Abstract
Background:
Intravenous immunoglobulin (IVIg) and therapeutic plasma exchange (TPE) are among the main immunotherapies for neurological disorders. Their benefit is greatest in immune-mediated conditions, but their distinct efficacy cannot be simply explained.
Objectives:
This review aimed to systematically identify studies comparing the efficacy of TPE and IVIg treatments for selected autoimmune neurological disorders and identify optimal therapies for each condition.
Data Sources and Methods:
PubMed, MEDLINE and Embase databases were searched for original publications from 1990 to 2021. Additional publications were identified via expert recommendations. Conference abstracts older than 2017, review articles and articles without information on TPE and IVIg comparison in title and abstract were excluded. Risks of bias were descriptively addressed, without a meta-analysis.
Results:
Forty-four studies were included on Guillain–Barré syndrome (20 studies – 12 adult, 5 paediatric, 3 all ages), myasthenia gravis (11 studies –8 adult, 3 paediatric), chronic immune–mediated polyradiculoneuropathy (3 studies –1 adult, 2 paediatric), encephalitis (1 study in adults), neuromyelitis optica spectrum disorders (5 studies –2 adult, 3 all ages) and other conditions (4 studies – all ages). TPE and IVIg were mostly similarly efficacious, measured by clinical outcomes and disease severity scores. Some studies recommended IVIg as easy to administer. TPE procedures, however, have been simplified and the safety has been improved. TPE is currently recommended for management of neuromyelitis optica spectrum disorder relapses and some myasthenia gravis subtypes, in which rapid removal of autoantibodies is crucial.
Conclusion:
Despite some limitations (e.g. the low evidence levels), this review provides an extensive 30-year-long overview of treatments for various conditions. Both IVIg and TPE are usually comparably efficacious options for autoimmune neurological disorders, with few exceptions. Treatment choices should be patient-tailored and based on available clinical resources. Better designed studies are needed to provide higher-level quality of evidence regarding clinical efficacy of TPE and IVIg treatments.
Keywords
Introduction
Intravenous immunoglobulin (IVIg) and therapeutic plasma exchange (TPE) have been used to treat several immune-mediated neurological conditions.1,2 While these treatments are often reported to have similar efficacies, there are some nuances and differences in their utility in clinical practice.
IVIg contains purified human immunoglobulins, manufactured from large pools of plasma from thousands of healthy donors.1,3 IVIg is thought to achieve its therapeutic effects by inactivating the immune mediators (e.g. immune complexes and antibodies targeting nervous system components) and modulating immune responses [e.g. fragment crystallisable (Fc) receptor modulation, impeded activation of the complement system, altered B- and T-cell activation, decreased inflammatory cell adhesion and migration].1,4
In contrast to IVIg, TPE aims to eliminate the immune mediators.1,3 During TPE, the patient’s blood is separated into plasma and cellular components. The plasma fraction is replaced with fresh frozen plasma from a donor or a colloid solution (e.g. albumin), which is re-infused into the patient together with autologous blood cells.1,3,5 Between 66% and 85% of the original noncellular intravascular constituents are removed after exchange of 1.0–1.5 plasma volumes. 3 Removal of antibodies that are directly pathogenic may be particularly important in some conditions, such as myasthenia gravis (MG)6,7 and neuromyelitis optica spectrum disorders (NMOSD). 8
Both IVIg and TPE decrease the levels of circulating autoantibodies and immune mediators, although via different mechanisms and to varying degrees. As each approach brings specific benefits but also risks,1,3 the therapy of choice must be tailored to each patient and their condition. For instance, neurologists may prefer IVIg over TPE in patients with severe infections. Furthermore, it may not always be feasible to administer each of these therapies. TPE requires planning and provision (e.g. trained medical personnel); TPE-associated complications may be catheter-related (e.g. suboptimal placement of central venous lines, line sepsis, hematoma) or exchange-related (e.g. electrolyte imbalance or hypotension).1,3 Similarly, IVIg may cause adverse events (AEs), which are rarely severe (e.g. renal impairment, haemolytic anaemia, arterial or venous thrombosis) but may occur more frequently, particularly if patients had already experienced adverse effects or if IVIg is administered for the first time. 9 The choice between TPE and IVIg often depends on several factors: availability, ease of administration and cost in local settings. 10 Specific circumstances such as the ongoing COVID-19 pandemic may further impact availability and usage. 10
The recommendations for IVIg or TPE treatment of neurological disorders vary locally, and many were published more than 5 years ago.2,3,5,11 Establishing robust recommendations for IVIg and TPE use in autoimmune neurological disorders is limited by low disease incidence leading to underpowered therapeutic study designs, often enrolling fewer than 100 patients. This systematic literature review (SLR) aims to summarise data on IVIg and TPE treatment efficacies in several autoimmune diseases over a 30-year period.
Material and methods
Review objectives
To conduct an SLR of studies comparing the efficacy of TPE versus IVIg to treat autoimmune neurological diseases.
To identify those conditions in which both treatments show similar efficacies and those where one is preferred due to superior clinical benefit.
To recommend how to apply available evidence for treatment of rare autoimmune neurological diseases and recommend strategies for future studies.
Search strategy and selection criteria
This SLR focused on neurological disorders treated with IVIg or TPE. The PubMed, MEDLINE and Embase databases were screened over two rounds: the first in 2020 (period: 1990–2020) and the second, as an update of the first one, in 2021 (period: January 2020–June 2021), each with the same set of four search strings (Supplementary Table 1). Three searches (one search of PubMed, Embase and Medline each; Supplementary Table 1) were initially run more ‘globally’, that is, including all references describing IVIg and TPE treatments in all neurological conditions. This strategy, however, yielded few records directly comparing IVIg and TPE, which prompted us to run a fourth search (in PubMed) specifically designed to retrieve papers comparing both therapies (Supplementary Table 1). Immunoadsorption was not included as a search term, even though it is an alternative to TPE in some pathophysiological backgrounds. Immunoadsorption is a distinct technique, used to a significant extent only in some countries (e.g. Japan and Germany). 12 As the present review focuses on the widely available TPE and IVIg comparison, studies on immunoadsorption were not within its scope. The authors also provided input on relevant publications.
