Abstract
Background:
Prior to the conduct of the Head Position in Stroke Trial (HeadPoST), an international survey (n = 128) revealed equipoise for selection of head position in acute ischemic stroke.
Objectives:
We aimed to determine whether equipoise exists for head position in spontaneous hyperacute intracerebral hemorrhage (ICH) patients following HeadPoST.
Design:
This is an international, web-distributed survey focused on head positioning in hyperacute ICH patients.
Methods:
A survey was constructed to examine clinicians’ beliefs and practices associated with head positioning of hyperacute ICH patients. Survey items were developed with content experts, piloted, and then refined before distributing through stroke listservs, social media, and purposive snowball sampling. Data were analyzed using descriptive statistics and χ2 test.
Results:
We received 181 responses representing 13 countries on four continents: 38% advanced practice providers, 32% bedside nurses, and 30% physicians; overall, participants had median 7 [interquartile range (IQR) = 3–12] years stroke experience with a median of 100 (IQR = 37.5–200) ICH admissions managed annually. Participants disagreed that HeadPoST provided ‘definitive evidence’ for head position in ICH and agreed that their ‘written admission orders include 30-degree head positioning’, with 54% citing hospital policies for this head position in hyperacute ICH. Participants were unsure whether head positioning alone could influence ICH longitudinal outcomes. Use of serial proximal clinical and technology measures during the head positioning intervention were identified by 82% as the most appropriate endpoints for future ICH head positioning trials.
Conclusion:
Interdisciplinary providers remain unconvinced by HeadPoST results that head position does not matter in hyperacute ICH. Future trials examining the proximal effects of head positioning on clinical stability in hyperacute ICH are warranted.
Introduction
The optimal head position for various types of stroke may be important to ensure clinical stability, especially in the hyperacute stage of management. The Head Position in Stroke Trial (HeadPoST) 1 was a large cluster-randomized trial that attempted to determine whether head position could impact 3-month functional outcome in both acute ischemic stroke and acute intracerebral hemorrhage (ICH), finding no difference in outcomes between those managed with the head at 0° or 30° height. The methodology used and the conclusions drawn by HeadPoST investigators have been heavily criticized for a number of reasons, including enrollment of relatively minor stroke patients [National Institutes of Health Stroke Scale (NIHSS) median score 4 (interquartile range, IQR) = 2–8] who were many hours into stroke symptoms [median 14 (IQR = 5–35) h], unknown intervention fidelity, the potential for enrollment bias particularly among the ICH cohort, lack of vascular imaging and serial clinical monitoring, and use of a 3-month endpoint that had not been previously studied and therefore lacked data to support an understanding of effect size for the intervention in each of the patient populations enrolled. 2 Currently, it remains unknown whether HeadPoST has resulted in clinical practice changes for hyperacute ischemic stroke or ICH patients, although a multi-center clinical trial is underway in large vessel occlusion (LVO) ischemic stroke patients to study the effect of head position on clinical stability prior to mechanical thrombectomy. 3
Few studies have explored head positioning in the hyperacute ICH population using a serial monitoring approach to understand benefit or harm associated with the intervention.1,4–15 Unlike the majority of ischemic stroke patients, increased intracranial pressure (ICP) is a real concern in patients with ICH, although the incidence of increased ICP varies significantly in this patient population16–19 and is likely driven by hemorrhage volume, pre-existing intracranial capacity tied to factors such as atrophy, and hemorrhage location. Positioning the head of bed at 30° is theoretically sound20–31 especially in the absence of ICP monitoring where actual pressures are unknown. However American, European, Canadian, and Australian guidelines are silent on recommending ICH head positioning.32–35 We sought to understand whether clinical equipoise existed in selection of head position for patients with hyperacute ICH in the years following publication of HeadPoST, and to identify methods and endpoints that practitioners believed would best support a clinical trial of head positioning in hyperacute spontaneous ICH.
Methods
Survey aims
Institutional review board approval was obtained for the conduct of an international, web-distributed survey focused on head positioning in hyperacute ICH patients. The three main aims of the survey were (1) to explore whether clinical equipoise existed following publication of HeadPoST in selection of head positioning in the hyperacute ICH population; (2) to determine what interdisciplinary practitioners deemed to be the most appropriate primary endpoint and the timing for endpoint measurement for future hyperacute ICH head positioning clinical trials; and (3) to determine what serial clinical monitoring parameters and device measurements interdisciplinary practitioners deemed most important to definitively determine best head position for hyperacute ICH patients.
