Abstract
Background:
Alemtuzumab is a disease-modifying therapy for highly active relapsing-remitting multiple sclerosis (RRMS). Sustained efficacy up to 9 years was observed in the phase IIIb/IV open-label TOPAZ clinical trial and assessed in the real-world retrospective and prospective study, TREAT-MS.
Objectives:
To examine long-term efficacy and safety of alemtuzumab in participants with multiple sclerosis (MS) and highly active disease (HAD) by combining up to 13 years of TOPAZ data and TREAT-MS interim data.
Design:
TOPAZ: Randomized participants completing core CARE-MS I and II could receive additional alemtuzumab (12 mg/day, 3 consecutive days; ⩾12 months apart) for 11–13 years after initiating treatment. TREAT-MS: Participants from German MS clinics were observed for 4 years after last alemtuzumab treatment phase.
Methods:
Efficacy outcomes (annualized relapse rate (ARR), change in Expanded Disability Status Scale (EDSS), 6-month confirmed disability worsening/improvement, magnetic resonance imaging), and adverse events (AEs) were examined. Primary HAD definition (⩾2 relapses in the year prior to baseline and ⩾1 gadolinium-enhancing lesion at baseline), and two alternative HAD definitions were assessed.
Results:
More participants from CARE-MS I (28%) and II (24%) met primary HAD criteria than TREAT-MS (~14%). Mean ARR for alemtuzumab-treated HAD participants was significantly reduced in CARE-MS I and II (0.14 and 0.15, respectively, Years 3–13) and in TREAT-MS (0.24,
Conclusion:
Sustained efficacy of alemtuzumab was observed for clinical and radiological outcomes in participants with HAD in the TOPAZ clinical trial and real-world TREAT-MS study with no new safety signals.
Trial registration:
ClinicalTrials.gov (CARE-MS I, NCT00530348; CARE-MS II, NCT00548405; CARE-MS Extension Study, NCT00930553; TOPAZ, NCT02255656). Paul-Ehrlich-Institut (TREAT-MS, NIS 281).
Introduction
Timely treatment of highly active disease (HAD) in people with relapsing-remitting multiple sclerosis (RRMS) is critical. 1 HAD is characterized by disability accumulation, frequent relapses, and ongoing magnetic resonance imaging (MRI) activity, 2 which necessitates highly effective treatment early in the disease course. However, there is a lack of consensus on the criteria for defining HAD, with some using Expanded Disability Status Scale (EDSS) scores and relapse frequency (“aggressive multiple sclerosis 1–3”) and others using MRI activity and relapse frequency to define “highly active RRMS.” 3 These various HAD definitions complicate the establishment of a consensus on the treatment paradigm for this highly active multiple sclerosis (MS) phenotype.
Alemtuzumab (Lemtrada®; Sanofi, Cambridge, MA, USA) is a humanized anti-CD52 monoclonal antibody approved as a disease-modifying therapy (DMT) for people with highly active RRMS.4,5 Alemtuzumab targets B and T lymphocytes expressing CD52, resulting in selective depletion and a distinct pattern of lymphocyte repopulation with long-term immunological benefits.6–8
Alemtuzumab (12 mg/day) is administered by intravenous (IV) infusion for two initial treatment courses: 5 consecutive days for the first course and 3 consecutive days for the second course, given 12 months after the first, with additional courses administered as required. 9 The recommended pulsed treatment with prolonged intermittent treatment-free phases for alemtuzumab 10 contrasts with the continuous dosing regimen for the majority of DMTs for RRMS, 11 resulting in sustained treatment-free efficacy.12,13
In the phase III, randomized, rater-blinded trials CARE-MS I 14 (NCT00530348) and CARE-MS II 15 (NCT00548405), IV alemtuzumab demonstrated significantly greater improvements in efficacy, compared with subcutaneous (SC) interferon β-1a (IFNB-1a) over 2 years, resulting in decreased annual relapse rates in both trials and reduced disability accumulation in CARE-MS II. A 4-year CARE-MS extension study (NCT00930553) followed, enabling CARE-MS I and II participants to receive additional courses of alemtuzumab as required.16–18 Participants completing the CARE-MS extension study were eligible to enroll in TOPAZ: a phase IIIb/IV, open-label, multicenter long-term trial, during which alemtuzumab efficacy was maintained for up to 13 years, with safety risks declining over time, and no new safety signals were observed. 19
The ongoing noninterventional long-Term study foR obsErvAtion of Treatment with alemtuzumab in active relapsing-remitting MS (TREAT-MS; Paul Ehrlich Institute registry: 281) is the largest real-world evidence study of long-term alemtuzumab efficacy and safety to date.20,21 TREAT-MS is an observational, retrospective, and prospective cohort study that seeks to address the need for comprehensive real-world evidence in participants with active RRMS from approximately 400 centers in Germany.
In this manuscript, we aim to better understand the long-term efficacy and safety of alemtuzumab treatment in MS among participants with HAD, by combining up to 13 years of clinical trial extension data from TOPAZ and real-world interim results from TREAT-MS.
Methods
Study design
CARE-MS core and extension studies: TOPAZ
The CARE-MS and TOPAZ trials have been described previously.14,15,19 In brief, CARE-MS I and II comprised the 2-year core phase III trials that compared alemtuzumab with SC IFNB-1a in participants with active RRMS who were either treatment-naïve (CARE-MS I) or had an inadequate response to prior therapy (CARE-MS II).14,15 In the core studies, participants were treated with either IV alemtuzumab (12 mg/day, for 5 consecutive days at baseline followed by treatment for 3 consecutive days after 12 months), or SC IFNB-1a 44 μg (3 times per week) (Figure S1(a)).
Participants from CARE-MS I and II were able to enter a 4-year extension trial (CARE-MS extension study) to receive additional alemtuzumab as required (12 mg/day for 3 consecutive days ⩾12 months apart), or other DMTs at the investigator’s discretion, upon evidence of disease activity.17,18 Participants treated with SC IFNB-1a were also able to enter the extension and receive alemtuzumab. Outcomes for these participants beyond the 2-year core study are not presented herein due to discontinuation of SC IFNB-1a treatment. Participants who completed the core and 4-year extension studies were eligible to enter the TOPAZ extension (>6 years from baseline), allowing participants to receive additional alemtuzumab courses (12 mg/day for 3 consecutive days ⩾12 months apart) or another DMT per investigator’s discretion at any time (no specified criteria).22,23
TREAT-MS
TREAT-MS is a prospective and retrospective, open-label, uncontrolled, multicenter study including people with RRMS treated with alemtuzumab. The study design has been described previously.20,21 The ongoing planned period of observation of participants is 4 years after the last treatment phase with alemtuzumab (up to a total of 60 months) (Figure S1(b)), with data collection undertaken in specialized MS clinics at approximately 400 sites throughout Germany. 20 The retrospective data collection was completed from October 2013 to June 2014. The prospective inclusion period began from December 2014, with participant documentation continuing until December 2025; database closure is planned for March 2026. Additionally, regular interim evaluations of the data collected were undertaken, and the interim analysis is presented herein. The data cut-off date was February 2, 2023.
