Abstract
Background:
Several pharmacological pathways have revealed statin to have a positive role in patients with for intracranial aneurysms. However, prior studies regarding the association between statin use and patients’ outcomes after pipeline embolization device (PED) treatment were not completely supportive.
Objectives:
To investigate whether statin medication following PED treatment would improve the outcomes of intracranial aneurysm patients in a real-world setting.
Design:
A retrospective multicenter cohort study.
Methods:
Patients were selected from the PLUS registry study conducted from November 2014 to October 2019 across 14 centers in China. The population was divided into two groups: those who received statin medication after the PED treatment and those who did not receive statin medication after PED treatment. Study outcomes included angiographic evaluation of aneurysm occlusion, parent arteries stenosis, ischemic and hemorrhage complications, all-cause mortality, neurologic mortality, and functional outcome.
Results:
1087 patients with 1168 intracranial aneurysms were eligible; 232 patients were in the statin user group and the other 855 were in the non-statin user group. For the statin user group versus the non-statin user group, no significant difference was found for the primary outcomes of complete occlusion of aneurysm (82.4% versus 84.2%; p = 0.697). Of the secondary outcomes, none had a significant difference including stenosis of parent arteries ≥ 50% (1.4% versus 2.3%; p = 0.739), total subarachnoid hemorrhage (0.9% versus 2.5%; p = 0.215), all-cause mortality (0.0% versus 1.9%; p = 0.204), neurologic mortality (0.0% versus 1.6%; p = 0.280), excellent (95.5% versus 97.2%; p = 0.877), and favorable (98.9% versus 98.4%; p = 0.933) functional outcomes. The total ischemic complication rate (9.0% versus 7.1%; p = 0.401) was higher but not significant in the statin user group. The propensity score-matched cohort showed similar results. Results of binary multivariable logistic regression analysis and propensity score-matched analysis both showed that statin usage was not independently associated with an increased rate of complete occlusion or any other secondary outcomes. Subgroup analysis found the same result in patients who did not use statin before the procedure.
Conclusion:
Among patients with intracranial aneurysms, statin use after the PED treatment was not significantly associated with better angiographic and clinical outcomes. Well-designed studies are needed to further confirm this finding.
Introduction
As a new-generation therapy for cerebral aneurysms, flow diversion devices such as the pipeline embolization device (PED) have recently come to the fore. In contrast with other endovascular treatments, intracranial aneurysms would gradually occlude through intra-aneurysmal thrombosis and endothelial cell proliferation along the device at the aneurysmal neck after PED treatment. 1 According to the result from PLUS study, 2 the largest registry study of patients treated with PED to date, hyperlipidemia was a significant predictor of aneurysm occlusion. Due to the effective cholesterol-lowering effect, statins are considered the first-line medication for hyperlipidemia.3,4 Notably, statins are also regarded as the cornerstone treatment for atherosclerotic cardiovascular disease because of their essential role in improving endothelial function and reducing inflammation, which has an impact on various key vascular beds such as cerebrovascular arteries.5–7 In addition, several studies have demonstrated the protective impact of statin on in-stent thrombosis.8,9 Given all the evidence, the impact of statin might theoretically contribute to the occlusion of aneurysms and the reduction of in-stent stenosis.
However, prior studies regarding the association between statin use and angiographic and clinical outcomes post-PED treatment were not completely supportive of this hypothesis.10–12 Moreover, no information was provided from the Asian population, which appears to have a higher rate of intracranial atherosclerosis than the western population. 13 On the contrary, the Asian population was more sensitive to statins and might suffer more adverse outcomes from the long-term use of statin.14,15 It was crucial to comprehend the practical impact of statin medication following the PED procedure for the Asian population.
Based on the PLUS study data, we assessed whether statin medication following PED treatment would improve the angiographic and clinical outcomes of intracranial aneurysm patients in a real-world setting.
Methods
Study design and patient selection
Patients were selected from the PLUS study,2,16,17 which was a postmarket multicenter registry study investigating the embolization of intracranial aneurysms with the PED in China. The study included 1171 patients with 1322 intracranial aneurysms receiving embolization using PED treatment from November 2014 to October 2019 across 14 centers in China. Postoperative medication treatments such as antiplatelet and lipid-lowering drugs were collected. Follow-ups for angiographic and clinical outcomes were performed for up to 3 years.
