Abstract
Seizures are a common symptom of craniocerebral diseases, and epilepsy is one of the comorbidities of craniocerebral diseases. However, how to rationally use anti-seizure medications (ASMs) in the perioperative period of craniocerebral surgery to control or avoid seizures and reduce their associated harm is a problem. The China Association Against Epilepsy (CAAE) united with the Trauma Group of the Chinese Neurosurgery Society, Glioma Professional Committee of the Chinese Anti-Cancer Association, Neuro-Oncology Branch of the Chinese Neuroscience Society, and Neurotraumatic Group of Chinese Trauma Society, and selected experts for consultancy regarding outcomes from evidence-based medicine in domestic and foreign literature. These experts referred to the existing research evidence, drug characteristics, Chinese FDA-approved indications, and expert experience, and finished the current guideline on the application of ASMs during the perioperative period of craniocerebral surgery, aiming to guide relevant clinical practice. This guideline consists of six sections: application scope of guideline, concepts of craniocerebral surgery-related seizures and epilepsy, postoperative application of ASMs in patients without seizures before surgery, application of ASMs in patients with seizures associated with lesions before surgery, emergency treatment of postoperative seizures, and 16 recommendations.
Keywords
Seizures are a common symptom of craniocerebral diseases, and epilepsy is one of the comorbidities of craniocerebral diseases. Approximately 3%–50% of patients may suffer from their first seizure after craniocerebral surgery.1–4 How to rationally use anti-seizure medications (ASMs) in the perioperative period of craniocerebral surgery to control or avoid seizures and reduce their associated harm has long been a problem that neurosurgeons and related specialists must pay attention to. In 2006, the National Neurosurgical Seizure Prevention and Control Cooperative Group published the ‘Guidelines for the Prevention and Treatment of Neurosurgery Perioperative and Post-traumatic Seizure (Draft). 5 ’ In 2012, the China Association Against Epilepsy (CAAE) published ‘Expert Consensus on Application of Anti-Seizure Medications After Craniocerebral Disease Surgery (Trial Version). 6 ’ Apart from these, many societies and associations have published relevant consensuses or guidelines. These ‘consensuses or guidelines’ provide clinicians with an important theoretical basis and practice guidance for dealing with craniocerebral surgery-related seizures. However, there are certain differences in the application of ASMs between different consensuses or guidelines, which causes confusion in clinical practice. Furthermore, in the past 10 years, many new ASMs (especially injections) have been marketed in China. The definition, classification, and treatment of seizures, as well as the surgical methods and techniques, of craniocerebral surgery have evolved tremendously. Therefore, it is need to publish an official update guideline on the application of ASMs in the perioperative period of craniocerebral surgery.
Methods
Establishment of the expert panel
The CAAE united with the Trauma Group of the Chinese Neurosurgery Society, Glioma Professional Committee of the Chinese Anti-Cancer Association, Neuro-Oncology Branch of the Chinese Neuroscience Society, and Neurotraumatic Group of Chinese Trauma Society to set up a leading group and work group. Each professional association recommended potential experts for participating in this work, respectively. All experts were discipline leaders of their departments from tertiary A-level hospitals across China, and with over 20 years’ corresponding discipline work experiences. After reviewing their conflict-of-interest statements and curriculum vitae by the leading group, a multidisciplinary panel of experts in neurosurgery and epilepsy were set up.
Evidence search strategy
(1) A comprehensive search was conducted by members of working group with several English and Chinese databases including PubMed, Embase, and the Cochrane Central Register of Controlled Trials in English, and CNKI and Wanfang Data in Chinese. The last search was conducted in April 2021. The search strategy applied for PubMed was a combination of the following key words:
#1 (brain tumor) OR (craniocerebral tumor) OR (craniocerebral trauma) OR (brain injury) OR (cerebrovascular disease) OR (intracranial hemorrhage) OR (intracranial arteriovenous malformation) OR (intracranial aneurysm) OR (cranioplasty) OR (stereotactic brain biopsy) OR (deep brain stimulation) OR (stereotactic brain biopsy) OR (brain abscess) OR (cerebral cavernous vascular malformation);
#2 (epilepsy) OR (seizure) OR (status epilepticus);
#3 (surgery) OR (resection) OR (operation) OR (neurosurgery) OR (neurosurgical operation) OR (craniocerebral surgery) OR (craniocerebral operation);
#4 (anti-epilepsy drugs) OR (anti-epileptic drugs) OR (anti-seizure medications);
#5 #1 AND #2 AND #3 AND #4;
(2) ASMs and their instruction, including indications, approved by Chinese-FDA.