Exclusion criteria are not defined due to an unselected population, though participants older than 55 years of age were excluded. Participant data are documented at regular intervals via the multidimensional Multiple Sclerosis Documentation System (MSDS 3D).
Clinical efficacy assessments
CARE-MS core and extension studies: TOPAZ
Efficacy assessments were secondary endpoints in TOPAZ. 24 Relapses were defined as new MS symptoms with an objective change on neurological examination lasting at least 48 h and were verified at each 6-monthly study visit. Annualized relapse rate (ARR) and proportions of participants free of relapse were reported. EDSS score was assessed every 6 months and at the time of a suspected relapse by raters blinded to treatment. Changes in EDSS scores were analyzed, according to changes from core study baseline and were classified as improved (⩾1.0-point decrease), stable (⩽0.5-point change in either direction), or worsened (⩾1.0-point increase). Confirmed disability worsening (CDW) was defined as a ⩾1.0-point EDSS score increase (or ⩾1.5-points if baseline EDSS = 0) confirmed over 6 months. Confirmed disability improvement (CDI) was defined as a ⩾1.0-point EDSS score decrease from baseline confirmed over 6 months (assessed only in participants with baseline EDSS score ⩾2.0). MRI was assessed annually by imaging specialists blinded to core study treatment, with blinded brain MRI scans undertaken at NeuroRx Research (Montréal, Canada) for lesion examinations and the Cleveland Clinic (Cleveland, OH, USA) for brain parenchymal fraction (BPF) analyses. Participants were classified as free of MRI disease activity with the absence of new gadolinium (Gd)-enhancing T1 lesions on current MRI and the absence of new/enlarging T2 hyperintense lesions since the last MRI. No evidence of disease activity (NEDA-3) was defined as the absence of relapse, 6-month CDW, and MRI disease activity. Brain volume loss (BVL) was assessed by calculating the annualized percent brain volume change (PBVC; (% change from baseline in BPF from drug start to last BPF measure divided by the number of days of follow-up between drug start and last BPF measure) multiplied by 365).
TREAT-MS
Neurologists and MS nurses were guided by the MS documentation system through treatment management, including monitoring of infusions, examinations, and regular laboratory screenings.25,26 Relapses were assessed at baseline and then monthly. EDSS score was assessed at baseline and every 3 months thereafter. Changes from baseline in EDSS scores were analyzed, and the participant’s condition was classified as worsened if EDSS change from baseline ⩾1.0-point, stable if −1.0 < change from baseline ⩽ 1.0, and improved if change from baseline <−1.0-point. CDW was defined as a ⩾1.0-point EDSS score increase (or ⩾1.5-points if baseline EDSS = 0) confirmed over 6 months. Clinical disease progression or worsening was defined as an increase (and CDI a decrease) in EDSS score, from baseline of ⩾1.5 if the baseline EDSS score was 0, of ⩾1.0 if baseline EDSS score was 1–5, or ⩾0.5 if the baseline EDSS score was >5.0, all confirmed over 6 months. All available data were included in the analysis. Imputations were only conducted for missing dates for days (substituted by the 15th day of the month) and for months (substituted by June), while years were not substituted.
Participant-reported outcomes
CARE-MS core and extension studies: TOPAZ
The Health-Related Quality of Life (EuroQoL) questionnaire, EQ-5D, was completed at baseline, every 6 months until Year 2.5, then yearly. Satisfaction with treatment, cognition, state of health, health-related quality of life, and ability to work were examined. Participants’ health status was assessed using the EuroQol Visual Analogue Scale (EQ-VAS), which captures the self-rating of health status using a visual “thermometer” with the endpoints of 100 and 0 (best and worst imaginable health, respectively).
To obtain data on informal care and work capacity, the modified health-related productivity questionnaire (HRPQ) was administered every 12 months and at the end of the study.
TREAT-MS
The EuroQoL questionnaire was completed at baseline and every 6 months thereafter. The EQ-VAS score was reported, as described for TOPAZ. The work productivity and activity impairment (WPAI) questionnaire 27 was completed by participants at baseline and every 6 months thereafter. WPAI consists of six questions, which cover the effects of health problems (physical and psychological) on the ability to work and pursue normal activities.
Safety monitoring
CARE-MS core and extension studies: TOPAZ
Safety monitoring was the primary endpoint and occurred for at least 48 months since the last alemtuzumab infusion in line with the recommended risk-minimization protocol. Hematology tests (complete blood counts with differential) were undertaken at least once a month, renal examinations (serum creatinine and urinalysis with microscopy) were performed monthly, and thyroid function tests were performed at least every 3 months. All adverse events (AEs), serious AEs, and medical events of interest were recorded. Infusion-associated reactions (IARs) were defined as any treatment-emergent AE (TEAE) with onset during and within 24 h of alemtuzumab infusion.
TREAT-MS
Safety examinations, including complete blood count with differential count, serum creatinine, and urinalysis with microscopic sediment analysis, were undertaken monthly up to 48 months after the last infusion. Thyroid function tests were performed every 3 months up to 48 months after the last infusion.
HAD definitions
Three different criteria corresponding to alternative definitions of HAD were applied to the core CARE-MS I and II populations, resulting in three groups of HAD participants. 19 Some participants met more than one definition and were included in more than one group. The primary HAD definition (HAD) was applied to participants with ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline. Alternative HAD Definition 1 (HAD1) is relapse-focused and specifies participants with ⩾2 relapses in the year prior to baseline. Alternative HAD Definition 2 (HAD2) is MRI-lesion focused and defines participants with ⩾1 relapse in the year prior to baseline and ⩾3 Gd-enhancing lesions at baseline. Results presented herein are largely focused on participants meeting criteria for the primary HAD definition. Results corresponding to HAD1 and HAD2 are included in the supplement.
