Abstract
Background:
China has a large number of myasthenia gravis (MG) patients, creating an urgent need for rapid and tolerable treatment options. As the first-approved Fc receptor antagonist, efgartigimod has bright prospects for treating MG. However, real-world evidence on its application within the Chinese MG population are limited.
Objective:
This study aims to evaluate the rapid efficacy and safety of efgartigimod in Chinese MG population.
Design:
This single-center prospective study enrolled Chinese MG patients aged 18 and older who were treated with efgartigimod, classified as Myasthenia Gravis Foundation of America I–IV, with a baseline Myasthenia Gravis Activities of Daily Living (MG-ADL) score of at least 4.
Methods:
Patients received efgartigimod at a dose of 10 mg/kg infused once weekly for 4 weeks. During the treatment, the corticosteroids dosage could be adjusted as appropriate or the non-steroidal immunosuppressive therapies (NSISTs) added. Prior to each infusion, patients’ MG-ADL scores, IgG levels, and routine laboratory tests were evaluated, while also recording the prednisone tapering and any adverse events occurring during the treatment.
Results:
Twenty five Chinese MG patients were enrolled between November 2023 and June 2024, including 3 with ocular MG (OMG) and 22 with generalized MG (GMG). During the 8-week follow-up, in GMG patients, whether positive for acetylcholine receptor (AChR) or muscle-specific tyrosine kinase (MuSK) antibodies, the overall efficacy was significant. Within one treatment cycle, 18 (82%) patients showed a reduction of at least 2 points in MG-ADL scores and sustained for at least 4 weeks, and 6 (27%) attained minimal symptom expression (MSE) and sustained for at least 4 weeks. Only 1 patient experienced exacerbation. Among OMG patients, 1 achieved MSE within the treatment cycle, while 2 showed minor improvements. Patients who added tacrolimus concurrently with efgartigimod did not achieve better improvement in MG-ADL scores compared to others. The average reduction in prednisone dosage was 27.4%. Only one patient experienced transient vomiting and diarrhea, with no serious adverse reactions reported.
Conclusion:
This study confirmed the short-term efficacy and safety of efgartigimod in Chinese MG patients. However, in clinical practice, careful consideration is needed regarding its application in OMG and whether to add NSISTs regimen during the treatment. Efgartigimod could potentially serve as an alternative to long-term corticosteroids therapy.
Introduction
Myasthenia gravis (MG) is an immune-mediated disorder characterized by specific autoantibodies, 1 which can be detected in the majority of patients (approximately 85%). The most common autoantibodies are against acetylcholine receptors (AChRs), while antibodies against muscle-specific tyrosine kinase (MuSK) and low-density lipoprotein receptor-related protein 4 (LRP4) are relatively rare. These antibodies are directly involved in the pathogenic process of MG, 2 and intervening in the immune process is a principal strategy for MG treatment. However, conventional immunosuppressive drugs, particularly corticosteroids,3,4 have made us aware of the unavoidable side effects during long-term clinical treatment, creating an urgent need for more effective and better-tolerated treatment options.
With the advent of the era of biologics, various novel drugs targeting different nodes of the pathogenesis of MG have been developed, showing promising prospects. 5 Among these, neonatal Fc receptor (FcRn) inhibitors can occupy the IgG binding sites on FcRn, thereby increasing the catabolism of autoantibodies, resulting in reduced IgG levels. This represents a hopeful new direction for the treatment of MG. 6
Efgartigimod is the first approved FcRn antagonist in the world. As a humanized IgG1 Fc fragment, it competitively binds to FcRn, thereby rapidly and effectively inhibiting the circulatory recycling of IgG. In a phase III clinical trial (An Efficacy and Safety Study of ARGX-113 in Patients With Myasthenia Gravis Who Have Generalized Muscle Weakness, ADAPT), 7 efgartigimod demonstrated exceptional clinical efficacy and tolerability. This success has led to its continuous approval in the United States, Japan, and Europe. Subsequent real-world studies have also begun to validate the outstanding clinical performance of efgartigimod.8 –10
In September 2023, efgartigimod was also approved for marketing in China. However, our experience with the use of this emerging drug is still limited, and the sample sizes of Asian populations included in previously published studies have been quite small. With a large population base in our country, the number of MG patients can reach up to 200,000. The substantial patient population combined with limited medical resources drives our urgency to deliver rapid and prominent treatment effects for patients, leading us to have high expectations for the short-term efficacy of this novel drug. Therefore, we conducted a single-center prospective study at the First Affiliated Hospital of Chongqing Medical University to evaluate the short-term clinical efficacy and safety of efgartigimod in the Chinese MG population, aiming to provide more real-world experience for this novel FcRn inhibitor in the treatment of MG.
