Abstract
Background:
Very-late-onset myasthenia gravis (VLOMG) refers to myasthenia gravis (MG) with onset at age 65 or older. Current research on VLOMG prognosis remains limited, especially regarding factors influencing outcomes.
Objectives:
To identify the clinical factors that affect the short- and long-term prognosis of MG patients with an onset age ⩾65 years.
Design:
This was a single-center, retrospective cohort study of AChR-ab positive VLOMG patients, classified into two subgroups based on age of onset: sub-very-late-onset MG (S-VLOMG, onset age ⩾65 and <75 years), and super-late-onset MG (SLOMG, onset age ⩾75 years).
Methods:
A total of 93 patients were included, including 75 in the S-VLOMG group and 18 in the SLOMG group. Clinical, therapeutic, and prognosis data were reviewed, and the Cox regression model was used to identify factors influencing short- and long-term prognosis.
Results:
Patient characteristics were well balanced between the groups. Overall, 49.5% of patients achieved minimal symptom expression (MSE) within 6 months and 86% within 24 months. There was no significant difference between the groups in the proportion achieving MSE at 6 months (p = 0.635) or 24 months (p = 0.714). The median time to achieve MSE was also comparable between the S-VLOMG and SLOMG groups (199.0 days vs 280.5 days, p = 0.463). Low baseline MG-ADL score and steroid therapy were associated with better short-term prognosis (p = 0.007 and p = 0.002, respectively). For long-term prognosis, baseline bulbar and limb involvement, time to treatment initiation, and use of immunosuppressants were significant factors (p = 0.025, p = 0.004, p = 0.025, and p < 0.0001, respectively). There were no significant differences in side effects or drug withdrawal rates between two groups.
Conclusion:
This study demonstrated that AChR-ab positive VLOMG patients have a favorable prognosis and responded well to medication, with age and comorbidities showing no significant impact.
Plain language summary
The study aimed to understand how older adults with myasthenia gravis (a disease causing muscle weakness) recover in the short and long tewas to explore what factors impact their rm. Since research on people aged 65 and older with this condition is limited, the goal recovery and how treatment affects them.
The researchers reviewed medical data from 93 patients, divided into two age groups: 65-74 and 75+. They tracked how these patients responded to treatment over 6 months and 2 years. The study focused on factors like symptom severity, treatment timing, and medication type to see which helped or slowed recovery.
Nearly half of the patients showed significant improvement within 6 months, and over 85% improved within 2 years. Age and other health conditions (comorbidities) didn’t have a major effect on recovery. Instead, starting treatment early and using the right medication, particularly immunosuppressant therapy, were key factors for better outcomes.
The study shows that older patients with myasthenia gravis can recover well if they receive timely treatment. Doctors should focus on early intervention and appropriate medication to improve patient outcomes.
Introduction
Myasthenia gravis (MG) is an autoimmune disease characterized by the presence of autoantibodies targeting the neuromuscular junction and it manifests as a variable combination of fluctuating weakness in ocular, bulbar, trunk, limb, and respiratory muscles.1,2
MG can occur at any age, displaying with a bimodal distribution to the age of onset. There is an early peak in the 30s, primarily affecting females, and a late peak in the 60s, more commonly seen in males. 3 MG is typically categorized into early-onset MG (EOMG) and late-onset MG (LOMG), based on an age cut-off of 50 years.3–6 In recent years, due to the aging population and advances in diagnosis and treatment, there has been a notable increase in MG incidence among individuals over 65 years old. The distribution of age onset for MG has been significantly altered. 7 An epidemiological study in China identified the peak incidence of MG occurred in individuals aged 70–74 years. 8 Multiple studies have classified MG patients with onset age ⩾65 years as very-late-onset MG (VLOMG).9–12
The prognosis of VLOMG has varied across different studies. In a study by Cortés-Vicente, 424 out of 939 MG patients were VLOMG, characterized by anti-AChR antibodies and absence of thymoma. This study also suggested VLOMG patients may experience life-threatening events at onset, but they may achieve favorable long-term outcomes with fewer use of immunosuppressants (IS). 9 Meanwhile, Myasthenia Gravis Foundation of America (MGFA) Patient Registry point to a more positive prognosis that VLOMG patients showing better disease-specific quality of life and disease severity compared to younger individuals. 13 Vijayan et al. reported similar findings, noting that VLOMG patients responded well to treatment, with comorbidities having no significant impact on symptom severity or outcomes. 12 However, a Chinese study showed that VLOMG patients had a poorer prognosis, evidenced by a achieved minimal manifestations (MMS) rate, and a shorter maintenance time of MMS at the last follow-up and higher MG-related deaths compared to EOMG and LOMG. 14 These available studies suggest that patients with varying ages of onset exhibit distinct clinical features and immunological characteristics, but there is still a lack of systematic prognosis studies delving into subgroups within the VLOMG population.