Statistical analyses
Calculation and justification of sample size
Based on 1600 participants receiving alemtuzumab in TREAT-MS, the fraction of participants with premature discontinuation can be determined by a 95% confidence interval (CI) of length ⩽5 percentage points. In subgroups containing at least 25% of the total random sample, this fraction can be determined by a 95% CI of length <10 percentage points. Undesirable events with an incidence of at least 0.00187 (1:534) were observed with 95% certainty at least once in the random sample of 1600 participants.
Demographic and disease characteristics of participants
For continuous variables, the mean and standard deviation were reported. For categorical variables, the numbers and percentages were reported.
ARR among HAD participants
We further examined ARR for TOPAZ (CARE-MS I and CARE-MS II) and TREAT-MS in participants with HAD. In TOPAZ, the ARR from Year 3 to Year 13 was estimated through negative binomial regression models with robust variance estimation. In TREAT-MS, Poisson regression models adjusted for overdispersion were used to estimate ARR. These models included the number of relapses as the outcome and observational time in years after the start of alemtuzumab up to Year 11 and subgroup (alemtuzumab vs SC IFNB-1a).
Change in Expanded Disability Status Score
Changes in EDSS scores for participants treated with alemtuzumab versus SC IFNB-1a from baseline through Year 11 were analyzed, using ranked analysis of covariance adjusted for geographic region and baseline EDSS score. The trajectory of participants with improved, stable, or worse EDSS was visualized using Sankey flow diagrams from a subpopulation with all the time points (Year 2–Year 11) included in the analysis.
Free of 6-month CDW or CDI
The proportions of participants free of 6-month CDW or achieving 6-month CDI were evaluated using Kaplan–Meier analysis. 28 CDI was only assessed for participants with a baseline EDSS score ⩾2.0.
Free of MRI disease activity
Proportions of participants free of MRI disease activity, new Gd-enhancing T1 lesions, new/enlarging T2 hyperintense lesions, and new T1 hypointense lesions were evaluated using logistic regression models adjusted for baseline values, with 95% CIs obtained by normal approximation to the binomial distribution.
Annualized percent brain volume change
Percentage change in BPF between alemtuzumab and SC IFNB-1a from core study baseline up to Year 11 was evaluated at each time point among HAD participants; distribution-free estimates were obtained for the CI of the median for CARE-MS I and CARE-MS II.
EuroQol Visual Analogue Scale
A linear mixed-effects model with unstructured variance–covariance structure and random intercepts and slopes was used to evaluate change in EQ-5D over 72 months. This model included treatment, time, geographic region, baseline EQ-5D score, and a two-way interaction between treatment and time.
Work and household productivity
Mean (SD) scores for work and household productivity were analyzed for participants with HAD, according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline. Participants completed the following questionnaires: HRPQ for TOPAZ (CARE-MS I and II) and WPAI for TREAT-MS. Outcomes assessed from pooled CARE-MS I and II included the household productivity hours lost due to MS over time, and the percentage of the impact on work output for household chores over time. For TREAT-MS, the percentage of work time missed, percentage of impairment while working, and percentage of activity impairment due to MS were calculated.
Results
Participant disposition and baseline characteristics
About one-quarter of the CARE-MS I and II populations met the primary HAD definition (28% and 24%, respectively), with most participants with HAD entering the extension study (94%, CARE-MS I; 89%, CARE-MS II) (Figure 1). Close to half of the CARE-MS I (44%) and CARE-MS II (45%) HAD populations remained in the TOPAZ trial through the end of Year 12. Prior to Year 12 completion, 54% of CARE-MS I and 50% of CARE-MS II participants with HAD who entered the extension discontinued (Figure 1). The main known reasons for discontinuation were withdrawal of consent (13 participants CARE-MS I; 8 CARE-MS II), lost to follow-up (3 CARE-MS I; 5 CARE-MS II), and physician decision (3 CARE-MS I; 2 CARE-MS II).
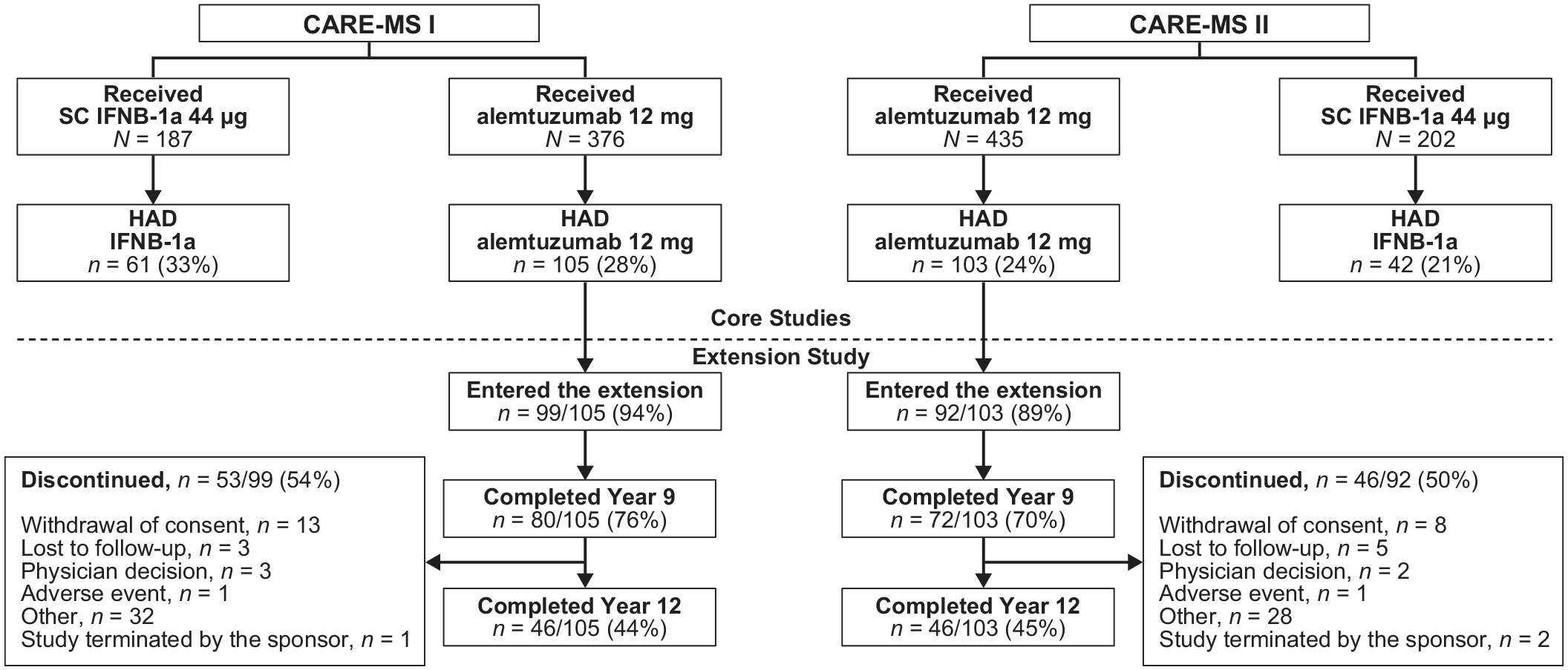
Disposition of CARE-MS I and CARE-MS II participants meeting HAD definition according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
