Abstract
Background:
Magnetic Resonance-guided Focused Ultrasound (MRgFUS) is an innovative therapeutical approach for medically refractory tremor. It is currently under investigation for other neurological diseases including refractory neuropathic pain (NP).
Objective:
The objective of this systematic review is to analyze available evidence about the effectiveness and safety profile of MRgFUS in the treatment of refractory NP.
Methods:
Eligible studies were identified by searching published studies in PubMed and Scopus databases from inception to December 2022 and by identifying ongoing studies registered on the clinicaltrials.gov website. The study was registered in PROSPERO (ID: CRD42021277154).
Results:
We found three published observational studies and nine ongoing studies. In published studies, the involved population ranged from 8 to 46 patients with overall 66 patients being included with NP or trigeminal neuralgia. The target lesion was in the posterior part of the central lateral nucleus of the thalamus, bilaterally. Outcomes were assessed at different times through the Visual Analog Scale, showing a variable degree of improvement. Adverse events were rare, mild, and transient (vertigo, paresthesias, and dysesthesias) with intracerebral bleeding being reported as major adverse event in one case only. Among ongoing studies, we found three prospective, randomized, sham-controlled, crossover trials (RCTs) and six observational studies. Inclusion criteria are previous failure of more than three pharmacological treatments and NP duration longer than 6 months. The thalamus is the main proposed target and measured outcomes are accuracy of the procedure and pain relief, with a follow-up period ranging from 1 week to 1 year.
Conclusion:
This systematic review suggests that, although high-quality studies are lacking, available evidence endorses the effectiveness and safety of MRgFUS in the management of NP. Ongoing RCTs will provide more robust data to understand benefits and risks of the procedure.
Registration:
PROSPERO (ID: CRD42021277154)
Introduction
Chronic neuropathic pain (NP) is the direct consequence of a lesion or diseases affecting the somatosensory system with both peripheral and central sensitization mechanisms being involved. 1 NP may be spontaneous or evoked, either occurring as an increased response to a painful stimulus (hyperalgesia) or as a painful response to a normally nonpainful stimulus (allodynia). NP can be further classified as central or peripheral, depending on the main involvement of the spinal cord and/or of the brain or of the small unmyelinated C fibers and the myelinated A fibers, namely, the Aβ and Aδ fibers.1,2 Therefore, the diagnosis of NP requires a history of nervous system injury caused, for example, by a stroke, a nerve trauma, or a diabetic neuropathy, and a neuroanatomically plausible distribution of the pain. 1 The National Institute for Health and Clinical Excellence (NICE) guidelines for NP suggest the use of antidepressants and antiepileptic drugs as first-line medications. 3 Second-line management implies the switching to another first-line medication or the combination of two first-line drugs. 3 The third-line treatment includes opioids, singly or combined with second-line medications. 3 However, NP is often difficult to treat because it is resistant to many medications and/or because of adverse effects associated with effective medications, with many patients failing to enjoy meaningful benefits. 3 NP is considered resistant to pharmacotherapy when single medications or a rational combination of drugs proven efficacious in randomized controlled trials (RCTs) fail in inducing useful pain relief from the patient’s or the physician’s point of view after an appropriate duration of treatment with adequate dosage, or if intolerable side effects occur. 4 For the management of resistant NP, alternative or additional non-pharmacological approaches are under investigation, including Magnetic Resonance-guided Focused Ultrasound (MRgFUS) thalamotomy. The aim of this review is to systematically analyze the current knowledge about the use of MRgFUS in NP, in order to identify the clinical profile of patients potentially eligible for the treatment, the benefits and the risks associated with the procedure, the anatomical therapeutic targets, and strengths and limitations of the available studies in this field.