The aim of our study was to categorize patients with VLOMG into more specific groups based on the onset age and compare the clinical outcomes between subgroups. To seek potential factors that may affect their prognosis.
Patients and method
Patients
In this single-center, retrospective cohort study, patients’ medical records and follow-up data since February 2017 to July 2024 were extracted from Xuanwu Hospital Capital Medical University Myasthenia Gravis Trial Database. The Ethics Committee of Xuanwu Hospital approved the study ([2022]082 and [2017]084), located at No. 45 Changchun Street, Beijing 100053, China. Participants provided written informed consent to participate in the study before taking part, and all data were fully de-identified. This manuscript adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Inclusion and exclusion criteria
The inclusion criteria included: (1) Patients were diagnosed with MG. The diagnosis of MG was based on fluctuating weakness symptoms and required evidence of at least one of the following: pharmacological, serologic, and electrophysiologic tests; (2) Patients’ onset age was ⩾65 years; (3) Seropositivity for autoantibodies against AChR. (4) Follow-up time no less than 2 years.
Patients were excluded if they met any of the following conditions: (1) Thymectomy or immunomodulatory therapies including steroids and nonsteroids IS were used before enrollment. (2) Baseline MG-activities of daily living (MG-ADL) score <2 or baseline Quantitative Myasthenia Gravis (QMG) score <3. (3) Insufficient baseline data or loss to follow-up.
Main content of observation
Clinical features were collected, including demographic characteristics (gender, age at onset); MGFA clinical classification at onset and at baseline: ocular MG (OMG), corresponding to Class I, defined as symptoms restricted to the ocular muscles for at least 1 month; generalized MG (GMG), corresponding to Classes II–V, defined as involving weakness of the limb, neck, bulbar, or respiratory muscles; symptoms at baseline; generalization, defined as patients with OMG at onset progress to GMG; baseline MG-ADL scores and QMG score; MG-ADL scores at 3rd, 6th, 9th, 12th, 18th, 24th month; frequency of myasthenic crisis, relapse and deaths; thymoma; complications; treatment, which were divided into four groups including pyridostigmine monotherapy, IS therapy (with or without pyridostigmine), steroid therapy (with or without pyridostigmine), and combination of steroids and IS (with or without pyridostigmine); diagnostic delay, defined as time from onset to diagnosis date; time before treatment, defined as the duration from disease onset to the he initiation of IS or steroids, if these were not used, defined as the duration from disease onset to the use of pyridostigmine; time from treatment to minimal symptom expression (MSE); side effects, including the proportion of drug side effects and the withdrawal rate.
Therapeutic regimens
All patients were initially treated with pyridostigmine. If symptom control was inadequate, steroids and/or immunosuppressants were added. Myasthenic crises during the disease course were managed with intravenous immunoglobulin or plasma exchange. In patients with steroid therapy, most of them were treated with oral prednisone, with only three cases requiring intravenous methylprednisolone due to the severity of their condition. Oral prednisone was initially administered at a dose of 1 mg/kg/day, then tapered by 5 mg every 2 weeks until 20 mg, followed by a further reduction of 5 mg every 1–3 months. For intravenous methylprednisolone, the initial dose was 500 mg/day, which was halved every 3 days until achieved a dose of 120 mg/day for 3 days. Treatment was subsequently transitioned to oral prednisone at a dose of 60 mg/day, with further tapering to a maintenance dose based on clinical response. When oral prednisone was combined with IS, the initial dose was typically 20 mg/day or 0.5 mg/kg/day.
