Abstract
Background:
Outcome data regarding the administration of tenecteplase (TNK) to acute ischemic stroke (AIS) patients presenting in the extended time window are limited.
Objectives:
We aimed to assess the current evidence regarding the efficacy and safety of TNK at a dose of 0.25 mg/kg for AIS treatment in the extended time window.
Design:
A systematic review and meta-analysis was conducted including all available randomized-controlled clinical trials (RCTs) that compared TNK 0.25 mg/kg versus no thrombolysis in AIS patients presenting in the extended time window (>4.5 h after last-seen-well or witnessed onset).
Data sources and methods:
Eligible studies were identified by searching Medline, Scopus, and international conference abstracts. The predefined efficacy outcomes of interest were 3-month excellent functional outcome [defined as the modified Rankin Scale (mRS) score ⩽1; primary outcome], 3-month good functional outcome (mRS ⩽ 2), 3-month reduced disability (⩾1-point reduction across all mRS scores). We determined symptomatic intracranial hemorrhage (sICH), any ICH and 3-month mortality as safety endpoints. A random-effects model was used to calculate risk ratios (RRs) and common odds ratios (cORs) with corresponding 95% confidence intervals (CIs).
Results:
Three RCTs were included comprising 556 patients treated with TNK versus 560 controls. TNK 0.25 mg/kg was associated with a higher likelihood of 3-month excellent functional outcome compared to controls (RR = 1.17; 95% CI = 1.01–1.36; I2 = 0%), whereas there was no difference regarding good functional outcome (RR = 1.05; 95% CI = 0.94–1.17; I2 = 0%) and reduced disability (adjusted cOR = 1.14; 95% CI = 0.92–1.40; I2 = 0%) at 3 months. The risks of sICH (RR = 1.67; 95% CI = 0.70–4.00; I2 = 0%), any ICH (RR = 1.08; 95% CI = 0.90–1.29; I2 = 0%) and 3-month mortality (RR = 1.10; 95% CI = 0.81–1.49; I2 = 0%) were similar between the groups.
Conclusion:
Based on data from three RCTs showing increased efficacy and a favorable safety profile of TNK in the treatment of AIS in the extended time window, continuing efforts of ongoing RCTs in the field are clearly supported.
Trial registration:
PROSPERO registration ID: CRD42023448707.
Keywords
Introduction
Tenecteplase (TNK) is a third-generation tissue plasminogen activator, which is created by the genetic modification of alteplase at three sites, allowing for higher fibrin specificity, increased resistance to plasminogen activator inhibitor 1, and improved feasibility of administration (in a single bolus injection).1 –4 Both data from randomized-controlled clinical trials (RCTs) and real-world data have shown a favorable safety and efficacy profile for TNK in the treatment of acute ischemic stroke (AIS) patients.5 –9 According to the recent European Stroke Organisation (ESO) expedited guidelines, TNK at a dose of 0.25 mg/kg may be used as a safe and equally effective alternative to alteplase for AIS patients within 4.5 h post symptom onset and this recommendation is based on strong evidence. 9 Importantly, an expert opinion suggests that TNK may even be preferred to alteplase, given the easy administration with single bolus dose. On the other hand, TNK at a dose of 0.4 mg/kg should not be offered, since it has been associated with numerically higher rates of symptomatic intracranial hemorrhage (sICH) without further improving the efficacy. 9
Patients presenting with wake-up stroke or with stroke of unknown onset represent a significant proportion of AIS patients being admitted. 10 Current evidence-based recommendations indicate that these patients should receive intravenous thrombolysis (IVT) with alteplase when certain neuroimaging criteria are fulfilled, showing either the presence of significant salvageable tissue (perfusion mismatch) or being interpreted as a temporal indicator of stroke onset (diffusion-weighted imaging/fluid-attenuated inversion recovery magnetic resonance imaging (MRI) mismatch).11,12 On the contrary, TNK for the treatment of AIS patients who present in the extended time window is not recommended by the ESO guidelines due to paucity of data. 9 Indeed, by the time of the writing of the ESO expedited recommendations on TNK, only one RCT was available addressing the use of TNK in the extended time window, the Tenecteplase in Wake-up Ischemic Stroke Trial (TWIST). 13 Yet, more recently, another RCT has been concluded and presented its final results during the ESO Conference 2023, the Tenecteplase in Stroke Patients Between 4.5 and 24 Hours (TIMELESS) trial14,15 and an additional one, the MRI-guided thrOmbolysis for Stroke bEyond Time Window by TNK (ROSE-TNK), has been published. 16
We sought to conduct a systematic review and meta-analysis with the aim to assess the current data on the efficacy and safety of TNK at a dose of 0.25 mg/kg for the treatment of AIS patients presented at the extended time window.