MRgFUS clinical mechanisms and applications
MRgFUS is a combination of focused ultrasound with magnetic resonance imaging (MRI). Intensity is a key determinant of ultrasound effects, with different outcomes being achieved through high-frequencies focused ultrasound (HIFU) and low-frequencies focused ultrasound (LIFU). 5 HIFU-MRgFUS is a non-invasive procedure for ablative purposes such as thalamotomy, pallidotomy, and subthalamotomy for movement disorders. On the contrary, LIFU-MRgFUS has been used in preclinical studies for blood–brain barrier opening (BBBO), making this procedure a potential future paradigm for controlled drug delivery in neurodegenerative diseases and neuro-oncology and for neuromodulation (see Figure 1). Approved Food and Drug Administration (FDA) indications for HIFU-MRgFUS include medically refractory essential tremor and Parkinson’s disease (PD)-related tremor, while other clinical applications are currently under investigation (see Figure 2). During HIFU-MRgFUS ablative sessions, the shaved head of the patient is fixed to a stereotactic frame and assembled with an MRI machine (see Figure 3). A flexible silicone membrane is applied to the head to seal the space between the head and the transducer. This space contains cool water (15–20°c) to reduce the heating of the scalp. After the preoperative image registration and the planning of the target to treat, high-power sonification is delivered to the target area under MRI and MR thermometry. 6 Once the target point has been established, subthreshold, low-power sonication is applied several times for 10–20 s to reach a peak temperature between 40°c and 42°c. High power sonification (under 54°c) create thermal coagulative necrosis of cells and permanent ablation. 7 The procedure is particularly promising because HIFUs pass through the intact skull and reach deep brain tissues with high precision and excellent tissue differentiation, thus providing immediate therapeutic effect with a quick return to normal activities. Moreover, the advantages of MRgFUS, which caused its rapid spread and popularity, lie with the possibility to perform intraoperative clinical assessment: in fact, HIFU lesions are created through consecutive sonications of increasing power between which the awake patient is typically assessed for efficacy and side effects.8,9 This stepwise approach allows functional neurosurgeons and neurologists to have a just-in-time performance feedback about efficacy and side-effects.8,9 So far, MRgFUS has mostly been used to treat medically refractory essential tremor and PD-related tremor through thermoablation of the thalamic ventral intermediate (VIM) nucleus.10–15 Neurological complications of MRgFUS VIM thalamotomy include reversible symptoms that occur during sonication such as headache, dizziness, vertigo, nausea, vomiting, sensation of warmth in the scalp or flushing, and paresthesia, and symptoms lasting after the procedure such as hypogeusia, gait disturbances and, more rarely, unilateral weakness. 16 However, most of the adverse effects are mild and usually resolve within 3 months.9,16

Possible mechanisms of MRgFUS.

Neurological diseases potentially treated by MRgFUS.

Components of the MR-guided transcranial focused ultrasound system.
HIFU-MRgFUS represents a suitable technique also to treat chronic NP, through a process of high-power sonication that caused a permanent ablative lesion into specific brain targets. Throughout the procedure, the patient remains awake and responsive and is neurologically evaluated immediately after the procedure. This approach might be particularly promising for patients with refractory trigeminal neuralgia (TN), for whom Gasserian ganglion percutaneous techniques, gamma knife surgery, and microvascular decompression are the available invasive treatment options, and for patients with debilitating pain not responding to common medications.17,18
Methods
This systematic review was performed according to the 2020 update of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 19 The study was registered in PROSPERO (ID: CRD42021277154) and the protocol can be found at the PROSPERO database.
Investigated outcomes included pain relief following the treatment and safety of the procedure.
Studies were identified by searching articles indexed on PubMed and Scopus from the beginning of indexing up to December 2022. We conducted a search on both databases using the search terms ‘focused ultrasound’, ‘MRgFUS’, or ‘thalamotomy’, combined with the terms ‘chronic pain’ or ‘neuropathic pain’. The search was restricted to humans, and to articles published in English. We focused on articles about HIFU MRgFUS while discarding those dealing with LIFU MRgFUS as, to the best of our knowledge, it is under investigation for other indications in humans.
Duplicate publications were removed by manual check.
Original studies, including clinical trials, observational studies, and case series addressing HIFU MRgFUS for NP were eligible for this review. Studies lacking a clear definition of study design and setting, case reports, letters, abstracts, studies not performed on humans, studies not written in English, and unpublished studies were excluded; studies concerning the use of the MRgFUS for indications other than NP were also excluded. Furthermore, non-neuropathic chronic pain conditions such as cancer pain were excluded. Reviews were not included in the systematic review; however, the references of suitable reviews were checked to maximize the inclusion of original studies and the reviews were considered for the critical discussion of the topic.