Targeted survey participants
Investigator consensus was reached that a variety of ICH interdisciplinary providers should be invited to complete the survey. We therefore targeted physicians, bedside nurses, and advanced practice providers (APPs) (i.e. nurse practitioners, clinical nurse specialists, and physician assistants) as these providers were most likely to assign/prescribe or assume responsibility for head positioning in hyperacute ICH patients.
Survey design
An English-language-only survey was constructed with a brief open-ended demographics section limited to country of practice, years of post-graduate clinical experience managing hyperacute ICH patients, annual hyperacute ICH patient volume, profession and title of clinical position, prescriber status, and primary unit where the respondent was employed or spent most time clinically (i.e. critical care, emergency, or stroke unit). We deliberately did not collect any identifying information about the hospital site or individual participants.
To understand uptake into practice of HeadPoST findings, we crafted the question, ‘Does your hospital have a policy or procedure that specifies placement of the head at 30° height in hyperacute ICH?’ (response options: yes, no, unsure). We also developed a Likert-type scale item (1 = strongly agree, 2 = agree, 3 = unsure, 4 = disagree, and 5 = strongly disagree) to further understand use of standard admission orders for head positioning, namely, ‘Written admission orders for hyperacute ICH patients always include positioning the head at 30-degrees elevation on my unit’.
Two Likert-type scale ICH ‘practice belief’ survey items were also developed; these items were accompanied by instructions stating clearly that there were no right or wrong answers to these questions, instead emphasizing that we were interested in respondents’ ‘beliefs’ about the items listed on the survey. The first practice belief item was focused on the clinical equipoise aim of the study, asking respondents to rate their beliefs to the statement, ‘HeadPoST findings provide definitive evidence that head positioning (elevated to 30-degrees or 0-degrees flat) does not matter in patients with hyperacute ICH’. The second practice belief item was focused on beliefs associated with the validity of head positioning as a method that could alone impact longitudinal patient outcomes: ‘I believe that head positioning ALONE can influence 6-month outcomes in ICH’. Use of ‘6-’ instead of ‘3-month’ was selected for this item given data demonstrating that ICH functional outcome is best measured at this point of time.36,37
Given that international guidelines remain silent on head positioning, one survey item was constructed to understand both the timing and preferred endpoint measures recommended by participants for use in future hyperacute ICH head positioning clinical trials. Specifically, respondents were asked to choose one of the following mutually exclusive options: (A) a proximal serial clinical stability endpoint using clinical measures such as the serial NIHSS or the Glasgow Coma Scale (GCS) during the head positioning intervention; (B) available instruments such as serial ICP or brain tissue oxygenation (PbtO2) monitoring in combination with clinical measures (NIHSS or GCS) during the head positioning intervention; (C) longitudinal measurement of modified Rankin Score (mRS) at 6 months; or (D) a combination of both the proximal and longitudinal measures described.
Prior to distribution, the survey was piloted by providers that mirrored the targeted interdisciplinary group, including some for whom English was a second language. During this phase, we encouraged open-ended comments related to the items to ensure our understanding of any limitations in how the items were developed and presented. We intently examined responses that could improve survey item content validity, and our findings were used to finalize the survey and ensure its applicability across international settings.
Survey assembly and distribution
The final survey was assembled in SurveyMonkey given its acceptance as a distribution platform worldwide, including more restrictive countries. Because email addresses for all possible interdisciplinary acute stroke clinicians are unavailable, a variety of survey distribution methods were used, including invitation by stroke-specific listservs, critical care listservs, and social media outlets (Twitter; Facebook). We also utilized purposive ‘snowball sampling’ with distribution of the survey to a sample of professional society leaders on each continent requesting these individuals identify and encourage participation of others known to them personally and through their professional organizations. The survey remained open from September 2018 through February 2019 and was reposted on social media and listservs twice after the initial posting; international leaders were also contacted twice with the second contact occurring 6 weeks after the initial contact. Participation in the survey was voluntary, and consent was implied by survey completion.
