Abstract
Background:
Cerebral small vessel disease is the most common cause of lacunar strokes (LS). Understanding LS pathogenesis is vital for predicting disease severity, prognosis, and developing therapies.
Objectives:
To research molecular profiles that differentiate LS in deep brain structures from those in subcortical white matter.
Design:
Prospective case–control study involving 120 patients with imaging-confirmed LS and a 120 control group.
Methods:
We examined the relationship between Alzheimer’s disease biomarkers [amyloid beta (Aβ1–40, Aβ1–42)], serum inflammatory marker (interleukin-6, IL-6), and endothelial dysfunction markers [soluble tumor necrosis factor-like weak inducer of apoptosis, and pentraxin-3 (sTWEAK, PTX3)] with respect to LS occurring in deep brain structures and subcortical white matter. In addition, we investigated links between LS, leukoaraiosis presence (white matter hyperintensities, WMHs), and functional outcomes at 3 months. Poor outcome was defined as a modified Rankin scale >2 at 3 months.
Results:
Significant differences were observed in levels of IL-6, PTX3, and sTWEAK between patients with deep lacunar infarcts and those with recent small subcortical infarcts (20.8 versus 15.6 pg/mL, p < 0.001; 7221.3 versus 4624.4 pg/mL, p < 0.0001; 2528.5 versus 1660.5 pg/mL, p = 0.001). Patients with poor outcomes at 3 months displayed notably higher concentrations of these biomarkers compared to those with good outcomes. By contrast, Aβ1–40 and Aβ1–42 were significantly lower in patients with deep LS (p < 0.0001). Aβ1–42 levels were significantly higher in patients with LS in subcortical white matter who had poor outcomes. WMH severity only showed a significant association with deep LS and correlated with sTWEAK (p < 0.0001).
Conclusion:
The pathophysiological mechanisms of lacunar infarcts in deep brain structures seem different from those in the subcortical white matter. As a result, specific therapeutic and preventive strategies should be explored.
Keywords
Introduction
Lacunar strokes (LS) constitute approximately 20–30% of all acute ischemic strokes and exhibit a recurrence rate of 20%, a 5-year mortality rate of 25%, and associated complications such as vascular cognitive impairment, as well as changes in gait and behavior.1 –3 These strokes typically result from the constriction or obstruction of small blood vessels that provide blood to the deep or subcortical regions of the brain. Given their small size and the specific brain regions they tend to affect, LS typically results in precise neurological symptoms and signs corresponding to lacunar syndromes. The primary cause of these vascular obstructions is cerebral small vessel disease (cSVD), with a common subtype being lipohyalinosis characterized by the deposition of hyaline material in the perforating arteries alongside diffuse arteriopathy. This condition is frequently linked to high blood pressure, diabetes, smoking, and the natural process of aging.4 –6 Atherosclerosis also plays a significant role in nearly 20% of LS cases. 7 Less common causes encompass blood clots (found in less than 10% of LS cases), vasculitis or inflammation of blood vessels, and vascular abnormalities associated with genetic mutations, including CADASIL, CARASIL, and COL4A1 mutations.8,9
Significant breakthroughs have emerged in the treatment of large vascular occlusions, which represent the majority of stroke cases, while focused therapy for LS has seen limited advancement.10,11 Consequently, advancing our understanding of LS pathogenesis could bring us closer to the attainable goal of targeted therapeutics for cSVD. The development of biomarkers specific to LS and cSVD holds immense importance for predicting disease severity, prognosis, halting disease progression, and identifying potential treatment targets. In this context, a meta-analysis study has pinpointed three clinically relevant mechanisms in the pathogenesis of LS.11,12 The researchers observed that LS patients exhibited altered serum levels of inflammatory, endothelial dysfunction, and coagulation/fibrinolysis markers, including but not limited to von Willebrand factor, homocysteine, angiogenic T cells, alkaline phosphatase, and C-reactive protein (CRP) when compared to other stroke subtypes or healthy individuals. Beyond these three categories of biomarkers, there is ongoing evaluation of novel lipid-based plasma biomarkers such as glucosylceramide, phosphatidylethanolamine, free fatty acids, and triacylglycerol.13,14 It is worth noting that these research findings underscore the complexity of interpreting biomarker results, primarily due to the diverse nature of stroke and the multitude of influencing variables.
In this context, our investigations have unveiled that elevated levels of serum inflammatory molecules, such as interleukin (IL-6) and tumor necrosis factor-alpha (TNF-α), may serve as predictive indicators for unfavorable outcomes in patients with LS, potentially leading to early neurological deterioration.15,16 Previous research conducted by our team has also established a link between increased concentrations of pentraxin-3 (PTX3), an acute phase reactant (APR), and adverse prognosis, along with elevated mortality rates in individuals with ischemic stroke.17,18 Reactive astrocytes have been posited as the source of PTX3 secretion, where they act on cerebral endothelial cells to contribute to the maintenance of blood–brain barrier (BBB) stability following an ischemic stroke. In addition, our recent findings have shown a higher prevalence of leukoaraiosis [white matter hyperintensities (WMHs) in T2-FLAIR magnetic resonance images] in symptomatic LS patients, and this was associated with the presence of the soluble TNF-like weak inducer of apoptosis (sTWEAK). 19 sTWEAK is a cytokine from the TNF superfamily, with implications for cardiovascular prognosis and endothelial dysfunction.