The systematic literature search was conducted by a separate member from the working group, then two other members screened the retrieved records and extracted the text, quality of evidence and strength of recommendations from all relevant literatures independently. Any possible conflicts would be resolved by group discussion with a member from the leader group.
Grade of the evidence
As mentioned above, all relevant literatures were evaluated and graded based on the Oxford Center for Evidence-based Medicine Levels of Evidence (Supplementary Table 1) by two members from the working group independently. Any possible conflicts would be resolved by group discussion with a member from the leader group.
Process of guideline preparation
4.1 The member of leading group and working group build the outline of the guideline after discussion based on the evidences.
4.2 The working group finished the first draft of guideline according the outline and evidence. A meeting of all panel members was held online to discuss the contents of the guideline, and the members of panel get the first draft and literature through email 1 week in advance.
4.3 The working group re-searched the literatures, and second meeting of leading group and working discussed the comments and confirm the revision of the draft.
4.4 A meeting of all panel members was held face to face to discuss the guideline, and the members of panel get the second draft, supplementary literature, and suggest of leading group on the comments on first online meeting through email 1 week in advance.
4.5 A questionnaire was performed to grade the items with controversy or lack of high-level evidences from strongly disagree to strongly agree by all members of panel. The items with 75% agree or strongly agree without strongly disagree, or items with 85% agree or strongly agree and no more than 5% strongly disagree was list in the guideline. The working group finished the third draft after discussion with the members of leading group according the results of questionnaire and comments on second meeting of all members of panel.
4.6 The third meeting of all expert group members was held online to revise the third draft of guideline. The members of panel get the third draft, results of questionnaire, and suggest of leading group on the comments on second meeting through email 1 week in advance.
4.7 The working group finished the fourth draft after discussion with the members of leading group according the comments of members in the third meeting. All members of panel approved the guidelines online at last.
Results
Application scope of guideline
Seizures following craniocerebral surgery usually occur after supratentorial craniotomy, while the incidence of seizures after infratentorial craniotomy is minimal. Therefore, this guideline is mainly applicable to the following two types of patients undergoing supratentorial craniotomies: (1) patients with no history of seizures before surgery; and (2) those with a history of preoperative seizures which are a clinical symptom of craniocerebral diseases and will be treated by this surgery (such as supratentorial meningioma, glioma, and arteriovenous malformations).
Due to significant differences between perioperative treatment for some special patients and that in patients receiving conventional craniocerebral surgery, this guideline does not apply to the following patients: (1) patients undergoing surgery specifically aimed at controlling seizures; (2) those with seizures complicated by craniocerebral disease, for whom the craniocerebral disease treated by this surgery has no causal relationship with the seizures; (3) those undergoing intravascular interventional therapy and stereotactic radiosurgery for craniocerebral diseases; (4) those receiving non-surgical treatment after craniocerebral trauma or stroke; and (5) and those undergoing infratentorial craniocerebral surgery.
Craniocerebral surgery-related seizures and epilepsy
Craniocerebral surgery-related seizures refer to a clinical symptom that appears in patients undergoing craniocerebral surgery. It is caused by excessive abnormal hyper-synchronous firing of neurons. These seizures are paroxysmal, repetitive, transient, and stereotypical 7 (Supplementary Table 2). Seizures occurring after craniocerebral surgery are divided into three categories according to the time of occurrence: immediate seizures occurring within 24 hr (inclusive), early-onset seizures occurring within 24 hr to 14 days (inclusive), and late-onset seizures occurring after 14 days.8–10
Epilepsy is a disease that is associated with persistent changes in the brain and is susceptible to repeated onsets. The diagnosis of craniocerebral surgery-related epilepsy generally requires one of the following conditions: (1) two or more unprovoked seizures within an interval of > 24 hr and (2) an unprovoked seizure with obvious electroencephalogram (EEG) abnormalities, craniocerebral structural abnormalities associated with seizures, or seizure-related gene mutations as well as other high-risk factors for recurring seizures at the same time11,12 (Supplementary Table 2). Provoked seizures are mainly seizures provoked by conditions such as craniocerebral surgery, trauma, hemorrhage, and electrolyte disturbances. A seizure associated with a disease before craniocerebral surgery could be diagnosed as epilepsy, while immediate seizures and early-onset seizures after craniocerebral surgery are mostly provoked seizures, and are generally not diagnosed as epilepsy.