More participants from CARE-MS I (28%) and CARE-MS II (24%) met the primary HAD criteria relative to TREAT-MS participants (124/921; ~14%) (Table 1). Baseline demographics and characteristics of participants with HAD were broadly similar between CARE-MS trials and TREAT-MS. CARE-MS II participants with HAD had twice as many mean years since initial relapse (4.0 years) as those in CARE-MS I (1.7 years), whereas TREAT-MS participants first displayed MS symptoms 7.2 years prior to baseline. Most CARE-MS II participants (67%) received only one previous DMT, whereas most TREAT-MS participants (~59%) received two or more DMTs (the CARE-MS I population was treatment-naïve). TREAT-MS participants with HAD previously received a wider range of DMTs relative to CARE-MS II, including fingolimod (34.7%), dimethyl fumarate (16.1%), and teriflunomide (3.2%), which were not received by CARE-MS II participants. A larger proportion of TREAT-MS participants previously received natalizumab (33.1%) relative to CARE-MS II (6.8%). Additionally, a small proportion (16.1%) of the TREAT-MS population with HAD was treatment-naïve (Table 1).
Demographic and disease characteristics of participants.

Data are expressed as mean (SD) unless otherwise stated. CARE-MS I and II are part of the TOPAZ clinical trial. For some parameters, the number of participants differs from the overall population as follows: CARE-MS I, BPF n = 103; CARE-MS II, BPF n = 102; TREAT-MS, previous DMTs received n = 103.
BMI, body mass index; BPF, brain parenchymal fraction; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; Gd, gadolinium; HAD, highly active disease; IFNB-1a, interferon β-1a; MS, multiple sclerosis; NA, not applicable; NC, not collected; SD, standard deviation.
Efficacy outcomes
Annualized relapse rate
In the core clinical trials, ARR over 0–2 years was significantly reduced with alemtuzumab treatment versus SC IFNB-1a in participants with HAD (p ⩽ 0.01, CARE-MS I and II) (Figure 2(a) and (b)). ARR remained low over Years 3–13 in CARE-MS I (0.14), and CARE-MS II (0.15), with 54% and 48% of participants remaining free of relapses, respectively. In TREAT-MS, ARR was similarly reduced from baseline at Years 0–2, and remained low (0.24) after 2 years (Figure 2(c)). Similar results for CARE-MS I and II, and TREAT-MS were obtained for alternative definitions of HAD and are shown in the Supplemental Material (Figure S2).

ARRs for (a) CARE-MS I, (b) CARE-MS II, and (c) TREAT-MS participants with HAD treated with 12 mg alemtuzumab with the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Expanded Disability Status Scale
At Year 2, mean changes from baseline in EDSS scores did not differ significantly in CARE-MS I participants with HAD treated with alemtuzumab versus those treated with SC IFNB-1a (Figure 3(a)). By contrast, mean changes from baseline in EDSS scores were significantly improved in CARE-MS II for participants with HAD treated with alemtuzumab versus SC IFNB-1a (p < 0.05; Figure 3(b)). At Year 11, 74% of CARE-MS I and 67% of CARE-MS II participants with HAD achieved stable or improved EDSS scores, while 79% of participants with HAD from TREAT-MS achieved stable or improved EDSS at Year 3.6 (Figure 3(a)–(c)). Similar results were obtained for participants with alternative definitions of HAD (HAD1 or HAD2), and are shown in the Supplemental Material (Figure S3). A similar proportion of participants from CARE-MS I (26%) and TREAT-MS (27%) had improved EDSS scores at Year 4 and Year 3.6, respectively, compared with 19% for CARE-MS II at Year 4.

Percentage of HAD participants with improved or stable EDSS scores compared to baseline over time is shown for (a) CARE-MS I, (b) CARE-MS II, and (c) TREAT-MS, according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Change in EDSS
The flow of participants in TOPAZ with improved, stable, or worsened EDSS scores was visualized using Sankey diagrams at Years 2, 4, 6, and 11 (Figure S4). In both CARE-MS I and II, the majority of participants including those remaining in the study follow-up, were stable or improved at Year 2 through Year 11. The percentage of participants who improved was more consistent through Year 11 in CARE-MS I relative to CARE-MS II (CARE-MS I, stable: 59% Year 2, 51% Year 11; improved: 28% Year 2, 22% Year 11; CARE-MS II, stable: 58% Year 2, 49% Year 11; improved: 33% Year 2, 18% Year 11).
CDW and CDI
Further, 67% of CARE-MS I and 54% of CARE-MS II participants with HAD were free of 6-month CDW at Year 11 and Year 10, respectively (Figure 4). TREAT-MS had the highest proportion of participants with HAD free of 6-month CDW (89%), followed by CARE-MS I (83%) at Year 4, during which 77% of CARE-MS II participants with HAD were free of 6-month CDW. However, at Year 8, a significantly greater proportion of participants from CARE-MS I (70%) and CARE-MS II (60%) were free of 6-month CDW relative to TREAT-MS (5%). Similar results were also obtained for participants with HAD1 or HAD2 (Figure S5). A similar proportion of participants with HAD achieved 6-month CDI at Year 4 from CARE-MS I (39%) (Figure 5(a)), CARE-MS II (43%) (Figure 5(b)), and TREAT-MS (32%) (Figure 5(c)), with about half of participants with HAD achieving 6-month CDI at Year 11 (CARE-MS I, II). Further, over half of CARE-MS II participants with HAD2 achieved 6-month CDI at Year 11 (Figure S6).