Tacrolimus was initiated at a dose of 2 mg/day; if well tolerated, the dose was increased to 3–4 mg/day after 2–3 weeks, with a maximum daily dose not exceeding 5 mg. Azathioprine was started at 50 mg/day or 1 mg/kg/day and increased 50 mg/day every 2–3 weeks, reaching a target dose of 3 mg/kg/day over 2–3 months. Mycophenolate Mofetil was started at 0.5–1.0 g/day, with escalation to 1.0–1.5 g/day. Methotrexate was initiated at a dose of 10 mg/week, with gradual escalation to 20 mg/week. Novel biologics such as efgartigimod and eculizumab were not yet available in China during follow-up period. Additionally, no patients were identified as refractory MG, and thus none required rituximab treatment.
Outcome and follow-up assessments
The main outcome was defined as reaching MSE, defined as MG-ADL score 0 or 1. 15 Short-term prognosis was defined as achieving MSE within 6 months, while long-term prognosis was defined as achieving MSE within 24 months. Follow-up assessments were measured every 3 months during the first year and every 6 months thereafter. Every assessment must include clinical status and MG-ADL scores. If patients could not attend the follow-up visits, telephone follow-ups were performed. All patients were followed-up at least 2 years.
Statistical analysis
Continuous variables with a normal distribution were presented as mean ± SD, while continuous variables with non-normal distribution were presented as median and interquartile range (IQR). Differences in characteristics between two subgroups were evaluated using the Mann–Whitney U Test for continuous variables and Fisher’s exact test for categorical variables. The Cox regression model was performed by the time from medication to MSE for all patients. Variables with p < 0.2 in univariate analysis, along with clinically relevant variables identified in previous studies, were included in the multivariate analysis. A stepwise selection method was used to build the multivariate Cox regression model. Kaplan–Meier curves stratified by age were plotted to illustrate the cumulative incidence of reaching MSE. X-tile (Version 3.6.1) was used to determine cut-off value for MG-ADL and time before treatment data. Sample size calculations were performed using the PASS 2021 (v21.0.3) with Logrank Tests. Based on a 2:1 group allocation ratio (N2/N1 = 2), anticipated survival rates of 0.8 in the S-VLOMG group and 0.5 in the SLOMG group, a power of 90%, and a significance level (α) of 0.05, the required sample size was estimated to be 58 (S-VLOMG: 39, SLOMG: 19). All data were analyzed using SPSS (Version 26, IBM, Armonk, NY, USA) and R (Version 4.1.1).
Results
Demographics
A total of 188 patients met inclusion criteria were diagnosed prior to July 2024. However, 95 of them were excluded from the study due to the use of steroids or IS before enrollment (n = 68), thymectomy before enrollment (n = 14), loss to follow-up (n = 1) or baseline MG-ADL <2 or baseline QMG <3 (n = 12). Finally, 93 patients were included in this study, with 75 classified as S-VLOMG and 18 as SLOMG (Figure 1).

Flowchart of patients included in the study.
Basic and disease characteristics
Patient characteristics were well balanced between the S-VLOMG and SLOMG groups, including sex distribution, comorbidities, MGFA classification at onset and baseline, symptoms at baseline, MG-ADL and QMG scores, thymoma, and treatment approaches, etc., showed no statistically significant differences between the two age groups (p > 0.05 for all comparisons; Table 1).
Baseline demographic and clinical characteristics.