Likewise, endothelial dysfunction resulting from the deposition of amyloid beta (Aβ) has been associated with LS and WMHs caused by cerebral amyloid angiopathy.20,21 WMHs are intricately connected to the pathological progression of cSVD, which serves as a precursor to more severe neurological conditions, including early dementia, Alzheimer’s disease, LS, as well as ischemic and hemorrhagic strokes. 22 The interplay between LS and endothelial dysfunction is characterized by its complexity and multifaceted nature, and ongoing research endeavors aim to provide deeper insights into the underlying mechanisms. Therefore, given that WMHs have been observed in a significant proportion of individuals with Alzheimer’s disease, we propose a hypothesis suggesting a potential connection between Alzheimer’s disease markers and subcortical vascular involvement. In addition, we postulate that blood biomarkers that could be directly associated with the presence/development of WMHs, which are linked to arterial hypertension, could serve as risk factors for the development of deep vascular manifestations. In a prospective cohort study involving patients with confirmed imaging of WMHs, our primary objective is to assess serum levels of markers associated with Alzheimer’s disease, systemic inflammation, and endothelial dysfunction in both deep brain structures and small infarcts within the subcortical white matter. In addition, we investigated risk factors associated with WMHs, including cardiovascular factors.
Materials and methods
Standard protocol approvals, registrations, and patient’s consent
This prospective case–control study was conducted in accordance with the Declaration of Helsinki of the World Medical Association (2008) and approved by the Ethics Committee of the Servizo Galego de Saúde (ID: 2016/399). Informed written consent was obtained from each patient after a full explanation of the procedures. All patients were treated by expert neurologists according to national and international guidelines in the Stroke Unit of the University Clinical Hospital of Santiago de Compostela (Spain). The Strengthening the Reporting of Observational Studies in Epidemiology guidelines were included in our research. 23
Outcome endpoints
The main objective of this study was to analyze the association between serum levels of IL-6, Alzheimer’s disease biomarkers (Aβ1–40, Aβ1–42), and endothelial dysfunction markers (sTWEAK, PTX3) with lacunar infarcts in deep brain structures and small infarcts in subcortical white matter. Our secondary objectives included evaluating the connection between LS, WMH presence, and functional outcomes at 3 months.
Inclusion and exclusion criteria
Inclusion criteria for this analysis were as follows: (1) authorization for the anonymous use of individuals’ data for research purposes; (2) patients with clinical lacunar syndrome of <6 h from the onset of symptoms; (3) clinical LS syndrome with an anatomically corresponding lacunar infarct confirmed on magnetic resonance imaging/computed tomography (MRI/CT; radiographic criteria for cSVD); and (4) follow-up (face-to-face or telephone) within a minimum of 3 months. Exclusion criteria were as follows: (1) age <18 years; (2) comorbidity with a life expectancy of less than half a year; (3) lost to follow-up; (4) no CT or MRI image confirmation of lacunar infarct in a second study performed during the first week after admission; (5) immunomodulatory medication; (6) acute or chronic inflammatory disease; and (7) decline to participate in the study.
The final number of included LS patients was 120 according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification, 24 and 120 healthy controls without any type of cSVD matched by age and sex among patients’ relatives. Control subjects were excluded if they had a history of neoplasia, acute or chronic inflammatory disease, brain or cardiovascular disease, or cognitive impairment (any persistent and present clinical neurological manifestation at the time of inclusion).
Chronic inflammatory disease involves all patients with autoimmune diseases or processes associated with an inflammatory response of more than 6 months of evolution. An acute inflammatory response is associated with the presentation of clinical manifestations of inflammation (tumor, pain, flushing, heat) or infectious processes in the last 6 months. For both cases and controls, demographic and medical information were obtained employing a questionnaire.
Clinical variables
The study includes demographic variables, vascular risk factors, blood pressure, and serum biomarkers. At admission, neuroimaging and ultrasonographic evaluations were completed. The protocol for the neurological exam has been written up in detail elsewhere (including T1-weighted, T2-weighted, DP-weighted, and FLAIR).25,26 Body mass index (BMI) was calculated using the following formula: weight (kg)/height (m2) and classified according to the World Health Organization cut-off points. Hypertension was defined as a systolic blood pressure >140 mmHg and/or diastolic blood pressure >90 mmHg, or treatment with antihypertensive drugs. Hyperlipidemia was defined as serum cholesterol level >240 mg/dL, or treatment with lipid-lowering medication. 27 A modified Rankin scale (mRs) was used to evaluate the outcome at 3 months. 25 Stroke etiology was classified according to TOAST criteria. Poor outcome was defined as mRs >2 at 3 months.