Seizures after craniocerebral surgery can cause early damages such as intracranial hemorrhage, vasospasm, cerebral hypoxia, and cerebral edema, and can also lead to long-term damage such as stigma (caused by social misconceptions) and cognitive impairment. ASMs themselves can also bring potential risks such as allergic reactions and drug interactions8,13 (Supplementary Table 3). Therefore, ASMs should be selected carefully and rationally based on the specific conditions of patients and the characteristics of ASMs (Table 1), in accordance with the principles of long-term efficacy, adequacy, individualization, and the type of seizure.
Introductions of anti-seizure medications.

ASM, anti-seizure medications; CBZ, Carbamazepine; LCM, Lacosamide; LEV, Levetiracetam; LTG, Lamotrigine; OXC, Oxcarbazepine; PB, Phenobarbital; PER, Perampanel; PHT, Phenytoin; PR,; SJS, Steven-Johnson Syndrome; TEN, toxic epidermal necrolysis; TPM, Topiramate; VPA, Valproic acid; ZNS, Zonisamide.
Level C: Animal studies have shown that the drug has teratogenic or embryo-killing effects on fetuses, but there are no adequate and well-controlled studies on pregnant women; or there are no studies on pregnant women and no animal studies. Such drugs must be evaluated by a physician and weighed against the pros and cons before they can be used. Level D: There is clear evidence of harm to human fetuses, but in some cases (such as pregnant women with serious and life-threatening diseases, no safer drugs are available, or drugs are safe but ineffective). The benefits of medication for pregnant women Greater than harm. +++: obvious, ++ medium, + mild, – no.Postoperative application of ASMs in patients without seizures before surgery
Postoperative application of ASMs in patients without seizures before surgery
Indications for early application of ASMs after surgery
For patients with no history of seizures before surgery, whether the prophylactic application of ASMs is needed after craniocerebral surgery has always been controversial. The selection of cases for the postoperative prophylactic use of ASMs should be based on the following three criteria: (1) patients with a high risk of seizures, (2) suffering from seizures that may cause serious injury, and (3) patients in whom ASMs have minimal side effects. It is generally believed that when the risk of postoperative seizures exceeds the risk of using ASMs, they can be used prophylactically. When the risk of seizures is significantly higher than the risk associated with the use of ASMs, they should be used prophylactically (Table 1, Supplementary Table 3).
Most previous clinical evidence does not support the postoperative prophylactic use of ASMs in patients with supratentorial tumors and cerebrovascular diseases.1,14–17 In practice, preventive application of ASMs is extensive.18–21 Studies in the recent years have shown that postoperative prophylactic application of ASMs can significantly reduce the occurrence of seizures, 22 and new ASMs can better control postoperative seizures and have fewer adverse reactions. 23 Therefore, the prophylactic use of ASMs is recommended for patients at high risk of postoperative seizures.
Craniocerebral trauma
It is not recommended to routinely prophylactic useing ASMs after mild and moderate craniocerebral trauma surgery.24–26 However, in the case of one or more of the following conditions, ASMs should be used prophylactically after craniocerebral trauma surgery:24–30
Severe craniocerebral trauma, especially open craniocerebral trauma (intracranial abnormal residue and firearm injuries), depressed skull fracture; multiple cortical contusions, intracranial hematoma, and obvious midline shift (>5 mm);
Multiple craniocerebral operations;
If EEG shows epileptic discharge or is susceptible to seizure (pathogenic gene mutation, family history of seizure, age >40 years, and chronic alcoholism).
