Abstract
Background:
Minimal manifestation status (MMS) is an important landmark in the treatment of myasthenia gravis (MG), and predictors of MMS induction have rarely been identified in previous studies.
Objective:
The objective of this study is to evaluate the clinical factors associated with MMS induction among patients with MG.
Design:
This two-step retrospective cohort study with a single center investigated the factors that may be associated with MMS induction and retested these predictors in a test cohort.
Methods:
A total of 388 diagnosed MG patients who visited Xiangya Hospital between 1 July 2015 and 1 July 2019 were involved. We performed detailed chart reviews and recorded all cases achieving MMS. Demographics and clinical characteristics were also collected and their relationships to achieving MMS were investigated.
Results:
MMS was achieved in 124 patients (50.2%), and the median time to achieve MMS was 26 months. Several factors were found to be associated with MMS induction in exploring cohort, including muscle-specific tyrosine-protein kinase receptor (MuSK) antibody positivity (adjusted hazard ratio, HR = 4.333, 95% confidence interval, CI: 1.862–10.082), isolated ocular involvement (adjusted HR = 1.95, 95% CI: 1.284–2.961), and low baseline quantitative myasthenia gravis score (QMG score; adjusted HR = 2.022, 95% CI: 1.086–3.764). These factors were then retested in the test cohort. Isolated ocular involvement or low baseline QMG scores were factors found to be beneficial for MMS induction were confirmed.
Conclusion:
Isolated ocular involvement and low baseline QMG score are predictors of MMS induction in MG patients.
Background
Myasthenia gravis (MG) is a T-cell dependent, antibody-mediated, neuromuscular junction–involved autoimmune disease. Pathogenic antibodies impair postsynaptic structures through complement activation, promoting anti-acetylcholine receptor (AChR) internalization, blocking AChR, or disturbing the agrin/low-density lipoprotein-receptor-related protein 4 (LRP4)/muscle-specific tyrosine-protein kinase receptor (MuSK)/Dok7/rapsyn pathway. 1 These processes impair neuromuscular junction transmission, characterized by fluctuating muscle weakness and fatigability.2,3 With the development of intensive care and immunotherapy, the mortality of MG has significantly decreased, and many patients can achieve minimal manifestation status (MMS) or better status. 4
MMS is an important milestone in the treatment of MG; once it is achieved, the target of immunotherapy is to maintain MMS for a period of 6–12 months and then progressively retreat to avoid adverse effects and it is recommended as a therapeutic goal in MG. Therefore, for MG patients, induction of MMS as soon as possible would be a good start for treatment. Furthermore, MG is a heterogeneous disease.5–7 Patients with different antibodies have different clinical presentations, and thymic abnormalities can be diverse. Therapy for MG patients should be tailored and prognostic markers would be helpful in treatment decisions and predicting clinical outcome. 7 However, previous studies to determine prognostic factors used various measurements for the outcome, and MMS was rarely adopted.4,5 In addition, these studies focused on long-term outcome rather than a short-term one, namely, achieving MMS. Thus, to identify clinical factors associated with MMS induction, we retrospectively collected and analyzed MG cases from the Outpatient Department of Neurology in Xiangya Hospital.
Subjects and methods
This study was a two-step retrospective cohort study of patients with MG. In the first step, some MG patients constituted an exploring cohort, and their data were analyzed to identify the factors that may affect MMS induction. Subsequently, others constituted a test cohort, and MMS was defined as the primary outcome. The identified factors in the first step were regarded as exposure and further retesting was done for the test cohort.