The following studies were included: original publications (from 1990 to 2021) or congress/conference abstracts (from 2017 to 2021), comparing efficacy of IVIg and TPE therapies for neurological disorders; studies in humans; no restrictions for language or geographical region. Studies in which patients were switched from one treatment to another were also included.
Publications on organ transplantation or non-neurological conditions, review papers, noncomparative TPE or IVIg efficacy studies (with the exception of NMOSD studies, see below) and studies with combined TPE and IVIg treatments were excluded. While review articles (narrative, systematic and meta-analyses) were excluded, additional studies of interest were identified by checking their reference lists.
During the first search round, four reviewers screened the identified records for inclusion in the review, and conflicts were resolved by consensus. Three reviewers were involved during the second round, in which conflicts were resolved by consensus and expert opinion. Selected records included papers and abstracts meeting all inclusion criteria. An exception was made for studies on NMOSD treatment, because of the relevance of this group of disorders, for which the search identified no comparative studies. Data from selected studies were extracted by two independent reviewers in the first and by one reviewer in the second search round. Oxford Centre for Evidence-Based Medicine criteria were used to determine study robustness [level of evidence (LoE)] and grade of recommendation.13,14
The following characteristics were tabulated from each included study: country of study, study design, LoE, treatment protocols, objectives, main outcomes and conclusions. A meta-analysis of extracted data could not be done, due to the overall low LoE and differences in protocols of the included studies. Risks of bias were thus descriptively assessed following expert input.
Results
Search results
After conducting both search rounds and applying the selection criteria, a total of 2262 records (2067 publications and 195 conference abstracts) were identified in PubMed, MEDLINE and Embase databases; 1189 duplicates were removed yielding 1073 unique records. After excluding an additional 933 records, 140 publications and abstracts were assessed for eligibility. Of those, 33 were analysed in this review. Expert input provided 11 additional records of interest (Figure 1).

PRISMA flow diagram.
Efficacy of IVIg and TPE therapies per category
Guillain–Barré syndrome
Guillain–Barré syndrome (GBS) is an autoimmune disease of the peripheral nervous system, often preceded by a bacterial or viral infection and diagnosed more often in men. 15 It is characterised by rapidly progressing, ascending flaccid limb paresis and areflexia, usually starting in the lower limbs, and worsening over 1–4 weeks. 15
GBS is usually classified into four subtypes: (1) acute inflammatory demyelinating polyradiculoneuropathy (AIDP), in which immune targeting of Schwann cells or myelin results in segmental demyelination with secondary axonal loss; (2) acute motor axonal neuropathy (AMAN), in which axonal degeneration occurs as a consequence of an autoimmune reaction against ganglioside epitopes; (3) acute motor and sensory axonal neuropathy (AMSAN); (4) Miller-Fisher syndrome, characterised by ataxia, ophthalmoplegia and areflexia but usually not limb weakness. The subtype prevalence differs between continents. 16
The systematic search identified 20 studies comparing TPE versus IVIg (Table 1), in which treatment efficacy was measured by improvement on established disability scales, mostly the Hughes and GBS disability scale; one study also measured compound Functional Independence Measure and Montebello Rehabilitation Factor Score. 30
TPE and IVIg in treatment of Guillain–Barré syndrome.
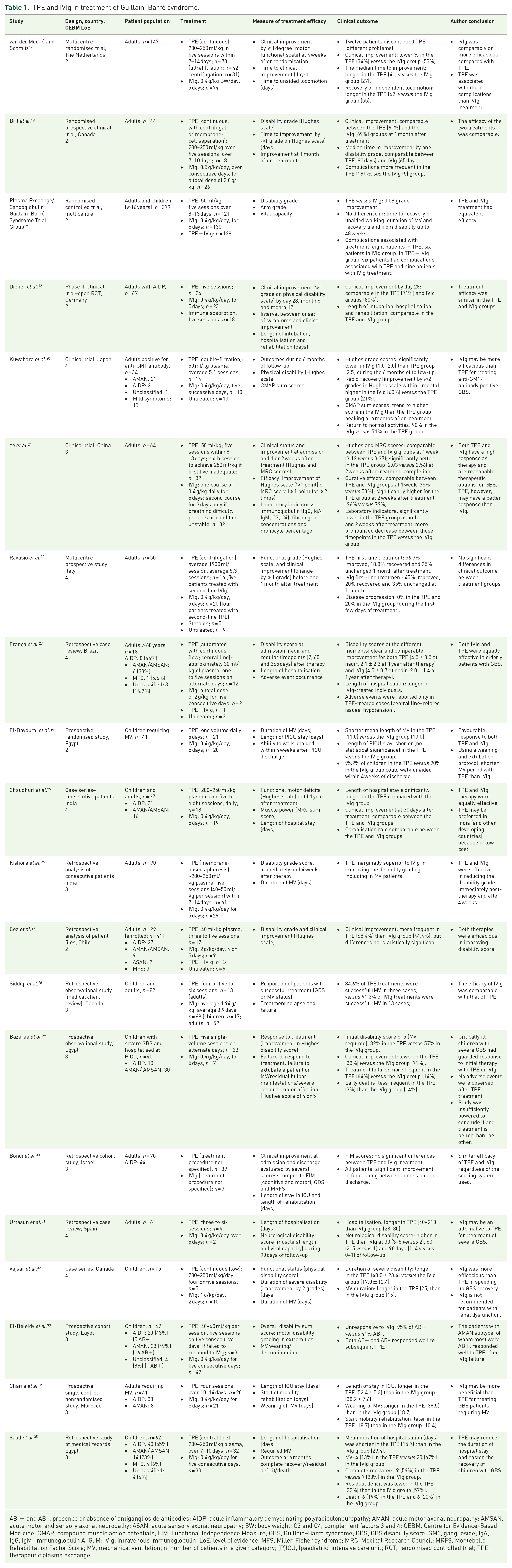
AB + and AB–, presence or absence of antiganglioside antibodies; AIDP, acute inflammatory demyelinating polyradiculoneuropathy; AMAN, acute motor axonal neuropathy; AMSAN, acute motor and sensory axonal neuropathy; ASAN, acute sensory axonal neuropathy; BW: body weight; C3 and C4, complement factors 3 and 4; CEBM, Centre for Evidence-Based Medicine; CMAP, compound muscle action potentials; FIM, Functional Independence Measure; GBS, Guillain–Barré syndrome; GDS, GBS disability score; GM1, ganglioside; IgA, IgG, IgM, immunoglobulin A, G, M; IVIg, intravenous immunoglobulin; LoE, level of evidence; MFS, Miller-Fisher syndrome; MRC, Medical Research Council; MRFS, Montebello Rehabilitation Factor Score; MV, mechanical ventilation; n, number of patients in a given category; (P)ICU, (paediatric) intensive care unit; RCT, randomised controlled trial; TPE, therapeutic plasma exchange.