Percentage of HAD participants free of 6-month CDW over time is shown for (a) CARE-MS I, (b) CARE-MS II, and (c) TREAT-MS with the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.

Percentage of HAD participants achieving 6-month CDI over time for (a) CARE-MS I, (b) CARE-MS II, and (c) TREAT-MS with the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Free of MRI disease activity
Significantly more CARE-MS I (Year 2) and CARE-MS II (Years 1 and 2) alemtuzumab-treated participants with HAD were free of MRI disease activity than those treated with SC IFNB-1a (p < 0.001 and p < 0.0001, respectively). In CARE-MS I (Year 2), similar results were observed for participants with HAD1 (p < 0.0001), whereas in CARE-MS II, significantly more alemtuzumab-treated participants with HAD1 or HAD2 were free of MRI disease activity than those treated with SC IFNB-1a (p < 0.0001 (HAD1, Years 1 and 2), p < 0.01 (HAD2, Year 1), and p < 0.001 (HAD2, Year 2)) (Figure S7). At least 60% of CARE-MS participants treated with alemtuzumab with HAD (Figure 6(a) and (b)) or HAD2 (Figure S7) were free of MRI disease activity from Years 3 through 11.
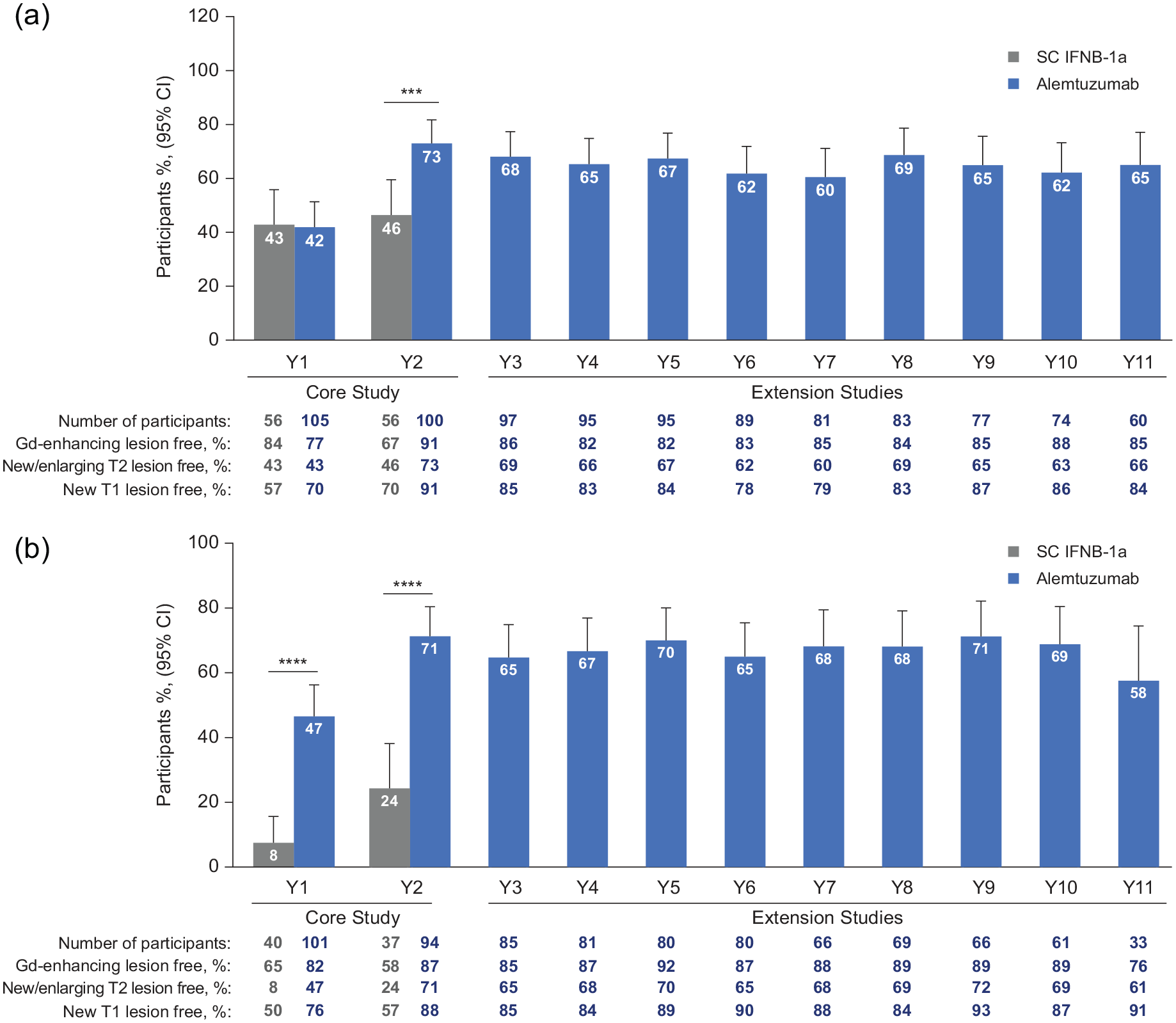
Percentage of participants with HAD free of MRI disease activity for (a) CARE-MS I and (b) CARE-MS II is shown with the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
No evidence of disease activity-3
In CARE-MS I and II, about one quarter of participants achieved cumulative NEDA-3 by Year 2 (26% and 23%, respectively). Through Year 11, a small proportion of participants with HAD achieved sustained cumulative NEDA-3 (8% CARE-MS I and 5% CARE-MS II) (Figure 7). Similar results were observed for participants with HAD1, although a smaller percentage of participants with HAD2 achieved cumulative NEDA-3 through Year 11 (Figure S8).

Percentage of participants with HAD achieving NEDA-3 for (a) CARE-MS I and (b) CARE-MS II with the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Percent brain volume change
In CARE-MS I (Figure 8(a)), the annualized change in median PBVC decreased over time for the alemtuzumab-treated cohort from −0.81% (95% CI, −1.02 to −0.61) in Year 1 to −0.26% (95% CI, −0.29 to −0.2) in Year 11. Similarly, in CARE-MS II (Figure 8(b)), the annualized change in median PBVC decreased from −0.57% (95% CI, −0.82 to −0.33) in Year 1 to −0.17% (95% CI, −0.25 to −0.13) in Year 11. Similarly, median PBVC was decreased in participants with HAD1 or HAD2 (Figure S9).

