Abstract
Background:
Adeno-associated virus (AAV) vectors are a promising platform for
Objective:
To evaluate prevalence of total binding antibodies (neutralizing and non-neutralizing) against rAAVrh74 in patients with DMD.
Methods:
Eligible individuals (
Results:
A large preponderance (86.1%) of patients with DMD in this data set was seronegative for anti-rAAVrh74 total binding antibodies. These patients would potentially meet the antibody status eligibility criterion for entry into rAAVrh74-based gene therapy clinical trials.
Conclusion:
Measuring total binding antibodies is a more comprehensive approach to assess pre-existing immune response
Introduction
Adeno-associated virus (AAV) is a leading gene delivery platform for the treatment of monogenic diseases, with > 200 ongoing or completed clinical trials.1,2 Attributes of AAV include replication deficiency, no known human pathogenicity, and generally weak immunogenicity.3–5 Recombinant AAV vectors used for gene therapy are classified by serotypes that may vary significantly in relative transduction efficiency, tissue tropism, and safety profiles.6–8 Pre-existing humoral immunity to the AAV capsid has the potential to limit therapeutic efficacy and poses a potential safety risk for treatment with AAV-based gene transfer therapies. Specifically, pre-existing AAV antibodies can inhibit transduction
9
and promote inflammatory responses
There is currently no standardized assay for measuring pre-existing immunity against AAV vectors. Rather, entry into a gene transfer therapy program requires a specific antibody test, endorsed by the gene therapy sponsor.2,17 Importantly, program-specific assays can differ in the methodologies used for evaluating pre-existing immunity. Specifically, some sponsors will measure only antibodies that prevent AAV-mediated transduction of target host cells, known as neutralizing antibodies (NAbs), whereas other sponsors will choose a more comprehensive approach that measures total binding antibodies (TAbs), which includes non-neutralizing antibodies (non- NAbs) that can still bind to the vector and promote pro-inflammatory responses in addition to NAbs. Of additional importance in terms of the varied approaches that sponsors take in assessing pre-existing immunity is the cut-off threshold for what is considered to be an ‘elevated’ level of antibodies, or seropositivity, which would preclude participation in a gene therapy program. These thresholds must be experimentally determined through both preclinical and clinical studies and will be unique for each gene therapy program.
As previously mentioned, NAbs are a subset of TAbs capable of inhibiting transduction and thus reducing the efficacy of gene transfer therapy.10,18 NAbs are typically measured using an
Prevalence of pre-existing antibodies against AAV is dependent on multiple factors, including the serotype, type of antibodies assessed, method of assessment, and geographic location and age range of the population evaluated.17,22 rAAVrh74, which was first identified in rhesus macaques, 23 has demonstrated widespread transgene delivery to muscle – including skeletal, diaphragm, and cardiac – in animal models following intravenous administration.5,24,25 Moreover, because rAAVrh74 was isolated from monkeys, it may be associated with less pre-existing immunity compared with AAV serotypes isolated from humans.23,26 These features of rAAVrh74 have made it an attractive platform for development of gene transfer therapies targeting neuromuscular diseases. Indeed, one study that examined seroprevalence of AAV antibodies in various populations, including patients with DMD, found rAAVrh74 to be among the serotypes with the lowest rates of pre-existing immunity.5,23 Several rAAVrh74-based gene transfer therapies are under investigation, including delandistrogene moxeparvovec (SRP-9001) for gene transfer in patients with DMD [phase I (NCT04626674), phase I/II (NCT03375164), phase II (NCT03769116), and phase III (NCT05096221)] and SRP-9003 for the gene transfer of β-sarcoglycan into patients with limb-girdle muscular dystrophy type 2E/R4 [phase I/II (NCT03652259)].