Six records were (randomised) clinical trials mostly conducted in the 1990s, with numbers of randomised and treated patients ranging from 24 to 379.12,17–21 Four of these trials concluded that IVIg and TPE were comparably efficacious in treating GBS (LoE: 2–4).12,18,19,21 In addition, van der Meché et al. 17 concluded that IVIg is at least comparably effective or may even be superior to TPE for treating GBS in patients aged 4 or more. Notably, the median treatment delay (TD; time elapsed between symptom onset and start of treatment) in the same study was longer in the TPE than IVIg group [1.0 (0–4) versus 0.0 (0–3) days]. This study also reported significantly lower complication rates in IVIg- versus TPE-treated patients, 17 which was observed as a nonsignificant trend in two other trials.12,18 Specifically, Diener et al. 12 observed comparable rates of clinical AEs, but lower rates of treatment-related AEs with IVIg than TPE treatment. This is the only trial that distinguished between disease- versus treatment-specific AEs in GBS patients. 12 While the above-mentioned four trials12,17–19 included unselected patients or those with moderate or severe GBS, Kuwabara et al. 20 examined treatment efficacies in patients positive for antiganglioside antibodies and reported significantly faster clinical recovery after IVIg than TPE treatment. Of note, this trial included 24 patients, and the authors stated that efficacy of TPE may have been decreased by double plasma filtration. 20
Nine other studies (four prospective, five retrospective) concluded that TPE and IVIg were both viable options or that TPE was slightly more efficacious than IVIg for GBS treatment (LoE: 2–4).22–30 Similar to the clinical trials above, most of these studies agreed that the safety of TPE and IVIg was acceptable. One study indicated that TPE may be more frequently associated with complications. 23 Of note, the route of TPE administration (central versus peripheral), the TPE device and the IVIg producer were often insufficiently detailed. Ravasio et al. 22 reported having used IVIg from six different producers in their study, but did not state which product was used in which patients. Also, only three studies specified the TD duration.22,26,29 The median TD was comparable between IVIg and TPE groups, except in Ravasio et al. 22 [9.0 (4–65) versus 5.5 (2–40) days]. 22
The remaining five studies on GBS came to different conclusions on how IVIg and TPE compare with each other in treating GBS. Two studies in adults reported shorter hospital stay periods and higher recovery rates with IVIg,31,34 although the study of Urtasun et al. 31 included only six patients.
Two studies in Egyptian children favoured TPE: 95% of IVIg-refractory patients positive for antiganglioside antibodies (AB+) responded to TPE treatment, 33 and the second reported shorter hospitalisation and higher recovery rates in patients treated with TPE versus IVIg. 35 Of note, the predominant GBS subtype differed: 49% of patients had AMAN 33 and 65% had AIDP. 35 A Canadian study, however, reported longer mechanical ventilation and duration of two-grade clinical improvement after TPE treatment. 32 The paediatric studies differed in their sizes (e.g. 15 patients in the Canadian study compared with the 47 and 62 in the Egyptian studies), geographical regions and selection of patient populations (e.g. AB-status stratification, time elapsed between symptom onset and enrolment).
The grade of recommendation for superiority of TPE over IVIg and vice versa was low (grade C). The grade of recommendation for TPE being equally efficacious to IVIg was moderate (grade B).
MG
MG is an autoimmune disorder in which antibodies, mainly against the nicotinic acetylcholine receptor (AChR) or muscle-specific tyrosine kinase (MuSK), cause defective neuromuscular signal transduction.7,36,37 The suboptimal signal transduction results in fluctuating and transient muscle weakness and fatiguability. MuSK-antibody-associated disease (MuSK MG) occurs in about 5% of MG patients and is phenotypically distinct from MG with AChR antibodies (AChR MG). 7 While AChR MG is often accompanied by asymmetry of ptosis, divergent gaze and distal limb weakness, the dominant feature of MuSK MG is bulbar weakness. 7 Thymic comorbidities (e.g. hyperplasia and thymoma) are common in patients with MG and result in referral of selected patients for thymectomy.36–38
Some patients experience a myasthenia crisis – an acute episode of respiratory muscle weakness ending in respiratory failure.6,38 Myasthenia crisis may be precipitated by systemic physiological changes (e.g. medication, pregnancy or illness).6,38
Out of 11 comparative studies (Table 2), 8 concluded that TPE and IVIg were equally efficacious (LoE: 2–4)40,42–47,49 and 2 concluded that TPE was more efficacious than IVIg (LoE: 2 and 3).39,48 One study favoured IVIg for the shorter patient intubation times necessary during treatment of MG (LoE: 2). 41 The grade of recommendation for either therapy was moderate (grade B).
TPE and IVIg in treatment of myasthenia gravis.

ab, antibody; AChR, acetylcholine receptor; CEBM, Centre for Evidence-Based Medicine; IA, immune adsorption; ICU, intensive care unit; IgG and IgM, immunoglobulin G and M; IVIg, intravenous immunoglobulin; LoE, level of evidence; MG, myasthenia gravis; MGFA, Myasthenia Gravis Foundation of America; MV, mechanical ventilation; n, number of participants in each category; PrsmR, presynaptic membrane receptor; QMGS, quantitative MG score; TPE, therapeutic plasma exchange; VAS, visual analogue scale.
