Abstract
Background:
Vigabatrin (VGB) is currently the most widely prescribed first-line medication for individuals with infantile spasms (IS) and especially for those with tuberous sclerosis complex (TSC), with demonstrated efficacy. Meanwhile, its adverse events, such as vigabatrin-associated brain abnormalities on magnetic resonance imaging (MRI; VABAM), have also been widely reported.
Objectives:
The objectives of this study were to observe the occurrences of VABAM in patients with IS caused by TSC (IST) and further explore the associated risk factors.
Methods:
Children with IS receiving VGB were recruited from our institution; clinical, imaging, and medication data were collected. Cerebral MRI was reviewed to determine the occurrence of VABAM. Group comparisons (IS caused by TSC and other etiologies) were performed; subgroup analyses on IST were also performed. Next, a retrospective cohort study of children taking VGB was conducted to explore risk/protective factors associated with VABAM.
Results:
The study enrolled 172 children with IS who received VGB. VABAM was observed in 38 patients (22.1%) with a peak dosage of 103.5 ± 26.7 mg/kg/day. Subsequent analysis found the incidence of VABAM was significantly lower in the 80 patients with IST than in the 92 patients with IS caused by other etiologies (10% versus 32.6%, p-value < 0.001). In subgroup analyses within the IST cohort, VABAM was significantly lower in children who received concomitant rapamycin therapy. Univariate and multivariate logistic regression analysis of the 172 IS children showed that treatment with rapamycin was the independent factor associated with a lower risk of VABAM; similar results were observed in the survival analysis.
Conclusion:
The incidence of VABAM was significantly lower in IST patients. Further research is needed to examine the mechanisms that underlie this phenomenon and to determine if treatment with rapamycin may reduce the risk of VABAM.
Highlights
Rapamycin is potentially a protective factor associated with VABAM.
The incidence of VABAM in patients with infantile spasms caused by tuberous sclerosis complex was lower than in other etiologies.
The risk factors associated with VABAM were unclear, but VABAM may occur even at the conventional dosage of VGB (i.e. ≤150/kg/day).
Introduction
Infantile spasms (IS), also identified as Infantile epileptic spasms syndrome by the International League Against Epilepsy, is characterized by the onset of epileptic spasms between 1 and 24 (peak 3 and 12) months of age and caused by more than 200 etiologies, and can be divided into two major categories based on etiology: known (symptomatic) and unknown (cryptogenic).1–4 Among the known etiologies, tuberous sclerosis complex (TSC) is among the leading causes of IS.2,4 Standard first-line systemic therapy for IS includes hormonal therapy (e.g. adrenocorticotropic hormone and prednisolone) and vigabatrin (VGB).4,5 VGB has shown clinical effectiveness in children with IS, especially when caused by TSC.6–8
With general use, however, adverse events associated with VGB were reported, the most common being sedation, somnolence, irritability, insomnia, sleep disorder, constipation, lethargy, decreased appetite, and hypotonia. 9 VGB-specific side effects also include peripheral visual field defects and recently reported vigabatrin-associated brain abnormalities in magnetic resonance imaging (MRI; VABAM). 10 Typical VABAM manifest as reversible high T2-weighted imaging (T2WI) signal and diffusion restriction of the thalamus, basal ganglia, brainstem, and cerebellar dentate nucleus.11–14 Although some studies have shown that VABAM is not accompanied by clinical symptoms, other studies on VABAM cases provide evidence that VABAM is associated with movement disorders, encephalopathy, dysautonomia, or death.13,15 Currently, there are no effective therapeutic drugs available to prevent or treat VABAM, and clinicians wait for the spontaneous regression of VABAM by discontinuation of VGB. 13
The incidence of VABAM in infants can be as high as 21–32.5%.13,16–19 Given the clinical efficacy of VGB in seizure control of IS, especially in IS caused by TSC (IST),4,6,7,20 discontinuation caused by VABAM may result in the exacerbation or relapse of seizures. Therefore, it is essential to further explore VABAM in IST. Regrettably, only scattered cases are reported11,12,14,21,22 and no study has focused on this aspect. In our previous study, we discovered that the peak dosage of VGB is the risk factor for VABAM, and the incidence was 32.5%. 19 However, patients with IST were not incorporated into the study. When VABAM was observed in a larger patient cohort in our medical center, we found that the incidence in IST was much lower than in patients with IS caused by the other etiologies (ISO). Our medical center registered and carried out the first clinical study using rapamycin (Reference No. ChiCTR-IPR-15007241), an mTOR inhibitor, to treat patients with TSC in China. As a result, most of the IST patients above had received the combination treatment with rapamycin.