Neuroimaging studies
LS was diagnosed when the patient had one of the clinical lacunar syndromes lasting, no evidence of cortical dysfunction, and a normal deep focal infarction with a diameter ⩽15 mm or subcortical infarct <20 mm, in an appropriate location visualized by CT/MRI [Figure 1(a) and (b)]. Silent infarcts, or asymptomatic brain infarcts, were defined as the presence of a lacunar infarct on the baseline CT when the topography did not match the current clinical condition. 28 The presence and severity of leukoaraiosis (WMHs) were assessed using Fazekas’ scale29,30 by MRI/CT [CT was performed in 64 (53.3%) patients, MRI in 56 (46.7%)]. The presence of an atheroma plaque was defined as a focal structure that encroaches into the arterial lumen by at least 0.5 mm or 50% of the surrounding intima-media thickness value or exhibits a thickness >1.5 mm as measured from the media–adventitia interface to the intima–lumen interface. The evaluation of atheromatous involvement was conducted using ultrasonography at the supra-aortic trunk level. Patients eligible for angioplasty or endarterectomy underwent examinations, including CT angiography, MR angiography, or carotid arteriography, based on the year and the neuroradiologist’s recommendation. For individuals with suspected embolic heart disease, transthoracic echocardiography was conducted, and in specific cases, transesophageal echocardiography and a Holter study (ranging from 48 h to 1 week) were performed on an outpatient basis. Expert neuroradiologists, blinded to clinical data, performed neuroimaging evaluations and classification of WMHs deep brain structures and subcortical white matter. 26
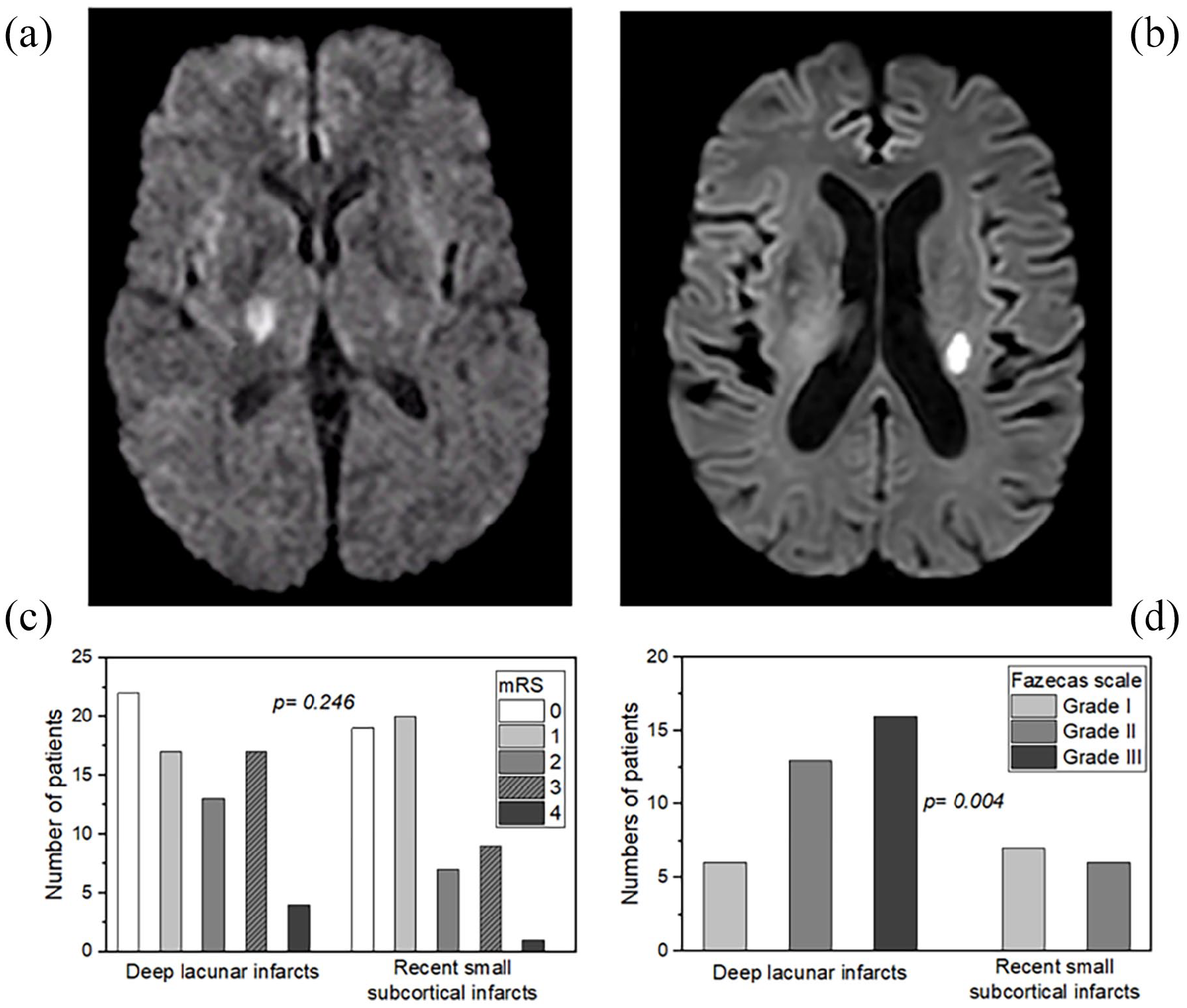
Example of MRIs obtained during the first week after admission (a) deep lacunar infarct, (b) recent small subcortical infarcts. (c) Distribution of WMH patients in both groups concerning the mRs score at 3 months ± 15 days. (d) Correlation between the presence/degree of WMHs and deep lacunar infarcts versus recent small subcortical infarcts patients.
Biomarkers
Circulating biomarkers were selected based on existing evidence from experimental and clinical studies. The patient group had fasting blood samples collected within the first 48 hours after admission, while the control group had fasting samples collected at any time during the study inclusion period. After extraction, serum samples were immediately frozen and stored at −80°C until the assay. Serum levels of all biomarkers were measured by enzyme-linked immunosorbent assay (ELISA) technique following manufacturer instructions. IL-6 ELISA kit (Proteintech™, Manchester, UK) minimum assay sensitivity was 3.8 pg/mL with an intra-assay coefficient of variation (CV) of 5.0% and inter-assay CV of 6.4%; PTX3 ELISA kit (Rockland Antibodies and Assays, Limerick PA, USA) minimum assay sensitivity was 10 pg/mL, with an intra-assay CV of 6.9% and inter-assay CV of 7.0%; sTWEAK ELISA (Thermo Scientific, MA, USA) kit minimum assay sensitivity was 10 pg/mL, with an intra-assay CV of 5.0% and interassay CV of 2.3%; Aβ1–40 ELISA kit (Elabscience®) minimum assay sensitivity was 9.38 pg/mL, with an intra-assay CV of 4.8% and inter-assay CV of 6.5%; and Aβ1–42 ELISA kit (Elabscience®) minimum assay sensitivity was 9.38 pg/mL, with an intra-assay CV of 6.0% and inter-assay CV of 6.8%. Determinations were performed in the Clinical Neurosciences Research Laboratory.