Methods
Standard protocol approvals, registrations, and patient consents
The prespecified protocol of the present systematic review and meta-analysis has been registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO (registration ID: CRD42023448707) and is reported according to the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 17 No ethical board approval or written informed consent by the patients were required due to the study design (systematic review and meta-analysis).
Data sources, searches, and study selection
Following the PICO format, a systematic literature search was conducted to identify available RCTs evaluating adult patients with AIS presenting at the extended time window (P: population) treated with IVT with TNK at a dose of 0.25 mg/kg (I: intervention) versus no IVT (C: comparator). Functional outcome at follow-up (O: outcome) was required for studies to be considered eligible for inclusion. The extended time window was defined as a presentation within 4.5–24 h after witnessed onset or last-seen-well, including wake-up stroke or stroke of unknown onset. The literature search was performed independently by three reviewers (LP, AHK, and AT). We searched MEDLINE, and Scopus, using search strings that included the following terms: ‘tenecteplase’ and ‘stroke’. No language or other restrictions were applied. Our search spanned from inception of each database to 12th November 2023. We additionally searched reference lists of published articles and international conference abstracts manually, to ensure the comprehensiveness of bibliography.
Observational studies, cohort studies, non-controlled studies, case series, and case reports were excluded. Commentaries, editorials, and narrative reviews were also discarded. All retrieved studies were independently assessed by the three reviewers (LP, AHK, and AT) and any disagreements were resolved after discussion with the corresponding author (GT).
Quality control, bias assessment, and data extraction
Eligible studies were subjected to quality control and bias assessment employing the Cochrane Collaboration tool (RoB 2) for RCTs. 18 Quality control and bias assessment was conducted independently by three reviewers (LP, AHK, and AT), and disagreements were settled by consensus after discussion with the corresponding author (GT).
Data extraction was performed on structured forms, including trial names, period of enrollment, inclusion criteria, patient sample, patients’ characteristics, and outcomes of interest among the intention to treat population.
Outcomes
The primary efficacy outcome of interest was excellent functional outcome at 3 months, defined as the modified Rankin Scale (mRS) score ⩽1. 9 Good functional outcome (defined as mRS ⩽ 2) and reduced disability (defined as ⩾1-point reduction across all mRS scores) at 3 months were assessed as secondary efficacy outcomes.
The primary safety outcome of interest was the risk of sICH, as defined by each study. Any ICH and 3-month mortality were also evaluated as secondary safety outcomes.
Statistical analysis
For the comparison of outcome events among patients receiving TNK versus no thrombolysis, we calculated for each dichotomous outcome of interest the corresponding risk ratios (RRs) with 95% confidence intervals (95% CI) using dichotomous inverse variance meta-analysis. For the evaluation of reduced disability at 3 months, the adjusted common odds ratio (cOR) was calculated using generic inverse variance meta-analysis. The random-effects model (DerSimonian and Laird) was used to calculate the pooled estimates. 19 The inverse variance method was selected because it performs well when the random-effects model is used for meta-analysis and may be preferable when included studies have different sample sizes. 9 Heterogeneity was assessed with the I2 and Cochran Q statistics. For the qualitative interpretation of heterogeneity, I2 values >50% and values >75% were considered to represent substantial and considerable heterogeneity, respectively. The significance level for the Q statistic was set at 0.1. Furthermore, in order to evaluate for potential sources of heterogeneity, an exploratory subgroup analysis was conducted stratified by the administration of endovascular treatment (EVT). Publication bias across individual studies was assessed when more than four studies were included in the analysis of the outcomes of interest, using both funnel plot inspection and the Egger’s linear regression test, 20 and the equivalent z test for each pooled estimate with a two-tailed p value <0.05 was considered statistically significant. All statistical analyses were performed using the R software version 3.5.0 (package: meta). 21
Results
Literature search and included studies
The flow diagram for the selection and inclusion of studies in this systematic review is presented in Figure 1. After excluding duplicates, the systematic literature database search yielded a total of 514 records. Following the initial screening process, the full texts of 12 records were retrieved, whereas 502 manuscripts were excluded because they did not fulfill the inclusion criteria of our meta-analysis. After reading the full-text articles, 10 records were further excluded (Supplemental Table 1). Additionally, five records were identified through the international conference abstracts’ search, three of which were excluded being duplicate to studies already identified through databases, whereas an additional one was excluded due to inclusion of wrong population (within 4.5 h after symptom onset) and wrong comparator (alteplase), leaving one study eligible for inclusion. Finally, we included three eligible studies13 –16 in the systematic review and meta-analysis, comprising a total of 556 patients that were treated with TNK versus 560 controls.