To better understand VABAM, we conducted this study in a larger cohort by incorporating IST patients into the previous study cohort. A retrospective cohort and case–control study were designed and conducted to identify risk factors for the occurrence of VABAM.
Methods
Study subjects
All children with IS who visited our hospital (including patients who received an initial dose of VGB and those with a VGB exposure history referred by the other institution) were included in this study. Criteria for inclusion or exclusion of subjects were synthesized based on our previous study. 19 Inclusion criteria were: (1) clear diagnosis of IS based on the clinical symptoms and electroencephalogram results (as Infantile epileptic spasms syndrome defined by International League Against Epilepsy in 2022), 3 (2) exposure more than 30 days and still taking VGB at the time of this study, and (3) at least one cerebral MRI examination completed before or during VGB exposure. The exclusion criteria were: (1) time of first cerebral MRI review after VGB exposure is more than 1 year, (2) time of first cerebral MRI review after VGB exposure has not reached 30 days, (3) children with IS caused by congenital metabolic diseases, and (4) children with VGB exposure history referred by another institution that did not perform cerebral MRI before an initial dose of rapamycin therapy in our hospital.
Observation indicators, diagnosis of TSC, and identification of VABAM
Clinical data were recorded, including IS etiology; age at first VGB exposure; duration, cumulative, and peak dosage of VGB exposure; other anti-seizure medications (ASMs); hormonal therapy when taking VGB; and concomitant rapamycin therapy of all children.
The diagnosis of TSC was established in a proband with one of the following diagnostic criteria: (1) presence of any two of the major clinical features listed by the International Tuberous Sclerosis Complex Consensus Group, 23 (2) one major clinical feature and two or more minor clinical features, 23 or (3) identification of a heterozygous pathogenic variant in TSC1 or TSC2 by molecular genetic testing. 23
VABAM was identified when a comparison of cerebral MRI before and after VGB exposure showed new onset bilateral symmetrical thalamus, basal ganglia, brainstem, or cerebellar dentate nucleus T2WI, or diffusion-weighted imaging (DWI) high signal in these areas. 24 The imaging data of subjects were reviewed independently by two highly experienced senior pediatric neurologists (Drs. Xiu-Yu Shi and Guang Yang, with more than 20 years of clinical experience). When results differed between the two raters, the final identification of VABAM was confirmed by the chief pediatric neurologist (Dr. Li-Ping Zou, with more than 40 years of clinical experience). In addition to knowing that all patients had VGB exposure, all the other treatment protocols were blinded to the above three assessors.
When VABAM was identified on review of MRI, progress notes in the medical record were serially reviewed in the search for new-onset symptoms consistent with VABAM (i.e. movement disorders, lethargic, unresponsive, and respiratory distress or arrest). If the patient had the above symptoms, he (or she) would be considered symptomatic VABAM, otherwise would be considered asymptomatic VABAM.
Statistical analysis
SPSS 26.0 statistical software and the R programming language (version 4.1.2) were used for analysis, and the data description was expressed in the form of means (standard deviations) for normally distributed variables or medians (interquartile ranges) for non-normally distributed variables. The independent two-sample t-test, chi-square test, or Wilcoxon rank-sum test was conducted to analyze the above observation indicators. Univariate logistic regression analysis was conducted to assess for collinearity and filter variants; multivariate logistic regression analysis was performed to clarify the risk factors of VABAM. Variable selection was conducted by least absolute selection and shrinkage operator (LASSO) in multivariate logistic regression. Kaplan–Meier (KM) curves and log-rank tests were used for the survival analysis of VABAM. A p-value of less than 0.05 indicated statistical significance.