Subjects and outcomes
MG patients diagnosed at the Neurology Department, Xiangya Hospital, during the period from 1 July 2015 to 30 June 2018, were included in the exploring cohort, and the MG patients’ data from 1 July 2018 to 30 June 2019 were retrieved as a test cohort to verify the factors that were screened out in the first step. Data regarding all suspected patients were extracted from the outpatient clinic system of Xiangya Hospital by searching the diagnostic items, such as ‘Myasthenia Gravis’, or ‘ocular Myasthenia Gravis’, or ‘generalized Myasthenia Gravis’, or ‘MG’, or ‘suspected Myasthenia Gravis’. The outpatient identity numbers were used to identify the individuals. The clinical data of these cases were reviewed. The diagnosis of MG was confirmed based on the following criteria: (1) fluctuating weakness and fatigability in skeletal or extraocular muscles, (2) positive results in serum test, and (3) positive neostigmine test or amplitude pathological decrement after repetitive nerve stimulation. Diagnosed patients should present criteria 2 and/or 3, in addition to criterion 1. The exclusion criteria included (1) patients who were younger than 15 years; (2) patients had an onset more than 5 years prior to visiting our center because of the less accurate recall of medication from these patients, and this study primarily focused on MMS induction in newly onset patients; (3) patients accompanied with neoplasms (except thymoma); and (4) patients with incomplete baseline records or those who could not provide treatment information before or reject immunotherapy.
Demographics and clinical data were extracted from medical records. Serum tests were performed in Daan Clinical Laboratory Central (Guangzhou, China) using a radioimmunoprecipitation assay (MuSK antibody) or enzyme-linked immunosorbent assay (AChR antibody). The quantitative myasthenia gravis score (QMG score) was performed by specialized physicians after discontinuation of the cholinesterase inhibitor for at least 12 h. Thymus data for patients were derived from imaging scans [contrast-enhanced computed tomography (CT) scan] or histopathology. Ocular myasthenia gravis (OMG; isolated ocular involvement) was defined as a weakness that did not transform into generalized myasthenia gravis (GMG) for more than 2 years after onset. Patients with insufficient follow-up time, but with isolated ocular involvement at present, were excluded due to possible misclassification. The MMS was defined as the status in which the patient had no symptoms or functional limitations from MG but had some weakness upon examination of certain muscles. 8
Exposure to a certain medicine was defined as taking the drug more than 3 months with a sufficient dose: prednisone (20–60 mg per day or equivalent), tacrolimus (TAC; target blood concentration: 4.8–10 pg/ml), mycophenolate mofetil (MMF; 1–2 g per day), and azathioprine (AZA; 100–200 mg per day). For patients who started immunotherapy before visiting our center, previous therapeutic regimens were carefully asked about. Intravenous immunoglobulin (IVIG) and plasma exchange (PE) were used to rescue patients with crises or impending crises. Follow-up started when patients received immunotherapy, up to 30 months or the time achieving MMS. MG patients usually revisit our center every 1–3 months for follow-up and evaluation. They were asked about their symptoms and recorded in their outpatient charts. For the few patients who accepted the evaluation but symptoms were not recorded, we connected with the patients and confirmed their symptom condition if their clinical scales met the PASS criteria (QMG⩽7, ALD⩽2, MGC⩽3, MG-QOL15⩽8). 9 The time for which immunotherapy was received before visiting our center was also calculated. For patients who did not come back for follow-up, their last postintervention statuses were recorded and included in the survival analysis. However, the cases who only had their first record were excluded.
Statistical analysis
Descriptive statistics, including mean and standard deviation (SD), percentages, medians, and interquartile ranges, were used to describe demographic and clinical characteristics. Student’s t test or Mann–Whitney U test was used for quantitative variables, while the chi-square test or Fisher’s exact test was used for categorical variables when appropriate.
In the exploring cohort, a multivariate analysis method, the Cox proportional hazards model, was used to identify the independent factors related to MMS achievement. Univariate survival analysis was performed, and factors with a p value <0.1 or considered clinically relevant were included in the Cox proportional hazard models. The age of onset (<50 and ⩾50 years), antibody status, baseline QMG scores (⩽16 points regarded as low QMG score group and >16 points regarded as high QMG score group),10,11 purely ocular involvement (OMG), glucocorticoids (GCS) exposure, TAC exposure, AZA exposure, needing IVIG/PE due to crisis, and presence of thymoma or thymectomy were included in the Cox proportional hazards model.