Flow chart of the systematic review.
ROSE-TNK was a phase II, investigator-initiated, multicenter, open-label with blinded endpoint assessment RCT, that included patients in the extended time window (within the 4.5- to 24-h time window since stroke onset) who additionally presented a MRI mismatch (diffusion-weighted imaging – fluid-attenuated inversion recovery mismatch). 16 Furthermore, patients should have a National Institutes of Health Stroke Scale (NIHSS) score between 6 and 25 at admission to be eligible for inclusion, whereas those being planned for endovascular thrombectomy were excluded. Finally, 80 patients were included in the trial and were equally randomized into receiving TNK at a dose of 0.25 mg/kg versus standard of care. The majority of the efficacy and safety outcomes were recorded at a similar rate between the two groups, whereas higher odds of early neurological improvement (defined as a decrease in NIHSS within 24 h of more than four points) were observed among the TNK-treated patients.
TWIST was a phase III, multicenter, open-label with blinded endpoint assessment RCT that included AIS patients who presented within 4.5 h of awakening with stroke symptoms (i.e. wake-up stroke). 13 No advanced neuroimaging selection was required for inclusion. A non-contrast computed tomography (CT) scan excluding ICH or large infarction [defined as hypoattenuation in more than 1/3 of the middle cerebral artery (MCA) territory] was the sole imagine prerequisite. Regarding clinical stroke severity, patients with limb weakness and a NIHSS score ⩾3 or aphasia were considered eligible for inclusion. Patients were randomized in receiving TNK at a dose of 0.25 mg/kg (n = 288) versus standard of care (no IVT; n = 290). Further administration of EVT was allowed per indications in both treatment groups. With regard to the results of TWIST, neither efficacy nor safety differences were found between the two groups.
The TIMELESS trial was a phase III, multicenter, double-blind, placebo-controlled RCT that included AIS patients presenting within the 4.5- to 24-h time window since stroke onset and who had evidence of large vessel occlusion (LVO; internal carotid artery, M1 segment, or M2 segment of the MCA) and salvageable brain tissue. 14 Salvageable brain tissue was defined as an ischemic core volume of less than 70 ml, a mismatch ratio of 1.8 or more and a mismatch volume of 15 ml or more, as demonstrated in advanced neuroimaging (CT perfusion or MRI/MR perfusion). Extensive early ischemic changes involving more than 1/3 of the MCA territory, demonstrated either on non-contrast CT (hypodensity) or perfusion imaging (Tmax > 6 s) were used to exclude patients from participation. Furthermore, patients with NIHSS score of 5 or more were considered eligible for inclusion in the trial. Patients were randomized in receiving TNK at a dose of 0.25 mg/kg (n = 228) versus placebo (n = 230), whereas EVT could be offered per indications in both treatment groups. According to the main results of the total population, efficacy and safety outcomes were similar between the groups. 15 Finally, when the patients receiving mechanical thrombectomy were separately evaluated, TNK treatment was associated with higher odds of complete recanalization at 24 h.
The characteristics of the included studies are summarized in Table 1.
Characteristics of randomized-controlled clinical trials (RCTs) included in the systematic review and meta-analysis.

Continuous variables are expressed as mean (standard deviation).
ACA, anterior cerebral artery; AIS, acute ischemic stroke; ASPECTS, Alberta stroke program early CT score; CT, computed tomography; DWI, diffusion-weighted imaging; EVT, endovascular treatment; FLAIR, fluid-attenuated inversion recovery; LVO, large vessel occlusion; MCA, middle cerebral artery; NIHSS, National Institutes of Health Stroke Scale; NR, not reported; PCA, posterior cerebral artery; TNK, tenecteplase.
Quality control of included studies
The risk of bias among the included studies was assessed by the Cochrane risk-of-bias (RoB 2) tool 18 and is presented in Supplemental Figures 1 and 2. The included studies were of excellent quality regarding the randomization process, the low proportion of missing data, the assessment, and the reporting of the outcomes. Yet, ROSE-TNK and TWIST, being open-label trials, carried a high risk of performance bias due to lack of blindness among investigators and participants.13,16 TWIST trial additionally presented minor deviations from intended interventions. 13 Finally, the TIMELESS trial presented some concerns due to minor deviations from intended intervention.14,15
Quantitative analyses
An overview of analyses for all primary and secondary, efficacy, and safety outcomes is summarized in Table 2.