Generalised MG (including moderate and severe forms)
Out of three clinical trials in adults with MG (LoE: 2),39–41 two found that TPE and IVIg treatments comparably reduced disease severity [measured by, e.g., the quantitative MG score (QMGS)].40,41 Katzberg et al. 40 described that baseline QMGS was predictive of IVIg but not TPE response, although they stated that neither disease duration nor underlying medical conditions were decisive factors for the use of either therapy. Alipour-Faz et al. 41 suggested that IVIg may be advantageous to reduce the period of intubation in MG patients undergoing thymectomy. Disease duration, however, was significantly longer in the TPE- versus the IVIg-treated group in this study. Liu et al. 39 reported on higher short-term clinical effectiveness and more efficient removal of immune mediators (anti-titin, -AChR and serum IgG antibodies) by TPE than IVIg in adults with late-onset MG. TPE-treated patients required relatively less respiratory support and were hospitalised for a shorter time than IVIg-treated patients, but AEs were relatively more frequent in the TPE- than the IVIg-treated group. 39
In agreement with the mentioned clinical trials of Alipour-Faz et al. 41 and Katzberg et al., 40 four other studies in patients with generalised MG found TPE and IVIg to be comparably efficacious treatment options.42–45 Only one study by Barth et al. 45 relied on the Osserman classification as a disease severity measure, but still concluded that IVIg was a similarly efficacious alternative to TPE in patients with MG who were referred for thymectomy. This study in 84 patients found that IVIg and TPE similarly improved electrophysiological characteristics 2 weeks after treatment, but this improvement persisted until day 28 only for IVIg. 45 The authors nevertheless concluded that both immunomodulatory therapies were viable options to treat moderate to severe MG. 45 Mandawat et al. 44 found that mortality and complication rates were comparable, but that length of hospital stay was longer with TPE. One retrospective study in children and adolescents reported on a 100% response rate to TPE (7/7 patients) versus 50% for IVIg (5/10 patients) treatment. 46 While response rates to TPE treatment were found to be more consistent, both TPE and IVIg were comparably efficacious as maintenance therapy in juvenile MG (LoE: 3). 46
MG exacerbation and myasthenia crisis
Treatment responses to TPE and IVIg were comparable with respect to severity score, intubation and hospital stay length in adults.47,49
A single clinical trial by Gajdos et al. 47 reported on similar response rates and muscular score improvement with TPE and IVIg in adults with MG exacerbation. Similar to Liu et al., 39 this trial found that AEs were relatively more frequent after TPE than IVIg treatment. Comparable percentages of TPE- and IVIg-treated patients, however, had a decrease in anti-AChR antibodies. 47 Of note, the study by Gajdos et al. 47 was limited due to unmasked analyses and biased towards IVIg benefits through failure to administer a full course of TPE.
In agreement with the above, two other studies found that, while TPE was efficacious in treating patients with MG exacerbations or myasthenia crises, it was also associated with relatively more AEs and complications.48,49 Qureshi et al. 48 reported on more favourable ventilatory status (percent of extubated patients at 2 weeks) and higher proportion of patients without disability at 1 month after TPE versus IVIg treatment. The same study observed higher efficacy with respect to severity score improvement, but longer (although not statistically significant) intensive care unit and hospital stay times for patients treated with TPE versus IVIg (LoE: 3, grade: B). 48 While the studies of Pittayanon et al. and Qureshi et al. support the use of TPE as an effective treatment of MG exacerbation and myasthenic crisis, the low numbers of recruited patients limit data interpretation and are insufficient to support superiority of TPE. The efficacy of TPE and IVIg treatments was comparable for juvenile myasthenia crisis (LoE: 3). 46
Additional considerations regarding studies in MG
Of the 11 studies in patients with MG, five included information on autoantibody serological status.39,40,45–47 All five reported comparable seropositivity rates for anti-AChR antibodies (range: 61–80%)39,40,45–47 and three on anti-MuSK antibody seropositivity (range: 4–18%).40,45,46 Aside from Katzberg et al., 40 none of these studies stratified clinical outcomes and treatment efficacy by autoantibody serological status; effects of IVIg and TPE on MuSK MG were investigated only in noncomparative studies. The degree of MG-associated autoantibody reduction was similar or favoured TPE where this was studied in comparative trials on MG.39,42,45,47 Patients with MuSK MG have been consistently shown to respond well to TPE,50,51 but less so to IVIg. Anti-MuSK antibodies mainly belong to the immunoglobulin G4 (IgG4) subclass and are therefore not complement fixing 52 and are directly pathogenic on a passive transfer mouse model. 53 Their pathogenicity probably underpins the benefit of direct removal by TPE. Acute treatment of MuSK MG crisis has TPE as the mainstay 7 and transfer to a hospital unit with an available TPE device is warranted in this MG variant.
Chronic inflammatory demyelinating polyradiculoneuropathy
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is an acquired, mostly treatable, immune-mediated disorder of the peripheral nervous system with slow symptom progression, exceeding 8 weeks, followed by a progressive or relapsing course. 54 CIDP is usually treated with corticosteroids, TPE, IVIg or a combination thereof; chronic treatment with immunotherapies is a valuable alternative in patients with certain CIDP subtypes refractory to corticosteroid treatment. 54 CIDP has been classified into different subtypes falling under typical and atypical CIDP. 54
The present literature search identified three records, one clinical trial in adult patients (LoE: 3) 55 and two paediatric studies (LoE: 4)56,57 comparing IVIg and TPE efficacy in treating CIDP (Table 3). Treatment responses and outcomes were measured by changes in functional disability: modified Rankin scale (MRS)56,57 or a combination of several scores. 55
TPE and IVIg in treatment of chronic inflammatory demyelinating polyradiculoneuropathy.