Median annualized PBVC with 95% CI for (a) CARE-MS I and (b) CARE-MS II participants with HAD according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Participant-reported outcomes
Mean baseline VAS, assessing self-rated health status, was higher for participants with HAD treated with alemtuzumab from CARE-MS I (75.4) relative to those from CARE-MS II (72.0) and TREAT-MS (67.5) (Figure 9(a)–(c)). In Year 1, alemtuzumab-treated CARE-MS I participants had a higher mean VAS (82.8) relative to those treated with SC IFNB-1a (77.2, p < 0.01). In Year 2, alemtuzumab-treated CARE-MS II participants had a higher mean VAS (78.2) relative to those treated with SC IFNB-1a (70.6, p < 0.01). There was a significant increase in the mean VAS relative to baseline for alemtuzumab-treated participants in CARE-MS I (Years 0.5 and 5.0, p < 0.01; Years 1.0 and 4.0, p < 0.0001; Years 1.5 and 2.5, p < 0.001; Year 2.0, p < 0.05), and in CARE-MS II (Years 1–2, p < 0.001; Year 2.5, p < 0.01; Years 4–5, p < 0.05). At Year 5, CARE-MS I and II participants with HAD reported higher mean VAS values (81.6 and 75.9, respectively), whereas TREAT-MS participants reported a lower mean VAS (70.2) at Year 4.5 and a mean VAS of 77.8 at Year 5.

Mean VAS (SD) scores for participants with HAD from (a) CARE-MS I, (b) CARE-MS II, and (c) TREAT-MS according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
Work and household productivity
Work and household productivity were assessed using the modified HRPQ for TOPAZ (pooled CARE-MS I and II), and WPAI questionnaire for TREAT-MS. In TOPAZ, there was a small trend toward improved household productivity in participants treated with alemtuzumab, seen as a decrease in the mean hours lost in Year 5 (6.11) compared with baseline (8.57), and an increase in mean work output in household chores in Year 2 (21.22%) relative to baseline (16.08%) (Figure 10(a) and (b)). Similarly, a trend toward improved work productivity was observed in participants from TREAT-MS by Year 2, shown as a decrease in mean missed work time (baseline (BL) 36.4% vs Year 2 3.9%), mean work impairment (BL 31.6% vs Year 2 23.7%), and mean activity impairment (BL 34.2% vs Year 2 27.5%). The decreased trend relative to baseline also extended to Year 4 (Figure 10(c)–(e)).

Mean (SD) scores for work and household productivity for participants with HAD according to the following criteria: ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline. Participants completed the following questionnaires: HRPQ for TOPAZ (CARE-MS I and II) and WPAI for TREAT-MS. (a) Household productivity hours lost due to MS over time, pooled CARE-MS I and II. (b) Percentage of the impact on work output for household chores over time, pooled CARE-MS I and II. (c) Percentage of work time missed due to MS, TREAT-MS. (d) Percentage of impairment while working due to MS, TREAT-MS. (e) Percentage of activity impairment due to MS.
Efficacy analyses for alternative HAD definitions
Sensitivity analyses of pooled CARE-MS populations were undertaken to evaluate the effect of restricting HAD criteria on clinical outcome using the primary HAD definition (HAD) and two alternative definitions of HAD (HAD1 and HAD2) (Figure 11). At baseline, a higher proportion of participants met the HAD1 (53%) criteria relative to HAD (25%) and HAD2 (21%) (Table 2). Some efficacy outcomes were similar for participants meeting HAD, HAD1, and HAD2 criteria. In particular, ARR (95% CI) over Years 3–13 was 0.14 (0.12–0.17) for HAD, 0.17 (0.14–0.19) for HAD1, and 0.15 (0.12–0.19) for HAD2. The proportion of participants with improved or stable EDSS was also similar (HAD, 71%; HAD1, 70%; HAD2, 67%), as was the proportion of participants free of 6-month CDW or with 6-month CDI (Table 2). Relative to the HAD group, participants meeting the first alternative definition (HAD1) had a similar mean EDSS score increase through Year 11 and were equally likely to be relapse-free or to attain annual NEDA-3 during Year 12. There was an increase in the proportion of HAD1 participants free of MRI disease activity and a similar proportion achieving cumulative NEDA-3 over Years 3–11 relative to HAD. HAD1 participants also experienced less cumulative BVL over Years 0–11.

Three different definitions of HAD were applied to the core CARE-MS I and II populations, resulting in three groups of HAD participants. Some participants met more than one definition and were included in more than one group.
Efficacy up to 13 years among alemtuzumab-treated participants meeting primary and alternative HAD definitions (pooled CARE-MS I and II).