To select eligible articles, we adopted a two-step procedure. In the first step, titles and abstracts were screened by two independent investigators (V.T., R.O.) to verify study eligibility; disagreements were resolved by discussion with a third party (F.P.). Duplicate records were removed by manual check. Full texts of selected studies were then evaluated, if appropriate. The reference lists and Google Scholar citations for all primary and review articles were also searched. Discrepancies between reviewers were resolved by discussion among all the study investigators.
To cover ongoing research in the field, one researcher (V.T.) made an additional search on the clinicaltrials.gov database from the beginning of indexing up to December 2022. Search terms were the same used for database search as well as inclusion and exclusion criteria regarding treatment and clinical conditions (HIFU MRgFUS in NP treatment).
The evaluation was done at study level and the included studies were described individually, with attention being paid to pain relief following the treatment and safety of the procedure. Data were summarized according to the type of pre-treatment pain, type of treatment, pain relief, side effects of the treatment.
Results
Literature search
A total of 272 records were identified by searching in PubMed and Scopus. After duplicate removal, 187 records were screened; 153 of which were deemed irrelevant by the reviewers. Finally, three observational studies were deemed eligible. The search on clinicaltrials.gov retrieved 31 records; nine ongoing studies were considered eligible for this review. The PRISMA flow diagram is presented in Figure 4.

Flow diagrams of studies and ongoing clinical trials selected.
Published studies
All the three published studies had an observational design; two were prospective and one was retrospective (see Table 1).20–22 The prospective observational study of Gallay involved eight subjects with TN. 20 In detail, six patients had idiopathic TN (of which one patient previously treated with microvascular decompression) and two secondary TN (one with multiple sclerosis and one with surgically treated trigeminal schwannoma). All patients suffered from pain with paroxysmal character. Six of them also reported associated continuous pain. The pain syndrome was unilateral in all the patients, mainly involving the right side. Overall, the pain duration was 17 ± 12 years (range 4–37). Bilateral central lateral thalamotomy (CLT) in one session was performed in six patients. The procedure was performed unilaterally in two patients for the following reasons: in the first patient, previously treated with bilateral radiofrequency CLT, unilateral CLT complement as well as one centrum medianum (CM) target were performed, while the second patient received a complementation on the right side of his bilateral MRgFUS CLT performed 14 months previously. Primary outcome of the study was pain relief evaluated through the Visual Analog Scale (VAS) and the McGill questionnaire, which were administered before surgery and at the 3-months and 1-year follow-up. There was an improvement in the VAS scale from 70 ± 20 to 27 ± 30/100 at 1 year following surgery (p = 0.0008). Another primary outcome was the frequency in pain paroxysms. Specifically, there was a change in the frequency of mean pain paroxysms that decreased from 84 per day before surgery to 3.9 at the 1-year follow-up (p = 0.003). Secondary outcomes included: the change in anxiety and depression feelings, as explored by the Hospital Anxiety and Depression Scale (HADS), at 3-month and 1-year follow-up as compared with preoperative scores; the change in cognitive status, as explored through the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) test, at 2 days and at 1-year follow-up as compared with preoperative scores. The HADS score was 14.4 ± 5.8 (5–21) preoperatively, 8.5 ± 3.7 (2–14) at 3 months (p = 0.03) and 7.6 ± 4.1 (2–16; n = 8, p = 0.017) at the 1-year follow-up. Mean MMSE scores were 29.3 ± 0.6 preoperatively, 29.7 ± 0.6 at 2 days, and at the 3-month and 1-year follow-up. Mean MoCA scores were 26.8 ± 4.1 (20–30) before surgery, 28.0 ± 2.5 at 2 days (24–30) and 28.6 ± 2.1 (25–30; p = 0.28) at the 1-year follow-up. There were no serious adverse events: one patient showed a postoperative frontal scalp swelling and two patients had mild transient vertigo.
