Abstract
Background:
Antiseizure medications can have negative effects on plasma lipid levels.
Objectives:
To evaluate plasma lipid changes in patients with newly diagnosed focal epilepsy treated with eslicarbazepine acetate (ESL) or controlled-release carbamazepine (CBZ-CR) monotherapy during a phase III, randomized, double-blind (DB) trial and 2 years of ESL treatment in an open-label extension (OLE).
Design:
Post hoc analysis of a phase III trial and OLE study.
Methods:
Proportions of patients with elevated levels of total cholesterol and low-density lipoprotein (LDL) cholesterol were assessed at DB baseline, OLE baseline (last visit of DB trial), and end of OLE.
Results:
A total of 184 patients received ESL monotherapy during the OLE: 96 received ESL monotherapy in the DB trial and 88 patients received CBZ-CR monotherapy. The proportions of patients with elevated total cholesterol and LDL cholesterol increased significantly during the DB trial in those treated with CBZ-CR monotherapy [total cholesterol, +14.9% (p < 0.001); LDL cholesterol, +11.5% (p = 0.012)] but decreased significantly after switching to ESL monotherapy in the OLE [total cholesterol, −15.3% (p = 0.008); LDL cholesterol, −11.1% (p = 0.021)]. No significant changes were observed in those treated with ESL monotherapy during the DB trial and OLE. At the end of the DB trial, between-group differences (ESL–CBZ-CR) in the proportions of patients with elevated total and LDL cholesterol were −13.6% (p = 0.037) and −12.3% (p = 0.061), respectively; at the end of the OLE, these between-group differences were −6.0% (p = 0.360) and −0.6% (p = 1.000), respectively.
Conclusion:
A lower proportion of patients with newly diagnosed focal epilepsy had increased levels of total and LDL cholesterol, compared to baseline, following monotherapy with ESL versus CBZ-CR; after switching from CBZ-CR to ESL, the proportions of patients with increased levels decreased significantly.
Registration:
ClinicalTrials.gov NCT01162460/NCT02484001; EudraCT 2009-011135-13/2015-001243-36.
Patients with epilepsy have an increased risk of having cardiovascular and cerebrovascular diseases (e.g., myocardial infarction and stroke). Treatment with antiseizure medications can have a negative effect on blood cholesterol levels [such as total cholesterol and low-density lipoprotein (LDL) cholesterol], which can further increase the risk of cardiovascular and cerebrovascular diseases.
We examined the impact of monotherapy treatment (i.e., treatment with only one antiseizure medication) using either eslicarbazepine acetate (ESL) or a controlled-release formulation of carbamazepine (CBZ-CR) in 184 patients with newly diagnosed focal epilepsy (ESL, 96 patients; CBZ-CR, 88 patients). Patients received monotherapy with ESL or CBZ-CR for approximately 1 year in a phase III clinical trial. After this, the patients could continue into a 2-year extension study during which they all received monotherapy with ESL. We assessed the proportions of patients with elevated levels of total cholesterol and LDL cholesterol at the beginning and end of the phase III trial, and at the end of the extension study. At the beginning of the phase III trial, the proportions of patients with elevated total cholesterol and elevated LDL cholesterol were similar between treatment groups. During the phase III trial, the proportions of patients with elevated total cholesterol and elevated LDL cholesterol increased in those treated with CBZ-CR monotherapy (total cholesterol, +14.9%; LDL cholesterol, +11.5%) but decreased after switching to ESL monotherapy in the extension study (total cholesterol, −15.3%; LDL cholesterol, −11.1%). By contrast, the proportions of patients with elevated levels of total cholesterol and LDL cholesterol remained relatively stable in those treated with ESL monotherapy during the phase III trial and extension study.
These findings indicate that ESL monotherapy may be an appropriate treatment option for patients with newly diagnosed focal epilepsy who either already have, or who are at risk of developing, high levels of cholesterol, since this may reduce their likelihood of having cardiovascular and cerebrovascular diseases.
Keywords
Introduction
Eslicarbazepine acetate (ESL) is a once-daily (QD) antiseizure medication (ASM) that is approved in Europe and the USA for the treatment of focal seizures as monotherapy or adjunctive therapy.1,2 ESL was approved by the European Medicines Agency as monotherapy based on a phase III, randomized, double-blind (DB), active-controlled, non-inferiority study (Study BIA-2093-311), which demonstrated that monotherapy treatment with ESL QD was non-inferior to monotherapy treatment with twice-daily (BID) controlled-release carbamazepine (CBZ-CR). 3
Patients with epilepsy have an increased risk of cardio- and cerebrovascular (CCV) diseases and ASMs have been shown to exert different effects on a range of CCV risk factors, including plasma lipid levels (total cholesterol, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol and triglycerides).4–9 These effects are thought to arise from the variable enzyme-inducing properties of certain ASMs, which increase a number of serological vascular risk factors, particularly cholesterol.10–13 The use of enzyme-inducing ASMs is associated with an increased risk of CCV diseases over the long term,14,15 while the use of enzyme-inhibiting ASMs has been shown to increase the risk of type 2 diabetes over the long term. 16 Choice of ASM monotherapy in the post-stroke epilepsy setting has also been shown to affect the relative risks of subsequent cardiovascular and all-cause death.17,18 Since epilepsy is a chronic condition, often requiring life-long ASM treatment, the impact of an ASM on CCV risk factors is an important consideration when choosing treatment in order to reduce the risk of stroke and myocardial infarction over the long term. 10
As a potent enzyme inducer, carbamazepine (CBZ) can lead to changes in lipid parameters via effects on enzymes involved in endogenous metabolic pathways. 19 In comparison, ESL is a less potent enzyme inducer than CBZ, and patients who have switched from CBZ to ESL have experienced improvements in lipid parameters.6,19–26 This post hoc analysis evaluated changes in plasma lipid parameters for patients treated with ESL or CBZ-CR monotherapy during a randomized DB trial comparing CBZ-CR with ESL for newly diagnosed epilepsy, 3 and during the following 2 consecutive years of ESL treatment in an open-label extension (OLE) study. 27
Methods
Study design
Study BIA-2093-311 was a phase III, randomized, DB, non-inferiority trial, in which adult patients with newly diagnosed focal epilepsy received monotherapy with ESL QD (800, 1200 or 1600 mg/day) or CBZ-CR BID (400, 800 or 1200 mg/day) over a 26-week evaluation period, followed by a 26-week maintenance period (Figure 1). 3 Patients completing the DB trial (i.e., who remained seizure-free for ⩾6 months at the last evaluated dose) were eligible to enter a 2-year OLE study, during which all patients received flexibly dosed ESL (800–1600 mg/day) (Figure 1). 27 The switching strategy used for transitioning patients from CBZ-CR to ESL at the start of the OLE study has been described previously (Trinka et al. 27 ; Supplemental Material). According to the investigator’s opinion, concomitant ASMs could be added during the OLE study, if required. 27

Study design. Figure reproduced from Trinka et al. 27 with permission from Wiley.