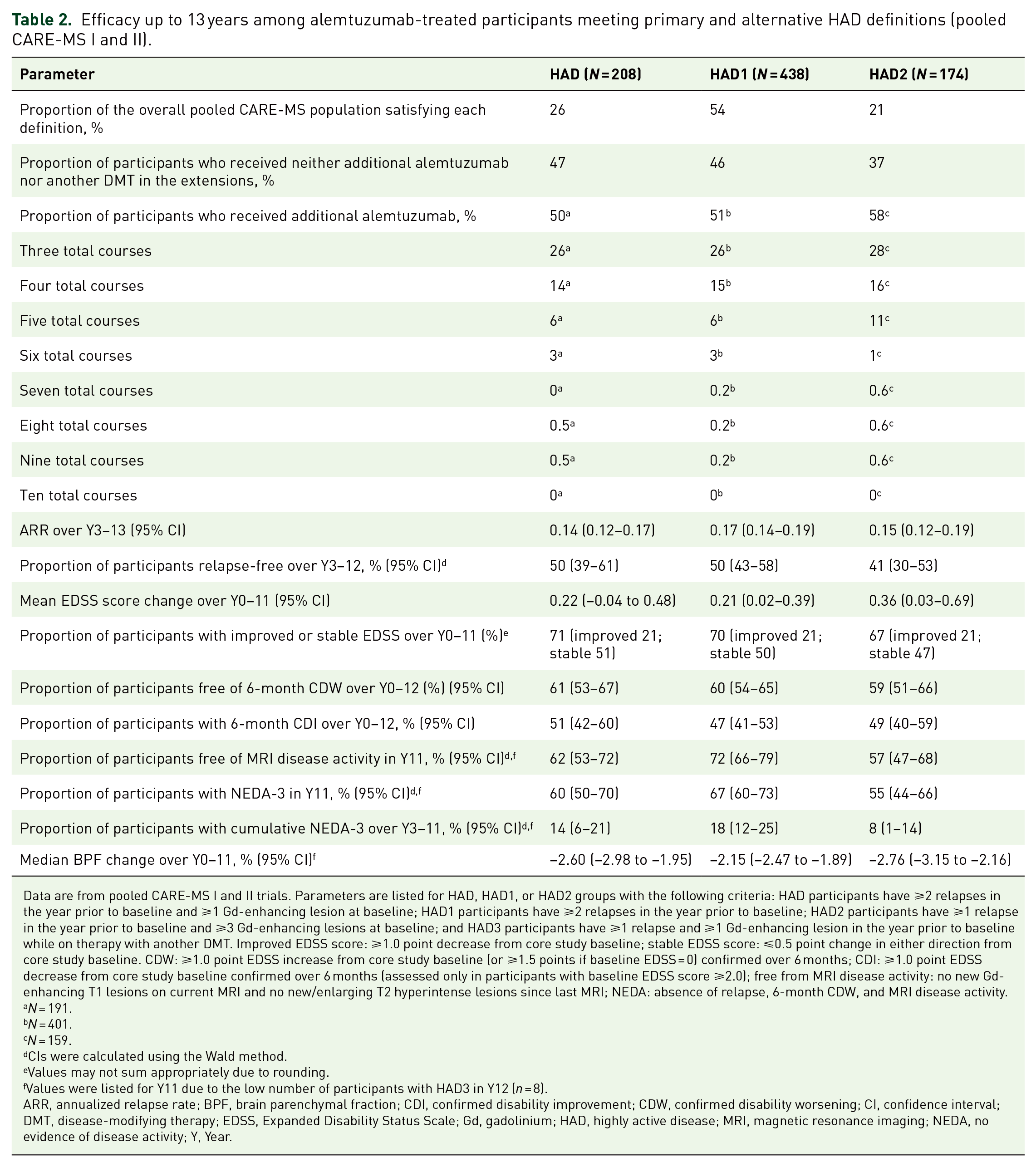
Data are from pooled CARE-MS I and II trials. Parameters are listed for HAD, HAD1, or HAD2 groups with the following criteria: HAD participants have ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline; HAD1 participants have ⩾2 relapses in the year prior to baseline; HAD2 participants have ⩾1 relapse in the year prior to baseline and ⩾3 Gd-enhancing lesions at baseline; and HAD3 participants have ⩾1 relapse and ⩾1 Gd-enhancing lesion in the year prior to baseline while on therapy with another DMT. Improved EDSS score: ⩾1.0 point decrease from core study baseline; stable EDSS score: ⩽0.5 point change in either direction from core study baseline. CDW: ⩾1.0 point EDSS increase from core study baseline (or ⩾1.5 points if baseline EDSS = 0) confirmed over 6 months; CDI: ⩾1.0 point EDSS decrease from core study baseline confirmed over 6 months (assessed only in participants with baseline EDSS score ⩾2.0); free from MRI disease activity: no new Gd-enhancing T1 lesions on current MRI and no new/enlarging T2 hyperintense lesions since last MRI; NEDA: absence of relapse, 6-month CDW, and MRI disease activity.
N = 191.
N = 401.
N = 159.
CIs were calculated using the Wald method.
Values may not sum appropriately due to rounding.
Values were listed for Y11 due to the low number of participants with HAD3 in Y12 (n = 8).
ARR, annualized relapse rate; BPF, brain parenchymal fraction; CDI, confirmed disability improvement; CDW, confirmed disability worsening; CI, confidence interval; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; Gd, gadolinium; HAD, highly active disease; MRI, magnetic resonance imaging; NEDA, no evidence of disease activity; Y, Year.
Relative to the HAD group, participants meeting the second alternative definition (HAD2) had a higher mean EDSS score increase through Year 11, and were less likely to be relapse-free in Year 12. Almost half of participants with HAD2 achieved cumulative NEDA-3 over Years 3–11 relative to those with HAD, though participants with HAD and HAD2 experienced a similar cumulative BVL over Years 0–11 (Table 2).
Similar to the TOPAZ overall population, 24 approximately 50% of participants with HAD and HAD1 from pooled CARE-MS trials received additional alemtuzumab following the first two courses (Table 2). A somewhat higher percentage of participants (58%) with the MRI-focused HAD2 criteria required additional alemtuzumab treatment.
Safety
Safety events were examined for participants in CARE-MS trials and TREAT-MS meeting the primary definition of HAD. Incidences of AEs/TEAEs were lower for alemtuzumab-treated participants with HAD in TREAT-MS (61.3%) relative to CARE-MS I and II (99% and 100%, respectively, Table 3). In TOPAZ, annual incidences for overall TEAEs, IARs, and infection declined over time until none was reported at Year 13 (data not shown). Incidences of serious TEAEs and infections were lower for alemtuzumab-treated participants with HAD in TREAT-MS relative to CARE-MS I and II. Thyroid events comprised the majority of autoimmune disease events reported for the CARE-MS trials and TREAT-MS. Three cases of immune thrombocytopenia were reported in CARE-MS I and five cases in TREAT-MS, while none was reported in CARE-MS II. One case of nephropathy was reported in each of CARE-MS I and II, with none in TREAT-MS. Malignancies were reported for 4 CARE-MS I participants and 1 CARE-MS II HAD participant, with none reported in TREAT-MS. Death of one participant in CARE-MS I was of an unknown cause and deemed unrelated to alemtuzumab. Deaths of two participants in CARE-MS II were due to general disorders and administrative site conditions, and cardiac disorder (atrioventricular block); both were deemed unrelated to alemtuzumab. No deaths were reported for the TREAT-MS HAD population.
Safety parameters for participants meeting the primary definition of HAD.

TEAEs are listed for HAD participants with the following criteria (primary definition): ⩾2 relapses in the year prior to baseline and ⩾1 Gd-enhancing lesion at baseline.
TOPAZ safety events were classified as TEAEs.
TREAT-MS safety events were classified as AEs.
First occurrence of AE for a participant.
Death of a participant in CARE-MS I was of an unknown cause and deemed unrelated to alemtuzumab. Deaths of two participants in CARE-MS II were due to general disorders and administrative site conditions, and cardiac disorder (atrioventricular block); both were deemed unrelated to alemtuzumab.