Methods
Study design
This study is a single-center prospective investigation conducted at the neurology clinic and ward of the First Affiliated Hospital of Chongqing Medical University. From November 2023 to June 2024, patients were screened and enrolled with the aim of exploring the short-term clinical efficacy and safety of efgartigimod in the Chinese MG population, providing real-world experience for the treatment of MG patients with efgartigimod. The reporting of this study conforms to STROBE guidelines. 11
Inclusion and exclusion criteria
Eligible patients must meet the following inclusion criteria:
(a) Be at least 18 years old at the time of enrollment.
(b) Meet one of the following diagnostic criteria: (1) abnormal single-fiber electromyography or positive repetitive nerve stimulation; (2) positive anti-acetylcholinesterase test; (3) improvement in MG symptoms following oral administration of acetylcholinesterase inhibitors, as assessed by the physician.
(c) Classified as type I–IV according to the Myasthenia Gravis Foundation of America (MGFA) classification.
(d) Myasthenia Gravis Activities of Daily Living (MG-ADL) score of ⩾4 points.
(e) Currently undergoing MG maintenance therapy (acetylcholinesterase inhibitors, corticosteroids, or NSISTs) or not receiving MG drug treatment.
Patients meeting the following criteria will be excluded:
(a) MGFA Class V.
(b) Received treatment with novel biologics (including rituximab, ofatumumab, eculizumab or efgartigimod) within 6 months prior to screening.
(c) Underwent thymectomy within 3 months prior to screening.
(d) Received IVIg or plasma exchange (PE) treatment within 1 month prior to screening.
(e) Pregnancy.
(f) Presence of autoimmune diseases other than MG.
(g) Presence of other serious chronic diseases or mental disorders that may affect the assessment.
Procedures
Each patient’s decision to use efgartigimod was based on the clinical judgment of the neurologist regarding the patient's condition and the patient’s personal willingness. The dosage of efgartigimod is 10 mg/kg, with a maximum single infusion limit of 1200 mg for patients weighing ⩾120 kg, administered via intravenous infusion over 1 h, once a week for a total of 4 weeks, constituting one treatment cycle. After initiation of treatment, NSISTs could be added as a combination therapy, and corticosteroids that have been used stably prior to enrollment could have their dosage adjusted as needed based on the patient's condition. Patients who required rescue treatment or needed to undergo PE or IVIg therapy due to significant worsening of symptoms would be discontinued from the study. However, to ensure safety, patients who were withdrawn would continue to be followed up.
Baseline information was collected at the time of enrollment, including age, gender, comorbidities, disease duration, baseline medication, presence of thymoma, and whether thymectomy has been performed. The type of autoantibody was detected by the cell-based assay method. Efficacy was assessed using the MG-ADL scores.12,13 Initially, a baseline evaluation of each patient was conducted at enrollment, followed by MG-ADL scoring at each efgartigimod infusion, and follow-up scoring 1 week and 5 weeks after the end of one treatment cycle. The MG-ADL scores at each time point were compared with the baseline to calculate the score change.
Blood samples were collected at baseline and before each infusion of efgartigimod for immunoglobulin level testing by ELISA, along with routine tests including complete blood count, liver function, kidney function, electrolytes, and electrocardiogram assessments.
Outcomes
The efficacy assessment indicators included changes in MG-ADL scores compared to baseline at each follow-up, the rate of MG-ADL improvement (defined as a decrease of at least 2 points from baseline), the proportion of patients achieving minimal symptom expression 13 (MSE, defined as MG-ADL score ⩽ 1), and the proportion of patients with deterioration (defined as new symptoms or an increase of ⩾2 points in MG-ADL score). Additionally, we assessed changes in patients’ IgG levels and the reduction of corticosteroid dosage during the follow-up period.
The safety of the drug was evaluated through adverse events (AEs), including infusion-related AEs, infection-related AEs, severe AEs, and other common AEs (such as headache, nasopharyngitis, nausea, diarrhea, upper respiratory infections, urinary tract infections, etc.), as well as changes in patients’ vital signs and electrocardiogram findings.
