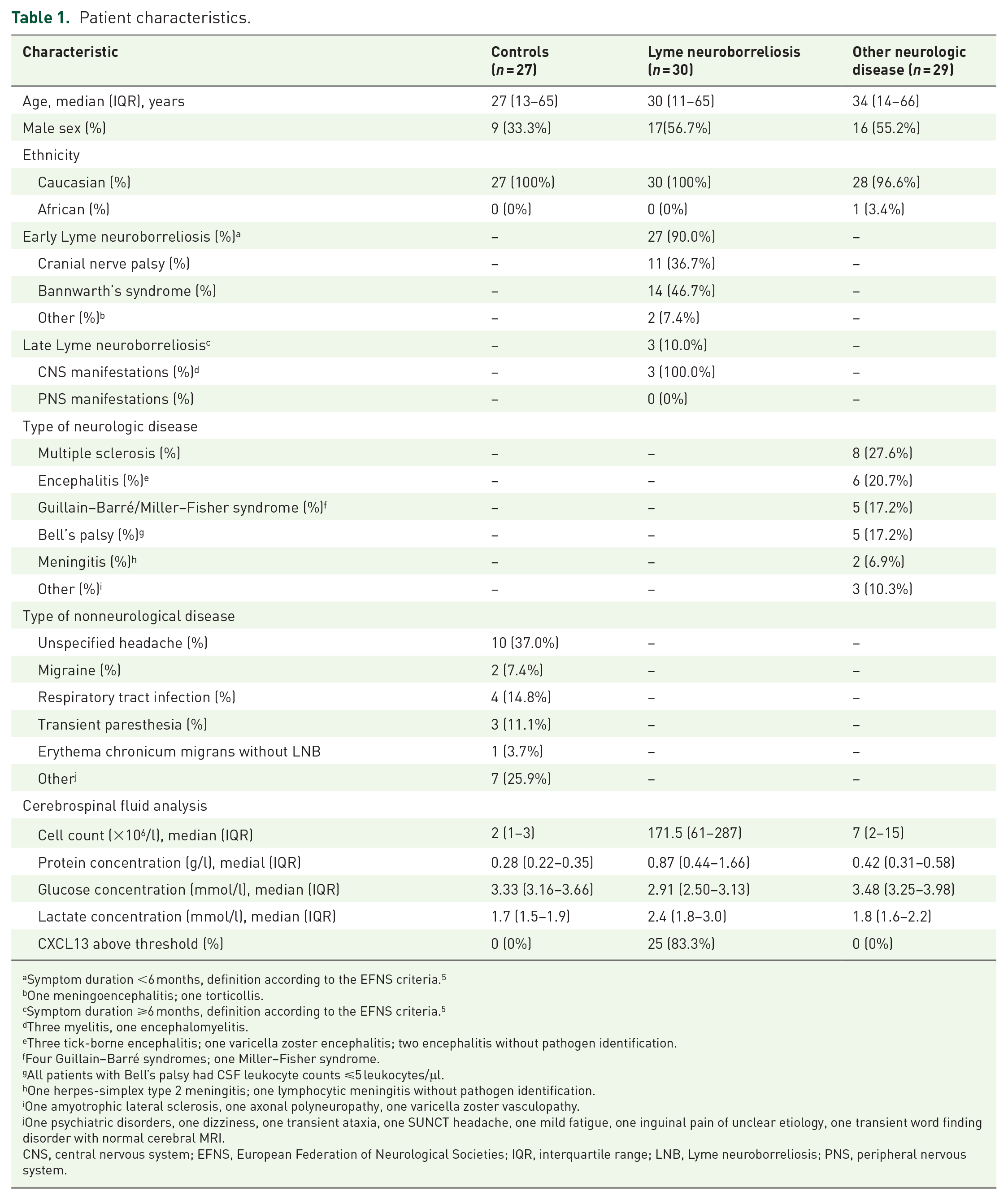
Symptom duration <6 months, definition according to the EFNS criteria. 5
One meningoencephalitis; one torticollis.
Symptom duration ⩾6 months, definition according to the EFNS criteria. 5
Three myelitis, one encephalomyelitis.
Three tick-borne encephalitis; one varicella zoster encephalitis; two encephalitis without pathogen identification.
Four Guillain–Barré syndromes; one Miller–Fisher syndrome.
All patients with Bell’s palsy had CSF leukocyte counts ⩽5 leukocytes/μl.
One herpes-simplex type 2 meningitis; one lymphocytic meningitis without pathogen identification.