Summary of studies included in the systematic review.

TN, trigeminal neuralgia; VAS, Visual Analog Scale.
The CLT was performed unilaterally for the following reasons: complement to radiofrequency treatment and as complement of previous bilateral MRgFUS CLT performed.
Described for the whole sample [neuropathic pain (NP) and other indications].
The CLT was performed unilaterally for the following reasons: complement to radiofrequency treatment, appearance of bleeding, immediate 100% pain relief after treatment of the first side, and intolerance of a longer intervention time
The total number of patients involved in the study was 180 (including treatments for chronic therapy-resistant idiopathic Parkinson’s disease, essential tremor, cerebellar tremor). The information reported in this table are only about the NP.
In the study of Jeanmonod, MRgFUS was successfully used in 11 patients for chronic uncontrolled NP due to amputation (phantom leg pain, one patient), root compression in failed back surgery syndrome (one patient), root compression by neurinoma (one patient), postherpetic neuralgia (one patient), traumatic trigeminal nerve injury (one patient), spinal cord lesion (two patients), putaminal lesion (one patient), avulsion of brachial plexus (two patients), and thalamic infarct (one patient). 19 The initial cohort included 12 patients but one of them was excluded by further analyses as the intervention did not result in a thermolesion due to a low applied temperature (42°C). The mean duration of pain was 8.5 years (1.5–21 years). The CLT was performed unilaterally (contralateral to the pain location) in five patients and bilaterally in six patients. The CLT was performed unilaterally for the following reasons: complement to radiofrequency treatment (two patients), appearance of bleeding (one patient; see Clinical Results), immediate 100% pain relief after treatment of the first side (one patient), and intolerance of a longer intervention time (one patient). Technical success, that is, occurrence of a thermolesion, was high (75% to 100%). In two patients, the therapeutic lesions, as seen 48 h after treatment on T2-weighted and DT images, were too small. Thus the MRgFUS treatment was considered complete in only 9 of the 11 patients. Outcomes were evaluated as changes in the VAS scores for pain intensity at 48 h, three months, and at the 1-year follow-up. Mean VAS score was 35.3/100 at the 1-year follow-up as compared with the preoperative score of 59.5/100. An additional outcome was the precision of MRgFUS targeting, expressed as values of dorsoventral, anteroposterior, and mediolateral millimetric differences between centers of the sonication lesions and presurgical target coordinates. Target reconstructions demonstrate precision within a millimeter for all the 3 target coordinates. Adverse events included vestibular manifestations with or without vegetative symptoms (eight patients), paresthesias (four patients), or dysesthesias/pain (nine patients). One serious adverse event was recorded: a CLT bleeding followed by possible blood-induced vasospasm and ischemia occurred in one patient, resulting in neglect and dysmetria. 21