Results
A total of 533 children with IS who had VGB exposure were screened. Of these, 390 underwent cerebral MRI before VGB exposure and during follow-up. Among these 390 children, 18 with VGB exposure for less than 30 days, 193 with VGB exposure for more than 1 year during cerebral MRI review, and 7 without MRI review before the initiation of rapamycin were excluded. All patients who needed to withdraw VGB for suspected symptomatic VABAM had performed MRI reexamined and were enrolled in the study. Ultimately, 172 children were included in this study (Figure 1), and 38 (22.1%) developed VABAM (The two senior investigators agreed on the evaluation results of VABAM in 170 children, the other 2 children were identified as non-VABAM by the chief investigators). VGB peak dosage was 103.5 (mean) ± 26.7 (standard deviation) mg/kg/day (37 with the peak dosage ⩽ 150 mg/kg/day), and the duration of VGB exposure was 170 ± 12 days. Of the 172 children, IS in 80 was caused by TSC (IST), and IS in 92 was caused by other etiologies (ISO). In agreement with our previously reported data, treatment of IS commonly exhibits a particular preference for concomitant ASM therapies; Valproate (VPA, n = 88, 51.2%) and topiramate (TPM, n = 103, 59.9%) were used more frequently than other ASMs (less than 20%).

Flow chart of screening of the patients with infantile spasms.
In the IST group (56 males and 24 females), 8 children (10%) developed VABAM. Age at first VGB exposure was a median of 10 months (interquartile range or IQR: 6–17.75 months); 51 cases (63.8%) started taking VGB for the first time in infancy (⩽ 12 months). VPA and TPM were used concomitantly with VGB in 32 (40%), and 30 (37.5%) patients, the dosage of VPA and TPM was 35.72 ± 3.65 and 6.26 ± 0.70 mg/kg/day, respectively. The peak dosage of VGB was 99.1 ± 30.1 mg/kg/day, the duration of exposure before MRI review was 261 ± 102 days, and the corresponding cumulative dosage was a median of 256 g (IQR: 130 g, 365 g). Thirteen cases (16.3%) received hormonal therapy when taking VGB. Concomitant rapamycin therapy was observed in 60 patients.
In the ISO group (53 males and 39 females), wherein 41 cases (44.6%) had known etiologies and 51 (55.4%) had unknown etiology, 30 cases (32.6%) developed VABAM. The age at first VGB exposure was 9 months (6.1, 14.875 months); 62 (67.4%) patients started taking VGB for the first time in infancy (⩽ 12 months). VPA and TPM were used when taking VGB in 56 (60.9%) and 73 (79.3%) patients, and the dosage of VPA and TPM was 35.55 ± 2.83 and 6.37 ± 0.32 mg/kg/day, respectively. The peak dosage of VGB was 96.7 ± 36.7 mg/kg/day, the duration of exposure before MRI review was 134 ± 92 days, and the corresponding cumulative dosage was 97.6 g (46.1 g, 200.5 g). A total of 37 cases (40.2%) received hormonal therapy when taking VGB. There were no children in the ISO group who received concomitant rapamycin therapy.
The incidence of VABAM was much lower in the IST group than in the ISO group (p < 0.001, Table 1). Significant differences emerged for the medication strategy between the IST and ISO groups; the use of VPA, TPM, and hormonal therapy were significantly less frequent in the IST group, but the IST group had more concomitant rapamycin therapy than the ISO group (p = 0.006, < 0.001, < 0.001, and < 0.001, Table 1). The dosage of VPA and TPM were not significantly different between IST and ISO groups (both p > 0.05, Table 1). The duration of VGB exposure and the cumulative dosage was significantly increased in the IST group (p < 0.001, both, Table 1), but the age at first exposure and peak dosage were not significantly different between IST and ISO groups (both p > 0.05, Table 1).
Comparison of clinical characteristics between the IST and ISO groups.