Supratentorial tumors
There is no need for the routine prophylactic use of ASMs after supratentorial tumor surgery; 21 however, ASMs should be used prophylactically in the following situations:10,31–37
Incomplete tumor resection; recurrent/progressive tumor;
The tumor involves the temporal lobe or motor cortex; the tumor size is large (maximum diameter of the tumor exceeds 3.5–4.5 cm);
Susceptibility to seizure: family history of seizures, history of febrile seizures, young age (standards vary between patients with different types of tumors; for example, for glioma, the patient’s age should be <45 years);
Long surgery duration (cortical exposure time >4 hr); postoperative imaging examination suggests cerebral infarction;
Local placement of slow-release chemotherapeutics for malignant tumors;
Supratentorial cerebrovascular disease
The routine prophylactic use of ASMs is not required after surgery for supratentorial cerebrovascular disease; 14 however, ASMs should be used prophylactically in the following situations:
Hemorrhagic stroke: the hematoma involves the cortical area; the hematoma volume is large (>10 ml); the cortex is obviously damaged by the operation; EEG reveals epileptic discharge.38,39
Intracranial aneurysm: age <40 years, male sex, or middle cerebral artery aneurysm (especially near the medial temporal lobe); the ruptured aneurysm is bleeding heavily (>15 cm3 of volume of teratoma), or there is loss of consciousness for > 1 h before treatment, Hunt-Hess grade is above grade III or computed tomography suggests that the Fisher grade is grade III or higher; postoperative imaging examination reveals brain contusion or cerebral infarction.40–43
Intracranial arteriovenous malformations: the malformed vascular mass is too large (>4 cm in diameter); it involves the frontal or temporal lobe; it is accompanied by intracranial hemorrhage or local neurological deficits; there is deep vein drainage.44–48
Cerebral cavernous vascular malformation: It is recommended that ASMs be routinely preventively used, especially in the case of the following conditions: the malformed vascular mass involves the cortex or medial temporal lobe; lesion diameter >1.5 cm; multiple lesions; incomplete resection of lesions or band with hemosiderin deposits.46,49–52
Stereotactic surgery
Deep brain electrode implantation: ASMs are not recommended for routine prophylactic use after deep brain stimulation;53,54 however, intracranial vascular events (hemorrhage, infarction, and edema) during the perioperative period can increase the risk of seizures, and ASMs should be used after surgery. 55
Stereotactic brain biopsy: ASMs are not recommended for routine prophylactic use after stereotactic biopsy;56,57 however, when there are high-risk factors for seizures after biopsy (there is visible bleeding on imaging and the penetration point is located in the functional area), ASMs can be used prophylactically. 58
Other surgeries that the risk of postoperative seizures is high and ASM can be used prophylactically
Prophylactic application of ASMs after surgery
Principles of drug selection
In the early stage, ASMs that have less impact on consciousness, fewer adverse reactions, faster onset, fewer drug interactions, and do not require a slow titration of the dosage should be selected. Later, they can be adjusted according to clinical needs. For the elderly and other patients with liver and kidney dysfunction, attention should be paid when choosing ASMs and those having little effect on liver and kidney function should be selected (Table 1). In principle, the later orally administrated ASMs should be the same as the postoperative intravenous ASMs.
Commonly used drugs
Commonly used injectable ASMs: Currently, the available domestic intravenous ASMs include sodium valproate injections (the first dose of 400 mg for adults is administered slowly via intravenous injection or instillation, and continuous administration via intravenous pumping at 1200 mg/d starts within 30 min; the first dose of 15 mg/kg for children is administered slowly via intravenous injection or instillation, and is continuously administered via intravenous pumping at 1 mg/kg/h), 62 levetiracetam injection (10–20 mg/kg administered via intravenous instillation once every 12 h),9,37 and lacosamide injection (100 mg/dose, twice a day, with the first dose doubled for adults or children weighing > 50 kg; 1–2 mg/kg/dose, twice a day for other children).63–65
Commonly used oral ASMs: Sodium valproate, levetiracetam, oxcarbazepine, 66 lacosamide, and topiramate. 67 If ASM usage needs to be adjusted later, lamotrigine, 68 perampanel, 69 and so on, can be selected. For patients with malignant tumors that require chemotherapy, the use of ASMs that induce hepatic microsomal enzymes should be avoided.10,70,71 Levetiracetam (non-enzyme inducing ASM) and sodium valproate (enzyme-inhibitor), which have a synergistic effect with chemotherapeutic agents like temozolomide, are recommended.62,68,72
Application method
Principles of application: Injectable ASMs should be first used on the day of surgery, and when the role of the gastrointestinal tract in food intake is restored, it should be changed to oral ASMs at a selected time; prophylactic ASMs need to reach the therapeutic dose, and blood concentration monitoring is recommended.