Study population
The DB trial included adult patients (⩾18 years) with newly diagnosed epilepsy, providing they had at least two focal seizures (with or without focal to bilateral tonic-clonic seizures) within 12 months previous to screening and at least one seizure during the previous 3 months. 3 Patients were also required to have: (i) had an electroencephalogram (EEG) and brain computerized axial tomography or magnetic resonance imaging (to exclude a progressive neurological lesion) in the previous 12 months; (ii) provided written informed consent; and (iii) demonstrated cooperation and willingness to complete the study. 3 Key exclusion criteria included: (i) indicators of generalized genetic epilepsy (based on clinical history, specific seizure types and EEG); (ii) history of a clinically significant or currently uncontrolled medical condition that would interfere with participation in the study (e.g. renal disorder, hepatic disorder, uncontrolled hypertension, psychiatric illness); and (iii) previous treatment with CBZ or oxcarbazepine. 3 For inclusion in the OLE study, patients had to participate in the DB trial, with treatment ongoing at the time of unblinding treatment allocation. 27 Key exclusion criteria for the OLE study included: (i) the presence of any major protocol violation during the initial DB trial that could impact compliance in the OLE study; (ii) suicidal risk (based on the investigator’s opinion and the Columbia Suicide-Severity Rating Scale); (iii) occurrence of an adverse event in the initial trial indicating suspected presence of atrioventricular block (second degree or above) or contraindicative to further participation (investigator’s judgment); (iv) events of alcohol/drug/medication abuse during the initial trial; (v) presence of clinically relevant laboratory abnormalities (e.g. sodium <125 mmol/L); and (vi) pregnancy or lactation. 27
Patients who required additional ASM treatment during the OLE study were excluded from the analysis described below in order to evaluate a true monotherapy population.
Study assessments
Plasma levels of total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides were assessed at baseline of the DB trial (DB baseline), at baseline of the OLE study (OLE baseline, which corresponds to the end of the DB trial), and at the end of study (EOS) in the OLE (OLE EOS).
Statistical analysis
Due to high variability in the data, it was not possible to conduct meaningful analyses of mean change in lipid parameters. Instead, an approach more aligned with clinical practice was adopted, in which the proportions of patients with high/low plasma levels of lipid parameters (according to reference laboratory values) were assessed to provide a clinically meaningful understanding of changes in lipids parameters over time. The proportions of patients with high/low plasma levels of lipid parameters (Lipid Reference Laboratory: LKF – Laboratorium für Klinische Forschung – POB 7134 – D-24171 Kiel, Germany) were calculated for the total monotherapy population, and for the subset of patients not treated concomitantly with statins [hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors], since evidence has demonstrated that some ASMs, such as CBZ and phenytoin, significantly affect the lipid-lowering effects of statins.7,28 Plasma levels were considered high for total cholesterol if > 6.22 mmol/L and normal if ⩽6.22 mmol/L. For LDL cholesterol, plasma levels were considered high if >4.14 mmol/L and normal if ⩽4.14 mmol/L. For HDL cholesterol, plasma levels were considered high if > 2.07 mmol/L, normal if ⩾1.04 and ⩽2.07 mmol/L, and low if <1.04 mmol/L. For triglycerides, plasma levels were considered high if > 1.70 mmol/L and normal if ⩽1.70 mmol/L. Qualitative data were statistically evaluated using the McNemar test (for comparison between visits inside each treatment arm) and Fisher’s test (for comparisons between treatment arms at each visit). Additional analyses were conducted to estimate the risk of developing high levels of total cholesterol or LDL cholesterol separately during the DB trial, and to assess the relative risk reduction for ESL versus CBZ-CR. These were assessed by analyzing cumulative risk (cumulative incidence), risk differences and relative risks [with 95% confidence interval (CI)] with their corresponding p values. No multiplicity adjustment of p values was conducted for any statistical testing due to the exploratory nature of the study. The standard alpha level of 0.05 was used, but statistically significant p values should be interpreted carefully and only within the context of this post hoc clinical evaluation.
Results
Study population
Of 206 randomized patients, 184 (89.3%) were treated with ESL monotherapy during the OLE study, of whom 96 had received ESL monotherapy in the DB trial (‘ESL/ESL’ treatment group) and 88 patients had received CBZ-CR monotherapy in the DB trial (‘CBZ-CR/ESL’ treatment group) (Figure 2). The remaining 22 randomized patients (10.7% of the randomized population) required concomitant ASM treatment during the OLE study and were therefore excluded from the analysis. The median (range) exposures to ESL and CBZ-CR during the DB trial were 1089 (675–1834) and 1165 (716–1904) days, respectively. During the OLE study, the median (range) exposures to ESL in the ESL/ESL and CBZ-CR/ESL treatment groups were 734 (39–769) and 730.5 (39–758) days, respectively.