Data analysis
Data were downloaded, assembled, and cleaned in Microsoft Excel, and imported to SPSS 26.0 © (IBM) for analysis. Data were carefully reviewed to identify duplicate entries; specifically, four survey items were reviewed to identify potential duplicate submissions: (1) IP address, (2) country of submission, (3) annual ICH patient volume, and (4) responses to whether an ICH head positioning policy/procedure existed in the practice site. One participant response was identified as a potential duplicate and removed prior to analysis. Nursing and APP participant titles were carefully reviewed and grouped into appropriate categories, with the final APP category reflecting only those providers with at least an earned masters’ degree and licensure commensurate with prescribing and ensuring delivery of evidence-based practice for a particular hospital service or unit; participants placed in the ‘bedside nurse’ category consisted only of non-APP nursing participants. Descriptive statistics were used to summarize the responses and describe the sample; χ2 tests were used to determine differences in years of experience, and beliefs and practices by profession; statistical significance was prespecified at less than or equal to 0.05. Study findings are presented according to SURGE guidelines. 38
Results
A total of 181 responses were received representing 13 countries with the majority from North America (79%), although 11% were from Europe, 7% from Australia/New Zealand, and 3% from Asia. Participants were 38% APPs, 32% were bedside nurses, and 30% were physicians. Participants reported a median of 100 (IQR = 37.5–200) hyperacute ICH admissions/year at their facilities.
Overall, participants had a median of 7 (IQR = 3–12) years of post-graduate clinical experience managing acute stroke patients. Table 1 describes participants’ years of experience and clinical role. Physician respondents (n = 54) practiced primarily in North America (46%), Europe (30%), or Australia/New Zealand (17%) with a small volume (7%) representing Asia. The majority of physicians were vascular neurologists (57%), although 15% were neurocritical care physicians and 28% were general neurologists/hospitalist stroke physicians. Physicians reported a median 6 (IQR = 2–15) years of acute stroke management experience. APPs (n = 69) were from North America (80%), Europe (16%), and Australia/New Zealand (4%) with a median 7 (IQR = 3.25–10) years clinical experience, and bedside nurses (n = 58) were from North America (84%), Australia (12%), and Europe (4%) with median 7 (IQR = 3–13) years of experience; 90% of bedside nurses responding to the survey were neurocritical care nurses, whereas the remainder were stroke unit nurses. Overall physicians had less years of clinical experience with hyperacute ICH after training, compared with bedside nurses and APPs (p = 0.009).
Description of survey participants.

IQR, interquartile range.
Physicians had significantly fewer post-graduate years of experience managing acute stroke patients compared with APPs and bedside nurses: χ2 = 581; p = 0.009.
Overall, 54% of providers indicated that policies and procedures were in place at their practice sites to maintain head position at 30° in all hyperacute ICH patients. Table 2 provides responses to Likert-type scale items about practice beliefs and management of hyperacute ICH patients. Participants ‘agreed’ with the statement, ‘Written admission orders for hyperacute ICH patients always include positioning the head at 30-degrees elevation on my unit’, with no difference by provider type. In responding to the statement, ‘HeadPoST findings provide definitive evidence that head positioning (elevated to 30-degrees or 0-degrees flat) does not matter in patients with hyperacute ICH’, physicians and bedside nurses ‘disagreed’ with the statement, while APPs were ‘unsure’ (χ2 = 55.4; p = 0.09) about agreement; overall, 53% of participants disagreed and 24% were unsure about the value of HeadPoST findings on head positioning in hyperacute ICH. In response to the second practice belief statement, ‘I believe that head positioning ALONE can influence 6-month outcomes in ICH’, overall, respondents were unsure (3; IQR = 2–3) about agreement with this statement, with no differences in belief found by profession.
Participants’ practices and beliefs about head positioning in hyperacute intracerebral hemorrhage.

ICH, intracerebral hemorrhage; IQR, interquartile range.
Likert-type scale: 1 = strongly agree; 2 = agree; 3 = unsure; 4 = disagree; 5 = strongly disagree.
Table 3 presents participants’ beliefs about ‘best’ endpoint and endpoint timing for future hyperacute ICH head positioning clinical trials providing breakdown by profession. Overall, 82% of participants recommended use of proximal endpoint measures: Use of all available technology (ICP and PbtO2) in combination with serial clinical measures (NIHSS and GCS) was recommended by 68% of participants overall, and 14% overall preferred use of proximal serial clinical measures (NIHSS and GCS) alone. Importantly, only 14% of respondents believed that a longitudinal measure of functional status using the modified Rankin Scale (mRS) would be the best measure for a hyperacute ICH head positioning clinical trial, and 4% believed both proximal and longitudinal endpoints should be considered. No differences in participants’ beliefs about endpoints and timing were found by profession.