Propensity score matching was used in the test cohort to balance the baseline in the exposure and non-exposure groups. All main characteristics such as sex, age of onset, antibody status, immunotherapy (including GCS exposure, TAC exposure, MMF exposure, and AZA exposure), IVIG or PE due to crisis, thymoma, and thymectomy were considered as predictors. The matching ratio was 1:1, and the matching algorithm was nearest neighbor matching. The caliper was set to 0.15. Kaplan–Meier curves were used to confirm whether the factors filtered before could affect MMS achievement in the matched groups. Subjects who were lost to follow-up, who died, or did not achieve MMS throughout the study period were censored. A p value <0.05 was considered meaningful, and statistical analyses were performed using SPSS 22 (IBM, Armonk, NY, USA).
Results
Clinical characteristics of MG patients included in the exploring cohort
MG patients who visited the outpatient department of Xiangya Hospital from 1 July 2015 to 30 June 2018 were included in the exploring cohort. A total of 388 MG patients aged ⩾15 years and onset within 5 years were found. Sixty patients with incomplete records and 61 patients having only their first records were excluded. Nine patients refusing immunotherapy and six cases without details of treatment information before were abandoned. Two patients presenting with ocular involvement but an onset less than 2 years ruled out (because they could not be classified accurately). And three patients accompanied with neoplasm were also excluded. A total of 247 MG patients were available at the last visit (Figure 1(a)). Among them, 24 (9.7%) patients received immunotherapy before visiting our center. The median follow-up time was 20 months (interquartile range, 8–27 months). MMS was achieved in 124 patients (50.2%), and the median time to achieve MMS was 26 months.

Flowchart detailing the process of patient inclusion in exploring cohort: (a) and test corhort (b).
The study population consisted of more females (55.1%) than males (44.9%). The median age of onset was 44 years (interquartile range, 29–54 years). AChR antibody was the predominant subtype, with up to 202 cases (81.8%), while anti-MuSK antibody was rare; only 9 cases (3.6%) and 7 of them achieved MMS. Thirty-six patients (14.6%) were double-negative. Sixty-nine patients (27.9%) had isolated ocular involvement. Patients who achieved MMS had lower baseline QMG scores than those who did not achieve MMS (8 points versus 12 points). Thymoma was detected in 66 patients (26.7%). Fifty-one patients (20.6%) underwent thymectomy. Fifty patients (20.2%) were accompanied with other autoimmune diseases. Up to 44 patients (17.8%, 12 in MMS group and 32 in non-MMS group) needed PE or IVIG due to crisis or impending crisis, and more patients in non-MMS group needed emergency rescue therapy (MMS group 9.7% versus non-MMS group 26%). GCS (88.2%) and TAC (38.9%) were the most common drugs for therapy, and TAC exposure was more frequently found in non-MMS patients (29% versus 48.8%). The details of the clinical and demographic characteristics of the study population are presented (Table 1).
Clinical characteristics of MG patients included in exploring cohort.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; GCS, glucocorticoids; GMG, generalized myasthenia gravis; IVIG, intravenous immunoglobulin; MG, myasthenia gravis; MMF, mycophenolate mofetil; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; OMG, ocular myasthenia gravis; PE, plasma exchange; QMG score, quantitative myasthenia gravis score; TAC, tacrolimus.
Nine patients lost the record of GCS dose in MMS group.
Eleven patients lost the record of GCS dose in non-MMS group.
p < 0.05.