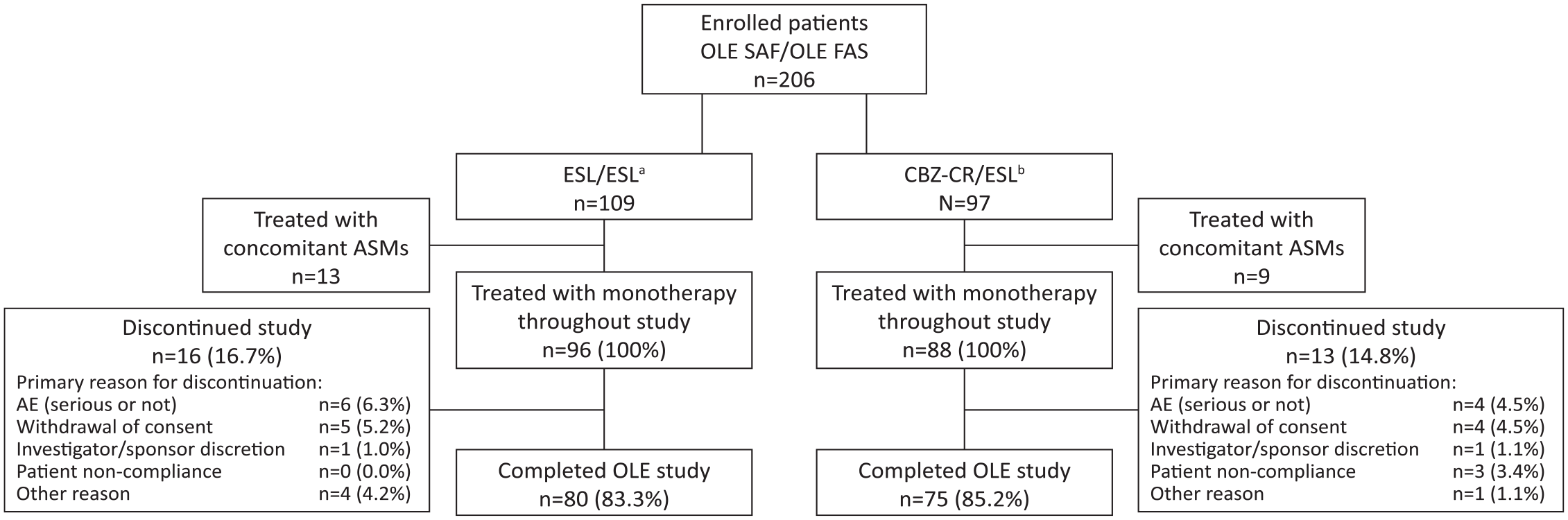
Patient disposition. Figure adapted from Trinka et al. 27 with permission from Wiley (a) Patients treated with ESL throughout the study (DB trial and OLE study). (b) Patients treated with CBZ-CR monotherapy in the DB trial and ESL in the OLE study.
The mean [standard deviation (SD)] age of the study population was 42.1 (15.8) years and 54.3% were male (Table 1). The mean (SD) age of onset of epilepsy was 39.2 (15.8) years. Patients had experienced a mean (SD) of 8.6 (21.7) seizures during the previous 3 months before DB baseline. Patient demographic and baseline characteristics were generally well balanced between treatment groups, including relevant medical conditions, such as CCV diseases. Overall, 10/96 (10.4%) patients in the ESL/ESL treatment group and 11/88 (12.5%) patients in the CBZ-CR/ESL treatment group were treated concomitantly with statins.
Demographic and baseline characteristics. Adapted from Trinka et al. 27 with permission from Wiley
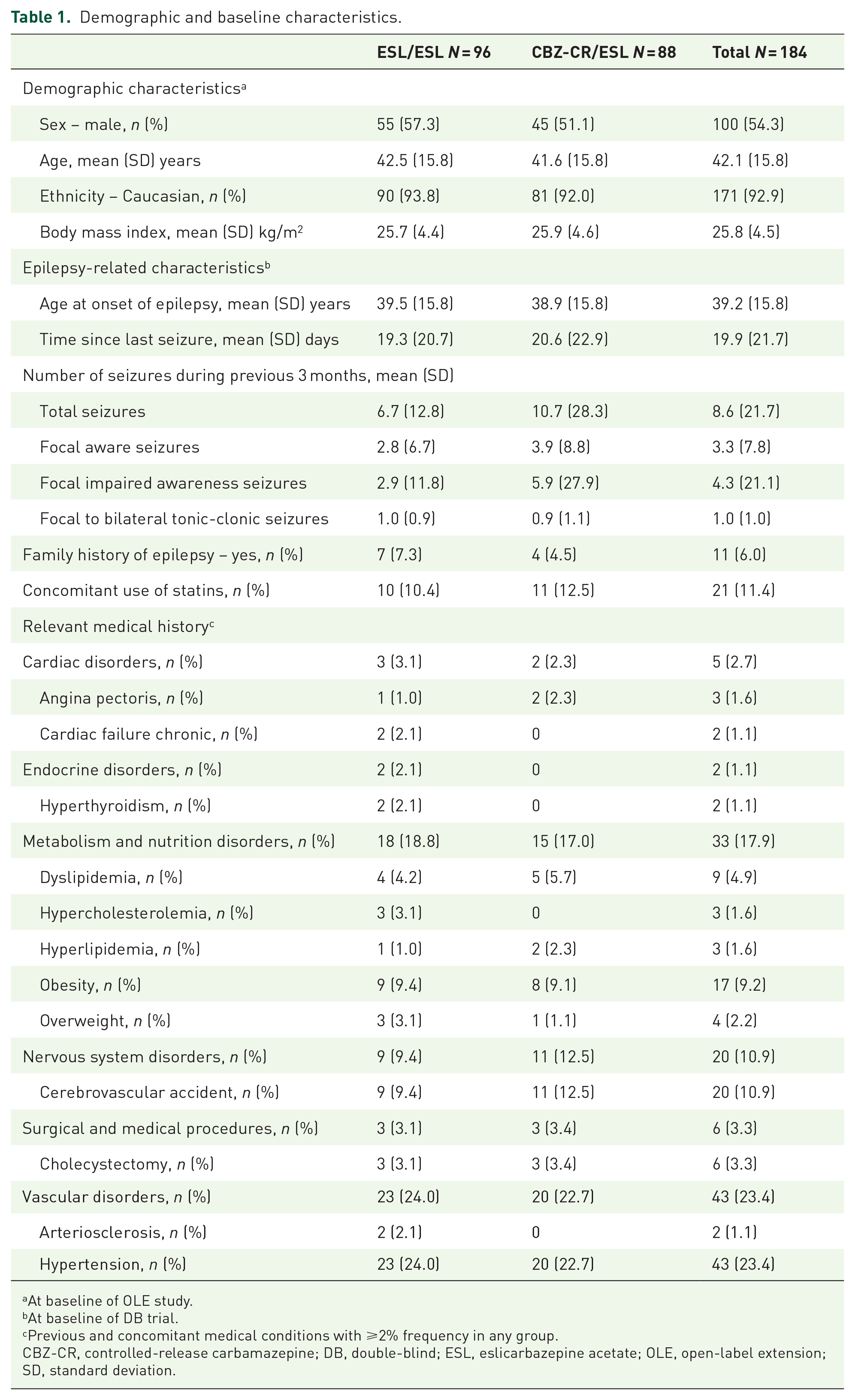
At baseline of OLE study.