Statistical analyses
For the descriptive study, categorical variables were described with frequency and percentage. Quantitative variables were described with the mean ± standard deviation or median and interquartile range according to the type of distribution measured by the Kolmogorov–Smirnov test for a sample with the significance correction of Lilliefors. The significance of the differences was estimated using the χ2 test, Student’s t-test, or Mann–Whitney U test according to the nature of the contrast variable and its adjustment to normality. Analysis of variance was used to compare differences between more than two groups. Pearson’s correlation coefficient (r) was used to correlate biomarkers. Receiver operating curve (ROC) analysis was done to illustrate the capability of IL-6, sTWEAK, PTX3, Aβ1–40, and Aβ1–42 to diagnose deep lacunar infarcts versus recent small subcortical infarcts patients. Logistic regression analyses were performed to identify those variables independently associated with lacunar infarcts in deep brain structures or recent small subcortical infarcts. The results were expressed as odds ratio (OR) with 95% confidence intervals (95% CI). A p value <0.05 was considered statistically significant. All statistical analyses were performed with SPSS V.21.0 (IBM, USA).
Results
Sample description
The mean age was 66.5 ± 10 years in the LS cases and 64.9 ± 10 in the controls. Within the LS group, 73 (60.8%) were classified as deep lacunar infarcts: 49 (67.1%) pure motor stroke, 15 (20.5%) pure sensory stroke, 5 (6.8%) mixed sensorimotor stroke, 3 (4.2%) dysarthria/clumsy hand, and 1 (1.4%) ataxic hemiparesis. Besides, 28 (34.4%) showed silent brain infarcts and 35 (47.9%) WMHs. On the other hand, 47 (39.2%) were classified as infarcts of subcortical white matter: 22 (46.8%) pure motor stroke, 11 (23.4%) pure sensory stroke, and 14 (29.8%) mixed sensorimotor stroke. Besides, 19 (40.4%) showed silent brain infarcts and 13 (27.7%) WMHs. The univariate analysis of clinical and molecular variables obtained from both groups (Table 1) showed that arterial hypertension, diabetes, hyperlipidemia, heart failure, and carotid atheromatosis were significantly higher in LS patients. As expected, they also took more statins than non-LS subjects. No significant differences between groups were observed regarding age, sex, BMI, smoking habit, and alcohol consumption. When analyzing molecular variables, the serum inflammatory IL-6 (5.4 ± 1.2 versus 18.7 ± 6.6 pg/mL, p < 0.0001), and endothelial dysfunction markers sTWEAK (828.6 ± 519.9 versus 6204.1 ± 3072.3 pg/mL, p < 0.0001), PTX3 (557.0 ± 296.3 versus 2188.5 ± 1427.2 pg/mL, p < 0.0001), Aβ1–40 (33.1 ± 4.8 versus 39.3 ± 18.9 pg/mL, p = 0.001), Aβ1–42 (48.7 ± 9.9 versus 62.1 ± 26.9 pg/mL, p < 0.0001) were higher in the LS group.
Univariate analysis of variables obtained among controls and clinical lacunar syndrome patients.

Aβ1–40, amyloid beta-protein 1–40; Aβ1–42, amyloid beta-protein 1–42; IL-6, interleukin 6; PTX3, pentraxin-related protein PTX3; sTWEAK, soluble tumor necrosis factor-like weak inducer of apoptosis; WMH, white matter hyperintensities.