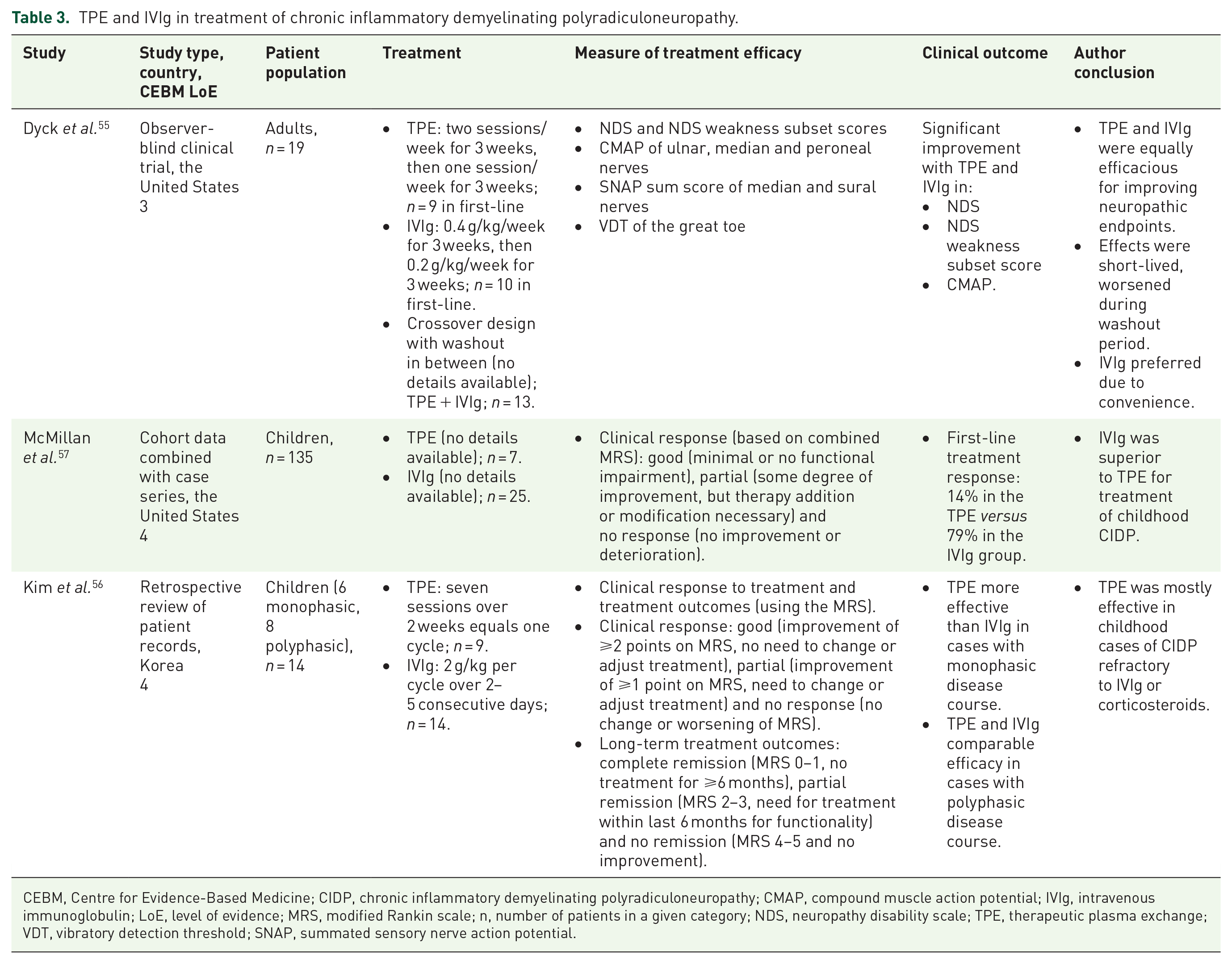
CEBM, Centre for Evidence-Based Medicine; CIDP, chronic inflammatory demyelinating polyradiculoneuropathy; CMAP, compound muscle action potential; IVIg, intravenous immunoglobulin; LoE, level of evidence; MRS, modified Rankin scale; n, number of patients in a given category; NDS, neuropathy disability scale; TPE, therapeutic plasma exchange; VDT, vibratory detection threshold; SNAP, summated sensory nerve action potential.
The single clinical trial in adult patients with CIDP found no differences between TPE and IVIg treatment in terms of neurological disability and weakness scores or summated muscle motor potentials. 55 The same trial reported that therapeutic effects were short-lived for both therapies and waned during the washout period. 55 The authors concluded that IVIg may be preferred due to its availability and ease of administration. The time of study conduct (1994), low number of enrolled participants and the complex and insufficiently clarified study design must be taken into account when interpreting the results of this study. 55
Data from the paediatric studies were inconsistent, with McMillan et al. 57 reporting a higher proportion of responders among IVIg- than TPE-treated patients; Kim et al. 56 found that TPE and IVIg were similarly efficacious, but TPE was likely a more favoured option for IVIg- and corticosteroid-refractory CIDP. These studies also reported different clinical experiences with TPE administration. McMillan et al. 57 claimed that TPE catheter placement is technically challenging and leads to complications (e.g. infection, hypotension); Kim et al. 56 described TPE administration without significant complications.
The studies in patients with CIDP found that TPE and IVIg were both efficacious.55–57 IVIg may, however, be preferable for its availability and relatively fewer AEs. 55 The AE occurrence should be interpreted cautiously as centrally administered TPE is more frequently associated with complications, and only one 56 of the cited studies specified the route of TPE administration.
Owing to low-level evidence (grade of recommendation for equal efficacy of TPE and IVIg: C) and limited number of participants and studies, it is difficult to conclude and recommend whether TPE or IVIg was superior for the treatment of CIDP. Previously, several rare variants of CIDP associated with IgG4 subclass autoantibodies to peripheral nerve proteins at the node and paranode (e.g. neurofascin) were found to respond poorly to IVIg. 58 There are no published data comparing IVIg with TPE in this group of CIDP patients.
Encephalitis
Several encephalitis types have been described so far. Autoimmune encephalitis (AIE) is a noninfectious, immune-mediated encephalitis, often characterised by antibodies against neuronal antigens and accompanied by various symptoms (behavioural and psychiatric changes, movement and autonomic disorders and seizures). 59 To date, at least 10 autoantibodies have been identified in different AIE subclasses, but a significant proportion of cases may have an immune origin even when no antibodies can be detected. 59
The AIE Alliance Clinicians Network has recently proposed AIE classification into four subclasses based on serological parameters: antibodies to intracellular antigens, antibodies for clinically relevant surface and other antigens, antibodies with surface antigens of low clinical relevance and seronegative AIE. 59 The 68 experts from 17 countries included in this multinational survey identified IVIg and TPE as possible immunotherapeutic options for both acute AIE and long-term management of AIE-associated symptoms.59,60 Of note, certain practicality issues may arise with the acute treatment of AIE in noncooperative (e.g. agitated) patients. 59 These patients will rarely tolerate a TPE procedure, making central venous access difficult unless intubated and sedated. They, however, may tolerate a peripheral venous line suitable for IVIg, as observed in clinical practice.