At baseline of DB trial.
Previous and concomitant medical conditions with ⩾2% frequency in any group.
CBZ-CR, controlled-release carbamazepine; DB, double-blind; ESL, eslicarbazepine acetate; OLE, open-label extension; SD, standard deviation.
Changes in plasma lipid parameters
Mean (SD) levels of lipid parameters (total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides) at DB baseline, OLE baseline and OLE EOS are presented in Table 2, and the proportions of patients with high/low levels of the lipid parameters at these timepoints are presented in Table 3 and Figures 3 and 4. Mean (SD) levels of lipid parameters in the subgroups of patients with high/low levels at DB baseline, OLE baseline and OLE EOS are presented in Supplemental Table S1. At DB baseline, there was no significant difference between treatment groups in the proportion of patients with high levels of total cholesterol [Figure 3(a) and Table 3]. At the end of the DB trial (OLE baseline), the proportion of patients with high levels of total cholesterol who had been treated with CBZ-CR had increased significantly from 15.9% to 31.0% (change from DB baseline to OLE baseline, 14.9%; p < 0.001), while in patients treated with ESL the proportion of patients with high levels of total cholesterol had increased non-significantly from 11.5% to 17.4% (change from DB baseline to OLE baseline, 6.5%; p = 0.109) [Figure 3(a) and Table 3]. This resulted in a significant between-group difference (ESL–CBZ-CR) of −13.6% (p = 0.037), demonstrating that a lower proportion of patients treated with ESL had high levels of total cholesterol at OLE baseline, in comparison with those treated with CBZ-CR. In patients treated with CBZ-CR during the DB trial, the proportion of patients with high total cholesterol levels decreased significantly by OLE EOS after switching from CBZ-CR to ESL (change from OLE baseline to OLE EOS, −15.3%; p = 0.008) [Figure 4(a) and Table 3]. By contrast, the proportion of patients with high total cholesterol levels remained relatively consistent in patients in the ESL/ESL treatment group during the DB trial and OLE study, without significant changes [Figure 4(a) and Table 3]. The cumulative risk of patients developing high levels of total cholesterol during the DB trial (having not previously had high levels) was 12.2% in those treated with ESL versus 19.2% in those treated with CBZ-CR [risk difference for ESL versus CBZ-CR, −7.0%; relative risk (95% CI), 0.64 (0.30–1.34); p = 0.235; Table 4(a)].
Mean (SD) levels of total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides at DB baseline, OLE baseline and OLE EOS (total monotherapy population).
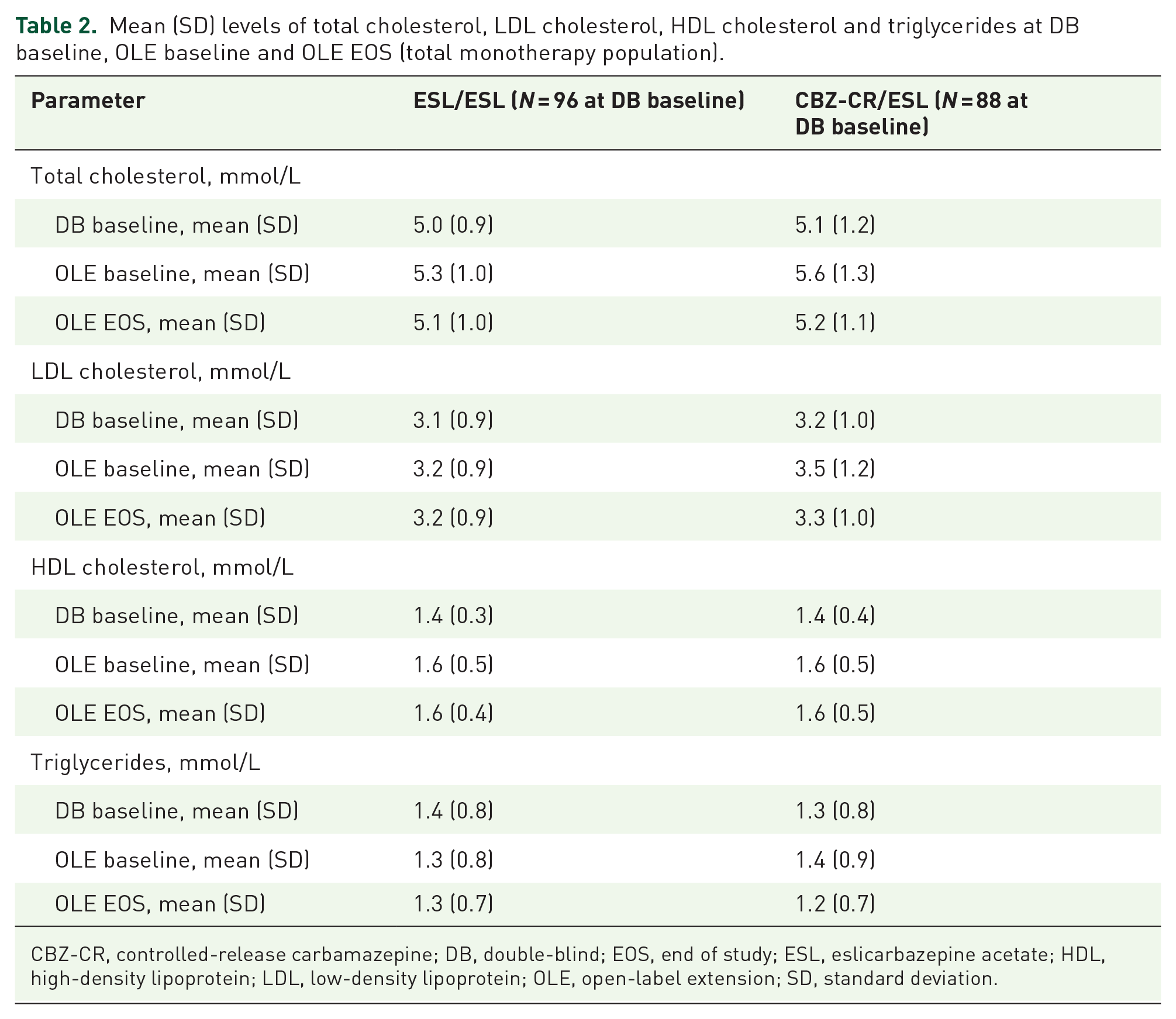
CBZ-CR, controlled-release carbamazepine; DB, double-blind; EOS, end of study; ESL, eslicarbazepine acetate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; OLE, open-label extension; SD, standard deviation.