Overview of analyses for efficacy and safety outcomes.
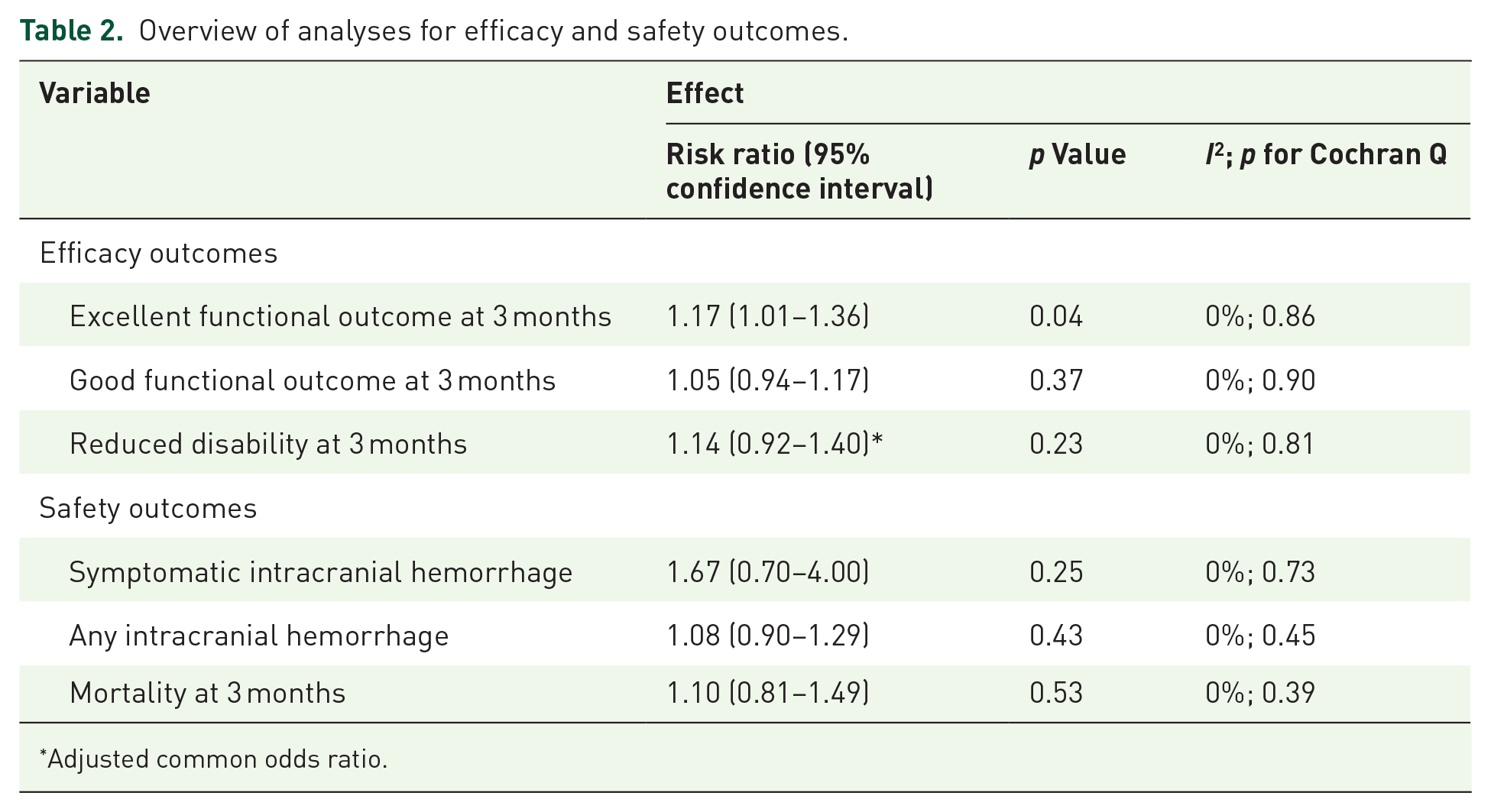
Adjusted common odds ratio.