Here, we report the total anti-rAAVrh74 antibody seroprevalence among 101 individuals with DMD. A total antibody level of < 1:400 was defined as seronegative (not elevated) and ⩾ 1:400 was defined as seropositive (elevated). This < 1:400 total antibody threshold was determined based on preclinical nonhuman primate studies 20 that found no inhibition of transduction nor safety events caused by antibodies at or below that level; these findings were subsequently clinically validated in human trials. 26
Methods
Study design
This study was conducted in the United States through a centralized virtual research coordination center and Principal Investigator, in accordance with the International Conference on Harmonisation Guidelines for Good Clinical Practice and the Declaration of Helsinki. The protocol was approved by a central institutional review board. Eligible patients were males, aged ⩾ 4 to < 18 years at the time of screening, with a definitive diagnosis of DMD based on clinical findings and confirmatory genetic testing. Key exclusion criteria were prior treatment with gene transfer therapy or living with a person who had previous known exposure to rAAVrh74 or other gene transfer therapies. Electronic consent and medical release from the eligible patients’ treating physicians were signed by the patients’ parents/legal guardians. Patients were provided with the choice of receiving their one-time blood draws either at a phlebotomy center or at home,
Statistical analysis
The sample size was calculated to obtain 8% precision around the overall seroprevalence estimate. With an estimated sample size of 100 patients, the margin of error [half width of the 95% confidence interval (CI)] would be < 8% for a rate of seroprevalence < 20. Demographic characteristics, including age (years), race, and ethnicity, were summarized for the full analysis set, defined as all patients who were enrolled in the study and had at least one sample evaluated with a valid result for immunogenicity. Analysis of seropositivity was performed on the full analysis set. Clopper–Pearson (exact) method was used to compute the CIs. 27
Results