Statistical analysis
Statistical analyses were performed by IBM SPSS Statistics version 27 (IBM Corp., Armonk, NY, USA). Continuous variables were compared by analysis of variance and independent t-tests, while categorical data were compared by the chi-squared tests. Probability values < 0.05 were considered statistically significant.
Results
Baseline features
This study cohort included 25 patients with MG, comprising 22 patients with generalized MG (GMG) and 3 patients with ocular MG (OMG). All patients completed at least 8 weeks of follow-up. Table 1 summarizes the main demographic and baseline clinical characteristics.
Demographic and baseline clinical characteristics of all patients.

Data are mean (SD) or n (%).
AChEi, acetylcholinesterase inhibitors; AChR-Ab, acetylcholine receptor antibody; MG-ADL, Myasthenia Gravis Activities of Daily Living; MGFA, Myasthenia Gravis Foundation of America; MuSK-Ab, muscle-specific tyrosine kinase antibody; NSIST, non-steroidal immunosuppressive therapy.
Among the enrolled patients, the majority were female (17/25, 68%). Most patients were classified as MGFA II (10/25, 40%) and III (10/25, 40%). The majority of patients were positive for AChR antibodies (22/25, 88%), with two patients being positive for MuSK antibodies (MuSK-MG) and both classified as MGFA IV. One patient had an undefined autoantibody type (MGFA II) due to a reluctance to complete the autoantibody testing (1/25, 4%). A total of four patients had previously undergone thymectomy. Less than half of the patients were receiving immunosuppressive therapy at the time of inclusion (10/25, 40%), among which seven patients (all AChR-GMG) were on a maintenance dose of prednisone, with a mean dose of 34.3 (SD 18.1) mg/d.
Efficacy in GMG
During the 8-week follow-up period, GMG patients showed significant efficacy overall (Table 2). Within the first week, 15 patients (68%) showed a reduction of at least 2 points in their MG-ADL scores, and 2 (9%) achieved MSE. During one treatment cycle, 18 patients (82%) showed a reduction of at least 2 points in their MG-ADL scores and sustained for at least 4 weeks, and 6 patients (27%) attained MSE and sustained for at least 4 weeks. At the end of one treatment cycle, the MG-ADL score for GMG patients decreased from 7.2 (±3.0) to 2.7 (±2.2), and at the conclusion of the 8-week follow-up period, it further decreased to 2.5 (±2.4). Only 1 patient (5%) experienced worsening symptoms during the treatment, but fortunately, after completing the treatment cycle, her clinical symptoms began to improve, and the MG-ADL score stabilized below 3.
Summary of the efficacy of efgartigimod in GMG patients.

Data are n (%).
GMG, generalized myasthenia gravis; MG-ADL, Myasthenia Gravis Activities of Daily Living; MSE, minimal symptom expression.
Among all the GMG patients included in the study, there were a total of 19 AChR-positive individuals. Their changes in MG-ADL, illustrated in Figure 1, showed the most substantial improvement in the first week, followed by sustained improvement in each subsequent assessment, demonstrating statistically significant differences compared to baseline. Among the two MuSK-MG patients, the MG-ADL score improvement was 9 points and 10 points by the end of the treatment cycle (Figure 2), respectively, and the former patient experienced a symptom rebound in the eighth week. The patient with unknown autoantibodies showed no decrease in MG-ADL score.