Deep lacunar versus subcortical white matter infarcts
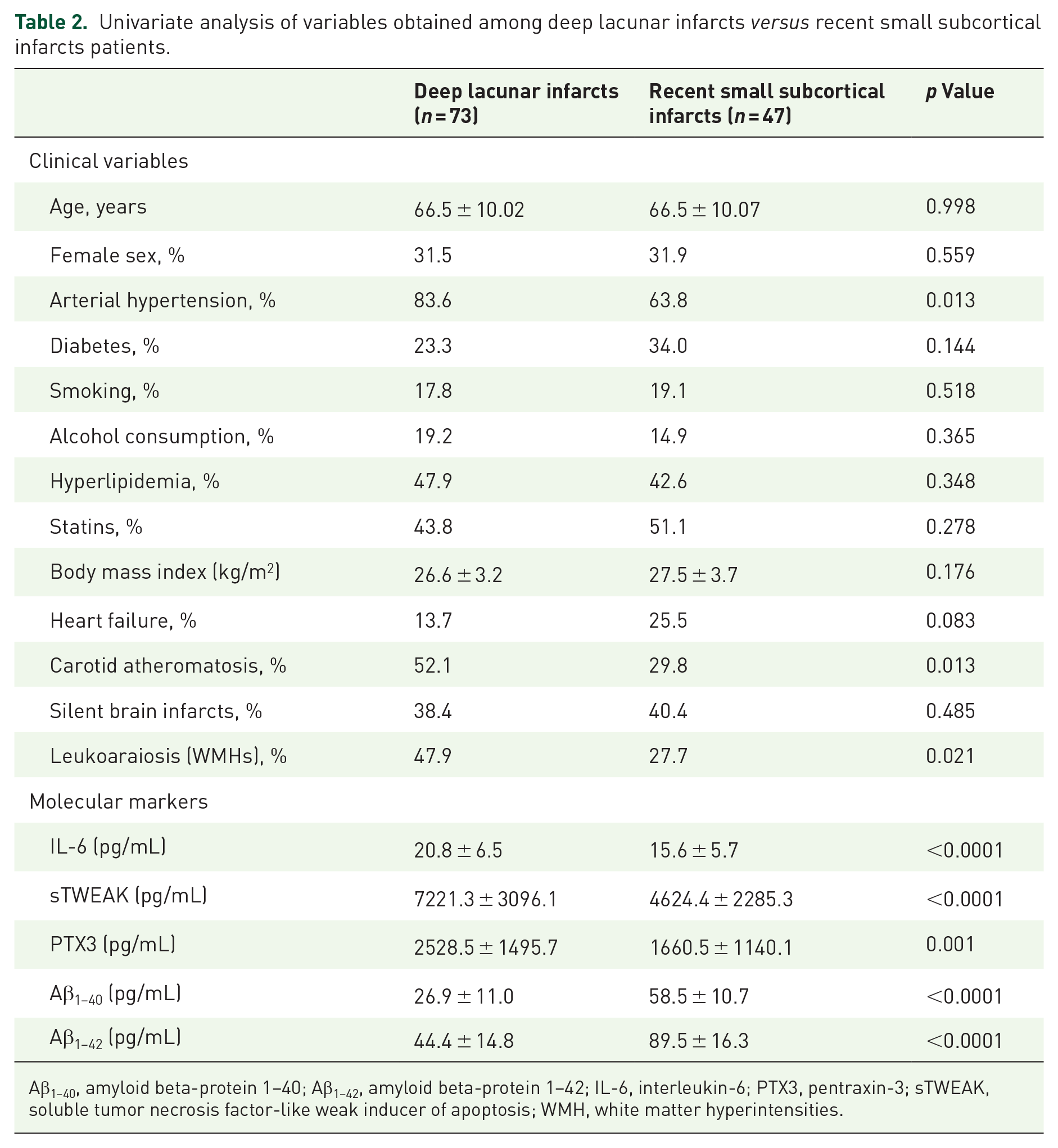
Analogous to the analysis performed for LS versus controls, a univariate analysis was carried out for deep lacunar infarcts versus recent small subcortical infarcts (Table 2). Deep LS patients had a higher frequency of hypertension and carotid atheromatosis compared to the other group (83.6 versus 63.8%, p = 0.013; 52.1 versus 27.7%, p = 0.021, respectively). No differences were observed regarding age, sex, diabetes, hyperlipidemia, heart failure, silent brain infarcts, BMI, smoking habits, and alcohol consumption. A neuroimaging variable, WMH, was observed in 47.9% of patients who suffered from deep lacunar infarcts, whereas this percentage decreased to 27.7% in those who developed recent small subcortical infarcts (p = 0.021). No significant differences between LS groups were observed regarding mRs at 3 months ± 15 days (p = 0.246). At 3 months, 29% of LS patients in the first group had a poor functional outcome, and 21% of patients in the other group [p = 0.246; Figure 1(c)]. When the degree of leukoaraiosis (WMHs) was included in the analysis, degree III was seen to be predominant in the presence of deep LS, while degree I was more prevalent [p = 0.004; Figure 1(d)] in recent small subcortical infarcts patients.
Univariate analysis of variables obtained among deep lacunar infarcts versus recent small subcortical infarcts patients.
Aβ1–40, amyloid beta-protein 1–40; Aβ1–42, amyloid beta-protein 1–42; IL-6, interleukin-6; PTX3, pentraxin-3; sTWEAK, soluble tumor necrosis factor-like weak inducer of apoptosis; WMH, white matter hyperintensities.
Association between serum levels of IL-6, Alzheimer’s disease (Aβ1–40, Aβ1–42), and endothelial dysfunction markers (sTWEAK, PTX3) with deep lacunar infarcts versus recent small subcortical infarcts
Serum levels of IL-6, PTX3, and sTWEAK were significantly elevated in patients with deep lacunar infarcts compared to the other group (20.8 ± 6.5 versus 15.6 ± 5.7 pg/mL, p < 0.001; 7221.3 ± 3096.1 versus 4624.4 ± 2285.3 pg/mL, p < 0.0001; 2528.5 ± 1495.7 versus 1660.5 ± 1140.1 pg/mL, p = 0.001, respectively). On the contrary, Aβ1–40, and Aβ1–42 serum concentrations were significantly lower in deep LS patients (26.9 ± 11.0 versus 58.5 ± 10.7 pg/mL, p < 0.0001, 44.4 ± 14.8 versus 89.5 ± 16.3 pg/mL, p < 0.0001, respectively) (Table 2).
According to ROC curve analysis, the serum levels of IL-6, sTWEAK, PTX3, Aβ1–40, and Aβ1–42 for deep lacunar infarcts showed an area under the curve (AUC) of 0.922; 95% CI: 0.890–0.953; p < 0.0001, 0.928; 95% CI: 0.898–0.958; p < 0.0001, 0.864; 95% CI: 0.811–0.916; p < 0.0001, 0.249; 95% CI: 0.178–0.231; p < 0.0001, and 0.321; 95% CI: 0.248–0.395; p < 0.0001, respectively [Supplemental Figure 1(A)–(E)].