MG-ADL, myasthenia gravis-activities of daily living; GMG, generalized myasthenia gravis; IS, immunosuppressant; IQR, interquartile range; MGFA, Myasthenia Gravis Foundation of America; MSE, minimal symptom expression; OMG, ocular myasthenia gravis; SLOMG, super-late-onset myasthenia gravis; S-VLOMG, sub-very-late-onset myasthenia gravis. Bold values in table indicate p<0.05.
Generalization rate was calculated during the whole follow-up period.
Bulbar symptom including dysarthria and dysphagia.
Prognosis
During the follow-up period, both S-VLOMG and SLOMG groups demonstrated substantial improvements in MG-ADL scores. Although the S-VLOMG group showed a more rapid decrease and ultimately achieved lower scores compared to the SLOMG group, a statistically significant difference between the groups was only observed at 24 months (p = 0.017), with the S-VLOMG group achieving lower MG-ADL scores. At all other time points, the differences between the groups were not statistically significant (Table 2).
Patients baseline MG-ADL/QMG score, MG-ADL changes during follow-up period, and prognosis.

IQR, interquartile range; MG-ADL, myasthenia gravis-activities of daily living; MSE, minimal symptom expression; QMG, quantitative myasthenia gravis score; SLOMG, super-late-onset myasthenia gravis; S-VLOMG, sub-very-late-onset myasthenia gravis. Bold values in table indicate p<0.05.
A significant proportion of patients in both groups achieved MSE within the designated time frames. By 6 months, 49.5% of all patients had reached MSE, with 50.7% in the S-VLOMG group and 44.4% in the SLOMG group. At 12 months, 72.0% had achieved MSE, including 73.3% in the S-VLOMG group and 66.7% in the SLOMG group. By 24 months, 86.0% of patients had reached MSE, with 86.7% in the S-VLOMG group and 83.3% in the SLOMG group. None of these differences were statistically significant (Table 2).
The time from treatment initiation to achieving MSE was comparable between the two groups, with a median of 199.0 days (IQR 102.0–456.75) in the S-VLOMG group and 280.5 days (IQR 107.5–545.25) in the SLOMG group(p = 0.463). Kaplan–Meier survival analysis also showed no statistically significant difference in the probability of achieving MSE between the two groups at either 6 months (p = 0.606; Figure 2(a)) or 24 months (p = 0.535; Figure 2(b)). Additionally, the time before treatment initiation was comparable between the groups, with a median of 112.0 days (IQR 54.0–239.5) for S-VLOMG and 93.0 days (IQR 59.5–167.25) for SLOMG (p = 0.475; Table 2).

Kaplan–Meier curves stratified by age. (a) Shows the probability of reaching minimal symptom expression (MSE) within 6 months, and (b) shows the probability of reaching MSE within 24 months. The curves compare patients with sub-very-late-onset myasthenia gravis (S-VLOMG) and super-late-onset myasthenia gravis (SLOMG). The Log-rank tests indicate no statistically significant difference between the groups at either time point (p = 0.606 for (a), p = 0.535 for (b)).
Factors correlative with prognosis
The Cox regression model was applied to assess factors influencing the short-term and long-term prognosis. For variables included in the multivariate Cox regression model, a collinearity diagnostic was conducted, confirming no significant collinearity among them (Table 3).
Variables associated with time to MSE in all patients predicted by Cox regression model.

Variables were included in multivariate analyses if p < 0.20 in univariate analyses or have clinical relevance. Bold values in table: p<0.2 (univariate), p<0.05 (multivariate).
HR, hazard ratio; IS, immunosuppressant; MG-ADL, myasthenia gravis-activities of daily living; MGFA, Myasthenia Gravis Foundation of America; OMG, ocular myasthenia gravis; QMG, quantitative myasthenia gravis score.
Short-term prognosis
The multivariate analysis identified the baseline MG-ADL score was a significant predictor (HR = 0.842, 95% CI = 0.744–0.954, p = 0.007). Additionally, patients who received steroid therapy was a protective factor of achieving MSE compared to those without steroids or IS (HR = 4.474, 95% CI = 1.712–11.696, p = 0.002). Furthermore, we determined the cut-off value for the MG-ADL score using X-tile software, and data were shown in (Figure 3(a)), which identified a cut-off value of 4. Patients with a baseline MG-ADL score ⩽4 were more likely to achieve MSE in 6 months.