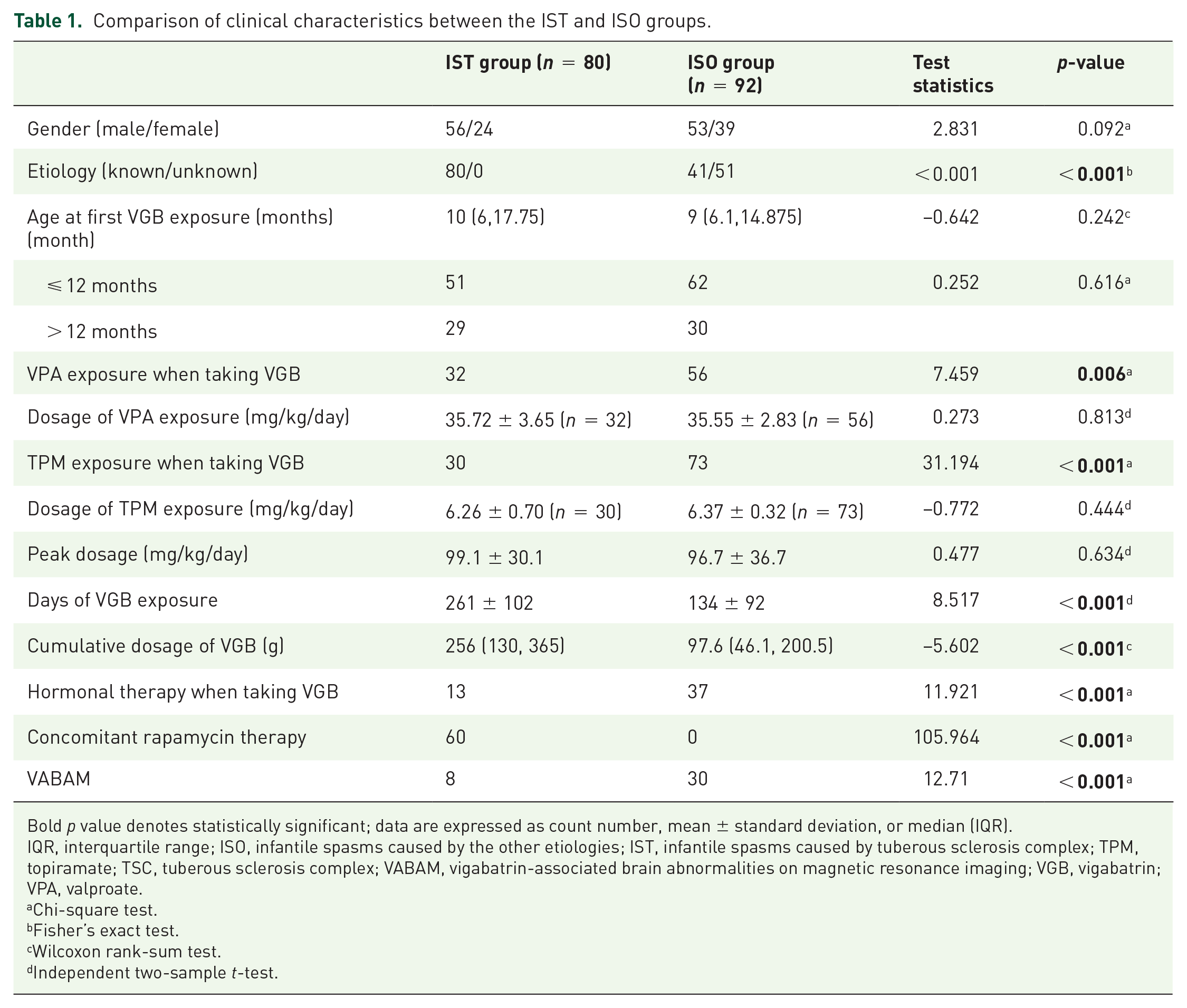
Bold p value denotes statistically significant; data are expressed as count number, mean ± standard deviation, or median (IQR).
IQR, interquartile range; ISO, infantile spasms caused by the other etiologies; IST, infantile spasms caused by tuberous sclerosis complex; TPM, topiramate; TSC, tuberous sclerosis complex; VABAM, vigabatrin-associated brain abnormalities on magnetic resonance imaging; VGB, vigabatrin; VPA, valproate.