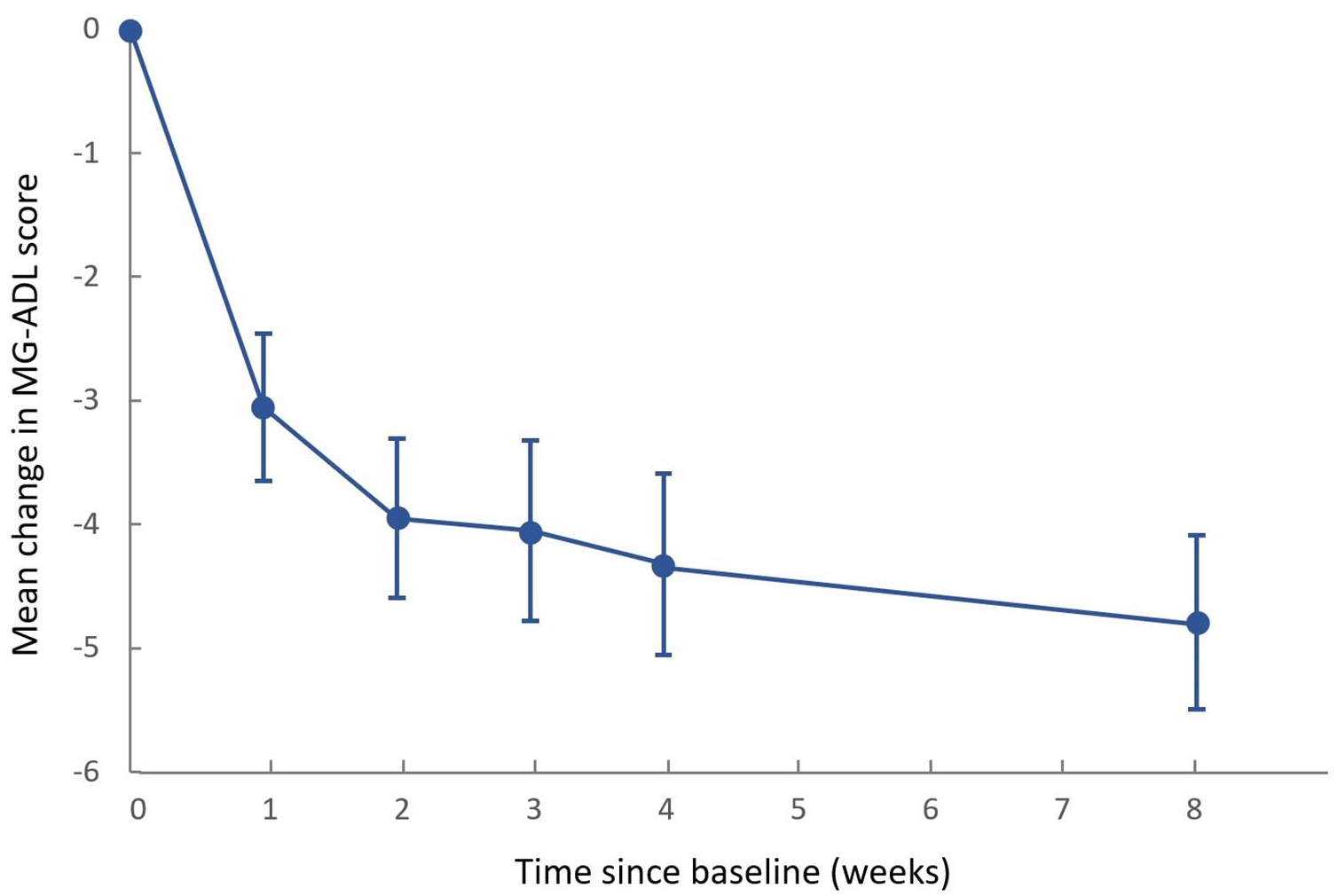
Mean change in MG-ADL score of AChR-GMG patients. Error bars show standard error.