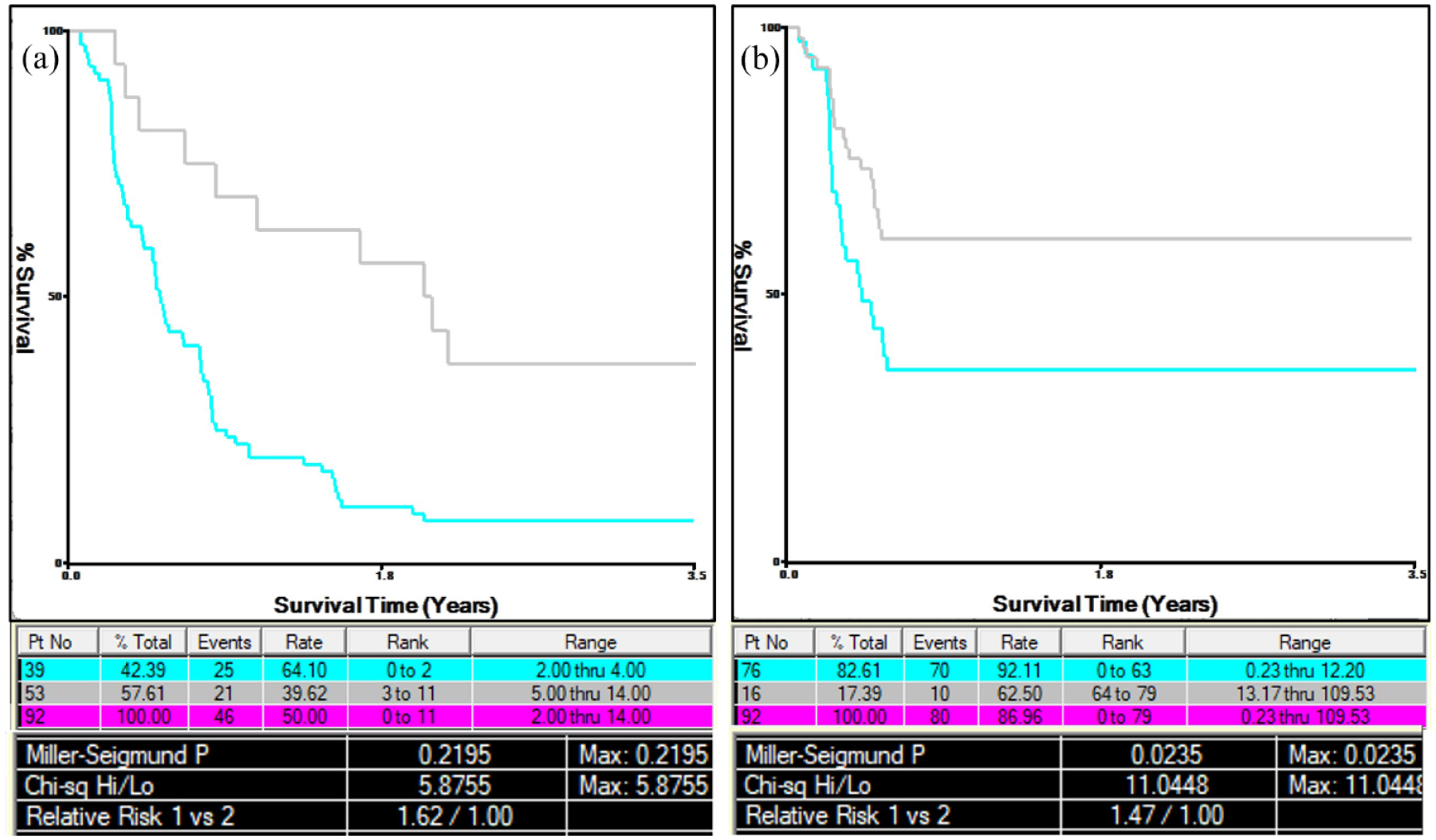
Cut-off value determination for MG-ADL score and time before treatment. (a) Kaplan–Meier survival curves used to determine the cut-off value for MG-ADL score. A cut-off value of 4 was identified. (b) Kaplan–Meier survival curves used to determine the cut-off value for time before treatment. The analysis identified a cut-off value of 12 months.