Patients receiving TNK in the extended time window had a higher likelihood of achieving an excellent functional outcome at 3 months [RR = 1.17; 95% CI = 1.01–1.36; I2 = 0%; p for Cochran Q = 0.86; Figure 2(a)] compared to controls. There were no differences between the two groups regarding the outcomes of good functional outcome [RR = 1.05; 95% CI = 0.94–1.17; I2 = 0%; p for Cochran Q = 0.90; Figure 2(b)] and reduced disability at 3 months [adjusted cOR = 1.14; 95% CI = 0.92–1.40; I2 = 0%; p for Cochran Q = 0.81; Figure 2(c)].
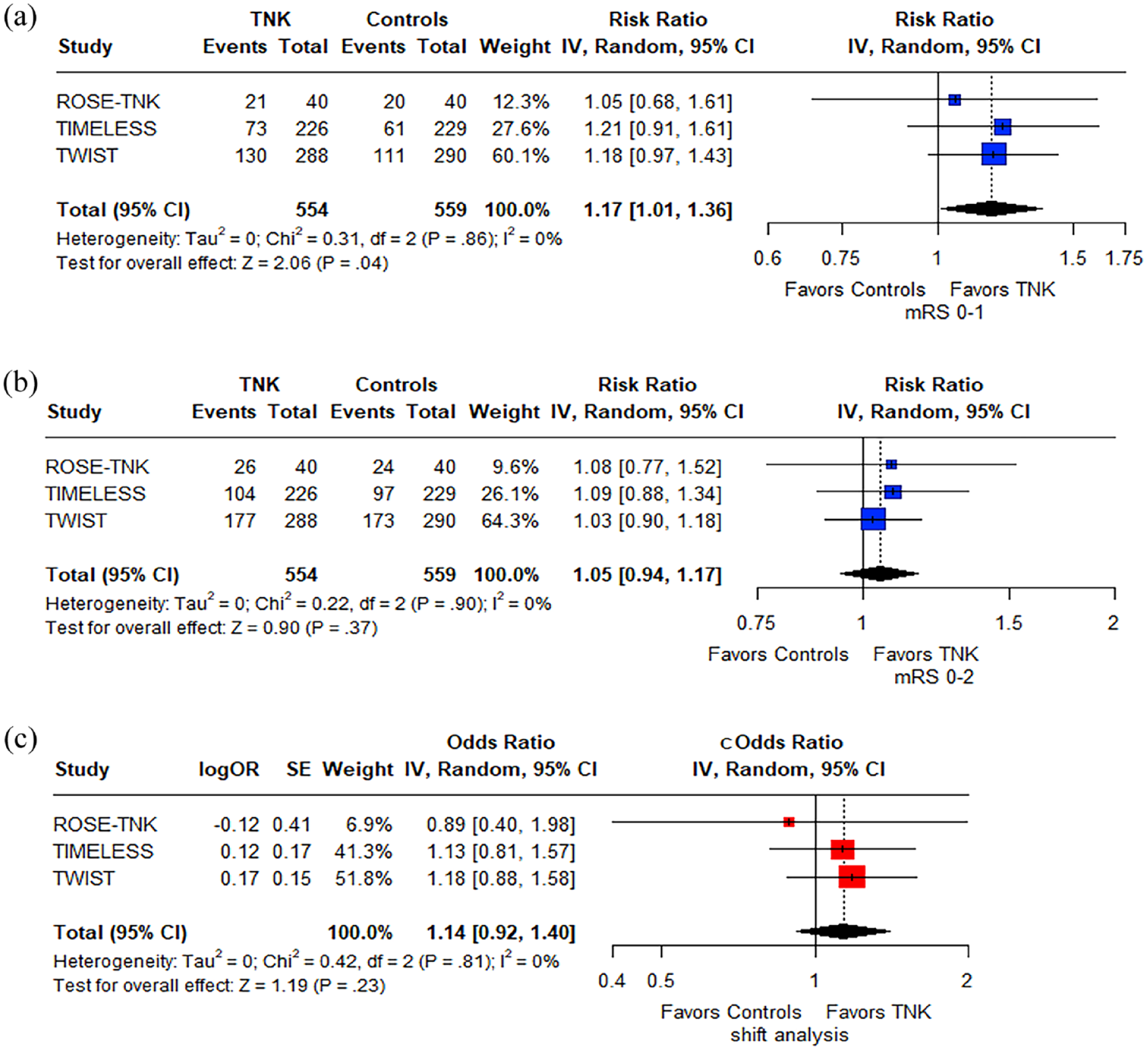
Analysis of efficacy outcomes. Forest plot presenting the association of tenecteplase versus control with excellent functional outcome (a), good functional outcome (b) and reduced disability (c) at 3 months among patients with acute ischemic stroke treated in the extended time window.