Patients were eligible if the perioperative statins medication strategy were available. The population was divided into two groups: those who received statin medication after the PED treatment and those who did not receive statin medication after PED treatment. The following baseline characteristics were collected: age, sex, family history of aneurysm, smoking status, alcohol abuse, comorbidities (hypertension, diabetes, hyperlipidemia, cerebral infarction, coronary disease, and cerebral atherosclerosis), regular statin usage, number of aneurysms, ruptured aneurysms, aneurysm characteristics [size, neck, type (saccular, fusiform, dissecting, or other), and location (anterior or posterior)].
Clinical and angiographic follow-up
Early postoperative follow-up was performed during the first 30 days. The first imaging follow-up verified through two-dimensional digital subtraction angiography was conducted 3–6 months after the PED procedure. 17 Additional angiography follow-up was not required for patients who showed complete occlusion at follow-up; while for patients who showed incomplete occlusion, further angiographic follow-up was performed. For each patient, information from last follow-up was used for analysis. Clinical follow-ups were conducted by telephone or email for all patients.
Study outcomes
The primary outcome was complete occlusion of aneurysm by imaging screening at the last follow-up. Secondary outcomes included stenosis of parent arteries, ischemic complications, hemorrhage complications, all-cause mortality, neurologic mortality, and favorable [modified Rankin Scale (mRS) ⩽1] and excellent (mRS ⩽2) functional outcome. 18 The imaging data and endpoint events were reviewed by a central review committee consisting of two neuroradiologists and a senior neurointerventionist.
Statistical analysis
Continuous variables and covariates are summarized as means ± SDs and frequency (percentages), respectively. Comparisons between groups for continuous variables were evaluated by using the unpaired t-test and Wilcoxon rank-sum test; for categorical variables, we used Pearson χ2 test with continuous correction. Except for aneurysm characteristics, most statistical analyses were performed on a per-patient basis. For patients with more than one aneurysm, we used the first aneurysm treated to calculate the average aneurysm size and aneurysm neck for statistical analysis and clarified the aneurysm type.
Propensity-score matching (PSM) was performed using the greedy nearest-neighbor matching algorithm. We matched the logit of the propensity score using calipers of width equal to 0.2 of the standard deviation of the logit of the propensity score. 19 The matched potential confounders included age, comorbidities (hypertension, hyperlipidemia, coronary disease, cerebral infarction), regular statin use, and aneurysm characteristics (number, size, neck).
Binary multivariable logistic regression analysis and PSM analysis were both used to evaluate whether statin use was independently associated with outcomes. Adjusted variables in the multivariable model were baseline characteristics that show significance between the two groups (i.e. age, hypertension, hyperlipidemia, coronary disease, multiple aneurysms, and aneurysm neck). The non-statin user group was used as the reference group. We also carried subgroup analysis to analyze the effect of statin initiation among patients who did not use statin before the procedure. Statistical analysis was performed using Statistical Package for the Social Sciences (SPSS, version 26.0.0; IBM Corp, Armonk, New York, USA) and R (version 4.1.2).
Results
Baseline characteristics
Among all patients in the PLUS study, 1087 patients with 1168 intracranial aneurysms treated with PEDs were eligible for this analysis; whereas no data on post-operative statin medication status was provided in the remaining 84 patients. A total 232 patients received statin after the PED treatment and the other 855 did not. The average age of the statin user group was significantly higher than the non-statin user group (55.95 ± 10.13 versus 53.06 ± 11.77; p = 0.001; Table 1). Statin users were more likely to have hypertension, hyperlipidemia, coronary disease, and to take statin regularly. The other factors of patients’ baseline characteristics were similar between the two groups. For aneurysm characteristics, the proportion of patients with multiple aneurysms (37.9% versus 21.5%; p < 0.001) was larger in the statin user group. While statin users had a smaller mean aneurysm size (11.88 ± 8.62 versus 14.04 ± 8.84; p = 0.001) and a thinner aneurysm neck (5.98 ± 4.03 versus 6.70 ± 3.99; p = 0.015). After PSM, all baseline characteristics were comparable between the two groups.
Baseline patient and aneurysm characteristics.