Chi-square test.
Fisher’s exact test.
Wilcoxon rank-sum test.
Independent two-sample t-test.
Sub-group analysis of the IST group showed that 3 in 60 cases with concomitant rapamycin therapy (5%) developed VABAM, compared with 5 in 20 patients without rapamycin therapy (25%; p = 0.021, Table 2). Children who received concomitant rapamycin therapy appear to have the lowest incidence of VABAM compared with other groups (Figure 2). Aside from having a longer duration of VGB exposure in those children with concomitant rapamycin therapy (p = 0.004, Table 2), no other significant differences were found between the two groups (p > 0.05, Table 2).
Comparison of clinical characteristics between the patients with and without rapamycin therapy.

Bold p-value denotes statistically significant; data are expressed as count number, mean ± standard deviation, or median (IQR).
IQR, interquartile range; TPM, topiramate; TSC, tuberous sclerosis complex; VABAM, vigabatrin-associated brain abnormalities on magnetic resonance imaging; VGB, vigabatrin; VPA, valproate.
Chi-square test.
Fisher’s exact test.
Wilcoxon rank-sum test.
Independent two-sample t-test.

Incidence rates of VABAM in various groups.
Considering VABAM as an outcome event, the univariate logistic regression analysis showed that TPM or hormonal therapy when taking VGB were risk factors associated with VABAM (p = 0.003 and 0.002, respectively; Table 3); concomitant rapamycin therapy and IST case were protective factors (both p < 0.001, Table 3). After LASSO regression analysis, the indicators of IST, TPM exposure when taking VGB, hormonal therapy when taking VGB, age at first VGB exposure, VGB exposure at infancy, cumulative dosage of VGB, peak dosage of VGB, and concomitant rapamycin therapy were entered in the final multivariate logistic regression analysis. This analysis showed that concomitant rapamycin therapy was the independent protective factor associated with VABAM (p = 0.019, odds ratio = 0.152, 95% confidence interval: [0.028, 0.715], Table 3).
Logistic regression analysis of VABAM risk factors in children with infantile spasms taking vigabatrin.

Bold p-value denotes statistically significant.
LASSO, least absolute selection and shrinkage operator; TPM, topiramate; TSC, tuberous sclerosis complex; VABAM, vigabatrin-associated brain abnormalities on magnetic resonance imaging; VGB, vigabatrin; VPA, valproate.
KM curves showed that cases who received concomitant rapamycin therapy had a higher percentage of freedom from developing VABAM than those cases without (95% versus 68.8%, p < 0.001, Figure 3).

Kaplan–Meier survival curves for VABAM in patients with vigabatrin treatment. (a) With or without concomitant rapamycin therapy and VABAM-free survival in patients with vigabatrin treatment. (b) Number of patients with or without concomitant rapamycin therapy at VABAM risk during VGB exposure.
There were 13 (34.2%) patients with symptomatic VABAM; the other 25 (65.8%) were asymptomatic. Among the symptomatic VABAM patients, seven had only movement disorder, three with motor and non-motor symptoms (lethargic and unresponsive), and three with non-motor symptoms (two lethargic and unresponsive and one respiratory distress/arrest). No group differences in the above-observed measures, which were used between IST and ISO groups, emerged between symptomatic and asymptomatic VABAM groups (Table S1). Logistic regression analysis showed no risk/protective factor associated with symptomatic VABAM in the patients with VABAM (n = 38, Table S2). In the whole VGB exposure cohort (n = 172), it showed that concomitant hormonal therapy (p = 0.012, odds ratio = 4.457, 95% confidence interval: [1.381, 14.385]) and TPM exposure (p = 0.037, odds ratio = 8.967, 95% confidence interval: [1.138, 70.64], Table S3) were the risk factors with symptomatic VABAM, but not confirmed by further final multivariate logistic regression analysis (Table S3).