Identifying independent clinical factors associated with MMS achievement
Age of onset, antibody status, OMG, baseline QMG score, thymoma and thymectomy, GCS exposure, TAC exposure, AZA exposure, and need for PE or IVIG were candidate factors associated with MMS induction and their Kaplan–Meier curves were displayed (Figure 2). Table 2 shows the odds of achieving MMS for each variate. In the univariate analysis, MuSK antibody (hazard ratio, HR = 2.851, 95% confidence interval, CI: 1.32–6.16), double-negative (HR = 1.626, 95% CI: 1.03–2.566), isolated ocular involvement (HR = 2.541, 95% CI: 1.779–3.63), low baseline QMG score (HR = 2.456, 95% CI: 1.471–4.099), and accepted thymectomy without thymoma (HR = 7.487, 95% CI: 1.798–31.101) were favorable for MMS induction. The analysis of the effect of thymectomy in non-thymomatous MG was not adopted due to few reported cases in this group (two cases). However, TAC exposure (HR = 0.513, 95% CI: 0.347–0.76) and needing PE/IVIG (HR = 0.444, 95% CI: 0.245–0.805) were disadvantage factors for MMS.

Kaplan–Meier curves for factors involved in exploring cohort.
Involved factors may be associated with MMS induction.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; CI, confidence interval; GCS, glucocorticoids; GMG, generalized myasthenia gravis; HR, hazard ratio; IVIG, intravenous immunoglobulin; MG, myasthenia gravis; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; OMG, ocular myasthenia gravis; PE, plasma exchange; QMG score, quantitative myasthenia gravis score; TAC, tacrolimus.
The Cox proportional hazards model was used to adjust the variables and identify independent predictors. After adjustment, the independent predictors were MuSK antibody-positive (adjusted HR = 4.333, 95% CI: 1.862–10.082), pure ocular involvement (adjusted HR = 1.95, 95% CI: 1.284–2.961), and low baseline QMG score (adjusted HR = 2.022, 95% CI: 1.086–3.764), as shown in Figure 3. TAC exposure and need for PE/IVIG were not independent predictors of MMS achievement.
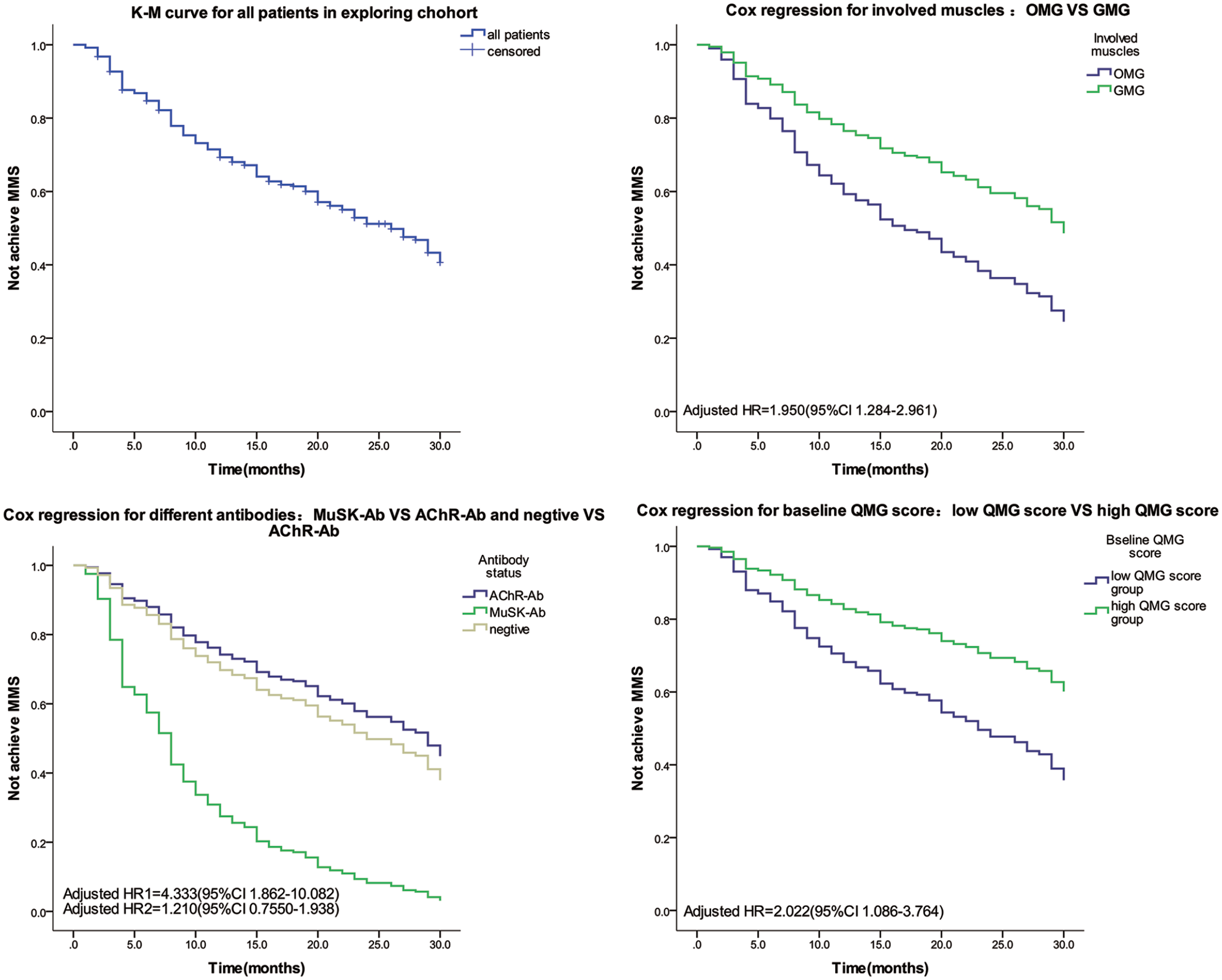
Kaplan–Meier curves for all patients in exploring cohort and Cox regression for independent factors favoring MMS induction after adjustment.
In short, MuSK antibody positivity, purely ocular involvement, and low baseline QMG score were potential predictors of MMS induction.
Confirm the independent predictors in a test cohort