One amyotrophic lateral sclerosis, one axonal polyneuropathy, one varicella zoster vasculopathy.
One psychiatric disorders, one dizziness, one transient ataxia, one SUNCT headache, one mild fatigue, one inguinal pain of unclear etiology, one transient word finding disorder with normal cerebral MRI.
CNS, central nervous system; EFNS, European Federation of Neurological Societies; IQR, interquartile range; LNB, Lyme neuroborreliosis; PNS, peripheral nervous system.
In age-matched patients, sNfL levels of individuals with LNB were significantly higher compared to individuals without neurologic disease (median (IQR): 36.3 pg/ml (19.3–112.0) vs 20 pg/ml (12.9–37.3), p < 0.001, Figure 1(a)). Likewise, patients with non-LNB neurological diseases exhibited higher sNfL concentrations compared to those without neurological diseases (median (IQR): 38.0 pg/ml (19.6–69.6) vs 20 pg/ml (12.9–37.3), p = 0.003, Figure 1(a)). sNfL levels did not differ between age-matched patients diagnosed with LNB and those with non-LNB neurological diseases (p = 0.405, Figure 1(a)). The comparisons of CSF NfL levels among the age-matched patient cohorts mirrored those observed in serum analysis (Figure 1(b)).

Serum and CSF neurofilament light chain levels. (a) Serum NfL levels in Lyme neuroborreliosis patients as compared to age-matched (±5 years) individuals without neurologic disease (Controls) and to patients with other neurologic disease than Lyme neuroborreliosis (Other neurologic disease). (b) CSF NfL levels in Lyme neuroborreliosis patients as compared to age-matched (±5 years) individuals without neurologic disease (Controls) and to patients with other neurologic disease than Lyme neuroborreliosis (Other neurologic disease). (c) Age-adjusted serum NfL levels in Lyme neuroborreliosis patients as compared to individuals without neurologic disease (Controls) and to patients with other neurologic disease than Lyme neuroborreliosis (Other neurologic disease). (d) Age-adjusted CSF NfL levels in Lyme neuroborreliosis patients as compared to individuals without neurologic disease (Controls) and to patients with other neurologic disease than Lyme neuroborreliosis (Other neurologic disease).
When analyzing age-adjusted sNfL levels, LNB patients (median (IQR): 29.7 pg/ml (17.1–57.3)) and individuals with non-LNB neurologic disease (median (IQR): 21.7 pg/ml (20.7–37.7)) proved to have higher sNfL levels than individuals without neurologic disease (median (IQR): 15.6 pg/ml (10.7–20.8), p < 0.001 for both comparisons; Figure 1(c)). Age-adjusted sNfL levels of LNB- and non-LNB neurologic disease were similar (p = 0.580, Figure 1(c)). Similarly, age-adjusted CSF NfL levels of LNB patients (median (IQR): 490.9 pg/ml (176.5–2406.9)) and non-LNB neurologic disease patients (median (IQR): 337.8 pg/ml (249.3–1141.5)) were increased compared to CSF levels of individuals without neurologic disease (median (IQR): 145.9 pg/ml (99.2–185.8), p < 0.001 for both comparisons; Figure 1(d)). Age-adjusted NfL levels corresponding to different manifestations of LNB are delineated in Supplemental Figure 2.
When including patients with LNB and without neurologic disease in a ROC analysis of age-adjusted sNfL- and CSF NfL levels, the area under the curve (AUC) was 0.78 (95% CI: 0.66–0.90) and 0.83 (95% CI: 0.71–0.94), respectively (Supplemental Figure 3). Comparable results were obtained when including patients with non-LNB neurologic disease and individuals without neurologic disease (AUC sNfL: 0.76 (95% CI: 0.63–0.89). AUC CSF NfL: 0.90 (95% CI: 0.81–0.98; Supplemental Figure 4). The ROC analysis for discriminating LNB patients and individuals with non-LNB neurologic disease by sNfL levels and CSF NfL concentrations revealed an AUC of 0.54 (95% CI: 0.39–0.69) and 0.51 (95% CI: 0.36–0.67; Supplemental Figure 5).
The optimal cut-off to distinguish LNB patients from individuals without neurological disease was 25.7 pg/ml for age-adjusted sNfL, yielding a sensitivity of 0.63 and a specificity of 0.89. For age-adjusted CSF NfL, the optimal cut-off was 253.5 pg/ml, with a sensitivity of 0.69 and a specificity of 0.96.