On the other hand, the results of the ROC curve analysis of these biomarkers for recent small subcortical infarcts obtained an AUC of 0.727; 95% CI: 0.667–0.787; p < 0.0001, 0.714; 95% CI: 0.653–0.775; p < 0.0001, 0.675; 95% CI: 0.603–0.747; p < 0.0001, 0.997; 95% CI: 0.993–1.000; p < 0.0001, and 0.979; 95% CI: 0.959–0.998; p < 0.0001, respectively [Supplemental Figure 1(F)–(J)].
Regression analyses
First, logistic regression models were used to assess the statistical significance of the biomarkers (IL-6, sTWEAK, PTX3, Aβ1–40, and Aβ1–42) between patients with lacunar infarctions and the control group (Supplemental Table 1). We observed that the levels of sTWEAK, PTX3, Aβ1–40, and Aβ1–42 (but not IL-6) are higher in patients with lacunar infarcts than in the control group. When all biomarkers are included in the logistic regression model, only sTWEAK and Aβ1–42 levels maintain statistical significance after adjusting for all the different clinical variables between controls and lacunar infarcts.
Second, the association of IL-6, sTWEAK, and PTX3 with the presence of deep lacunar infarcts was evaluated using logistic regression analysis (Table 3). After adjusting the logistic regression model for arterial hypertension, carotid atheromatosis, WMHs, and each biomarker separately, the following associations were observed:
Model 1: Arterial hypertension (OR: 3.17; 95% CI: 1.21–8.35; p = 0.019) and IL-6 (OR: 1.15; 95% CI: 1.06–1.24, p < 0.0001) were independently associated with the presence of deep lacunar infarcts.
Model 2: Arterial hypertension (OR: 3.26; 95% CI: 1.20–8.84; p = 0.020), carotid atheromatosis (OR: 2.6; 95% CI: 1.06–6.35; p = 0.037), and sTWEAK (OR: 1.00; 95% CI: 1.00–1.01, p < 0.0001) were significantly associated with the presence of deep lacunar infarcts.
Model 3: Arterial hypertension (OR: 2.69; 95% CI: 1.06–6.78; p = 0.037) and PTX3 (OR: 1.00; 95% CI: 1.00–1.01, p < 0.0001) were also independently associated with the presence of deep lacunar infarcts (Table 3).
Finally, a logistic regression model was carried out to evaluate the association of hypertension, carotid atheromatosis, WMHs, and the previous biomarkers with the presence of deep lacunar infarcts. We found that apart from arterial hypertension and carotid atheromatosis, only sTWEAK (OR: 1.00; 95% CI: 1.00–1.01, p = 0.032) was independently associated with deep lacunar infarcts (Table 3, Model 4).
Multivariate analysis including serum biomarkers of inflammation and endothelial dysfunction.

Dependent variable: deep lacunar infarcts.
CI, confidence interval; IL-6, interleukin-6; OR, odds ratio; PTX3, pentraxin-3; sTWEAK, soluble tumor necrosis factor-like weak inducer of apoptosis; WMH, white matter hyperintensities.
A positive and strong correlation was found between sTWEAK and IL-6 [r = 0.835, p < 0.0001; Supplemental Figure 2(A)], PTX3 [r = 0.554, p < 0.0001; Supplemental Figure 2(B)], or IL-6 and PTX3 [r = 0.595, p < 0.0001; Supplemental Figure 2(C)], in patients with deep lacunar infarcts.
With regard to recent small subcortical infarcts, a logistic regression model showed that the variables of arterial hypertension (OR: 0.04; 95% CI: 0.01–0.06; p = 0.022), and Aβ1–42 (OR: 1.44; 95% CI: 1.11–1.88, p = 0.007) were significantly associated with the presence of these infarcts (Table 4).
Multivariate analysis including serum biomarkers of inflammation and endothelial dysfunction.

Dependent variable: recent small subcortical infarcts.
Aβ1–40, amyloid beta-protein 1–40; Aβ1–42, amyloid beta-protein 1–42; CI, confidence interval; OR, odds ratio; WMH, white matter hyperintensities.
Association between the functional outcome of patients with deep lacunar infarcts and recent small subcortical infarcts, and biomarkers
We found a correlation between IL-6, sTWEAK, and PTX3 in patients with deep lacunar infarcts and mRs scores at 3 months [Figure 2(a)–(c)]. Concentrations of IL-6, sTWEAK, and PTX3 were significantly elevated in patients with poor outcomes compared to those with good outcomes (23.8 ± 5.1 versus 19.6 ± 6.6 pg/mL, p = 0.010; 8704.8 ± 2759.9 versus 6622.2 ± 3030.8 pg/mL, p = 0.008; 3334.8 ± 1659.5 versus 2202.8 ± 1304.7 pg/mL, p = 0.003, respectively). However, no differences were observed in relation to Aβ1–40 and Aβ1–42 levels.

Serum levels of biomarkers of LS patients according to functional outcome at 3 months. Correlation between different biomarkers in patients with deep lacunar infarcts and mRs scores at 3 months: (a) IL-6, (b) sTWEAK, and (c) PTX3. (d) Aβ1–42 in relation to mRs scores at 3 months of recent small subcortical infarcts patients.