To confirm the selected independent predictors in the first section, a test cohort was constructed. The MG patients who visited our center from 1 July 2018 to 31 July 2019, comprised the test cohort. The inclusion and exclusion criteria were the same as those in the first section. Up to 133 MG patients were included in the test cohort (Figure 1(b)). The exclusion criteria included incomplete records (31 patients), refusal of immunotherapy (4 patients), neoplasm (3 patients), lack of detailed prior treatment information (2 patients), and availability of only the first record (21 patients). Two patients with strict ocular involvement were also excluded because they had an onset of less than 2 years. Among the included cases, 106 patients (79.7%) were AChR antibody-positive, and 4 patients (3%) were MuSK antibody-positive. Twenty-eight patients (21%) had thymomas. The median follow-up time was 12 months, and 74 patients achieved MMS.
Screened predictors, isolated ocular involvement, and a low baseline QMG score were regarded as exposure. The test cohort was divided into an exposure group and a control group according to these predictors; demographic and clinical characteristics of the two groups were displayed (Tables 3 and 4). The MuSK antibody was not treated as exposure due to the few cases reported. Only four patients were MuSK antibody-positive. Considering confounding from other non-exposure variables, propensity score matching was used to balance the non-exposure variables. The propensity score matching model included sex, age of onset, antibody, use of drugs, interval between onset and treatment, thymoma and thymectomy, and PE/IVIG. This study obtained a dataset of 62 participants (when OMG was regarded as exposure) and a matched dataset of 40 participants (when low baseline QMG score was regarded as exposure), as shown in Tables 5 and 6. Statistical tests to evaluate the differences between the two groups revealed that the variables considered were not significantly different.
Clinical characteristics of OMG group and GMG group in test cohort.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; GCS, glucocorticoids; GMG, generalized myasthenia gravis; IVIG, intravenous immunoglobulin; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; OMG, ocular myasthenia gravis; PE, plasma exchange; MMF, mycophenolate mofetil; TAC, tacrolimus.