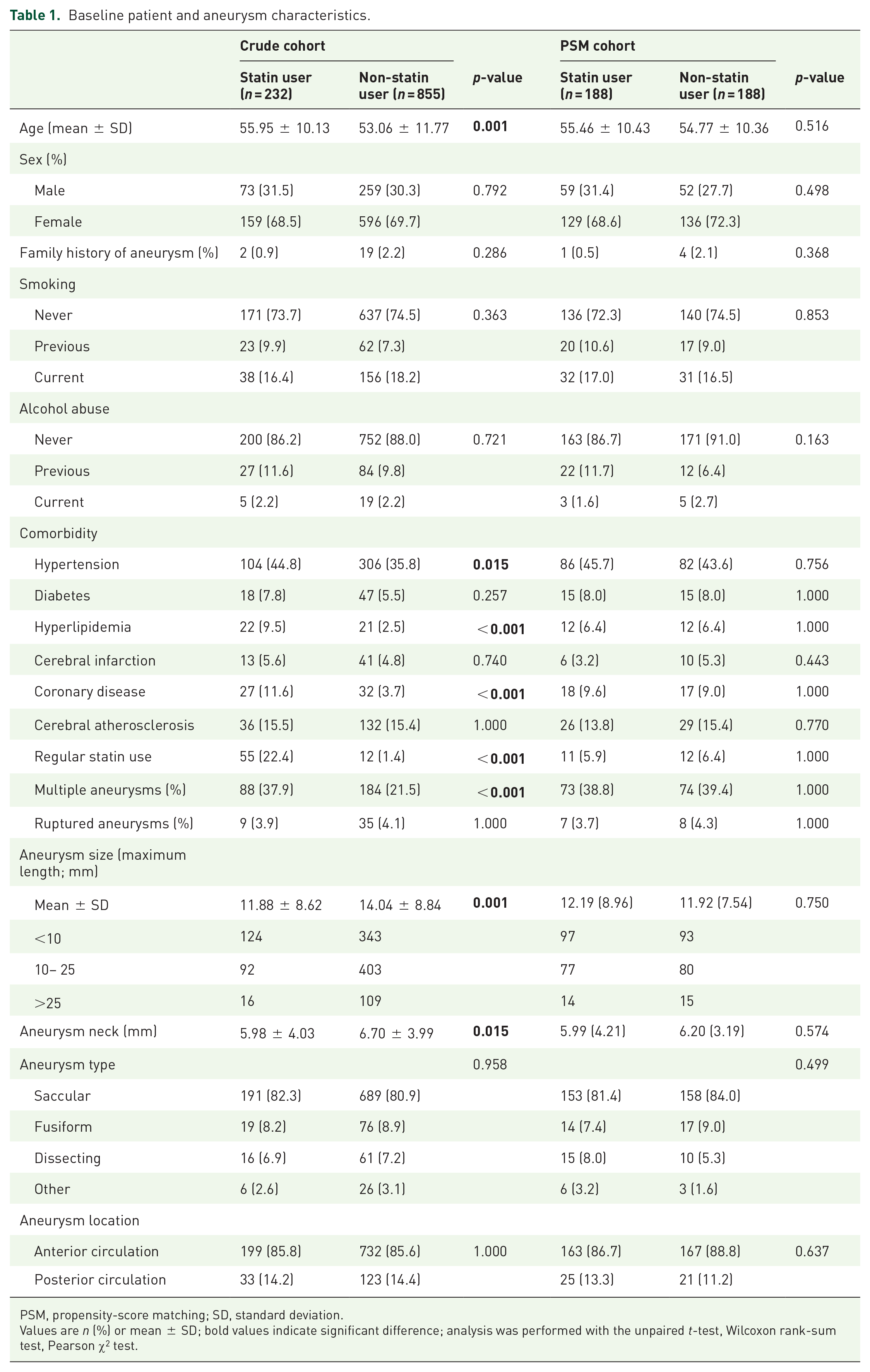
PSM, propensity-score matching; SD, standard deviation.
Values are n (%) or mean ± SD; bold values indicate significant difference; analysis was performed with the unpaired t-test, Wilcoxon rank-sum test, Pearson χ2 test.