Participants’ beliefs about best endpoints for future intracerebral hemorrhage head positioning clinical trials.
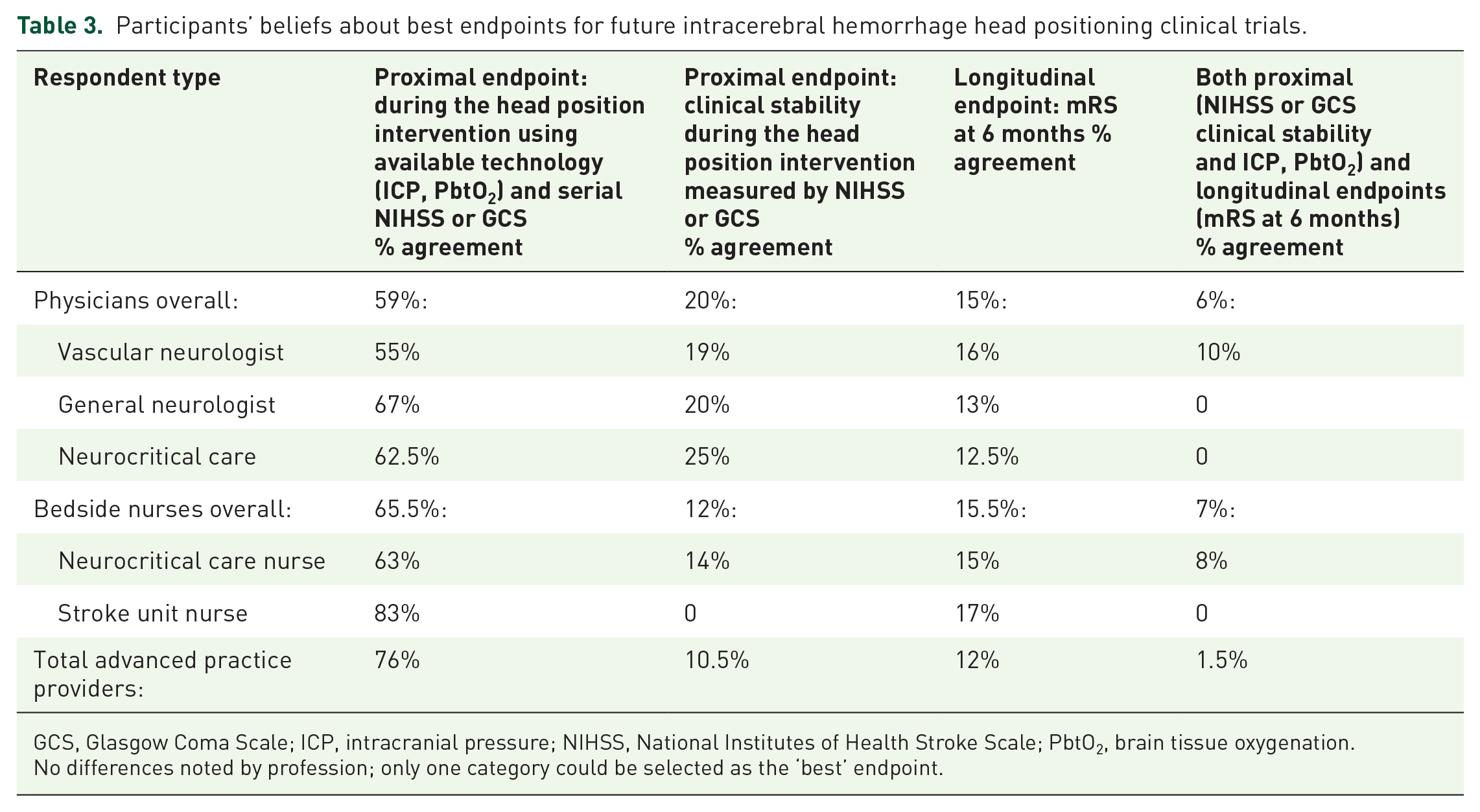
GCS, Glasgow Coma Scale; ICP, intracranial pressure; NIHSS, National Institutes of Health Stroke Scale; PbtO2, brain tissue oxygenation.
No differences noted by profession; only one category could be selected as the ‘best’ endpoint.