Clinical characteristics of low QMG group and high QMG group in test cohort.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; GCS, glucocorticoids; IVIG, intravenous immunoglobulin; MMF, mycophenolate mofetil; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; PE, plasma exchange; QMG score, quantitative myasthenia gravis score; TAC, tacrolimus.
Clinical characteristics of OMG and GMG after matching.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; GCS, glucocorticoids; GMG, generalized myasthenia gravis; IVIG, intravenous immunoglobulin; MMF, mycophenolate mofetil; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; OMG, ocular myasthenia gravis; PE, plasma exchange; TAC, tacrolimus.
Log-rank test for Kaplan–Meier curves.
Clinical characteristics of low QMG group and high QMG group after matching.

AChR-Ab, acetylcholine receptor antibody; AZA, azathioprine; GCS, glucocorticoids; IVIG, intravenous immunoglobulin; MMF, mycophenolate mofetil; MMS, minimal manifestation status; MuSK-Ab, muscle-specific tyrosine-protein kinase receptor antibody; PE, plasma exchange; QMG score, quantitative myasthenia gravis score; TAC, tacrolimus.
Log-rank test for Kaplan–Meier curves.
In the matched dataset, the association of exposures (simply OMG and low QMG scores) and MMS was tested using Kaplan–Meier curves. Patients with OMG were more likely to achieve MMS than patients with GMG (p = 0.009). Patients with a low baseline QMG score also had advantages in achieving MMS (p = 0.000), and the results are shown (Figure 4). The advantages of OMG and low QMG scores in MMS induction were verified by Cox regression. Considered as confounders, age of onset, antibody status, use of GCS or TAC or AZA, PE/IVIG, and thymoma and thymectomy were included in the Cox proportional hazards model. Patients with OMG or low QMG scores had a higher probability of achieving MMS (Figure 5). The HRs were 2.354 (95% CI: 1.386–3.998) and 5.391 (95% CI: 1.962–14.808).

Kaplan–Meier curves for OMG and QMG scores in propensity score matching cases.

Cox regression analysis for OMG and QMG scores in non-matching cases.
After the retest in the cohort, isolated ocular involvement and low baseline QMG scores were confirmed to be favorable factors for MMS induction.
Discussion
This study analyzed the clinical data of MG patients who visited the Department of Neurology in Xiangya Hospital, and identified several factors associated with MMS induction, MuSK antibody positivity, OMG, and low baseline QMG score. We then attempted to retest these predictors in a test cohort, and we confirmed that patients with isolated ocular involvement and low baseline QMG scores (⩽16 points) were more likely to achieve MMS. MuSK antibody positivity failed to be verified due to inadequate cases.
MMS, a description of the clinical state of postintervention status in MG patients, was first proposed in 2000 by the Myasthenia Gravis Foundation of America (MGFA). 12 It was then adopted as a primary or secondary outcome in many clinical studies of MG.4,13–16 MMS was also recommended as a therapeutic goal of MG according to the Japanese Clinical Guidelines (2018) and International Expert Consensus (2016).8,17 According to the guidelines or consensus, MG patients experience two phases of therapy: the MMS induction phase and the MMS maintenance phase. Thus, successful and early induction of MMS is a good start for MG therapy and may be benefit for patients. Utsugisawa et al. 18 found that intensive treatment strategies achieved MMS more frequently and earlier. Early fast-acting treatments avoid high-dose cumulative steroids and may improve the quality of life of patients with MG. 17 A recent multicenter, parallel, single-blind randomized trial also indicated MG patients with immediate high-dose daily prednisone had higher proportion to attainment of MMS without prednisone at 12 months and lower cumulative dose of prednisone. 19 However, previous studies have usually focused on long-term outcomes rather than MMS induction.4,20 In this study, we attempted to identify the factors associated with MMS induction and found that isolated ocular involvement and low baseline QMG score may be predictors of MMS induction.