Proportions of patients with high/low lipid parameters (total monotherapy population).

Fisher’s test was conducted to assess if the proportion of patients with high/low levels of corresponding lipid parameter differed significantly between treatment arms. p values use standard alpha level (0.05).
High level of total cholesterol defined as > 6.22 mmol/L.
McNemar test was conducted to assess if the treatment used between visits impacted the proportion of patients (using only completer subjects with data in both visits) with high/low levels of corresponding lipid parameter. p values use standard alpha level (0.05).
High level of LDL cholesterol defined as > 4.14 mmol/L.
Low level of HDL cholesterol defined as < 1.04 mmol/L.
High level of triglycerides defined as > 1.70 mmol/L.
Results in bold represent statistically significant change.
CBZ-CR, controlled-release carbamazepine; DB, double-blind; EOS, end of study; ESL, eslicarbazepine acetate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; OLE, open-label extension.

Between-group comparisons of the proportions of patients at DB baseline, OLE baseline and OLE EOS with (a) high levels of total cholesterol (>6.22 mmol/L), (b) high levels of LDL cholesterol (>4.14 mmol/L), (c) low levels of HDL cholesterol (<1.04 mmol/L), and (d) high levels of triglycerides (>1.70 mmol/L). (a) Patients who received ESL the DB trial and OLE study. (b) Patients who received CBZ-CR in the DB trial and ESL in the OLE study.

Within-group comparisons of the proportions of patients at DB baseline, OLE baseline and OLE EOS with (a) high levels of total cholesterol (>6.22 mmol/L), (b) high levels of LDL cholesterol (>4.14 mmol/L), (c) low levels of HDL cholesterol (<1.04 mmol/L), and (d) high levels of triglycerides (>1.70 mmol/L). (a) Patients who received ESL during the DB trial and OLE study. (b) Patients who received CBZ-CR in the DB trial and ESL in the OLE study.
Risk of patients with normal levels of total cholesterol and LDL cholesterol at DB baseline developing high levels of these lipid parameters following treatment with ESL and CBZ-CR in the DB trial: (A) total monotherapy population and (B) subgroup of patients not treated concomitantly with statins.
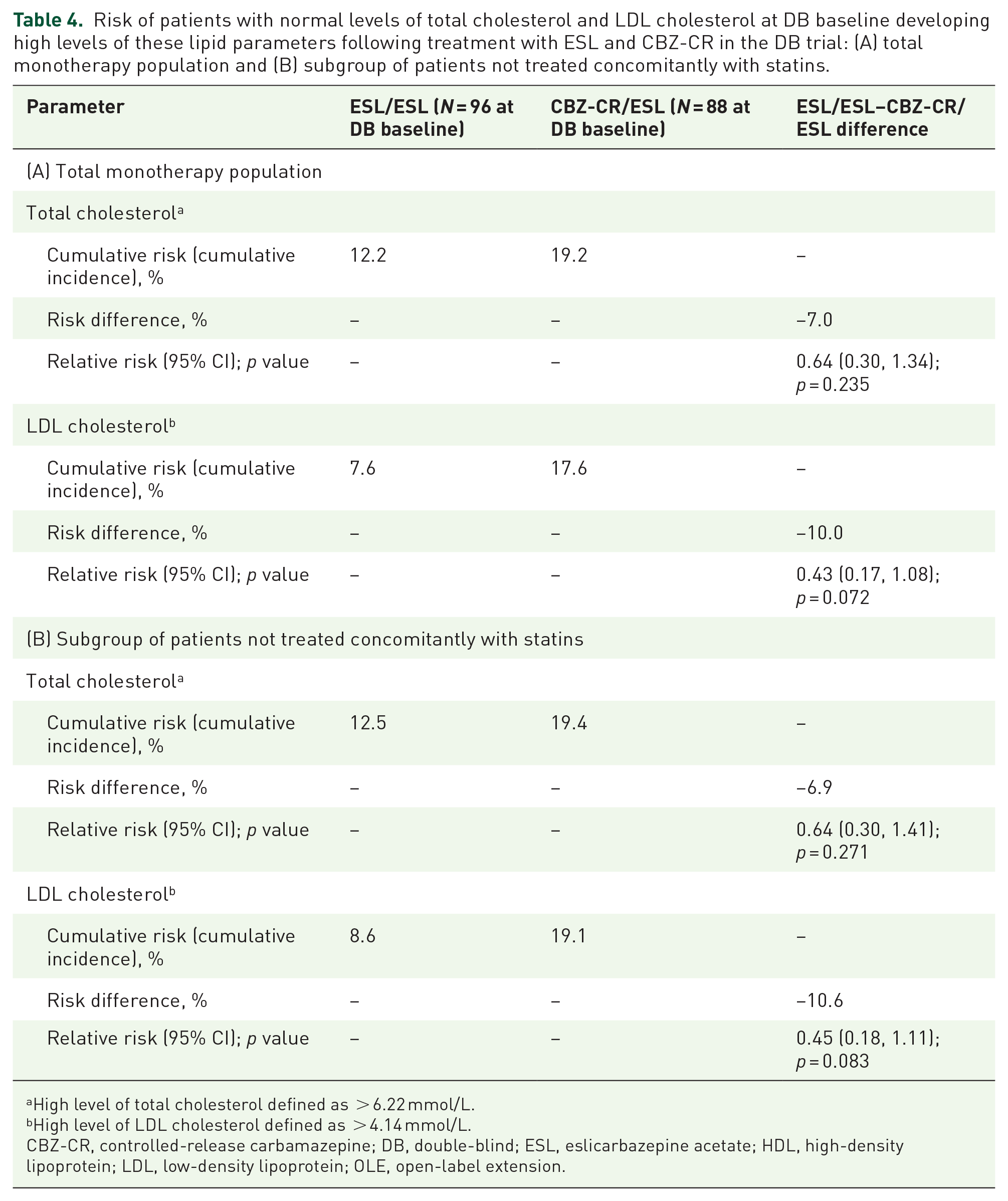
High level of total cholesterol defined as > 6.22 mmol/L.