Discussion
Our survey showed that clinical equipoise is lacking about optimal head positioning in patients with hyperacute ICH, with participants either not agreeing that HeadPoST results were definitive or participants being unsure of the trial’s implications for practice. Furthermore, head elevation in hyperacute ICH was supported by clinical policies and procedures in 54% of participants’ hospitals, and there was agreement overall that written admission orders called for 30° head positioning in hyperacute ICH. These findings reflect the silence found in the US and European guidelines on ICH head positioning, as well as the recommendations for 30° head positioning found in the Canadian ICH guidelines.32–35
We also documented that proximal endpoints measured during head positioning interventions in hyperacute ICH were overwhelmingly supported by our respondents. This may provide the rationale for why HeadPoST findings have not been widely translated into clinical practice in hyperacute ICH patients due to unanswered questions about how these patients actually fared during the positioning intervention. ICH is a dynamic process in which critical changes are known to evolve beyond the time of initial assessment 39 with the goal of any intervention being absence of harm. A proximal endpoint such as neurologic worsening, would provide crucial information on the effect of an intervention. This would be best accomplished by the use of serial neurologic assessments utilizing a validated scale such as the NIHSS 40 to detect clinically relevant changes in stability or deterioration.
The body of work examining the relationship between head positioning and intracranial hemodynamics demonstrates that among patients with increased ICP, positioning the head from 0° to 30° significantly reduces ICP,27–31,41–48 although most of these studies included traumatic brain injury patients with few ICH cases enrolled. Findings from a large cohort of consecutive ICH cases found that only 24% of patients underwent ICP monitoring; however, 70% of these experienced at least one episode of ICP reaching values greater than 20 mmHg, 49 raising concern that increased ICP may occur in many patients without objective confirmation of its presence. Pressure gradients may also vary in different areas of the brain depending on hematoma size as well as location, regardless of actual ICP measures, 16 and ICH patients with intraventricular hemorrhage that develop hydrocephalus are known to experience poor functional outcomes.17–19 Collectively, these findings suggest that head elevation may in fact be one of the most important first steps in managing hyperacute ICH patients, particularly in those with hematoma volume ⩾ 30 ml, active bleeding (spot sign positive), and intraventricular extension.17–19
Interestingly, we found no differences by profession in opinions about head positioning practices, beliefs about HeadPoST, or definitive endpoints for a future trial. This may reflect widespread dissemination across professions of the implications of increased ICP on clinical outcomes, including head elevation practices that have been long embedded as standard of care. In addition, as the characteristics of the HeadPoST ICH cohort have not been well described, little is known about these patients’ hematoma volume and location, presence of intraventricular extension, availability of ICP monitoring/measured ICP values, or ICH scores on enrollment, as well as the etiology of these hemorrhages. This may have contributed to the lack of clinical equipoise found by our study.
Our study has several limitations that must be acknowledged. First, our survey has not undergone psychometric evaluation and was developed using content expertise. However, it was constructed based on the literature and expert providers’ feedback and was pilot tested and refined prior to distribution. Second, although we cautiously examined each submission to identify potential duplicates from the same site, we may have inadvertently missed a duplicate response or eliminated a response that was not a duplicate. In addition, because we were most interested in individual opinions, any potential duplicates that were included would have only affected one demographic item tied to ‘annual admission volume of ICH’ and the item inquiring about whether a policy/procedure for head positioning existed at the practice site. While our survey distribution methods may have introduced bias, our approach was significantly less biased than that used for the pre-trial HeadPoST survey which included physicians known to the investigators as well as those who were part of the principal investigator’s stroke research network and participants in the Enhanced Control of Hypertension and Thrombolysis Stroke Study (ENCHANTED). 50 Finally, we acknowledge that our English-only survey may have limited our response rate. Despite these limitations, our survey return was considerably larger than that of the pre-trial HeadPoST survey, providing the largest international survey return to date on head positioning in hyperacute stroke. Consistent with our survey methods, we assume that respondents answered truthfully and accurately yet we cannot confirm this.
Conclusions
Despite HeadPoST findings suggesting that head position has no effect on acute stroke management, providers in multiple disciplines remain unconvinced and continue to favor 30° head elevation in hyperacute ICH patients. Future work in head positioning of hyperacute ICH patients should be supported by proximal serial assessment of both clinical and technology-based measures during the head position intervention to determine if 30° is indeed optimal, and if 0° head positioning is safe.