MuSK antibody-positive MG was thought to have more severe symptoms, higher frequency of bulbar muscle and axial muscle involvement, and higher risk of developing myasthenic crisis.21,22 Unlike anti-AChR antibody, MuSK antibodies are mostly IgG4. This subtype of IgG is functionally monovalent, with poor complement activation and low FcγR binding.23,24 Pathogenic IgG4s interfere with AChR clustering through blocking LRP4/MuSK signaling and cause dysfunction of the neuromuscular junction.23,24 Due to the unique pathomechanism of IgG4, MuSK-MG responds poorly to acetylcholinesterase inhibitors and IVIG, but particularly well to rituximab.23,25 In exploring cohort, we found that patients with MuSK antibody seemed to have an advantage in MMS induction, although they accepted non-rituximab treatment (namely, steroids and immunosuppressants). Properly speaking, our study was a single-center retrospective study with too little MuSK-MG cases to retest this result in the test cohort. This result should be taken with caution. But we thought the following reasons could probably explain this result. First, MuSK-associated MG was described to be responded well to GCS in many studies.15,21,23,26 In our cohort, almost all MuSK-associated MG patients accepted GCS (12 of 13 patients exposed to GCS), and GCS have a quicker onset of action, often beginning within 2–3 weeks. Selecting an effective and fast-action treatment may be an important factor in favorable result. Furthermore, defining ‘MMS or better’ as the primary outcome may be another reason. Due to earlier diagnosis and more timely treatment, the MGFA postintervention status has improved and this change was mostly related to the rise of MMS rate. 27 When we adopted ‘MMS or better’, a more flexible criterion, rather than complete stable remission (CSR) as a clinical outcome, the result may be in favor of MuSK-MG. For instance, both Jeffrey T Guptill et al. 28 and Fulvio Baggi et al. 29 reported MuSK-MG characterized by a significantly lower CSR rate in comparison with AChR-Ab (+) MG. But, when using ‘MMS or better’ instead (the rates of achieving MMS were also reported), it was found MuSK antibody-related MG was comparable or superior to AChR-Ab (+) MG. Using various criteria might result in different outcomes. In addition, a retrospective study from Japan (including 923 patients) also displayed a similar good outcome in MuSK-MG; 75% of patients achieved MMS, and the median time to achieve MMS was only 7 months. 15 Thus, further investigation with adequate MuSK-MG cases and prospective design is needed to verify this result. Besides, our study just focused on short-term outcome; there are still several problems in long-term therapy. MuSK-MG is likely to require long-term administration of multiple immunosuppressive agents. 26 The maintenance corticosteroid dose, which is associated with adverse effects, is higher than other disease subtypes, 20 and conventional immunosuppressants are thought to be comparatively less effective.24,30,31 Evoli et al.30–32 believed these patients often complain of increased weakness or suffer from severe disease relapses during steroid dosage tapering. Thus, maintaining MMS or better status without severe adverse effects in MuSK-MG may be a challenge.
OMG is generally considered to be a mild subtype, and the favorable prognosis of OMG has been described in several studies;15,33 79.8% OMG could achieve MMS or better, and the median time to achieve MMS was only 4 months. 15
Unsurprisingly, isolated ocular involvement was found to be a favorable factor for MMS indution in both exploring and test cohort after adjusted. Although it failed to validate in the sensitivity analysis (displayed in supplementary sensitivity analysis), the difference is likely to be attributed to small sample size in the sensitivity analysis. Our study also observed that the baseline QMG score was a predictor of MMS induction. In fact, previously published studies from different groups independently demonstrated that disease severity was a predictor of the outcome of MG. Kim et al. 34 reported that thymectomized MG patients with severe preoperative status (MGFA class IV–V) were less likely to achieve remission compared with patients with milder status. Salomi Salins et al. 35 found that the MGFA class, 3 years after diagnosis, is a predictor of long-term prognosis. And a post hoc analysis of data from the randomized trial of thymectomy in myasthenia gravis (MGTX) also found month 3 QMG scores improvement in the QMG scores between month 0 and 3 were significant covariates for achieving sustained MMS with complete withdrawal of prednisone. 36 Our data revealed similar result. However, a recently published work showed maximum severity during the first year was not a predictor of the satisfactory response (remission/MMS while on low-dose prednisone without PE or IVIG). 37 A previous study (including 268 cases) also found that prognosis is favorable for the majority of MG patients regardless of maximum disease severity. 4 We speculated this inconsistency may be explained by the maximum severity (such as QMG score or MGFA classification) usually obtained during exacerbation. There is always a trigger, such as infection, operation, or improper discontinuation of drugs, for the exacerbation of MG. Remove inducements quickly, and intensive medical care can effectively improve the symptoms in many patients. Therefore, the maximum QMG score just revealed an ‘instant severity’ rather than the ‘original severity’ in a period very well. Furthermore, we investigated the maximum QMG score in our data (see Supplementary Table 1) and found that the maximum QMG score was not associated with MMS induction.