Discussion
The major findings of our study were as follows: First, patients with definite LNB have increased serum and CSF NfL levels as compared to individuals without neurological disease. Second, serum and CSF NfL concentrations are similar in individuals with LNB and other neurological disorders. Third, the predictive accuracy, assessed by analyzing the area under the ROC curve of age-adjusted sNfL levels for diagnosing LNB, is deemed insufficient for clinical application.
A recent study reported that plasma NfL (pNfL) levels decreased following antibiotic treatment of LNB patients. 16 Using a different method for pNfL quantification (Quanterix® kits; Quanterix, Billerica, MA, USA. Simoa® NF-light Kit; Quanterix, Billerica, MA, USA), ~90% of patients (33/36) with LNB had pNfL levels above an age-defined reference at initiation of antibiotic therapy in this study. However, the median difference from the age-defined pNfL reference at the time of antibiotic therapy initiation was marginal (1.17 pg/ml; IQR 0.22–7.2). 16 In another study (using Simoa NF-light Kit for sNfL measurements) designed to investigate the value of sNfL as a prognostic marker in LNB patients, Skarstein et al. 11 report that only 63% (57/90) of their patients with definite or probable LNB had sNfL levels above an age-defined reference at the time of diagnosis. In our study, we used the ELLA (ProteinSimple) platform for NfL measurements. Both assays (ELLA and Simoa) utilize the same antibody for detecting NfL. Recent studies, however, have demonstrated that the ELLA platform yields systematically higher NfL values than the Simoa platform, but that the difference shows a linear relationship.17 –19 Due to the lack of age-specific reference values for sNfL generated using the ELLA platform, we employed an alternative approach for data analysis involving age-matching and age-adjustment. In line with the results of Mens et al. 16 and Skarstein et al., 11 we found that the sensitivity of sNfL values for reliable detection of LNB may be insufficient for clinical application and that the difference in sNfL concentration between LNB patients and healthy individuals is rather small.
Skarstein et al. 11 noted in their study that LNB patients with sNfL values ⩾95th percentile at baseline had significantly higher age. They emphasized that they did not include an age-matched control group in their analysis, and therefore, cannot exclude age as a factor of these observations. 11 To address this issue, we adopted their recommendation and implemented an age-matched analysis.
One notable strength of our study lies in its methodological approach to tackle the age-related rise in sNfL levels. To address this, we conducted both age-matched and age-adjusted analyses. In contrast to other studies, we investigated not only sNfL levels but also CSF NFL concentrations. Nevertheless, the study size is relatively small, and we included patients with early and late LNB. Due to the limited sample size, reasonable subgroup analyses are not possible, and patients with late LNB were underrepresented in our analysis. Additionally, we did not perform a sample size calculation; instead, we used all available samples.
Conclusion
Patients with LNB have increased sNfL and CSF NfL levels as compared to individuals without organic neurologic disease. However, we conclude that the discriminatory capabilities of sNfL concentrations as a diagnostic marker for LNB are insufficient for clinical use.
Supplemental Material
sj-pdf-1-tan-10.1177_17562864251314011 – Supplemental material for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis
Supplemental material, sj-pdf-1-tan-10.1177_17562864251314011 for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis by Cédric Hirzel, Annina Grütter, Denis Grandgirard, Robert Hoepner, Franziska Suter-Riniker and Stephen L. Leib in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-2-tan-10.1177_17562864251314011 – Supplemental material for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis
Supplemental material, sj-pdf-2-tan-10.1177_17562864251314011 for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis by Cédric Hirzel, Annina Grütter, Denis Grandgirard, Robert Hoepner, Franziska Suter-Riniker and Stephen L. Leib in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-3-tan-10.1177_17562864251314011 – Supplemental material for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis
Supplemental material, sj-pdf-3-tan-10.1177_17562864251314011 for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis by Cédric Hirzel, Annina Grütter, Denis Grandgirard, Robert Hoepner, Franziska Suter-Riniker and Stephen L. Leib in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-4-tan-10.1177_17562864251314011 – Supplemental material for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis
Supplemental material, sj-pdf-4-tan-10.1177_17562864251314011 for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis by Cédric Hirzel, Annina Grütter, Denis Grandgirard, Robert Hoepner, Franziska Suter-Riniker and Stephen L. Leib in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-5-tan-10.1177_17562864251314011 – Supplemental material for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis
Supplemental material, sj-pdf-5-tan-10.1177_17562864251314011 for Evaluating neurofilament light chain serum levels as a diagnostic marker for Lyme neuroborreliosis by Cédric Hirzel, Annina Grütter, Denis Grandgirard, Robert Hoepner, Franziska Suter-Riniker and Stephen L. Leib in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