This review included one study on unspecified encephalitis, 61 which reported on longer hospitalisation times and lower discharge possibility after treatment with TPE (Table 4). 61 The mean age of patients treated with TPE was higher than of those treated with IVIg, however.
TPE and IVIg in treatment of encephalitis.
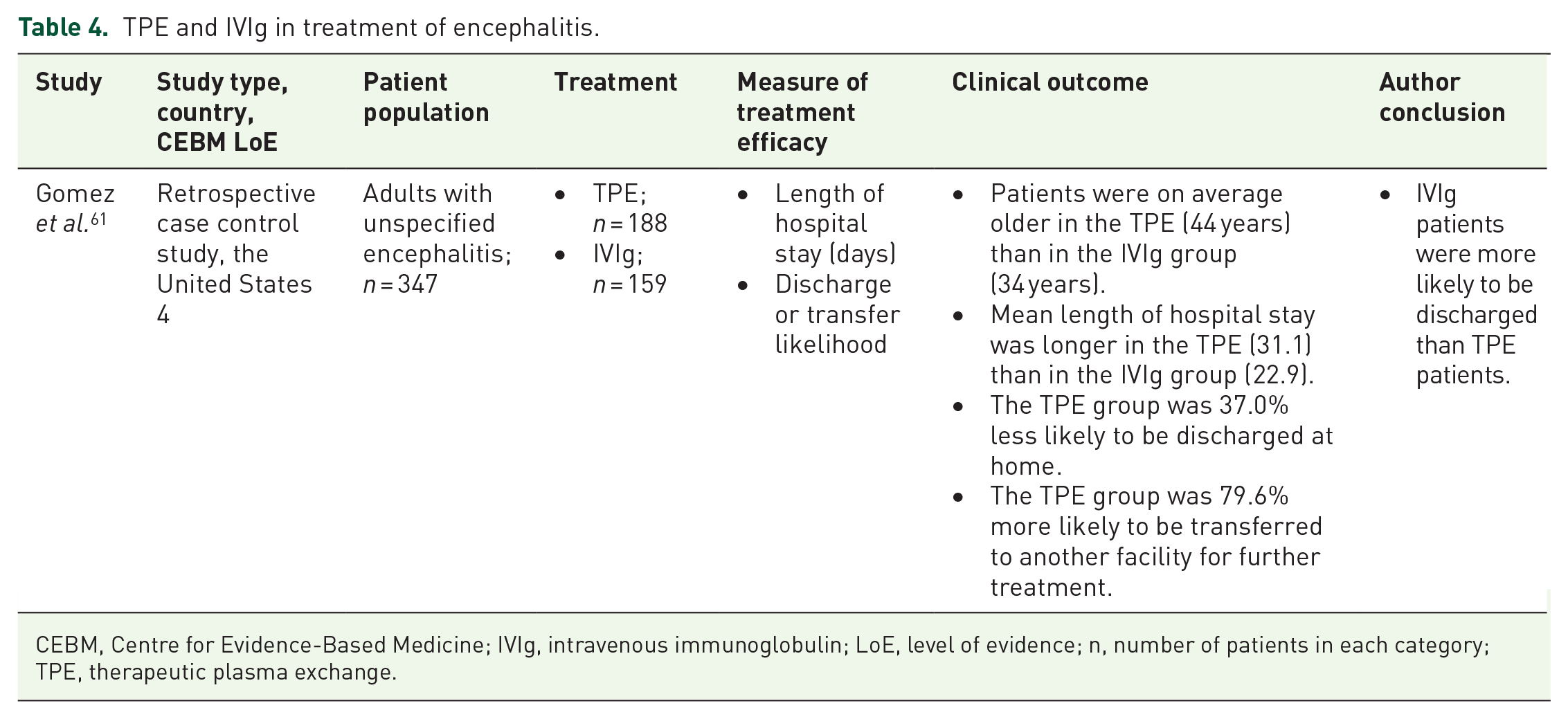
CEBM, Centre for Evidence-Based Medicine; IVIg, intravenous immunoglobulin; LoE, level of evidence; n, number of patients in each category; TPE, therapeutic plasma exchange.
NMOSDs
NMOSD is an autoimmune disease of the central nervous system, primarily affecting the spinal cord and optic nerve, characterised by the presence of antibodies against aquaporin 4 (AQP4). The clinical criteria for diagnosing NMOSD and distinguishing it from multiple sclerosis (MS) have been updated over the years.62,63 Aside from AQP4-specific IgG antibodies, most patients with NMOSD are usually characterised by longitudinally extensive transverse myelitis, severe optic neuritis and area postrema syndrome, and often have recurrent disease. 62 Although rare, NMOSD has been described across different ethnicities worldwide. 62
The present systematic search did not identify systematic studies that directly compare TPE and IVIg in NMOSD treatment (Table 5). Nevertheless, an exception was made for this group of disorders due to their relevance to the field and emerging clinical evidence. The selection of included publications in Table 5 was based on expert opinion.
TPE and IVIg in treatment of neuromyelitis optica spectrum disorders.