Overall, we investigated the factors affecting MMS induction and found that isolated ocular involvement and low baseline QMG score were predictors of MMS induction. These findings may be helpful in tailoring therapy and predicting treatment response. However, this study had several limitations. First, this was a single-center retrospective study; therefore, potential selection bias might have occurred with data missing from medical records. Second, data on thymic hyperplasia were not analyzed because of the low sensitivity and specificity of CT scans for thymic hyperplasia and a predictor might have been missed. Third, there were very few cases of MuSK-MG due to its low incidence in China. Fourth, propensity score matching used in the test cohort led to loss of cases and may introduce bias to the results. And the survival analysis was not flawless as the achievement of MMS, differently from death, is not permanent. A cohort study with sufficient cases would be more convincing. Therefore, further studies, particularly prospective studies in different centers, are needed to test whether these results are suitable for other populations.
Research Data
sj-7z-4-tan-10.1177_17562864221080520 – for Factors affecting minimal manifestation status induction in myasthenia gravis
sj-7z-4-tan-10.1177_17562864221080520 for Factors affecting minimal manifestation status induction in myasthenia gravis by Yi Li, Shumei Yang, Xiaohua Dong, Zhibin Li, Yuyao Peng, Wanlin Jin, Di Chen, Ran Zhou, Fei Jiang, Chengkai Yan and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-1-tan-10.1177_17562864221080520 – Supplemental material for Factors affecting minimal manifestation status induction in myasthenia gravis
Supplemental material, sj-docx-1-tan-10.1177_17562864221080520 for Factors affecting minimal manifestation status induction in myasthenia gravis by Yi Li, Shumei Yang, Xiaohua Dong, Zhibin Li, Yuyao Peng, Wanlin Jin, Di Chen, Ran Zhou, Fei Jiang, Chengkai Yan and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864221080520 – Supplemental material for Factors affecting minimal manifestation status induction in myasthenia gravis
Supplemental material, sj-docx-2-tan-10.1177_17562864221080520 for Factors affecting minimal manifestation status induction in myasthenia gravis by Yi Li, Shumei Yang, Xiaohua Dong, Zhibin Li, Yuyao Peng, Wanlin Jin, Di Chen, Ran Zhou, Fei Jiang, Chengkai Yan and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864221080520 – Supplemental material for Factors affecting minimal manifestation status induction in myasthenia gravis
Supplemental material, sj-docx-3-tan-10.1177_17562864221080520 for Factors affecting minimal manifestation status induction in myasthenia gravis by Yi Li, Shumei Yang, Xiaohua Dong, Zhibin Li, Yuyao Peng, Wanlin Jin, Di Chen, Ran Zhou, Fei Jiang, Chengkai Yan and Huan Yang in Therapeutic Advances in Neurological Disorders
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (grant number: 81771364).
Ethics statement
The study was approved by the Medical Ethics Committee of the Hunan Medical Science Research Institute of Xiangya Hospital (201703107). All participants were informed about and signed the consent form for the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