High level of LDL cholesterol defined as > 4.14 mmol/L.
CBZ-CR, controlled-release carbamazepine; DB, double-blind; ESL, eslicarbazepine acetate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; OLE, open-label extension.
Similarly, at DB baseline there was no significant difference between treatment groups in the proportion of patients with high LDL cholesterol levels [Figure 3(b) and Table 3]. By the end of the DB trial (OLE baseline), the proportion of patients with high levels of LDL cholesterol was higher in patients treated with CBZ-CR during the DB trial, compared with those treated with ESL, although the difference was not statistically significant [between-group difference (ESL–CBZ-CR), −12.3%; p = 0.061]. The proportion of patients with high LDL cholesterol was similar between treatment groups at the other two visits (DB baseline and OLE EOS), showing that the increased proportion of patients with high LDL after using CBZ-CR had decreased to the baseline proportion after switching to ESL [Figure 3(b) and Table 3]. During the DB trial, the proportion of patients with high LDL cholesterol increased significantly in patients treated with CBZ-CR (change from DB baseline to OLE baseline, 11.5%; p = 0.012), and additionally, after switching from CBZ-CR to ESL, decreased significantly by OLE EOS (change from OLE baseline to OLE EOS, −11.1%; p = 0.021) [Figure 4(b) and Table 3]. In contrast, the proportion of patients with high LDL cholesterol remained stable in patients in the ESL/ESL treatment group during the DB trial and OLE study, without significant changes [Figure 4(b) and Table 3]. The cumulative risk of patients developing high levels of LDL cholesterol during the DB trial (having not previously had high levels) was 7.6% in those treated with ESL versus 17.6% in those treated with CBZ-CR [risk difference for ESL versus CBZ-CR, −10.0%; relative risk (95% CI), 0.43 (0.17–1.08); p = 0.072; Table 4(a)].
At DB baseline, the proportion of patients with low HDL cholesterol was significantly higher in patients randomized to CBZ-CR than in those randomized to ESL [between-group difference (ESL–CBZ-CR), −11.9; p = 0.021] [Figure 3(c) and Table 3]. In patients treated with ESL during the DB trial, the proportion with low HDL cholesterol remained relatively stable during both the DB trial and OLE study. In contrast, in patients treated with CBZ-CR during the DB trial, there was a non-significant decrease in the proportion with low HDL cholesterol during the DB trial, which remained relatively stable after switching to ESL during the OLE study [Figures 3(c), 4(c) and Table 3]. However, between-group differences observed during the DB trial and OLE study are inconclusive, due to the differences at DB baseline originally presented.
There were no significant changes in the proportion of patients with high triglyceride levels within treatment arms during the course of the DB trial and OLE study, and no significant between-group differences [Figures 3(d), 4(d) and Table 3].
Analyses of the subset of patients who were not being treated concomitantly with statins were generally consistent with those observed for the total monotherapy population (Table 5). During the DB trial, the proportion of patients with high total cholesterol and high LDL cholesterol increased significantly in those treated with CBZ-CR but not significantly in those treated with ESL. Unlike in the total monotherapy population, the proportion of patients with high levels of total cholesterol at the end of the DB trial (i.e. at OLE baseline) did not differ significantly between treatment groups [between-group difference (ESL–CBZ-CR), −9.3%; p = 0.187]. During the OLE study, the proportions of patients with high total cholesterol and high LDL cholesterol significantly decreased in those switching from CBZ-CR to ESL, while remaining stable in those who received ESL throughout the DB trial and OLE study. The cumulative risk of patients developing high levels of total cholesterol during the DB trial (having not previously had high levels) was 12.5% in those treated with ESL versus 19.4% in those treated with CBZ-CR [risk difference for ESL versus CBZ-CR, −6.9%; relative risk (95% CI), 0.64 (0.30–1.41); p = 0.271; Table 4(b)]. The cumulative risk of patients developing high levels of LDL cholesterol during the DB trial (having not previously had high levels) was 8.6% in those treated with ESL versus 19.1% in those treated with CBZ-CR [risk difference for ESL versus CBZ-CR, −10.6%; relative risk (95% CI), 0.45 (0.18–1.11); p = 0.083; Table 4(b)].
Subgroup analyses of proportions of patients with high/low lipid parameters in patients not treated concomitantly with statins.

Fisher’s test was conducted to assess if the proportion of patients with high/low levels of corresponding lipid parameter differed significantly between treatment arms. p values use standard alpha level (0.05).
High level of total cholesterol defined as >6.22 mmol/L.
McNemar test was conducted to assess if the treatment used between visits impacted the proportion of patients (using only completer subjects with data in both visits) with high/low levels of corresponding lipid parameter. p values use standard alpha level (0.05).
High level of LDL cholesterol defined as >4.14 mmol/L.
Low level of HDL cholesterol defined as <1.04 mmol/L.
High level of triglycerides defined as >1.70 mmol/L.
Results in bold represent statistically significant change.
CBZ-CR, controlled-release carbamazepine; DB, double-blind; EOS, end of study; ESL, eslicarbazepine acetate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; OLE, open-label extension.
As in the total monotherapy population, the proportion of patients with low HDL cholesterol was significantly higher in patients randomized to CBZ-CR than in those randomized to ESL (Table 5). Within-group trends were consistent with those observed in the total monotherapy population. At OLE EOS, the proportion of patients with low levels of HDL cholesterol was significantly lower in patients randomized to ESL in the DB trial, in comparison with those who were randomized to CBZ-CR [between-group difference (ESL–CBZ-CR), −9.3%; p = 0.050]. However, as in the total monotherapy population, between-group differences were inconclusive due to the differences at DB baseline.
As in the total monotherapy population, there were no significant within-group changes or between-group differences in the proportion of patients with high triglyceride levels (Table 5).