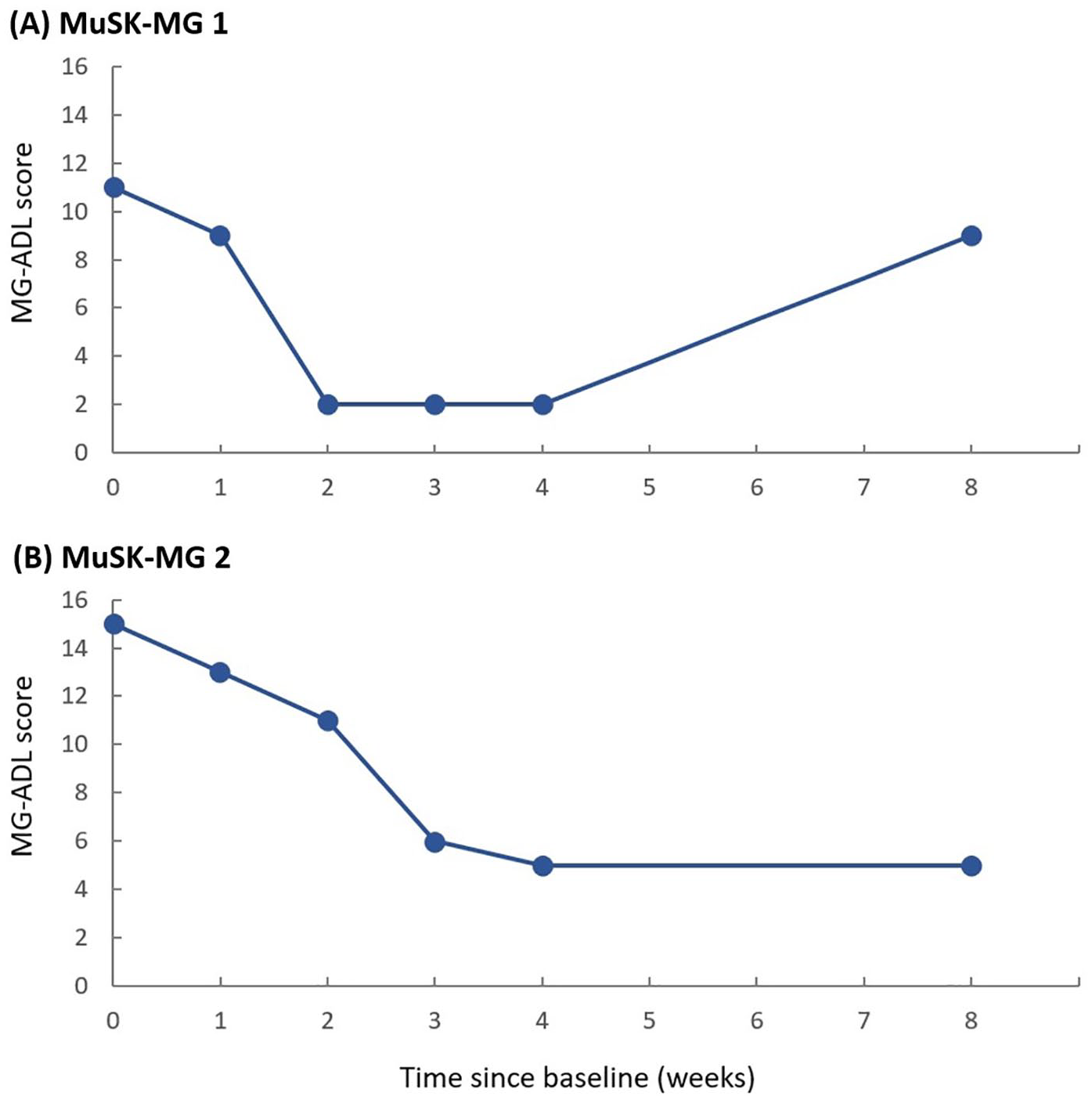
MG-ADL score changes in two MuSK-MG patients.
Efficacy in OMG
Three OMG patients who were AChR-Ab positive opted for treatment with efgartigimod. Figure 3 demonstrates their changes in MG-ADL scores. One patient revealed satisfactory efficacy after completing a cycle of efgartigimod infusion, reaching MSE (Figure 3(a)). In contrast, the other two patients did not achieve satisfactory results in either their MG-ADL scores or their subjective perceptions of improvement.