Despite that there was very low heterogeneity during analysis for all efficacy outcomes, we have further performed an exploratory subgroup analysis stratified by EVT administration, given the significant differences regarding EVT rates in the included studies (Table 1). Stratified data regarding EVT were available in both studies only for the outcome of reduced disability at 3 months. No subgroup differences were noted for the association of TNK with 3-month reduced disability among the patients who received EVT (adjusted cOR = 1.13; 95% CI = 0.80–1.61; I2 = 0%; p for Cochran Q = 0.49; Supplemental Figure 3) versus those who did not (adjusted cOR = 1.16; 95% CI = 0.89–1.51; I2 = 0%; p for Cochran Q = 0.72; Supplemental Figure 3; p for subgroup differences = 0.92).
Regarding the safety outcomes, there was no difference for sICH between the two patient groups [RR = 1.67; 95% CI = 0.70–4.00; I2 = 0%; p for Cochran Q = 0.73; Figure 3(a)]. More specifically, the pooled rate of sICH was 2% among the TNK-treated patients (95% CI = 1–4; I2 = 55%; p for Cochran Q = 0.11; Supplemental Figure 4A) and 1% in controls (95% CI = 1–3; I2 = 40%; p for Cochran Q = 0.19; Supplemental Figure 4B). Additionally, the risk of any ICH [RR = 1.08; 95% CI = 0.90–1.29; I2 = 0%; p for Cochran Q = 0.45; Figure 3(b)] and 3-month mortality [RR = 1.10; 95% CI = 0.81–1.49; I2 = 0%; p for Cochran Q = 0.39; Figure 3(c)] were also similar between the groups.

Analysis of safety outcomes. Forest plot presenting the association of tenecteplase versus control with symptomatic intracranial hemorrhage (a), any intracranial hemorrhage (b) and 3-month mortality (c) among patients with acute ischemic stroke treated in the extended time window.
Finally, evaluation for publication bias was not performed, since only three studies were included in the analysis.
Discussion
The present meta-analysis shows that IVT with TNK at a dose of 0.25 mg/kg for patients with AIS presenting in the extended time window is associated with a higher likelihood of achieving excellent functional outcome at 3 months compared to no thrombolysis, without any heterogeneity across included studies. There was no difference regarding good functional outcome and reduced disability at 3 months. Importantly, TNK treatment displayed a favorable safety profile, since the risk of sICH, any ICH and 3-month mortality were similar in the two treatment groups.
Although data regarding TNK use as an alternative thrombolytic agent in the 4.5-h window are accruing, administration of TNK in the extended time window has been investigated in a limited number of studies. 9 Patients with wake-up stroke have previously been included in the study of Tenecteplase Versus Alteplase for Thrombolysis in Acute Ischemic Stroke (NOR-TEST) trial. 22 This trial compared TNK at a dose of 0.4 mg/kg versus alteplase at the standard dose as the thrombolytic agents for patients that were eligible to receive IVT, meaning that a diffusion-weighted imaging/fluid-attenuated inversion recovery MRI mismatch was required for patients with wake-up stroke. 22 Neither in the total study population (n = 1091), nor for the small subgroup of wake-up stroke patients, that were presented as part of post hoc analysis, 23 was TNK at a dose of 0.4 mg/kg superior to alteplase. Although a higher rate of major neurological improvement within the first 24 h was shown for the wake-up stroke patients treated with TNK, this finding was severely limited by the fact that only 40 patients were included in this post hoc analysis. 23 The Norwegian Tenecteplase Stroke Trial 2 part A (NOR-TEST 2), that also investigated the efficacy and safety of TNK at a dose 0.4 mg/kg versus alteplase at a dose of 0.9 mg/kg, enrolled 16 patients, who presented in the extended time window (wake-up or stroke of unknown onset) and had a MRI mismatch. 24 This trial was prematurely terminated due to safety issues associated with bleeding complications in patients receiving TNK at a dose of 0.4 mg/kg. the TNK group had higher likelihood of sICH and 3-month mortality, whereas the rate of excellent functional outcome was lower compared to the alteplase group. No subgroup analysis has been presented for the patients presenting as wake-up or stroke of unknown origin, probably owing to the limited number of the included patients.