Long-term prognosis
Multivariate analysis showed that baseline bulbar and limb involvement were risk factors (HR = 0.515, 95% CI = 0.289–0.919, p = 0.025; HR = 0.335, 95% CI = 0.158–0.712, p = 0.004, respectively) for 2 years prognosis. Similarly, time before treatment was inversely associated with achieving MSE (HR = 0.968, 95% CI = 0.942–0.996, p = 0.025). Additionally, use steroids alone or in combination with IS were found to significantly promote the achievement of MSE (HR = 4.135, 95% CI = 1.826–9.362, p < 0.0001; HR = 2.382, 95% CI = 1.091–5.199, p = 0.029). We also determined the cut-off value for time before treatment using X-tile software and the data were shown in (Figure 3(b)), which identified a cut-off value of 12 months. Patients who began treatment within 12 months of disease onset were more likely to achieve MSE in 24 months.
Side effects
Table 4 shows the comparison of side effects and drug withdrawal rates between the two groups. A total of 46.2% of patients experienced side effects, with 44.0% in the S-VLOMG group and 55.6% in the SLOMG group (p = 0.377). Pyridostigmine-related side effects were reported in 36.6% of patients, with a higher incidence in the SLOMG group (50.0%) compared to the S-VLOMG group (33.3%) (p = 0.187). Steroid-related side effects were reported only in the S-VLOMG group (22.9%), while no cases were recorded in the SLOMG group (p = 0.232). IS-related side effects were similar between the two groups (p = 0.835). Drug withdrawal occurred in 27.9% of patients who experienced side effects, with a higher rate in the S-VLOMG group (33.3%) compared to the SLOMG group (10.0%) (p = 0.150). Overall, no statistically significant differences were observed between the two groups regarding side effects or drug withdrawal rates.
Side effects and drug withdrawal in two groups.
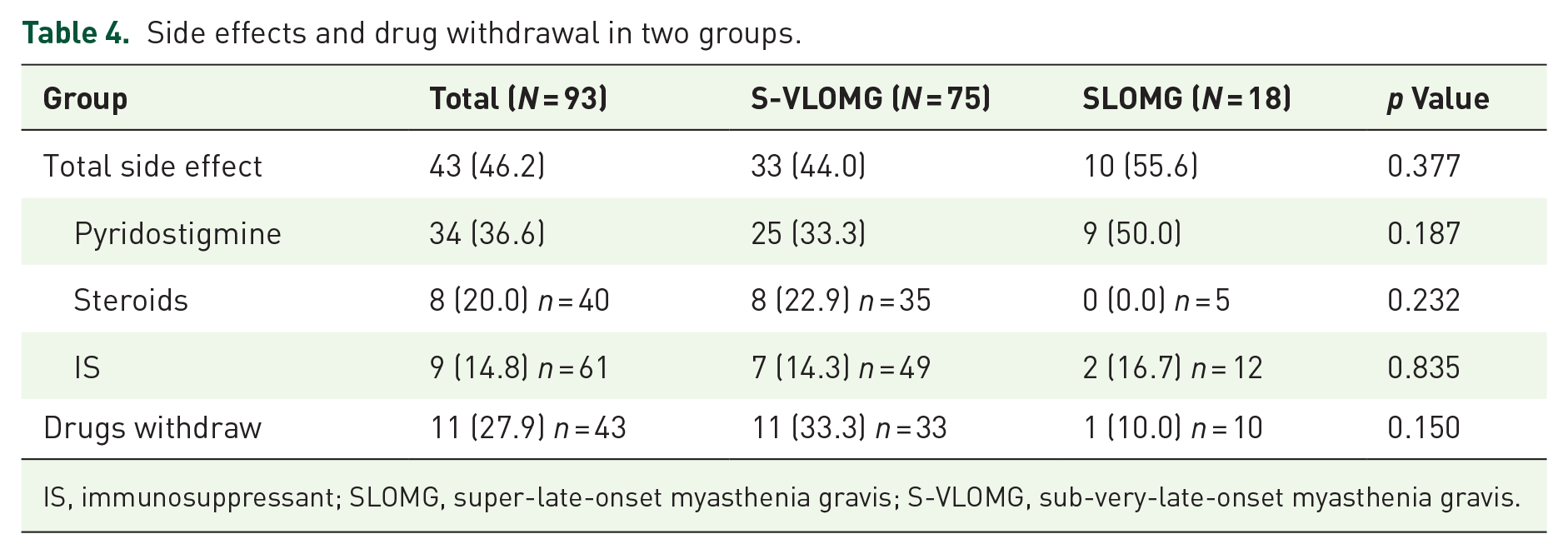
IS, immunosuppressant; SLOMG, super-late-onset myasthenia gravis; S-VLOMG, sub-very-late-onset myasthenia gravis.
Discussion
We found that AChR-ab positive VLOMG patients have favorable short-term and long-term outcomes, with 49.5% achieved MSE in 6 months and 86% in 2 years. Age and comorbidities did not significantly influence the outcomes. Additionally, there was no increasing trend in drug-related side effects.