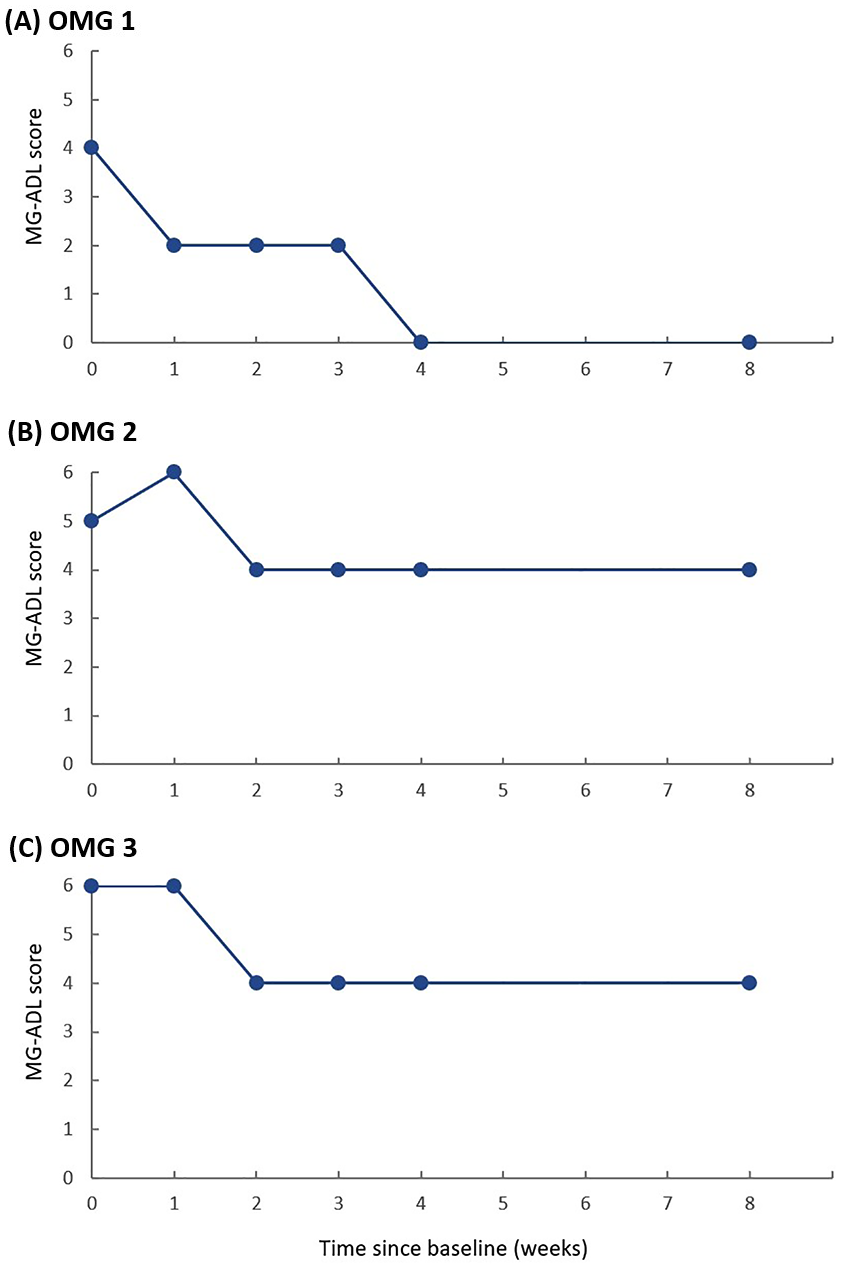
MG-ADL score changes in 3 OMG patients.
Impact of the combination with tacrolimus on efficacy
At the start of efgartigimod treatment, some patients (all were AChR-GMG) received additional NSISTs (six received tacrolimus, one received mycophenolate mofetil, and one received azathioprine). Considering that mycophenolate mofetil and azathioprine have a slower onset of action, 14 we conducted a separate analysis only on patients who were additionally treated with tacrolimus, and they maintained effective drug concentrations during regular follow-up. In contrast, seven patients did not use NSIST. There were no statistical differences in baseline characteristics between the two groups regarding age (p value 0.896), disease duration (p value 0.545), or MG-ADL scores (p value 0.772). However, it was unexpectedly found that patients who added tacrolimus did not have a more significant reduction in scores throughout the follow-up compared to the other group (p value > 0.05, Figure 4).

Mean change in MG-ADL score of AChR-GMG patients who added tacrolimus and without NSIST during treatment. Error bars show standard error.
Changes in IgG
Although not every patient’s IgG level test was completed, the available data showed that IgG levels decreased before each infusion compared to previous measurements, with the most rapid decline occurring in the first week. This trend was similar to the reduction observed in scores. By the eighth week, which was 1 month after a treatment cycle, IgG levels had significantly rebounded, but still remained below baseline (Figure 5). Additionally, we monitored IgM, IgA, and albumin levels, none of which showed statistical declines.

Mean change in IgG levels. Error bars show standard error.
Changes in corticosteroid dosage
Seven patients (all AChR-GMG) were receiving maintenance therapy with prednisone at the time of inclusion. At the end of the 8-week follow-up, the average dose of the prednisone had decreased from 34.3 (SD 18.1) mg/d to 21.4 (SD 7.5) mg/d, representing an average reduction of 27.4% (SD 39.3%), and one patient reduced to ⩽10 mg/d. Only one patient (autoantibody unknown) increased the prednisone dosage due to inadequate response.