Due the unfavorable clinical effect of the 0.4 mg/kg dose of TNK, ESO guidelines strongly recommend against this dose as an alternative to alteplase for the 4.5-h time window, whereas the dose of 0.25 mg/kg seems to be the dose of choice for TNK in AIS. 9 Recent network meta-analyses further support this, showing that TNK at the dose of 0.4 mg/kg is at the last rank concerning efficacy in comparison to TNK 0.25 mg/kg and alteplase 0.9 mg/kg, whereas the risk of sICH was the highest with TNK 0.4 mg/kg.25,26 Yet, these analyses were mostly limited to patients presenting within the standard time window (4.5 h). Another trial, the CHinese Acute Tissue-Based Imaging Selection for Lysis In Stroke – Tenecteplase (CHABLIS-T; https://www.clinicaltrials.gov; Unique identifier: NCT04086147) aimed to specifically investigate two different doses of TNK for the treatment of extended time window AIS. Patients with LVO and perfusion mismatch that presented within 4.5–24 h post stroke onset were included in this study and were randomized to TNK 0.25 mg/kg versus a higher dose of TNK at 0.32 mg/kg. The primary outcomes for this trial were the rates of averted EVT, achieved recanalization and sICH, while 3-month clinical outcomes were assessed as secondary outcomes only. This trial has been completed, and its results were presented during the International Stroke Conference in 2022. No increase in efficacy was noted in the arm of TNK 0.32 mg/kg compared to TNK 0.25 mg/kg, and the investigators have chosen to proceed to the next RCT, CHABLIS-T II (Unique identifier: NCT04516993) comparing the dose of TNK 0.25 mg/kg to standard of care.
Data regarding the efficacy and safety of TNK 0.25 mg/kg in the extended time window when compared to no thrombolysis are even more limited and only include the three trials that are presented in this systematic review. Considering the early signs of improved efficacy, while safety remains similar between the two groups, it seems that this approach is worth for further exploration. Another trial, the Randomization to Extend Stroke Intravenous ThromboLysis In Evolving Non-Large Vessel Occlusion With TNK (RESILIENT-EXTEND-IV; https://www.clinicaltrials.gov; Unique identifier: NCT05199662) trial is ongoing and randomizing AIS patients who present within 4.5–12 h from last-seen-well, do not have LVO, and demonstrate either a clinical-radiological or a perfusion mismatch, into TNK 0.25 mg/kg versus placebo. Furthermore, the ‘Randomized Controlled Trial of TNK-tPA Versus Standard of Care for Minor Ischemic Stroke With Proven Occlusion’ (TEMPO-2; https://www.clinicaltrials.gov; Unique identifier: NCT02398656) is currently recruiting patients presenting with a minor AIS (NIHSS < 6) within a 12-h time window from symptom onset. In order for the patients to be included, an acute intracranial occlusion or near occlusion either in the anterior or the posterior circulation should be demonstrated by either (multiphase) CT angiography or CT perfusion. Patients are randomized to TNK of 0.25 mg/kg or standard antiplatelet treatment. Results of this study are expected to shed light on two challenging clinical scenarios: presentation in the extended time window (without the need for mismatch presence), but also the management of minor strokes. The latter gains even more importance, considering the recent data showing that dual antiplatelet treatment is non-inferior to IVT with alteplase for minor, non-disabling strokes. 27
When a subgroup analysis stratified by EVT administration was performed, no difference was shown regarding the association between TNK and reduced disability at 3 months among patients that received EVT versus those who did not. This finding lends support to the hypothesis that TNK may even be used in an extended time window when bridging therapy is considered. The Extending the Time Window for Tenecteplase by Effective Reperfusion in Patients With Large Vessel Occlusion (ETERNAL-LVO; https://www.clinicaltrials.gov; Unique identifier: NCT04454788) is currently enrolling LVO patients who present within 24 h of stroke onset and are eligible for EVT, aiming to investigate whether pre-treatment with TNK at a dose of 0.25 mg/kg may be beneficial in comparison to standard of care, which may include treatment with alteplase when indicated. Similarly, patients with basilar artery occlusion will be enrolled in the ongoing Extending the Time Window for Tenecteplase by Recanalization of Basilar Artery Occlusion in Posterior Circulation Stroke (POST-ETERNAL; https://www.clinicaltrials.gov; Unique identifier: NCT05105633) trial and be randomized into TNK 0.25 mg/kg versus standard of care (which may include alteplase) in the extended time window (within the first 24 h post symptom onset) and before receiving EVT.
On the other hand, the Tenecteplase Reperfusion Therapy in Acute Ischemic Cerebrovascular Events-III (TRACE-III; https://www.clinicaltrials.gov; Unique identifier: NCT05141305) aims to enroll AIS patients with LVO presenting in the extended time window (within 4.5–24 h from last-seen-well) that are not planned to receive EVT. Treatment with recombinant human TNK 0.25 mg/kg will be compared to standard treatment (antiplatelet treatment). Advanced neuroimaging criteria, mandating the presence of perfusion mismatch, are also applied in this trial for enrollment of appropriate candidates.