Discussion
In this post hoc analysis of a phase III DB trial and OLE study, the proportions of patients with elevated levels of total cholesterol and LDL cholesterol increased significantly in those treated with CBZ-CR monotherapy in the DB trial and decreased significantly after switching from CBZ-CR to ESL monotherapy during 2 years of treatment in the OLE study. There were minimal changes in the proportions of patients with elevated levels of total cholesterol and LDL cholesterol in those treated with ESL monotherapy throughout the DB trial and OLE study (up to 30 months). The proportion of patients with high levels of total cholesterol at the end of the DB trial was significantly lower for those treated with ESL than for those treated with CBZ-CR, and there was a trend toward a lower proportion of patients with high levels of LDL cholesterol in those treated with ESL, in comparison with CBZ-CR. At the end of the OLE study, the between-group differences for both these parameters were not significant. Additional analyses to further understand changes in total cholesterol and LDL cholesterol were conducted by estimating the risk of developing high levels of total cholesterol and LDL cholesterol separately during the DB trial, and assessing the relative risk reduction for ESL versus CBZ-CR. This involved evaluating those patients who did not have high levels at DB baseline and assessing whether their levels of total and LDL cholesterol increased during the DB trial. Although the findings may indicate trends toward a lower risk of developing high total and LDL cholesterol when treated with ESL monotherapy, in comparison with CBZ-CR monotherapy, the limitation is that these analyses were only exploratory and no multiplicity correction of p values was conducted, and they should therefore be interpreted with caution and only considered within the context of this post hoc evaluation. Since the proportion of patients with low levels of HDL cholesterol differed significantly between treatment groups at baseline of the DB trial, it was not possible to draw meaningful conclusions from the findings for this lipid parameter. No notable within-group changes or between-group differences were observed for the proportions of patients with elevated triglyceride levels.
Statins are the most commonly used lipid-lowering agents and there is concern that the concomitant use of enzyme-inducing ASMs may affect the lipid-lowering efficacy of statins by inducing their metabolism.6,7 In the current study, the proportion of patients treated concomitantly with statins was too small to be analyzed separately [ESL/ESL subgroup, 10/96 (10.4%); CBZ-CR/ESL subgroup, 11/88 (12.5%)] and so it was not possible to assess the potential impact of ESL versus CBZ-CR on the lipid-lowering efficacy of statins. However, the findings from the subset of patients who were not being concomitantly treated with statins were generally consistent with those for the total monotherapy population.
Although both ESL and CBZ belong to the dibenzazepine family of sodium-channel-blocking ASMs, sharing a dibenzazepine nucleus bearing the 5-carboxamide substituent, 29 they differ pharmacologically in terms of metabolism, pharmacokinetics and pharmacodynamics.19,30 Pharmacodynamically, S-licarbazepine (the primary active metabolite of ESL, accounting for 94% of plasma drug exposure following oral administration) is thought to act primarily by enhancing the slow inactivation of voltage-gated sodium channels, whereas CBZ is thought to alter the fast inactivation of these channels.19,31 Moreover, S-licarbazepine appears to have greater selectivity than CBZ for inhibiting rapidly firing active ‘epileptic’ neurons.19,32 CBZ’s tolerability is compromised by being metabolized to carbamazepine-10,11-epoxide, which, in addition to its antiseizure effect, is known to cause adverse effects, including dizziness and somnolence.19,30 Furthermore, CBZ is a potent inducer of the cytochrome P450 (CYP450) enzyme system, which not only negatively affects the duration and action of many drugs but can also impact enzymes involved in endogenous metabolic pathways.10,12,19 By contrast, S-licarbazepine is a less potent enzyme inducer than CBZ, 14 being a weak inducer of CYP450 3A4 and uridine 5′-diphospho-glucuronosyl transferases.19,23,25 The impact of CBZ on endogenous metabolic pathways can lead to CCV diseases and other comorbidities, including sexual dysfunction and osteoporosis.10,12 Importantly, the CYP450 enzyme system is extensively involved in the synthesis and metabolism of cholesterol 33 and it is thought that potent CYP450 inducers, such as CBZ, increase cholesterol production by reducing levels of oxysterol intermediates, which are the main endogenous feedback inhibitors of HMG-CoA reductase (the rate-controlling enzyme in cholesterol synthesis).10,34 CBZ additionally causes alterations in thyroid hormone levels, which may also cause changes in serum lipid levels.35,36 Evidence for the impact of CBZ on lipid levels is substantial7,10; for example, in an analysis of data from a 12-month, phase III, international, randomized, DB trial of CBZ versus lacosamide for the initial treatment of focal epilepsy, CBZ-treated patients experienced increased levels of total cholesterol (+21.1 mg/dL), LDL cholesterol (+12.6 mg/dL), non-HDL cholesterol (+12.5 mg/dL) and HDL cholesterol (+8.5 mg/dL), whereas lacosamide-treated patients did not experience elevations in these parameters. 37 Moreover, the proportion of patients with elevated total cholesterol levels increased from 30.8% at baseline to 49.6% at 12 months in patients treated with CBZ but remained stable in those treated with lacosamide (37.0% at baseline; 34.8% at 12 months). 37 In a study that assessed the impact of discontinuation of CBZ due to concerns over the long-term consequences of enzyme induction, patients who discontinued CBZ were shown to experience significant reductions in total cholesterol, LDL cholesterol and HDL cholesterol, and to additionally experience a significant increase in free testosterone and a significant decrease in sex hormone-binding globulin levels, in comparison with those who continued to receive CBZ. 38 Another study demonstrated that CBZ significantly affected the ability of statins to lower total cholesterol, leading the authors to conclude that it is a poor choice for patients with hyperlipidemia or cardiovascular disease. 7 Such findings have led to recommendations regarding the consideration of switching patients treated with enzyme-inducing ASMs to non-enzyme-inducing ASMs,10,26,38,39 and to speculation regarding whether or not CBZ should be considered as first-line monotherapy in patients with newly diagnosed epilepsy. 10