Discussion
Previous studies have shown that the incidence of VABAM was between 22% and 32%,17,25 and a recent meta-analysis found a pooled rate of 21% in seven studies. 14 In our cohort of 172 cases in this study, 38 developed VABAM, and the incidence of 22.1% was generally consistent with previous studies.14,17,25 When all subjects were classified into two groups (IST and ISO), significant differences emerged in the distribution of children with VABAM. The incidence of VABAM in the ISO group (32.6%) is slightly higher than in previous reports but much lower than in the IST group (10%). Furthermore, subgroup analyses showed that concomitant rapamycin therapy seems likely to cause this discrepancy.
Previous studies reached inconsistent conclusions regarding risk factors associated with VABAM. Pearl et al. 17 concluded that young age and relatively high doses appear to be risk factors. Hussain et al. 13 pointed out that the occurrence of VABAM is related to the peak dosage of VGB, notably higher than 175 mg/kg/day. The meta-review performed by Biswas et al. 14 concluded that risk factors for VABAM were less than 12 months of age, unknown etiology of IS, and higher peak dosage of VGB (> 170 mg/kg/day). However, our previous study showed only peak dosage of VGB as an independent risk factor of VABAM, even using the conventional dosage of VGB (i.e. 50–150 mg/kg/day). 19 Although that study found a significant difference in the incidence of VABAM between the IST and ISO groups, no significant differences in the indicators mentioned above were observed. In addition, despite the ISO group having a shorter duration and cumulative dosage of VGB exposure, we had previously demonstrated that these indicators were not significantly associated with the occurrence of VABAM.13,19 The major therapeutic strategies for VABAM were VGB discontinuation; a higher rate of VABAM in the ISO group could lead to more discontinuation of VGB, which in turn would lead to decreased duration and cumulative dosage of VGB exposure in the ISO group.
In this study, treatment regimens differed in both groups, and increased usage of TPM, VPA, and hormonal therapy was observed in the ISO group. Current guidelines recommend VGB as the preferred first-choice medication for IST.4,6,7 According to antiepileptic-to-treat principles, ASM treatment must be initiated as monotherapy; then, combination therapy can be initiated when monotherapy fails.26,27 Furthermore, hormonal therapy, but not VGB, was the first treatment of choice for ISO, especially in the unknown etiologies.4,28–31 These reasons caused the disparities in pharmaceutical therapy between groups in this study.
In the final regression analysis, we found no risk factors associated with VABAM, but it is noted that 37 of 38 children with VABAM had the conventional dosage of VGB (i.e. ⩽ 150 mg/kg/day). We observed group differences in the concomitant rapamycin therapy between the two groups (ISO and IST). The final regression analysis also showed that concomitant rapamycin therapy could be the protective factor associated with VABAM. KM survival analysis indicated significantly different survival between children with or without concomitant rapamycin therapy. To our knowledge, this is the first study to report this phenomenon.
As we mentioned above, all the children with concomitant rapamycin therapy were IST. Hence, we attempted to understand the incidence of VABAM in children with TSC by reviewing the previous literature. Regrettably, only two case reports12,21 and three studies were found.11,13,17 Pearl et al. 17 reported that none of the seven TSC children developed VABAM; the count is 1 of 12 in the report by Hussain et al. 13 and 2 of 24 in that by Dracopoulos et al. 11 Although the incidence of VABAM in the IST group seemed consistent with the previous study, after sub-group analysis, 5 in those 20 without concomitant rapamycin therapy (25%) developed VABAM and 3 in those 60 with it (5%). The results of our study may have a weaker tendency for concomitant rapamycin therapy caused by the lower incidence of VABAM in the IST group, but it needs further verification in more cases. Fortunately, we note that there was a study of VGB use to prevent epilepsy in infants with TSC, 32 and we very much look forward to their follow-up work on VABAM.
Although the previous study demonstrated that VABAM is often reversible and mainly asymptomatic,6,11,13,14 a recent study showed there were 19 (43.2%) patients who had obvious symptoms in 44 VABAM patients, 33 which was also observed in our study (34.2%). For symptomatic VABAM, Hussain et al. 13 considered it might be associated with concomitant hormonal therapy. In our study, we did not observe any association between any factors and symptomatic VABAM; it could be possible interference by other treatment protocols (i.e. concomitant rapamycin therapy). Thus, conclusions from this subset should be taken with caution.