Based on our results and regarding the safety outcomes, it should be underscored that the pooled proportion of sICH among the patients receiving TNK was quite low (2%). The risk of sICH among the patients receiving IVT with alteplase within the standard time window ranges from 2% to 7%, 28 with similar rates presenting, when patients are being treated in the extended time window based on advanced neuroimaging. 11 Furthermore, the pooled proportion of sICH among the studies investigating TNK 0.25 mg/kg in the standard time window and included in the ESO guidelines is between 1% and 4%. 9 The low rates of sICH in AIS patients receiving IVT with TNK 0.25 mg/kg are also confirmed in the real-world setting. In a large, multicenter, retrospective, observational study by the Comparative Effectiveness of Routine Tenecteplase Versus Alteplase in Acute Ischemic Stroke (CERTAIN) collaboration, that included more than 9000 patients, it was shown that TNK treatment was complicated with sICH in 1.8%, whereas the proportion of sICH among alteplase-treated patients was 3.6%. 29 The same results persisted (i.e. lower likelihood of sICH among TNK versus alteplase), even after analysis was stratified according to EVT administration or not. This difference regarding sICH between TNK and alteplase should be expected, considering the pharmacological properties of the two thrombolytic agents: TNK has a fourfold higher fibrin selectivity compared to alteplase, resulting in a lower likelihood of systemic bleeding, whereas an effect of TNK on the blood–brain barrier has not been recorded, potentially explaining the favorable local safety profile as well (in comparison to alteplase that can lead to blood–brain barrier disruption). 30
Our present meta-analysis followed a prespecified protocol and included all available RCTs that have been completed to date, investigating efficacy and safety of TNK 0.25 mg/kg compared to no thrombolysis for the acute management of AIS patients presenting at an extended time window since symptom onset. However, there are some limitations to our study that should be acknowledged. First, our systematic search was able to detect only three RCTs available for inclusion, providing premature evidence for increased efficacy of TNK 0.25 mg/kg, while respecting safety. Our results should be interpreted as to further supporting and rationalizing the continuous efforts and investigation of this approach among ongoing RCTs. Second, the included population may be considered as heterogenous, since the three studies included patients that were selected based on different neuroimaging criteria. However, there was no heterogeneity across the three trials with regard to all prespecified safety and efficacy outcomes was very low in the assessment for all the outcomes of interest, whereas further subgroup analysis stratified for EVT administration did not disclose significant subgroup differences. Finally, one of the three included trials, the TIMELESS trial, has not presented its results within a peer-reviewed publication. The data extraction for this study was based on presented results during an international conference (ESO Conference 2023) and potential variations compared to the future publication cannot be excluded. It should be noted, though, that the protocol for this trial has been presented in a peer-reviewed publication, allowing for the quality assessment of the study to be performed without significant limitations. Of course, the results of future RCTs will provide further evidence for the use of TNK 0.25 mg/kg for IVT among AIS patients presenting in the late time window.
Conclusion
In conclusion, the current meta-analysis based on data from three RCTs provides evidence regarding the efficacy of TNK 0.25 mg/kg for the treatment of AIS patients presenting in the extended time window compared to no thrombolysis. This finding, coupled with the favorable safety profile, underscores the importance of the continuing efforts in already ongoing RCTs to provide more robust data regarding TNK 0.25 mg/kg as the thrombolytic agent in the extended time window.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231221324 – Supplemental material for Tenecteplase for the treatment of acute ischemic stroke in the extended time window: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864231221324 for Tenecteplase for the treatment of acute ischemic stroke in the extended time window: a systematic review and meta-analysis by Lina Palaiodimou, Aristeidis H. Katsanos, Guillaume Turc, Michele Romoli, Aikaterini Theodorou, Robin Lemmens, Simona Sacco, Georgios Velonakis, Charalambos Vlachopoulos and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864231221324 – Supplemental material for Tenecteplase for the treatment of acute ischemic stroke in the extended time window: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tan-10.1177_17562864231221324 for Tenecteplase for the treatment of acute ischemic stroke in the extended time window: a systematic review and meta-analysis by Lina Palaiodimou, Aristeidis H. Katsanos, Guillaume Turc, Michele Romoli, Aikaterini Theodorou, Robin Lemmens, Simona Sacco, Georgios Velonakis, Charalambos Vlachopoulos and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