Angiographic and clinical follow-up
Angiographic follow-up was available for 661 (60.8%) of the patients; with a mean follow-up duration of 9.0 ± 6.4 months. There were no differences in complete occlusion rates between the statin user group and the non-statin user group (82.4% versus 84.2%; p = 0.697; Table 2). Stenosis of parent arteries ⩾50% in the statin user group was lower than that in the non-statin group (1.4% versus 2.3%); however, the difference was not significant (p = 0.739). Rates of stenosis of <25% (6/142 versus 12/519; p = 0.342); ⩾25–50% (5/142 versus 6/519; p = 0.114); ⩾50–70% (1/142 versus 3/519; p = 1.000); and ⩾70–100% (1/142 versus 9/519; p = 0.615) were comparable between the two groups, respectively. After PSM, the complete occlusion rate of aneurysms remained not significant between the two groups (94/116 versus 94/111; p = 0.466). Four of 111 patients of the non-statin user group were found with stenosis of parent arteries ⩾50% while none was found in the statin user group, though it was not statistically significant (p = 0.119).
Angiographic and clinical outcomes.

PSM, propensity-score matching; mRS, modified Rankin Scale.
Analysis was performed with the Pearson χ2 test.
The total ischemic complication rate was higher in the statin user group (9.0%; 21/232) than in the non-statin user group (7.1%; 61/855), though not significant (p = 0.401). We found a lower total subarachnoid hemorrhage rate in the statin user group (0.9%; 2/232) compared with the non-statin user group (2.5%; 21/855) but not statistically significant (p = 0.215). No significant difference in perioperative and follow-up period ischemic complications and subarachnoid hemorrhage rate was found between the statin user group and the non-statin user group. All-cause mortality rate was not significantly different between the statin user group (0%; 1/232) and the non-statin user group (1.9%; 16/855; p = 0.204). Rates of neurologic mortality were comparable between groups (p = 0.280). No associations of excellent (p = 0.877) or favorable (p = 0.933) functional outcomes were observed between groups. The results of clinical outcomes after PSM were similar between the two groups (Table 2).
Multivariable analysis and subgroup analysis
The result of binary multivariable logistic regression analysis and PSM analysis showed that statin usage was not independently associated with an increased rate of complete occlusion of aneurysms or any other secondary outcomes (Figure 1).

Multivariable logistic regression and propensity score-matched analysis for the association between statin use and outcomes.
When including only patients who did not use statin before the procedure, we also observed no statistical association between the statin user group and non-user group for all outcomes (Table 3).
Subgroup analysis using multivariable logistic regression and propensity score-matched analysis for the association between statin use and outcomes among patients who did not use statin before pipeline embolization.

CI, confidence interval; mRS, modified Rankin Scale; OR, odds ratio.
Analysis was performed with the Pearson χ2 test.
Discussion
To our knowledge, this is the first study investigating the impact of statin medication after PED treatment on angiography and clinical outcomes. Our study included 1087 patients with 1168 intracranial aneurysms treated with PEDs and demonstrated that the statin medication following PED treatment was not associated with aneurysm occlusion rate, stenosis of parent arteries, ischemic complications, hemorrhage complications, or mortality. The finding indicated that intracranial aneurysm patients would not significantly benefit from statin use after the PED procedure.
With a usage rate prevalence of 18–45% in the population, statins are one of the most commonly prescribed medications all around the world. 20 Aside from its excellent impact on the regulation of cholesterol biosynthesis, 21 plenty of pharmacology pathways have been revealed to understand its pleiotropic effect. One main beneficial way of this effect on intracranial aneurysms was the improvement of endothelial function. By up-regulating the expression and the activity of endothelial nitric oxide synthase, statins could ameliorate endothelial dysfunction and improve vascular smooth muscle cell proliferation by promoting nitric oxide release.5,22,23 This effect was found to occur earlier than any significant reduction in serum cholesterol levels in the early period after statin initiation.24,25 Both in vitro and in vivo studies in a rabbit aneurysm model regarding statin-loaded stents showed favorable pro-endothelialization properties of statin. 26 Another protective effect of statin was its anti-inflammatory effect. Animal experiments have shown that statin inhibited leukocyte recruitment and the exudate production of interleukin-6, monocyte chemotactic protein-1, and RANTES (regulated on activation, normal T-cell expressed and secreted).27,28 It could suppress the development of aneurysms by inhibiting inflammatory reactions in aneurysmal walls and prevent the progression of preexisting aneurysms in rats. 29
The effect of statins on aneurysms observed in animal models on their overall benefit in patients cannot be linked directly due to the limitations of animal studies as models for human disease. While there was a recent randomized controlled trial conducted by Kang et al. 30 found that statin use could decrease aneurysm wall enhancement, indicating the potential modulation of alleviating aneurysm wall inflammation and prevention of rupture. The results of other observational findings did not completely support the hypothesis.31,32
To date, we still lack high-quality evidence on whether statin use could contribute to the outcomes of intracranial aneurysm patients who underwent endovascular intervention, especially for flow diversion devices. Flores-Milan et al. 11 performed a retrospective cohort study with 94 intracranial aneurysm patients to investigate factors associated with in-stent stenosis after PED treatment. The result of the multivariate analysis found statin usage to be the only variable to predictive and protective in-stent stenosis. However, the statistical power and generalizability of the results were limited by the modest sample size and single-center study design. By contrast, a study assessed the effect of statin use at the time of PED procedure by retrospectively reviewing the database data and did not identify any statistically significant association between statin therapy and higher aneurysm occlusion rate or clinical outcomes. 12 Similarly, a previous post hoc analysis study 10 that combined the data sets from three PED studies together in a per-patient set, demonstrated whether statin usage at the time of the procedure and follow-up duration following PED treatment was not associated with angiographic and clinical outcomes. Our study was consistent with the findings of two studies below10,12 and further confirmed that the use of the statin medication after PED treatment would not statistically significantly contribute to the outcomes.