AE, adverse event; Gd, gadolinium; HAD, highly active disease; ITP, immune thrombocytopenia; TEAE, treatment-emergent adverse event.
Discussion
This analysis extended the 2-year CARE-MS I and II core studies14,15,19 to assess the long-term efficacy and safety of alemtuzumab in participants with HAD over 13 years, and demonstrated sustained efficacy in treatment-naïve participants (CARE-MS I) and in those with inadequate response to prior therapy (CARE-MS II). These sustained outcomes were also demonstrated for participants with HAD from the TREAT-MS real-world study, which comprised both treatment-naïve participants and participants with prior DMTs at baseline. 21 The proportion of treatment-naïve participants (16.1%, Table 1) in this interim analysis in Germany was similar to that seen in observational studies in Spain (20.4% RMS) 29 and Denmark (12.7%), 30 and lower than that observed in Italy (29.3%). 31 The wider range of DMTs received by TREAT-MS participants was in line with real-world studies with similar proportions of treatment-naïve individuals. 30
Efficacy outcomes were generally consistent between CARE-MS I and II participants with HAD, where relapse rates were significantly reduced over 2 years with alemtuzumab relative to SC IFNB-1a, with sustained reduction through 13 years. Real-world data from TREAT-MS supported these outcomes, with a similar ARR to CARE-MS II in Years 0–2 (Figure 2). This similarity between clinical trial and real-world results was also observed for participants achieving stable or improved EDSS scores, for those achieving 6-month CDI (Figures 3 and 5), and for participants free of 6-month CDW (Year 4: 83% CARE-MS I, 77% CARE-MS II, and 89% TREAT-MS, Figure 4). On average, there was a similar proportion of participants free of MRI disease activity in CARE-MS I and II through Year 11 (Figure 4). As TREAT-MS did not collect brain volume data, we were unable to assess BVL for participants with HAD in this study. Efficacy outcomes through 13 years for TOPAZ CARE-MS I and II participants with HAD were generally in line with those from the overall CARE-MS population, and consistent with the 9-year interim data, which also demonstrated sustained efficacy of alemtuzumab in the HAD population. 19 The long-term follow-up of alemtuzumab treatment in this study is in contrast to the short-term efficacy and safety outcomes evaluated in most other DMTs for RRMS,32–34 and further consolidates the sustained benefits of alemtuzumab in highly active RRMS.
Participant-reported outcomes, assessed by EQ-VAS, were similar for CARE-MS II and TREAT-MS (Figure 9(b) and (c)). CARE-MS I participants with HAD demonstrated higher mean VAS at baseline and through Year 6 (Figure 9(a)). There was a trend toward improved household productivity relative to baseline in TOPAZ, using pooled data from CARE-MS I and II participants completing a modified HRPQ (Figure 10(a)), and a stronger trend toward improved work productivity in TREAT-MS from participants completing the WPAI questionnaire (Figure 10(c)–(e)). We were unable to assess work productivity in TOPAZ due to limited participation. Results from these participant-reported outcomes suggested a beneficial effect of alemtuzumab treatment on quality of life and productivity.
Treatment of participants with MS involves two distinct approaches that are considered early in the disease course. Escalation therapy begins with a low dose of the DMT that escalates to higher doses upon disease activity, thus prioritizing safety in minimizing treatment side effects. Induction therapy favors initiating higher efficacy treatment early to achieve the most beneficial long-term outcome.2,35 Findings from this study suggest induction therapy may be beneficial to participants with HAD, particularly regarding the similarities observed in the efficacy outcomes of CARE-MS I treatment-naïve participants, and the real-world population in TREAT-MS, which also comprises a treatment-naïve subpopulation. 21 This is consistent with a previous real-world study showing that participants with RRMS initiated with induction therapy had a lower long-term disability risk. 36
No new safety signals were observed in the HAD population in the CARE-MS studies or in TREAT-MS. The incidence of TEAEs in TOPAZ participants with HAD was consistent with previous findings in CARE-MS studies,14,15 with annual incidence declining over time through Year 13. Both TOPAZ and TREAT-MS had thyroid events as the predominant autoimmune disease in accordance with other alemtuzumab studies, 37 though there were no nephropathies, malignancies, or deaths reported for the real-world population with HAD.
This study was limited by the inherent differences in clinical trial and real-world studies, which may affect interpretation of results. There were also small variations between CARE-MS trials and TREAT-MS in the criteria used to define some of the efficacy outcomes (e.g., CDW/CDI, Figures 4 and 5). There was no sample size calculation in TOPAZ as the sample size was based on the elective inclusion of participants who were enrolled in, and completed at least 48 months of, the prior CARE-MS extension study (NCT00930553). The TREAT-MS study was undertaken in Germany; hence, restricting the population to a single demographic group. Due to limitations in the collection of brain volume data for TREAT-MS, we were unable to provide a comprehensive analysis of MRI disease activity in the real world.
However, one of the main strengths of this study is the combination of both real-world and clinical trial data from large populations consisting of both treatment-naïve participants and those with prior therapy. The extended study period of 13 years is critical in providing a better understanding of the long-term risk/benefit profile of alemtuzumab. Further, the main efficacy analyses using the primary definition of HAD are supported with sensitivity analyses with two alternative definitions (Table 2), which provide further insight into the effect of different HAD definitions on clinical outcome.
Conclusion
In the TOPAZ study, we demonstrated sustained efficacy of alemtuzumab in clinical and radiological outcomes in participants with highly active RRMS through 13 years, consistent with the overall population in TOPAZ. 24 These clinical trial findings were supported by real-world evidence from TREAT-MS with no new safety signals, further supporting alemtuzumab as an effective long-term treatment for RRMS participants with HAD.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241306575 – Supplemental material for Long-term efficacy and safety of alemtuzumab in participants with highly active MS: TOPAZ clinical trial and interim analysis of TREAT-MS real-world study
Supplemental material, sj-docx-1-tan-10.1177_17562864241306575 for Long-term efficacy and safety of alemtuzumab in participants with highly active MS: TOPAZ clinical trial and interim analysis of TREAT-MS real-world study by Tjalf Ziemssen, Ann D. Bass, Bart Van Wijmeersch, Sara Eichau, Stephan Richter, Frank Hoffmann, Nicole M. Armstrong, Magdalena Chirieac, Janete Cunha-Santos and Barry A. Singer in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