Overall safety
During the treatment and follow-up period, none of the patients experienced serious AEs. No patients suffered from myasthenic crisis or required rescue treatment due to worsening conditions. One patient did experience vomiting and diarrhea after the second infusion, but recovered shortly afterward and did not have any further symptoms. Other AEs, including respiratory infections, urinary tract infections, and infusion-related reactions, were not observed among the enrolled patients. All routine laboratory tests, electrocardiograms, and vital signs remained stable.
Discussion
It has been 1 year since efgartigimod was approved for use in China, few medical institutions that can effectively implement it in clinical practice, and reports on real-world studies of efgartigimod in Chinese patients are exceedingly scarce. In this study, we evaluated the efficacy and safety of efgartigimod in a real clinical setting involving a total of 25 MG patients with varying degrees of severity, minimizing bias while remaining close to the real clinical environment. Overall, we achieved results that were quite satisfactory.
For patients with GMG, efgartigimod demonstrated significant short-term efficacy. Most patients experienced clinically meaningful improvements after one treatment cycle, corroborating the conclusions from earlier clinical trials of efgartigimod.7,15 AChR-Ab is the most prevalent type of autoantibodies in MG patients, belonging to the IgG1 and IgG3 subtypes. 16 Based on the molecular characteristics of efgartigimod’s highest affinity for IgG1, 17 AChR-GMG has become its definitive indication, 18 and its efficacy in the AChR-GMG patients we included is unquestionable. We also enrolled two MuSK-MG patients with severe clinical manifestations. Both showed rapid improvement, with one experiencing symptoms worsening in eighth week but still responding well to a second treatment cycle in the subsequent follow-ups (data not shown). Notably, the other had undergone lymphoplasmapheresis for acute exacerbation 6 months ago, which yielded no obvious effect. Following this, he was treated with a cycle of efgartigimod immediately, resulting in marked improvement that persisted for several months. 19 This participation in the study was his second cycle. The experience of these two MuSK-MG patients provided evidence for the efficacy of efgartigimod in MuSK-MG. Despite the current lack of evidence-based medical evidence, efgartigimod appears to exhibit remarkable potential for rapid alleviation in MuSK-MG. Additionally, although our study did not involve MG patients with LRP4 antibody positive or even autoantibody negative, they had also shown a certain degree of positive feedback regarding efgartigimod in existing studies. 8 Further research is still needed to explore these rare types of MG more comprehensively. For patients with undetermined or antibody-negative types, if poor clinical efficacy is observed, we believe it may warrant reconsideration of the MG diagnosis.
In this study, we applied efgartigimod to the treatment of OMG patients, which has not been systematically reported before. Possibly it was due to the limitations of symptoms in OMG patients, resulting in a smaller improvement score compared to GMG patients. When patients do not achieve the expected effects, it is essential to promptly adjust the therapy, which may include corticosteroids, NSIST, or thymectomy,20 –22 in order to prevent progression to GMG. As in one of our patients (Figure 3(b)), symptoms improved after administering tacrolimus after week 8. Due to the limited sample size, the efficacy of efgartigimod in OMG warrants further investigation, and its potential role in preventing progression to GMG could become a major focus of research.
Phase III clinical study (ADAPT) 7 suggests that whether efgartigimod was combined with NSIST does not affect its efficacy, but this was based on maintaining the patient's original NSISTs regimen. In practice, however, clinicians often consider combination therapies to achieve as quick relief as possible. Our data suggest that the combination of efgartigimod and tacrolimus does not seem to yield better benefits. This may relate to molecular interactions between the drugs, as tacrolimus might influence the clearance of IgG by efgartigimod or accelerate the metabolism of efgartigimod. Furthermore, we cannot exclude the possibility of biases resulting from the small sample size, and challenges in ensuring strict medication adherence among patients. There is no relevant research currently, and the underlying mechanisms require further exploration. However, based on the preliminary conclusions, while the combination of efgartigimod and tacrolimus did not show a synergistic effect, it also did not increase the risk of infection-related AEs. We suggest that for patients with insufficient economic conditions who cannot opt for maintenance therapy with the new biological agent efgartigimod, tacrolimus can be selected for rapid transition.