The findings of the current study are consistent with previous reports.6,19–21,24 A single-center, observational, retrospective cohort study assessed the effects of ESL on lipid metabolism in 108 patients (without having newly diagnosed focal seizures, and including patients with add-on ASMs) over a median duration of 23.1 months (range 3–41 months), and, in the overall population, mean total cholesterol levels decreased significantly during follow-up, changing from previous increased levels (>200 mg/dL) to normal levels (<200 mg/dL) during treatment. 20 The proportion of patients with hypercholesterolemia also decreased during ESL treatment, although this change was not statistically significant. 20 Over half of the population had switched to ESL from prior treatment with CBZ or oxcarbazepine, and there was a marked decrease in mean total cholesterol and a significant decrease in both mean LDL cholesterol and median triglyceride levels in these patients. 20 Furthermore, after transitioning to ESL, there were significant reductions in the proportions of patients with hypercholesterolemia and hypertriglyceridemia. 20 In another retrospective cohort study, lipid levels were assessed in 36 adult patients before and after receiving treatment with adjunctive ESL over a mean follow-up duration of 11 months (range 6–18 months). 21 After at least 6 months of ESL treatment, there were significant reductions in the mean levels of total cholesterol and LDL cholesterol, and a significant increase in the mean level of HDL cholesterol. 21 As in the current study, no statistically significant change in triglyceride levels was observed. 21 In a post hoc analysis of pooled data from three phase III, multicenter, randomized, DB, placebo-controlled trials of adjunctive ESL therapy (400, 800, or 1200 mg QD) in patients with refractory focal seizures, changes from baseline in serum lipid levels were analyzed according to the use of enzyme-inducing ASMs and/or statins during the baseline period. 6 The most frequently used enzyme-inducing ASM in patients treated with ESL was CBZ (51%). 6 Changes from baseline were significantly greater for ESL 1200 mg/day versus placebo for total cholesterol and HDL cholesterol, but the effect sizes versus placebo were small. 6 There was also a small but significant difference in the change from baseline in LDL cholesterol for ESL 400 mg/day versus placebo. 6 ESL had no consistent effect on lipid levels in patients taking a concomitant enzyme-inducing ASM, and there were no clinically relevant changes in lipid levels following ESL treatment in patients who took statins during baseline. 6 In a similar post hoc analysis of pooled data from two phase III conversion to ESL monotherapy studies, lipid parameters were assessed at baseline, Week 18, and Month 12, and analyzed in terms of the use of enzyme-inducing ASMs at baseline. 24 The most frequently used enzyme-inducing ASMs were CBZ (77%) and phenytoin (24%). 24 Patients converting from enzyme-inducing ASMs had higher levels of total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides at baseline, compared with those converting from non-enzyme-inducing ASMs (significantly higher for total cholesterol and HDL cholesterol). 24 After converting to ESL monotherapy, plasma levels of total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides decreased in patients converting from enzyme-inducing ASMs and increased in those converting from non-enzyme-inducing ASMs, reaching very similar values that were intermediate between the baseline values for the subgroups of patients converting from enzyme-inducing and non-enzyme-inducing ASMs. 24 The authors concluded that ESL had only a modest and clinically insignificant impact on plasma lipid levels. 24 Other studies, conducted in the clinical practice setting, have demonstrated that ESL monotherapy can be effective in patients who have switched from CBZ due to inadequate efficacy,40,41 consistent with evidence demonstrating that ESL and CBZ have different mechanisms of action.19,31,32 Since no ASM has been shown to have protective CCV effects, in terms of improving lipid profiles in previously untreated patients, the relative impact of an ASM on CCV risk factors is an important consideration when choosing initial treatment, or when considering switching a patient from one ASM to another. Published guidance has provided recommendations for clinical scenarios in which it may be appropriate to switch patients from CBZ to ESL (such as patients experiencing or at risk of hypercholesterolemia and those with severe osteopenia-osteoporosis), along with pragmatic advice on how to do so.26,39
To our knowledge, this is the first prospective investigation of the impact of ESL on plasma lipid levels in patients receiving ESL as monotherapy for newly diagnosed epilepsy (i.e. without prior exposure to ASMs), since previous studies were conducted either in the adjunctive or conversion to monotherapy settings and/or were retrospective in design. A further strength is that we were able to analyze patients’ plasma lipid levels before starting ASM treatment, and after switching from CBZ monotherapy to ESL monotherapy. The nature of this post hoc study was exploratory. The sample size was small, and the original DB trial and OLE study were not designed or powered to assess the impact of treatment on plasma lipid levels. Therefore, the p values cited should not be considered confirmatory and the findings from the analyses should be interpreted with caution. Nevertheless, the results are consistent with those of previous studies.6,19–21,24 Since the sample size was small, it was not possible to conduct meaningful analyses of mean changes in lipid parameters, due to high variability in the data, but this was addressed by assessing the proportions of patients with high/low plasma levels of lipid parameters to provide a clinically meaningful understanding of changes in these parameters over time. A further limitation of the study was that information on alcohol use and smoking status, both of which can have an impact on lipid parameters, was not available, and it was therefore not possible to confirm whether there were between-group differences that could potentially have affected the results. However, relevant medical conditions, including CCV diseases, were well balanced between the treatment groups.
In summary, the findings from this study are consistent with current evidence regarding the impact of CBZ on plasma lipid levels, supporting recommendations for considering switching patients from CBZ to ESL to reduce the risk of hypercholesterolemia.19,38,39 The findings also suggest that ESL monotherapy may be a rational treatment option for patients with newly diagnosed focal epilepsy who have, or who are at risk of developing, hypercholesterolemia.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231193530 – Supplemental material for Lipid profile with eslicarbazepine acetate and carbamazepine monotherapy in adult patients with newly diagnosed focal seizures: post hoc analysis of a phase III trial and open-label extension study
Supplemental material, sj-docx-1-tan-10.1177_17562864231193530 for Lipid profile with eslicarbazepine acetate and carbamazepine monotherapy in adult patients with newly diagnosed focal seizures: post hoc analysis of a phase III trial and open-label extension study by Eugen Trinka, Rodrigo Rocamora, João Chaves, Mathias J. Koepp, Stephan Rüegg, Martin Holtkamp, Joana Moreira, Miguel M. Fonseca, Guillermo Castilla-Fernández and Fábio Ikedo in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Third party submissions: not applicable. Medical writing assistance (editing, collating of author comments, formatting to journal style) was provided by John Scopes of mXm Medical Communications and funded by Bial – Portela & Cª.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