In total, 107 patients with a definitive diagnosis of DMD were enrolled, of whom 101 completed the study (full analysis set) (Figure S1 in the Supplemental material). A summary of patient demographics and baseline characteristics is presented in Table 1. The mean (standard deviation) age of the evaluated patients was 9.1 (3.5) years. Most patients were ⩾ 8 to < 18 years old (65.3%) and White (78.2%). The study was not powered for ethnicity-related analysis. Eighty-one percent of patients selected the home phlebotomy option.
Demographic and baseline characteristics (full analysis set).
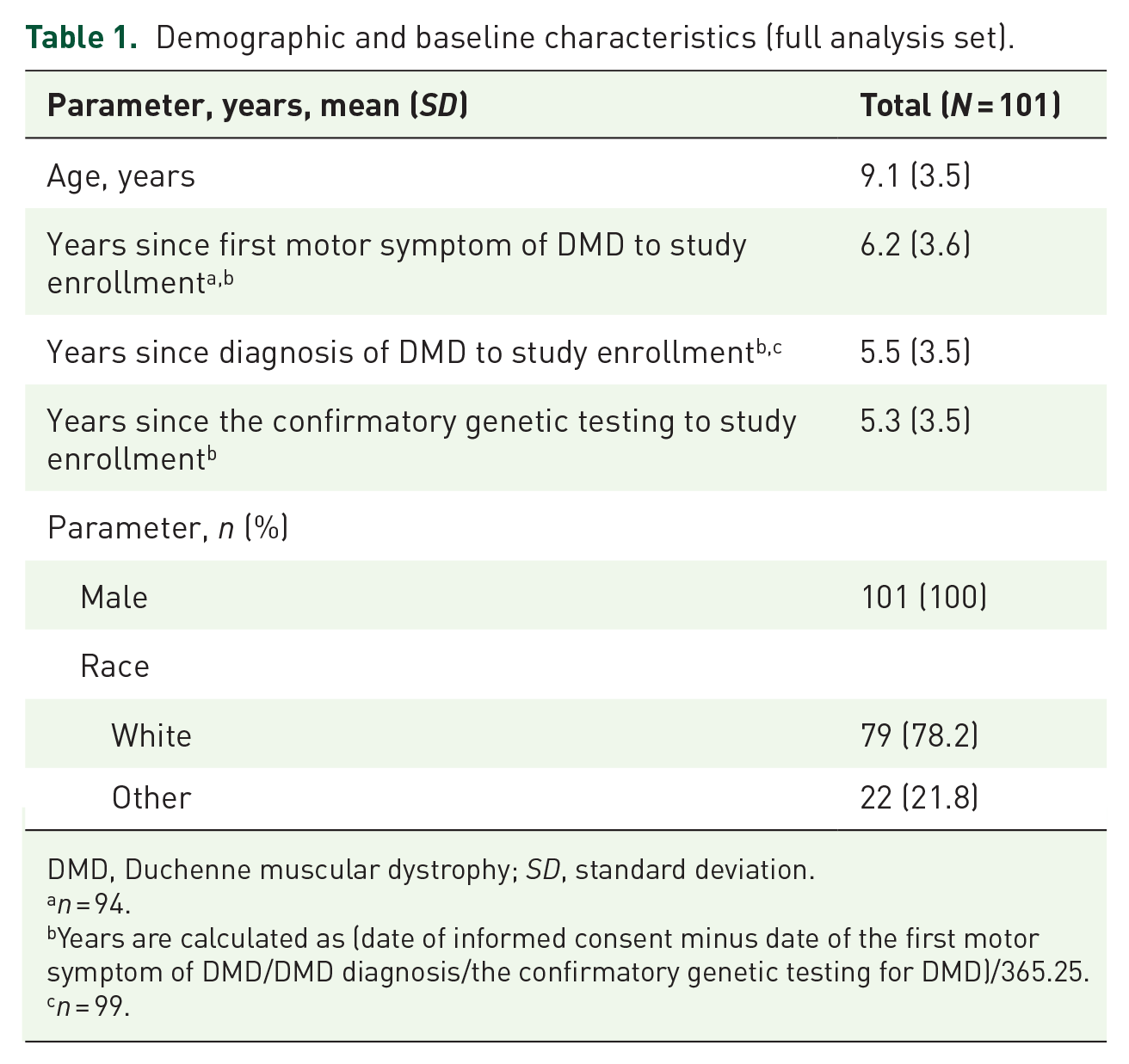
DMD, Duchenne muscular dystrophy;
Years are calculated as (date of informed consent minus date of the first motor symptom of DMD/DMD diagnosis/the confirmatory genetic testing for DMD)/365.25.
Results from the ELISA showed that a large preponderance of patients [87/101 (86.1%); 95% CI, 77.8–92.2] did not have elevated anti-rAAVrh74 antibody titers (⩾ 1:400) (Figure 1). In the 14 patients with elevated anti-rAAVrh74 antibodies, titers ranged from 1:400 to 1:3200 [1:400: 1.0% (1/101); 1:800: 4.0% (4/101); 1:1600: 5.9% (6/101); and 1:3200: 3.0% (3/101)].

Prevalence of anti-rAAVrh74 antibodies in 101 patients with Duchenne muscular dystrophy. Serum samples were assayed by binding ELISA for total immunoglobulin G against rAAVrh74. ELISA titer < 1:400 was considered as seropositive.
Discussion
Gene therapy has revolutionized the treatment of previously untreatable diseases. There is pressing need for the treatment of devastating, rare, progressive diseases, such as DMD, for which AAV-based gene transfer therapies could significantly impact large segments of the patient population. Currently, some patients are ineligible for AAV-based gene transfer therapies due to pre-existing antibodies to the AAV vector that may affect the safety and efficacy of treatment. In this study, patient samples from a cohort of 101 patients with DMD from across the United States were sent to a single-center laboratory and assessed for seroprevalence of total anti-rAAVrh74 antibodies, where seropositivity, or elevated antibody status, was defined as a titer ⩾ 1:400. This definition for seropositivity was based on preclinical nonhuman primate studies that established a threshold for impact on transduction efficiency. An overwhelming majority of patients were seronegative, suggesting the potential broad applicability of rAAVrh74-based precision genetic therapies for this population.