As a humanized IgG1 Fc fragment, 23 efgartigimod exhibits a selective clearance effect on IgG, particularly IgG1, 17 thereby having no impact on albumin and other types of globulins. However, the clearance effect of efgartigimod on IgG is reversible, with a notable increase in IgG levels observable within 1 month after a treatment cycle.7,8 Our data confirmed this conclusion, but it appeared not to affect the maintenance of therapeutic efficacy in patients.
The potential multiple side effects of corticosteroids pose a major obstacle to the treatment of MG. As with many other biologics that have steroid-sparing effects, 24 during efgartigimod treatment, as the patient's condition improves, corticosteroid dosage can be appropriately reduced to minimize drug-related side effects. Our practice has confirmed this conclusion in the short term. The adverse reactions observed in our patients during treatment were very few, suggesting that efgartigimod may have a more pronounced safety profile in real-world settings compared to randomized controlled trials. Nevertheless, due to various uncertainties in real-world research and the relatively short follow-up time, we cannot rule out the risk of underreporting of adverse reactions.
The prominent short-term efficacy and safety of efgartigimod make it a promising candidate for becoming one of the preferred therapies for many MG patients in China. The extension study of efgartigimod (ADAPT+) has further demonstrated its tolerability for long-term use and the sustained benefits it provides to patients. 25 Previous studies often regarded the exacerbation of symptoms as a condition for initiating subsequent cycles of efgartigimod treatment.7,8 However, patients often expressed a willingness to start subsequent cycles as a preventive treatment even when their symptoms had not yet worsened, aiming to maintain therapeutic efficacy, and the timing for initiating subsequent cycles was not uniform. It suggests that the long-term maintenance treatment plan for efgartigimod seems to be more inclined toward individualization.
The advantage of this study lies in its systematic evaluation of the short-term efficacy and safety of efgartigimod in real-world settings for Chinese patients with myasthenia gravis (MG) of varying severity and antibody types. Additionally, this study provided some reference value for the combination of NSISTs and the reduction of corticosteroids during efgartigimod treatment. Thanks to its unique mechanism of action, efgartigimod not only treats MG, but research has also found that it may have potential effects on other IgG-mediated related diseases and even autoimmune-related comorbidities. 26 The further potential applications of efgartigimod are worthy of more exploration.
The limitations lie in that our efficacy assessment may be limited by the following factors. Differences in patient age and the presence of comorbidities may affect the overall evaluation of efficacy. 27 Most of the patients we included were late-onset individuals, which could have somewhat limited the objective evaluation of drug effects. Additionally, the lack of a placebo control may have introduced bias into our conclusions, particularly in this non-blind study where participants' expectations might have influenced outcome assessments. Moreover, this study was conducted at a single center, did not calculate the sample size in advance, and had a short follow-up period, which presented certain limitations on the reliability of the results, the generalization of efficacy, and the assessment of adverse reactions. In summary, future real-world studies should involve a larger number of cases with control groups and longer follow-up periods to further explore the effectiveness and general applicability of efgartigimod.
Conclusion
The treatment of MG has entered a new era, and the arrival of efgartigimod has opened up clinical applications for FcRn antagonists in MG. Currently, the application of efgartigimod in the Chinese population is still in the exploratory stage. Our study confirmed its short-term efficacy and safety in the Chinese MG patients based on real-world data, particularly verifying its high response rate in AChR-GMG patients. Compared to generalized MG, the efficacy in ocular MG is less pronounced and requires further clinical investigation. Adding tacrolimus during efgartigimod treatment did not seem to be a beneficial factor, but it seems to serve as a rapid transition to maintenance therapy without increasing the risk of infection. Furthermore, efgartigimod can serve as an alternative to corticosteroids, and long-term maintenance therapy should adhere to the principles of individualization. At present, FcRn antagonists represented by efgartigimod are gaining momentum and are expected to bring more benefits and hope to a large number of MG patients.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241311127 – Supplemental material for Application of efgartigimod in Chinese patients with myasthenia gravis: a single-center real-world prospective study
Supplemental material, sj-docx-1-tan-10.1177_17562864241311127 for Application of efgartigimod in Chinese patients with myasthenia gravis: a single-center real-world prospective study by Geke Zhu, Han Zhou, Wanying Wang, Yongbo Ma, Xiangtao Nie, Wenjing Qi, Lei Hao and Xiuming Guo in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