The retrospective observational study of Gallay included 180 consecutive MRgFUS treatments performed between April 2011 and November 2016. 22 Among them, 46 procedures were performed to treat chronic and therapy-resistant NP. The duration of pain was 16 ± 13.6 years and CLT was the intervention performed. This study was different from the previous ones as aimed at investigating the safety and accuracy of MRgFUS rather than pain improvement. Pain dimension was evaluated as previously described by the same research group through the VAS and the McGill questionnaire, but analytics data about pain improvement are not available. 20 Changes in the cognitive status, estimated as an improvement in cognitive tests (MMSE and then MoCA) at 24–48 h following the intervention and at the 1-year follow-up were also explored. There was a significant improvement in the 24–48 h MMSE (29.0 ± 1.2 versus 29.5 ± 0.8; p = 0.06) and MoCa (27.1 ± 2.7 versus 28.1 ± 2.1; p = 0.002) scores as compared with preoperative scores. At the 1-year follow-up, the mean MMSE score (29.4 ± 1.3; p = 0.2) as well as the mean MoCA score (28.0 ± 2.8; p = 0.02) further improved. Concerning the precision of MRgFUS, target reconstructions demonstrated a precision within a millimeter for all the three target coordinates, as reported in the previous study. 22 General adverse events were listed for the whole sample and classified as it follows: non neurological side-effects occurring in the weeks following the procedure in five patients (one case of lung embolism, one case of cardiac decompensation, one case of bladder infection in a patient, one case of mild case of transient hiccups, and an episode of fall at home causing elbow fracture); procedure-related effects observed in four patients (two cases of subcutaneous swelling of the face, one case of transient right-sided frontal headache, and one case of temporary unilateral blindness during frame fixation after induction of supraorbital local anesthesia); disease-related effects, due to the worsening of pre-existing neurological or cognitive deficits, reported in seven patients in whom the procedure was planned for other indications than NP, mainly movement disorders; effects on neighboring structures, observed in three patients (one case of transitory hypesthesia on the lower lip and slight reduction of gustation, one case of transitory paresthesias around the mouth and on the hand, and one case of decreased mnestic performances in a PD patient). 22
Ongoing studies
There are nine ongoing studies, three RCTs and six observational studies (see Table 2). Conditions under investigations are NP due to radiculopathy or radicular injury, spinal cord injury, phantom limb pain and TN, all conditions refractory to the medical treatment. The targets are within the thalamus particularly in the central lateral (CL) nucleus, the ventral posteromedial (VPM) nucleus, and in the ventral posterolateral (VPL) nucleus. As reported in Table 2, studies show differences in outcomes and timing of follow-up.
Ongoing trials.
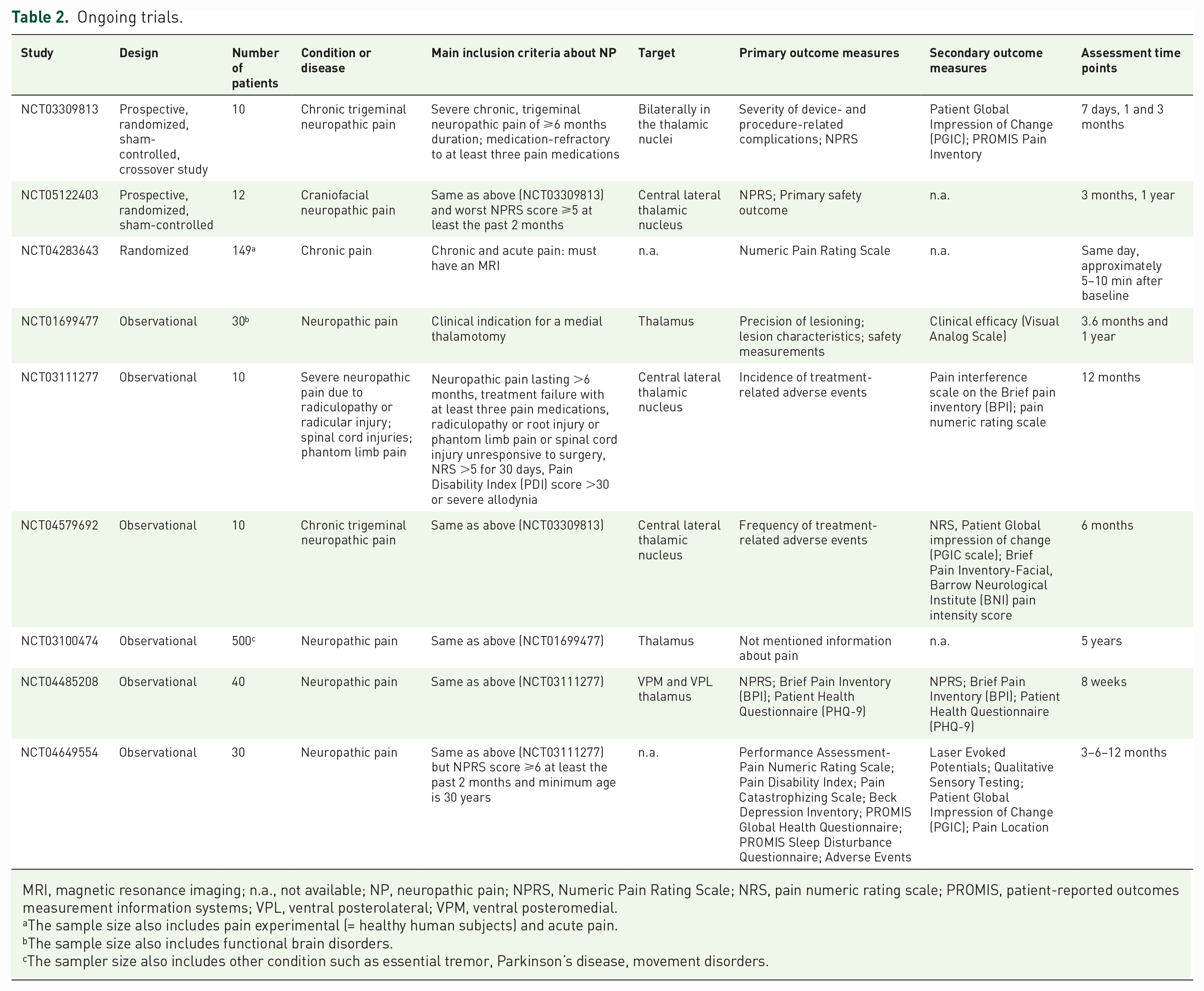
MRI, magnetic resonance imaging; n.a., not available; NP, neuropathic pain; NPRS, Numeric Pain Rating Scale; NRS, pain numeric rating scale; PROMIS, patient-reported outcomes measurement information systems; VPL, ventral posterolateral; VPM, ventral posteromedial.
The sample size also includes pain experimental (= healthy human subjects) and acute pain.
The sample size also includes functional brain disorders.
The sampler size also includes other condition such as essential tremor, Parkinson’s disease, movement disorders.
Discussion
MRgFUS represents a new potential treatment for medically refractory NP. This systematic review showed that MRgFUS targeting the thalamus is a promising approach with good effectiveness and safety. This technique could soon enrich the equipment of advanced management approaches for refractory NP. In fact, available studies show that the technical success of the procedure is high with rare adverse events, which are usually mild and transient despite the relative invasiveness of the procedure. The strength of the available studies are limited by several