There are several differences between the previous studies and the present one. Instead of focusing on the impact of statin use before or at the time of the PED treatment,10–12 we investigated the effects of statin use following the procedure. In our study, statin was primarily prescribed for patients who were not receiving routine statin treatment for hyperlipidemia or cardiovascular disease. Also, most of the patients were not regular statin users, so we could analyze the effect of statin initiation among these patients. The study also had the largest sample size to date and was conducted across multiple centers, which might better improve the statistical power. In addition, the results drawn in the Caucasian population cannot be directly applied to the Asian population who would typically have a higher risk of atherosclerosis and experiences a higher incidence of adverse effects from statins.14,15 We provided evidence for clinicians treating Asian patients who received PED treatment.
It’s crucial to understand that statin use after PED treatment should be considered a beneficial medication or an overtreatment practice. Although results from the previous PLUS study found that hyperlipidemia was a significant predictor of aneurysm occlusion, 2 statin use did not appear to have a beneficial effect on the outcomes. It could be proposed that it was the existing pathological vascular condition rather than the subsequent development of hyperlipidemia that affected aneurysm occlusion. Statins could not significantly reverse the formed arterial plaque on the PED-covered vessel. Also, the impact of statin may be limited if the parent arteries do not exhibit an inflammatory response or endothelial dysfunction.
There was evidence of the beneficial effect of statin for other endovascular procedures. According to a retrospective cohort study using the National Health Insurance Service–National Sample Cohort Database in Korea, statin treatment was shown to be significantly associated with a decreased pooled incidence of stroke, myocardial infarction, and all-cause death after coil embolization of cerebral aneurysms. 33 Brinjikji et al. 34 found that statin use was related to a decreased rate of aneurysm recurrence after small- and medium-sized ruptured aneurysms underwent endovascular coiling. The results of this study showed a slightly lower but not significant rate of stenosis of parent arteries ≥ 50% in the statin user group compared with the non-statin user group. One speculative explanation is that the influence of PED on vascular is weak, so this study with such a sample size was still insufficient to demonstrate the benefit of statin.
This study called for consideration of the necessity to initiate statin treatment for those who had not taken it before the PED treatment. As our subgroup analysis found no significant benefit of statin for patients who underwent PED procedures, clinicians should consider the benefits and drawbacks for patients because the research so far has not demonstrated clinically significant statin-specific improvement in clinical outcomes.
Nonetheless, we acknowledge that our study has several limitations. Limited by the retrospective design’s inherent bias, well-designed prospective studies were needed to further corroborate our findings. In addition, our angiographic follow-up relied on two-dimensional imaging, which may compromise the accuracy of stenosis percentage estimates. The use of three-dimensional imaging could provide more precise results in future studies. Another limitation is that our studies included only patients from China so the results cannot be readily extrapolated to other ethnicities or populations. We also lacked information on the type and dosage of statins as well as the relevant low-density lipoprotein cholesterol levels, which could allow us to evaluate the therapeutic effect of statins more accurately. Consequently, there might be an optimal type or dose of statin that would produce the desired outcomes. Future studies are demanded to clarify these matters.
Conclusion
Our finding indicated that statin medication after PED treatment is not significantly associated with improvement in angiographic or clinical outcomes. Well-designed studies are needed to further confirm this finding.