Regarding patients with recent small subcortical infarcts [Figure 2(d)], we observed that serum concentrations of Aβ1–42 were significantly higher in patients with poor outcomes compared to those with good outcomes (101.3 ± 11.4 versus 86.3 ± 16.1 pg/mL, p = 0.008). No differences were observed in relation to IL-6, PTX3, sTWEAK, and Aβ1–40. Finally, we conducted a logistic regression study to determine the relationship of all biomarkers (sTWEAK, PTX3, Aβ1–40, Aβ1–42) on the outcome of patients with the two types of lacunar infarctions. We found a statistically significant of the biomarkers with both locations (Supplemental Figure 3).
Influence of WMH presence and serum biomarkers in LS patients
A neuroimaging variable WMH was observed in 35 patients who suffered from deep lacunar infarcts, whereas this number decreased to 13 in those who developed recent small subcortical infarcts. We analyzed the association between LS presence and IL-6, sTWEAK, PTX3, or Aβ1–42 levels with the degree of WMHs. We found that the degree of WMH only had a significant influence on the patients with deep lacunar infarcts associated with sTWEAK (p < 0.0001), as observed in Figure 3.

Association between (a) IL-6, (b) sTWEAK, (c) PTX3, and (d) Aβ1–42 levels and WMHs severity in deep lacunar infarcts versus recent small subcortical infarcts patients.
Association with silent brain infarcts
In addition, the association of LS with silent brain infarcts presence (no versus yes) was determined through IL-6, sTWEAK, PTX3, and Aβ1–42 levels (17.9 ± 6.7 versus 20.1 ± 6.5, p = 0.088; 5781.1 ± 2994.7 versus 6861.1 ± 3107.3, p = 0.060; 1927.1 ± 1316.5 versus 2594.6 ± 1509.9, p = 0.012; 59.4 ± 25.7 versus 66.2 ± 28.4, p = 0.177 respectively), they appear to be APRs and thus show no significant relation to silent infarcts. In this sense, these results remain unchanged when deep lacunar infarcts are analyzed separately from recent small subcortical infarcts.
Discussion
In this study, we conducted a prospective case–control study involving 120 patients with LS and a control group. This clinical investigation aimed to pinpoint age-related risk factors (such as hypertension, diabetes, hyperlipidemia, heart failure, and carotid atheromatosis) associated with LS. Among the LS patients, 73 were categorized as having deep lacunar infarcts, and 47 had lacunar infarcts in the subcortical white matter.
Prior reports have indicated that the prevalence of LS increases with aging-related risk factors, but the connection with the location of the infarct has not been extensively explored.7,31 –33 When comparing patients with deep LS to those with lacunar infarcts in the subcortical white matter, we found that individuals with deep LS more commonly exhibited arterial hypertension and carotid atheromatosis. WMHs were identified in 47.9% of patients with deep lacunar infarcts, in contrast to 27.7% in the other group (p = 0.021). Degree III WMHs were predominant in the presence of deep LS, while degree I was more prevalent in patients with lacunar infarcts in the subcortical white matter (p = 0.004). However, no significant differences between the groups were observed in terms of the mRs score at 3 months. WMHs have been extensively associated with vascular irregularities, particularly cSVD. Our results align with previous clinical studies that have established a link between WMHs and lacunar infarction, as well as hemorrhagic and ischemic strokes, dementia, and Alzheimer’s disease.34 –37
Our analysis affirms a primary objective of this study; the pathophysiological mechanisms underlying lacunar infarcts in deep brain structures differ from those witnessed in subcortical white matter. We have observed that increased serum levels of IL-6, sTWEAK, and PTX3 are correlated with deep lacunar infarcts. Nevertheless, when these three markers are included in a logistic regression model alongside other clinical markers, only sTWEAK maintains a statistically significant association. A plausible hypothesis arises that IL-6 and PTX3 might be linked to elevated sTWEAK levels. Furthermore, we have established a connection between the presence and severity of WMHs and these three indicators. These findings align with previous research, where sTWEAK was associated with the presence and progression of WMHs in patients with ischemic stroke. 19 Elevated serum sTWEAK levels were also independently linked to hemorrhagic transformation and poor functional outcomes in patients with ischemic stroke who underwent reperfusion therapies.38,39
Among the potential markers under investigation, one of the central focuses is sTWEAK, a type II transmembrane glycoprotein belonging to the TNF superfamily. sTWEAK interacts with a transmembrane type I protein known as Fn14. Notably, Fn14 exhibits robust upregulation in endothelial cells, neurons, astrocytes, microglia, and progenitor cells under specific pathological conditions.40,41 Cultured endothelial cells express various tight junction proteins, matrix metalloproteinases, cytokines, and cell adhesion molecules at differing levels due to the presence of sTWEAK. Moreover, sTWEAK has been shown to influence the permeability of an in vitro model of the BBB.40,42 In this context, there is potential in inhibiting the activation of the sTWEAK-Fn14 system, which could emerge as a significant diagnostic and therapeutic option for impeding its progression and preventing the onset of other pathologies.
While our observations regarding the relationship between IL-6 or PTX3 and LS align with prior research, it is noteworthy that this connection to the location of the lesion has not been previously explored. IL-6 is recognized as a cytokine of particular significance in the acute phase of a stroke. It plays a role in stimulating the synthesis of acute-phase proteins (APPs), including CRPs and fibrinogen, primarily in the liver.43,44 Numerous authors have pointed out that an increase in IL-6 levels in the bloodstream within the first day following a stroke is associated with a decline in patients’ functional status and a larger volume of ischemic lesions. Furthermore, an IL-6 gene polymorphism, linked to heightened inflammation, is an independent risk factor for LS. In addition, both IL-6 and the concentration of TNF-α receptor 1 have been identified as predictors of the risk of recurrent vascular events in individuals who have recently experienced LS.