Rapamycin-based (an mTOR inhibitor) therapy has shown benefits for patients with TSC, especially those with renal cancer carcinoma, TSC, and lymphangioleiomyomatosis-related tumors.26,34–36 Beyond that, rapamycin could be useful for seizure control in TSC in both animal and human studies.37–40 Several ASMs are the substrate and inducer of CYP3A4 and phosphoglycolate phosphatase, which may influence the metabolism of mTOR inhibitor,41,42 but this effect was not observed with VGB. 43 Vogel et al. 44 showed that increased expression of transcripts in the mTOR pathway and autophagy was suppressed in the human retinal pigment epithelial cells ARPE19 cultured in VGB, and this effect could be attenuated by the mTOR inhibitor (Torin 2). Both animal and human studies have shown that VGB could induce intramyelinic edema,15,45–48 and studies showed the injury of myelin was associated with autophagy.49,50 Thus, we presume rapamycin could induce autophagy via partial mTOR inhibition to prevent the occurrence of VABAM. Paradoxically, the TSC patients with mTOR hyperactivation should have a higher incidence of VABAM based on that assumption, but this is not the case. We speculated that these seemingly conflictive results might be because TSC patients inherently have brain abnormalities on MRI, and therefore VABAM could be overlooked.
Disruption in the mTOR pathway is believed to enhance neuronal excitability and promote epileptogenesis in TSC patients due to an imbalance of GABAergic inhibition and glutamatergic excitation. 51 For this reason, mTOR inhibitors were introduced to treat various manifestations of TSC especially, epilepsy.37,52–55 Our early study had demonstrated that sirolimus treatment for TSC effectively modified the disease by preventing IS, delaying seizure onset, and relieving its severity. 56 Based on the above research results, rapamycin might be a beneficial therapeutic option in treating IST patients, especially in combination with vigabatrin, for its potential role in the prevention of VABAM.
This study has several limitations. First, this is a relatively small, single-center study; the strength of the conclusions is thus limited, and the results require confirmation by further investigations. Second, this was a retrospective study based on an observed clinical phenomenon, so some children with VGB exposure history but without cerebral MRI examination before an initial dose of rapamycin therapy were excluded; the sample number was small, and the bias should be small, but it should not be dismissed. Third, 143 patients with VGB but without cerebral MRI before VGB exposure or during follow-up were excluded from this study. Because of the study’s retrospective nature, this may be unavoidable and may bias the results; thus, the results need to be interpreted with caution. Fourth, symptomatic VABAM should be further carefully investigated, such as more detail of clinical symptoms and the association with the location of MRI abnormalities or treatment protocols. Finally, although interesting phenomena have been observed, we have encountered difficulties in elucidating specific mechanisms, and additional study is needed to verify our hypothesis. Detailed follow-up work along this line is underway in our team.
Conclusion
In conclusion, in this study with a larger patient cohort, no risk factor associated with VABAM was observed; consistent with our previous study, VABAM may occur even at the conventional dosage of VGB (i.e. ⩽ 150 mg/kg/day). The incidence of VABAM was much lower in IST patients. This finding may be related to treatment with rapamycin, which could reduce the risk of VABAM. We anticipate that our investigation will motivate future studies on the determinants identified in our research and that these studies can explore the mechanisms behind the protective effect.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221138148 – Supplemental material for Vigabatrin-associated brain abnormalities on MRI in tuberous sclerosis complex patients with infantile spasms: are they preventable?
Supplemental material, sj-docx-1-tan-10.1177_17562864221138148 for Vigabatrin-associated brain abnormalities on MRI in tuberous sclerosis complex patients with infantile spasms: are they preventable? by Lin Wan, Wen He, Yang-Yang Wang, Yong Xu, Qian Lu, Meng-Na Zhang, Qiu-Hong Wang, Shuo Dun, Li-Ying Liu, Xiu-Yu Shi, Jing Wang, Lin-Yan Hu, Bo Zhang, Guang Yang and Li-Ping Zou in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