Recent reports have indicated a rising incidence of elderly MG patients. An epidemiological survey in China indicated an incidence of MG was 0.68/100,000 person-years, with the highest incidence observed in the 70–74 years age group. 8 However, prognostic studies on elderly especially in VLOMG are limited and somewhat heterogeneous. For example, the studies by Cortés-Vicente et al., Gutter et al., and Vijayan et al. indicated a good prognosis for VLOMG patients, while Tang’s study showed a poor prognosis.9,12–14 This may be attributed to several reasons: first, earlier studies have used different cut-off ages to define the VLOMG population; second, many of these studies assessed prognosis based on the time of the last follow-up, leading to significant discrepancies in observation periods and resulting in inconsistent conclusions across different research centers. Furthermore, current research primarily compares the prognosis among EOMG, LOMG, and VLOMG, without delving into subgroups within the VLOMG population. 16 To address this research gap, we followed the approach of previous studies that categorized elderly MG patients in 10-year age increments, and based on this method, we defined SLOMG using a 75-year cut-off value. 7 Our study findings suggest that patients in the two age groups have comparable rates of achieving MSE and experiencing adverse effects. However, at 24 months, the median MG-ADL scores were higher in the SLOMG group, whereas no differences were observed at earlier follow-up points. This may be attributed to two possible reasons. First, the relatively small sample size could have caused the score changes from a few patients to significantly impact the median scores. Second, immunosenescence during prolonged treatment durations may lead to reduced drug tolerance and suboptimal therapeutic efficacy in elder patients. 17
Prognostic factors for VLOMG patients showed significant differences between short-term and long-term outcomes. Patients with milder severity disease (MG-ADL score ⩽4) and steroid therapy were more likely to achieve MSE within 6 months. Steroids are considered as one of the most effective medication in MG, and at a dose of 1 mg/kg/day can generally induce remission after a few weeks. 18 However, our study found that in the short-term period, IS, whether used alone or in combination with steroids, did not significantly affect the rate of achieving MSE. This may be due to the slower effect of IS and the lower dose of steroids used in combination. We also found an interesting observation: there was no significant difference between steroids combined with IS and using only pyridostigmine (no steroids or IS). This may be because patients using only pyridostigmine had milder disease onset and shorter disease duration, and they did not receive additional medications during the two-year follow-up period. The second reason may be that the results were limited by the small sample size.