factors: the lack of a randomized design with a control group managed with a sham procedure, the small number of patients involved, and the heterogeneity of patients included. Moreover, the wide heterogeneity in measured outcomes and in the timing of follow-up makes the results not easily comparable. With these limits, available studies show that the treatment is feasible and worth to be further investigated by ongoing prospective RCTs. Open issues that reserve to be investigated is the possible placebo effect of the treatment and the persistence of beneficial effects following the procedure: in this regard, sham surgery could serve an analogous purpose to placebo drugs, neutralizing biases such as the placebo effect. However, sham procedures, planned to counteract MRgFUS, are not easy to be planned. This explains why, although several RCTs are ongoing, most of them are observational. Regarding the target of the treatment for NP, the main targets addressed by the ongoing studies include CL, VPM, and VPL nuclei of the thalamus. CL nuclei are involved in sensorimotor functions and in salience and attention networks while VPM and VPL are engaged in the processing of somatosensory and nociceptive information. 23 Based on their anatomical location, as well as afferent and efferent connections, these nuclei project to brain areas that influence the pain matrix and perception. Moving from available evidence, the most suitable target for ablative treatment in medically refractory NP is the posterior part of the CL nucleus (CLp).24–27 CLp transfers nociceptive informations from spinothalamic and spino–reticulo–thalamic pathways to large domains of cortex, including areas involved in nociception, mainly the primary (SI) and secondary (SII) somatosensory cortices, the insula, the anterior cingulate cortex (ACC) SII, the prefrontal and posterior parietal cortex (see Figure 5).24,25 Integrated signals from various higher order areas of the brain are then transmitted via the periaqueductal gray to the spinal cord to act as descending modulatory pathways and influence the incoming nociceptive signals. The cells of the spinothalamic tract that project to the lateral thalamus generally have receptive fields on a restricted area of the contralateral skin and are, therefore, well suited to a signaling function of the sensory-discriminatory aspects of pain. 28 Cells from the spinothalamic tract that project to the CL nucleus region in the medial thalamus can also collateralize to the lateral thalamus; these cells have response properties identical to those of the spinothalamic tract cells, which project only to the lateral thalamus. 29 However, cells of the spinothalamic tract that project only to the CL nucleus have very large receptive fields, which often span the entire surface of the body and face. 29 So bilateral medial CLp-centered thalamotomies have been shown to be more efficient than unilateral contralateral ones in the treatment of medically refractory NP. This is in line with findings of previous studies showing that the presence of NP is associated with an increase in low- and high-frequency spectral electroencephalogram (EEG).30,31 Such correlation is further supported by the evidence of reduced theta overactivity in the EEG recordings of patients treated with CLT in the study of Jeanmonod. 21 An important component of the thalamic activity is the low threshold spike (LTS) that consists of a characteristic train of action potentials following an inhibitory event: LTS burst firing increases the synaptic efficacy of thalamo-cortical transmission related to somatic sensation and may be causally related to NP. 32 A causal role of a medial ‘essential thalamic center’ in the pathophysiology of neurogenic pain has been proposed first by Head and Holmes and then by Jeanmonod et al. 33 This hypothesis was later supported by the beneficial effects of stereotaxic medial thalamic lesions (medial thalamotomy) against positive symptoms in humans. 