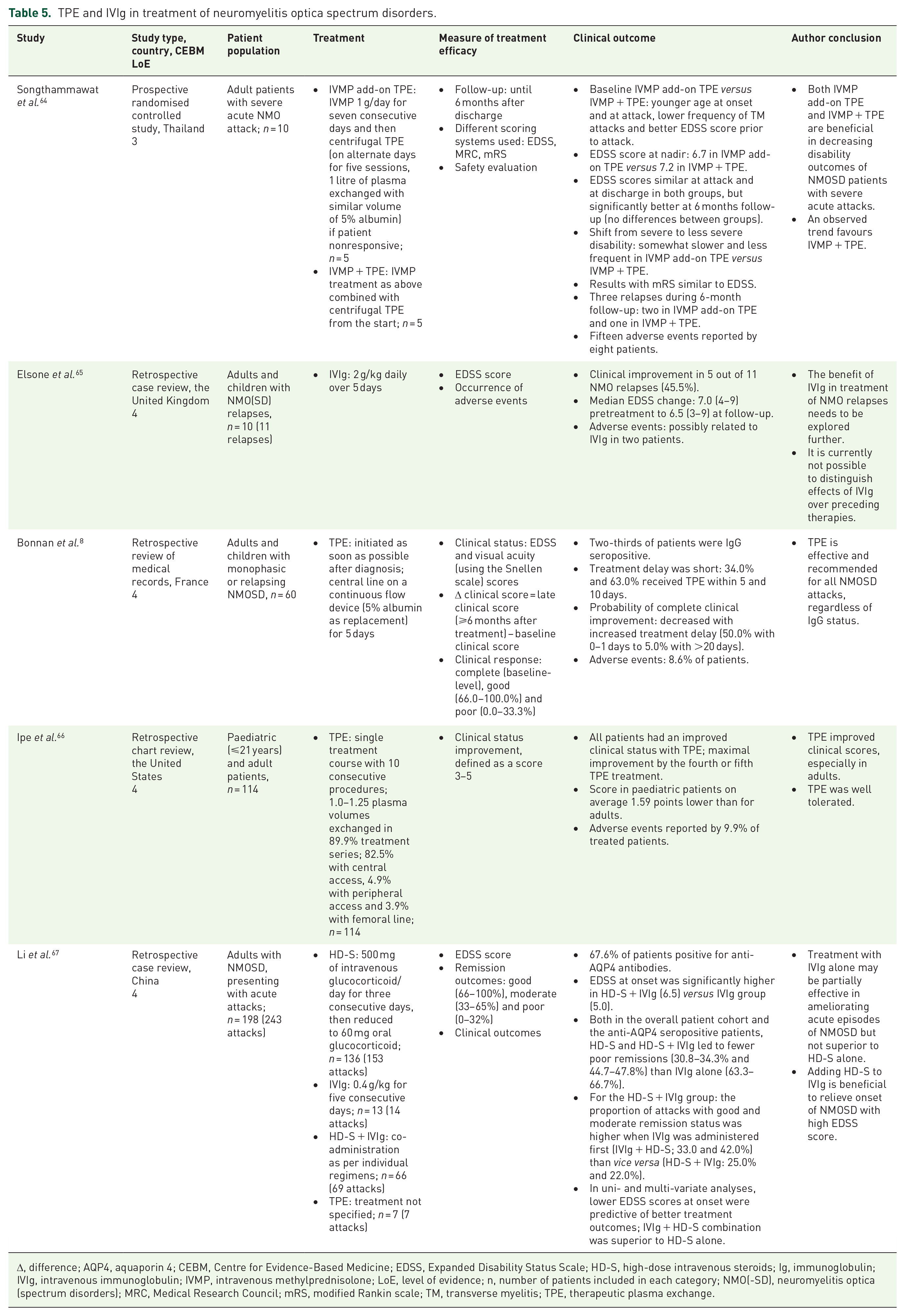
Δ, difference; AQP4, aquaporin 4; CEBM, Centre for Evidence-Based Medicine; EDSS, Expanded Disability Status Scale; HD-S, high-dose intravenous steroids; Ig, immunoglobulin; IVIg, intravenous immunoglobulin; IVMP, intravenous methylprednisolone; LoE, level of evidence; n, number of patients included in each category; NMO(-SD), neuromyelitis optica (spectrum disorders); MRC, Medical Research Council; mRS, modified Rankin scale; TM, transverse myelitis; TPE, therapeutic plasma exchange.
It is widely accepted that TPE is a mainstay treatment for NMO relapses, 68 as corroborated by three studies included in this SLR.8,64,66 The only included prospective controlled study, Songthammawat et al. 64 (LoE: 3), concluded that TPE was more efficacious when administered concurrently with intravenous steroids than as an add-on therapy in patients with NMOSD attacks. Bonnan et al. 8 found that TPE was efficacious for treating NMOSD attacks. The authors highlighted the importance of initiating TPE early in the attack. Longer TDs were associated with worse outcomes; administering TPE on day 0 translated into 50% probability to regain complete improvement, which was significantly higher compared with TPE administered after day 20 (1–5% probability for complete improvement).
In contrast, only a few case series have reported the possible benefit of IVIg in NMOSD. For the sake of balance and discussion, we have included selected studies on IVIg (Table 5).
Elsone et al. 65 observed a modest clinical benefit of IVIg in adult and paediatric patients with NMO relapses. Approximately half (5/11; 45.5%) of events improved with IVIg, with improvement of the Expanded Disability Status Scale score nonsignificantly. 65 Another study found that IVIg therapy conveyed moderate benefit to patients with acute NMOSD attacks and that combining IVIg with high-dose intravenous steroids helped to relieve onset of severe NMOSD. 67
Other conditions
This group includes several conditions for which only one study comparing TPE and IVIg treatment efficacies (per condition) was identified in the literature search (Supplementary Table 2). The conditions included MS, 69 acute immune-mediated polyradiculoneuropathy (AIMP; general condition that also includes GBS), 70 postinfectious polyradiculoneuropathy 71 and Sydenham’s chorea 72 (Supplementary Table 2). IVIg and TPE showed comparable efficacies for all these conditions.
Discussion
This SLR aimed to analyse available evidence from 1990 to 2021 on TPE and IVIg therapeutic efficacy for both peripheral and central nervous system autoimmune neurological disorders. GBS was the best represented (20 studies), followed by MG (11), NMOSD (5), CIDP (3) and encephalitis (1). Most studies (19) were retrospective, while 13 were clinical trials. After screening 44 comparative studies, we conclude that TPE and IVIg are efficacious in treating neurological conditions of largely autoimmune origin; no clear superiority of one treatment over the other has been clearly demonstrated thus far.