For long-term prognosis, the absence of limb and bulbar involvement at baseline were independent protective factors, indicating that elderly patients with the OMG subtype had better outcomes. OMG is considered as a mild subtype, and several studies have described its favorable prognosis. A Japanese study using Cox regression indicated that OMG patients were more likely to benefit from corticosteroids. 19 Another study also found that isolated ocular involvement or low baseline QMG scores were beneficial for achieving MMS. 20 In our study, the 2-year generalization rate of VLOMG was 63.8%, with no statistically significant difference observed between the S-VLOMG and SLOMG groups. This rate was comparable to that reported in previous Japanese VLOMG study but was significantly higher than the rates reported in EOMG and LOMG populations.9,21 This discrepancy may be attributed to the lower doses of immunosuppressive agents administered to elderly patients, which could possibly indicate a higher generalization rate.
The use of steroids, IS, and a shorter disease duration before treatment (⩽12 months) were also independent protective factors for long-term prognosis in our study. Clinical guidelines for MG in Japan have also recommended early immunotherapy intervention. The early, fast-acting treatment strategy enables more frequent and earlier attainment of the initial goal than other strategies. 22 Protracted MG can lead to damage of the neuromuscular junction, including loss of synaptic folds, widened clefts, and relocation of the nerve terminal, which may compromise the effectiveness of therapy. 23 Such structural changes highlight the importance and effectiveness of early immunotherapy intervention.
Managing the side effects of long-term immunotherapy in elderly MG patients remains a critical concern. Previous studies indicated an increased risk of corticosteroid side effects in elderly patients compared to younger patients.24,25 However, our study did not find an increase in side effects with age in the VLOMG population, this might be due to a lower proportion of using steroids (64.9%) and IS (64.5%) compared to other VLOMG studies (over 75%).9,12,14 Given the discrepancy between our findings and those of previous studies, additional research is warranted to corroborate the lack of increased side effects in the VLOMG population. A team developed an adverse event unit (AEU) to quantify and compare the burden of side effects among MG patients. They found that higher adverse event scores correlated with poorer quality of life, and patients treated with more than three long-term medications (including pyridostigmine) had significantly lower quality of life due to side effects. 26 Therefore, we generally recommend that elderly patients receive potent and rapid immunosuppressive therapy early in the disease course to minimize the total drug dose and occurrence of side effects. The consistent outcome in the S-VLOMG and SLOMG groups underscores the importance of maintaining ongoing management and intervention strategies to improve the quality of life for all MG patients.
This study has some limitations. First, it was a retrospective and single-center study, which may introduce selection bias. However, our research center is the largest in northern China, with a participant base drawn from across the country, offering a broad and comprehensive database that likely minimizes major selection bias. Data collection utilized standardized electronic forms, and patient assessments were conducted by experienced neurologists. Second, the relatively small sample size and the inclusion of only immunotherapy-naïve patients at baseline may impact the robustness of the conclusions. The immunotherapy-naïve status could have minimized the potential impact of immunosenescence on treatment outcomes in the early stages of therapy. Further investigation with longer follow-up periods and a broader patient cohort, particularly focusing on long-term immunotherapy response in elderly MG populations is needed to confirm these findings. Third, we only included AChR-ab positive patients with potentially missing relevant information on MuSK-ab positive MG. Fourth, due to the COVID-19 pandemic, many follow-up visits were conducted remotely, resulting in missing QMG score data. Consequently, we used the MG-ADL scale for prognostic evaluation, lacking dynamic QMG score assessments. Additionally, data from the Myasthenia Gravis-Quality of Life 15-Item Scale were not collected. Finally, the study did not compare patients with EOMG and LOMG, nor did it account for the total drug dose in the analysis. Due to limitations in available treatment options, novel biologics were not included in the therapeutic regimen.
Conclusion
In this study, AChR-ab positive VLOMG patients showed favorable short-term and long-term outcomes, with age and comorbidities might not have significant impact. Factors that influence short-term prognosis include baseline MG-ADL and steroid therapy. Factors associated with long-term prognosis include initial bulbar and limb involvement, time to treatment initiation, and the use of immunosuppressants.