33 The small therapeutic lesion within the CLp is believed to trigger a progressive normalization of the thalamocortical dysrhythmia hypothetically associated with NP. Future studies will establish which nucleus is more suitable to be targeted for the treatment of NP and which are the neurophysiological mechanisms involved in its development and disappearance.

Pathways underlying NP.
Another clinical issue deserving discussion is the improvement in the general cognitive status observed in patients treated with MRgFUS bilateral thalamotomy for NP. We observed the same cognitive improvement in a cohort of patients undergoing MRgFUS VIM nucleus thalamotomy for medical refractory essential tremor and Parkinson’s disease (PD)-related tremor. 9 We interpreted this improvement as the consequence of tremor extinction leading to enhanced well-being, self-esteem, and functional independence. 9 In this respect, MRgFUS may be more likely to preserve cognitive functions as compared with other traditional thalamotomy approaches or deep brain stimulation (DBS), due to its ability to produce well-circumscribed lesions with a real-time MRI monitoring. 9 The ability to target the nucleus of interest with extreme precision alongside the lack of trajectory-related damage may prevent the onset of even subtle detrimental effects on cognition. 9
In conclusion, MRgFUS may be an effective and safe alternative approach in the treatment of advanced NP. Our systematic review showed that MRgFUS is a promising technique in the treatment of NP although there is a general lack of high-quality studies investigating safety and efficacy. Preliminary data from observational studies on a limited number of subjects indicate that MRgFUS, with the thalamus as the main target, is safe and effective for managing NP with minor side-effects. However, HIFU thalamotomy is not feasible for every patient or every case of NP. Much work remains to be done to ascertain in which cases MRgFUS provides significant therapeutic and cost-effective value in the management of NP. RCTs are ongoing and will provide more robust data to understand the benefits and risks of the procedure. Future studies should include larger and more homogeneous samples of patients in order to make outcomes comparable. Objective assessment tools to evaluate pain should be used together with effective follow-up strategies and schedules. Moreover, concomitant drug abuse, that could influence the assessment, as well as the possible placebo effect experienced by patients, should be considered when evaluating the MRgFUS results on NP.
Another issue that complicates the assessment of NP following MRgFUS is the lack of an immediate behavioral feedback on the efficacy of the treatment: in fact, contrary to what happens in the treatment of tremor, where there is a just-in-time performance feedback about efficacy and side-effects, pain reduction may only be estimated in the subsequent follow-up.
Finally, MRgFUS is being investigated for numerous further applications, including blood–brain barrier disruption, targeted drug delivery, and neuromodulations. All these applications would be extremely useful for neurodegenerative diseases, whose prevalence is rapidly increasing worldwide. 34 This makes MRgFUS a novel therapeutic device with a growing potential in the treatment of NP, tremor, and other neurological disorders.