Until 2001, most evidence on therapeutic efficacy of TPE and IVIg was derived from studies on GBS, CIDP and MG in which both treatments have been established as mainstay, first-line therapeutic modalities.3,11,73 With some exceptions,17,19 however, these studies were limited by their retrospective approach and designed to test noninferiority of IVIg (the newer treatment) versus TPE. The noninferiority design warrants caution as it is only moderately accurate, subject to bias and different from superiority designs that require greater statistical power and typically greater participant numbers. 74 Other limitations of the mentioned studies were insufficiently defined and variable study designs,20,32,47 variable treatment outcome grading and small sample sizes.12,22,31,32,42,55
More recent studies offered further evidence on rare autoimmune neurological disorders, including more recently recognised conditions, such as AIE, NMOSD and MuSK MG. Although no comparative studies were identified in this review, both TPE and IVIg have led to clinical improvement in patients with AIE59,60 and, with a modest benefit of IVIg, NMOSD.8,64–67 Of note, in absence of larger comparator studies, IVIg benefit in NMOSD may have been underestimated by the expert choice of the included studies. Some other relevant diseases of autoimmune origin, such as the stiff-person syndrome, 75 were not included in the present review due to the lack of eligible studies.
Identification of immune mediators involved in disease pathogenesis (e.g. AQP4 in NMOSD,
62
N-methyl-
Despite limitations, the studies included in this SLR discuss clinical efficacy of IVIg and TPE in light of the trade-offs between clinical benefit and complications. Many studies reported higher complication rates and longer hospital stays with TPE versus IVIg treatment.17,25,31,34,44,47,48,61 Comparing complications between TPE and IVIg is fraught with difficulties because of the different techniques of TPE administration (e.g. central versus peripheral venous access) which are often not specified in the studies. TPE procedure has advanced, and the use of peripheral line TPE has reduced the number of both catheter- (e.g. deep vein thrombosis) and exchange-related (e.g. acute renal failure) AEs. 77 Newer TPE devices can accurately track fluid exchange, thereby minimising the risk of AEs. 77 Unlike membrane-mediated TPE with a central line access associated with many AEs, centrifugal TPE with a peripheral line access was shown to be acceptably safe and well tolerated. 77 TPE efficacy may have also been lowered by the suboptimal choice of replacement fluid. For instance, Kuwabara et al. 20 admitted that using double-filtered plasma may have negatively affected their treated patients with GBS. Central line placement may also delay TPE, significantly reducing its therapeutic efficacy, 8 so peripheral line TPE is advantageous as quicker to administer.
The main argument in favour of IVIg was its simplicity of use compared with the TPE protocols. Administering IVIg does not necessitate trained personnel and is preferred for patients in whom TPE is difficult to administer, such as psychotic or combative patients. 59 IVIg, however, does not directly remove immune mediators, although it may accelerate catabolism of autoantibodies or modulate downstream effects on complement and cellular antibody-dependent mechanisms. In IgG4-autoantibody-mediated diseases, such as MuSK MG7,50,51 or neurofascin-antibody CIDP, 58 that may be refractory or poorly responsive to IVIg, TPE remains the therapy of choice. Despite lack of comparative studies, the benefit of TPE is most prominent in NMOSD, in which it continues to be a mainstay therapeutic approach. 8 Also, some patients (e.g. patients with GBS seropositive for antiganglioside antibodies) 33 may be unresponsive or overly sensitive to IVIg or may have contraindications (e.g. renal failure). 9 Finally, the rapid clinical improvement observed after TPE favoured this treatment for the management of myasthenia crisis. 42
The main strengths of this review are its broad literature scope spanning 30 years and the variety of analysed neurological disorders. One significant limitation is the low quality of available evidence in many conditions, due to low numbers of participants in studies of autoimmune neurological disorders, patient factors influencing the choice of therapeutic modality and economic considerations. In addition, not all studies directly compared IVIg and TPE, and the benefit of other treatments (e.g. corticosteroids) was not considered in this analysis. Many included publications called for further and larger studies to validate the clinical benefit of TPE and IVIg treatment for these diseases, in agreement with opinions of expert authors of this review. Other limitations include changes in disease classification (e.g. separation of NMOSD from MS) 62 and strict exclusion criteria at title/abstract screening, possibly biasing the final selection of studies. Together with low LoE of the included studies, it was not possible to more thoroughly analyse the results discussed here.
The present review offers comprehensive and contemporary information on optimal TPE versus IVIg treatment for autoimmune neurological conditions. While the LoE may not be suitable for regulatory decision-making, 14 it may be satisfactory to guide physicians who need to treat patients with these disorders in real-world practice.
Conclusion
TPE and IVIg are both viable treatment options for several autoimmune neurological disorders: GBS, MG (including myasthenic crisis), CIDP and encephalitis. For NMOSD, the evidence from the targeted literature review favoured TPE over IVIg, although some studies point to a potential benefit of IVIg. The choice of the best therapeutic modality should be based on the available clinical resources and tailored according to individual patient conditions and needs. Future studies on autoimmune neurological disorders should ideally be prospective, with well-defined and standardised protocols and consensus evaluation criteria. We further propose the inclusion of quality-of-life assessments as a more meaningful way to evaluate the long-term benefit of different treatments to the patients.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231154306 – Supplemental material for Comparison of IVIg and TPE efficacy in the treatment of neurological disorders: a systematic literature review
Supplemental material, sj-docx-1-tan-10.1177_17562864231154306 for Comparison of IVIg and TPE efficacy in the treatment of neurological disorders: a systematic literature review by Ashwin A. Pinto, Jerome De Seze, Anu Jacob, Stephen Reddel, Anna Yudina and Kevin Tan in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors acknowledge Irena Zurnic Bönisch (Akkodis Belgium c/o Terumo Blood and Cell Technologies) for writing support and Sophie Timmery (Akkodis Belgium c/o Terumo Blood and Cell Technologies) for editing and coordination support. The authors also acknowledge the contribution of Rania Hassanein and Aicha Bah to the literature search and paper review. The authors would also like to acknowledge their colleagues in infusion and TPE teams and their commitment to clinical care. The listed authors have authorised the submission of their manuscript via a third party and approved any statements or declarations included in the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