PTX3, as the prototype member of the long pentraxin family, is a soluble pattern recognition molecule with versatile properties. Generally, PTX3 levels rise rapidly in response to the inflammatory process affecting vascular health. Moreover, it has been demonstrated that after experimental stroke in mice, brain PTX3 expression is upregulated in the ipsilateral hemisphere, promoting brain recovery by reducing edema and enhancing BBB integrity through improved glial scar formation.17,45
We noted a significant increase in serum concentrations of Aβ1–40 and Aβ1–42 in individuals with lacunar infarcts in the subcortical white matter. However, after utilizing logistic regression models, only Aβ1–42 could be firmly linked to these LS, with no correlation to the presence or severity of WMHs. In previous reports, endothelial dysfunction attributed to Aβ deposition has been associated with LS and WMHs.20,21,46 However, the specific relationship between Aβ and different subtypes of cSVD remains unclear.
It is worth noting that plasma Aβ1–40 concentrations were independently linked to the extent of WMH in individuals with Alzheimer’s disease, mild cognitive impairment, or cerebral amyloid angiopathy. The proteolytic processing of the amyloid precursor protein leads to the generation of plasma Aβ peptides, with two common variants, Aβ1–42 and Aβ1–40, consisting of 42 and 40 amino acids, respectively. Aβ1–42 is predominantly found in plaque amyloid, while Aβ1–40 species are a major component of vascular amyloid.
Having established the symptomatic vascular link with cSVD, we proceeded to investigate its connection with functional outcomes. Our findings revealed that patients with deep LS not only exhibited elevated levels of IL-6, PTX3, and sTWEAK but also experienced more unfavorable outcomes at the 3 months. By contrast, we observed that individuals with lacunar infarcts in the subcortical white matter, accompanied by poorer outcomes, displayed significantly increased serum concentrations of Aβ1–42. This underscores the notion that the underlying pathophysiological mechanisms for these two types of lacunar infarcts differ substantially. It becomes imperative to identify suitable strategies for post-hospital care, secondary prevention, and the development of new therapeutic approaches. The control of arterial hypertension and the potential blockade of sTWEAK should be the therapeutic focus for addressing deep lacunar infarcts, whereas interventions targeting Aβ1–42 deposition may prove effective in preventing subcortical lacunar infarcts. Further research is warranted to elucidate the potential connection between sTWEAK inhibition and LS in hypertensive animal models.
Limitations
The current study comes with a set of constraints. To begin with, we concentrated on sTWEAK and did not delve into the TWEAK–Fn14 connection, which might have shed light on the underlying mechanism of action. In future investigations exploring the multifaceted metabolic pathways of sTWEAK in various diseases, it would be prudent to incorporate multiple biomarkers in prospective studies. Second, it is crucial to acknowledge that WMHs are a progressive condition influenced by an array of risk factors, rather than being associated with a single pathological process. Neuroimaging has revealed that areas corresponding to WMHs display thickened and hyalinized walls of penetrating arteries, alongside narrowing, elongation, and tortuosity of small vessels, which could potentially result in reduced cerebral blood flow and enduring damage to the BBB. 47 Third, we must recognize that serum levels of sTWEAK do not serve as a specific marker for any particular process, as patients with conditions such as multiple sclerosis, heart failure, or atherosclerosis also exhibit fluctuations in sTWEAK levels. However, in our investigation, we delved into the potential relationship between sTWEAK, WMHs, and LS. Fourth, a potential interdependent relationship among the biomarkers might exist. To address this concern, we conducted several logistic regression models, including the biomarkers individually in one set and collectively in another. Fifth, the percentage of patients exhibiting WMHs appears notably low for a cohort with cSVD stroke, especially within the subcortical group. It is possible that small embolic infarcts, particularly in this subgroup, might have been included. Despite these limitations, the study boasts several strengths, including a well-structured prospective case–control design, a sizable cohort of enrolled patients, and a dedicated focus on the assessment of specific biomarkers.
Conclusion
Our findings reveal an association between serum concentrations of IL-6, sTWEAK, and PTX3 and the occurrence of deep lacunar infarcts, as well as the presence and severity of WMHs. Notably, only sTWEAK maintains a statistically significant relationship even after adjusting for all biomarkers and clinical variables. Conversely, serum concentrations of Aβ1–42 are linked to recent small subcortical infarcts but are independent of the presence and severity of WMHs. Given that these biomarkers might function as APRs, they show no correlation with silent infarcts. Furthermore, our analysis indicates that these biomarkers have a connection with the functional consequences of the infarcts at 3 months. This suggests that the pathophysiological mechanisms underlying lacunar infarcts in deep brain structures differ from those in subcortical white matter. Consequently, it becomes crucial to identify appropriate therapeutic and preventive strategies tailored to these specific mechanisms.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241243274 – Supplemental material for Differential blood-based biomarkers of subcortical and deep brain small vessel disease
Supplemental material, sj-docx-1-tan-10.1177_17562864241243274 for Differential blood-based biomarkers of subcortical and deep brain small vessel disease by Pablo Hervella, Maria Luz Alonso-Alonso, Ana Sampedro-Viana, Manuel Rodríguez-Yáñez, Iria López-Dequidt, José M. Pumar, Alberto Ouro, Daniel Romaus-Sanjurjo, Francisco Campos, Tomás Sobrino, José Castillo, Yago Leira and Ramón Iglesias-Rey in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