The innovative virtual design of this study capitalized upon a centralized virtual platform to identify, screen, and receive consent from patients. Moreover, patients were allowed to choose a preferred setting for blood collection (i.e. home
This seroprevalence study focused on patients with DMD because the rAAVrh74 vector is currently being evaluated in clinical trials of delandistrogene moxeparvovec (SRP-9001), an rAAVrh74-based investigational gene therapy for DMD. Seropositivity for rAAVrh74 is an excluding criterion for clinical trials of rAAVrh74-based therapies. To date, no observed serious adverse events in clinical trials of rAAVrh74-based gene therapies have been attributed to antibody-mediated inflammation or complement activation.26,28 Furthermore, no antibody-mediated inhibition of transduction has been observed in these studies to date. The clinical trials (NCT04626674, NCT03375164, NCT03769116, and NCT03652259) utilize an anti-rAAVrh74 antibody screening test and cut-off definition for elevated TAbs comparable with the test for seropositivity described in this report. The absence of adverse events attributed to anti-rAAVrh74 antibodies in these trials suggests that comprehensive antibody screening is appropriate for determining one aspect of patient eligibility for rAAVrh74 gene therapy.
Ongoing DMD gene therapy clinical development programs in the United States use different mini-/micro-dystrophin transgene constructs, muscle-specific promoters, and AAV serotypes, including rAAVrh74 and AAV9. 29 Each gene therapy program requires a unique and specific assay, designed to measure pre-existing antibodies to a particular AAV vector, with threshold values that have ideally been empirically determined and validated through preclinical and clinical studies specific to the vector platform used.
This study based seropositivity on a titer of total binding anti-rAAVrh74 antibodies, or TAbs, which includes NAbs and non-NAbs. Measurement of TAbs, as opposed to simply measuring NAbs, is a more comprehensive approach designed to reduce the risk of adverse events from pre-existing immunity.30,31 Other benefits to TAb assays include their ease of use, their fast turnaround time, and their simplicity to establish and validate compared with complex cell-based NAb assays. 30
Limitations of this study include that only United States patients were represented and the use of steroids, which was not assessed. While corticosteroid use has not been shown to impact seroprevalence in other studies of patients with DMD, 32 their effect on anti-rAAVrh74 antibody titer was not specifically evaluated in this study; thus, their impact on seroprevalence cannot be ascertained.
In conclusion, results from this study suggest that most patients with DMD in the United States are seronegative (86.1%) for pre-existing antibodies to rAAVrh74. This supports the broad applicability of rAAVrh74-based gene therapy, and those seronegative patients meeting other trial eligibility criteria would be eligible for entry into rAAVrh74-based clinical trials. Finally, because TAb assays offer a more comprehensive approach, this method of patient pre-screening may improve the safety and efficacy of AAV-based gene therapies.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221149781 – Supplemental material for Evaluation of rAAVrh74 gene therapy vector seroprevalence by measurement of total binding antibodies in patients with Duchenne muscular dystrophy
Supplemental material, sj-docx-1-tan-10.1177_17562864221149781 for Evaluation of rAAVrh74 gene therapy vector seroprevalence by measurement of total binding antibodies in patients with Duchenne muscular dystrophy by Natalie L. Goedeker, Sachi D. Dharia, Danielle A. Griffin, Jesantha Coy, Todd Truesdale, Rajan Parikh, Kasen Whitehouse, Sourav Santra, Damon R. Asher and Craig M. Zaidman in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
PPMD provided support in helping to enroll patients and offered optional counseling to patients and caregivers. Audrey Vandervelde of Sarepta Therapeutics, Inc., reviewed the manuscript for scientific accuracy. Medical writing support for this manuscript was provided by Paraskevi Briassouli, PhD, of Eloquent Scientific Solutions, and was funded by Sarepta Therapeutics, Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