Application of injections: Injectable ASMs should be continuously administered via intravenous pumping or intravenous instillation with a micro-injection pump in accordance with the package insert to ensure that the therapeutic concentration is reached quickly and remains stable.
Transformation from injection to oral use: According to the patient’s postoperative consciousness, the injection should be transitioned gradually to oral ASMs, and there can be an overlap for 12–24 h during the transition process. During the overlap, when using the same ASMs or ASMs having the same mechanism of action, attention should be paid to drug overdose and adverse reactions. Monotherapy is the first choice for early oral ASMs.
Time of use: The first intravenous injection should be administered as soon as possible after surgery to reduce the rate of occurrence of immediate seizures. The intravenous injection is usually used for 2–3 days. There is currently no evidence that the preventive use of ASMs can reduce the occurrence of late-onset seizures; thus, the long-term use of ASMs is not recommended. If no seizures occur after surgery, ASMs should be tapered off and discontinued 14 days after surgery.
Common adverse reactions and their treatment:73–79 (1) allergic reactions (phenytoin, carbamazepine, lamotrigine, zonisamide, phenobarbital, oxcarbazepine, etc.) are mainly manifested as skin rash and Steven-Johnson syndrome/toxic epidermal necrolysis/drug-induced hypersensitivity syndrome occurs in severe cases. Once common skin rash or critical ones like widespread maculopapular, erythema multiforme, and flaccid bullae are observed, ASMs that may cause allergy should be discontinued immediately and changed to ASMs that induce fewer allergic reactions (topiramate, levetiracetam, valproic acid, perampanel, etc.). (2) Patients with hyperammonemia (those who use valproic acid, phenytoin, lamotrigine, etc.) may have symptoms such as disturbance of consciousness, convulsive seizures, spontaneous respiratory arrest, and so on. The diagnosis can be confirmed by checking blood ammonia and EEG, and the related ASMs should be discontinued immediately and changed to ASMs that do not cause hyperammonemia. Patients with severe conditions need to use arginine injections and symptomatic supportive treatment. (3) mental symptoms mainly include irritability and aggressive behavior, which are mostly transient. Patients with severe conditions need to reduce the dose of related drugs or change the ASMs they are taking.
Control of postoperative seizures
Treatment of immediate and early-onset seizures after surgery.73,80
For those who prolaxylatic use ASMs after surgery: if immediate seizures or occasional early-onset seizures occur, after excluding the predisposing factors, the doses of ASMs can be increased or the condition can be observed. If no seizures occur for 3 consecutive months, ASMs could be tapered off and discontinued after considering EEG findings; If ⩾ 3 early-onset seizures occur, after excluding the predisposing factors, the doses of ASMs can be increased or they can be combined with other ASMs according to the situation, and blood concentration monitoring should be performed if necessary. If no seizures occur for 12 consecutive months, ASMs can be tapered off and discontinued after considering the EEG findings (except for glioblastoma).
For those who did not use ASMs preventively after surgery: For those with immediate seizure after surgery, after excluding the predisposing factors, intravenous injections of ASMs should be started, followed by oral ASMs; if no seizures occur for 3 consecutive months, ASMs can be tapered off and discontinued after considering the EEG findings. For early-onset seizures after surgery, intravenous injections of ASMs should be started, followed by oral ASMs, or oral ASMs may be started directly; if no seizures occur for 3 consecutive months, ASMs can be tapered off and discontinued after considering the EEG findings. If multiple immediate and/or early-onset seizures occurred after medication, the doses of ASMs can be increased, and blood concentration testing should be performed if necessary; if no seizures occur for 12 consecutive months, ASMs can be tapered off and discontinued after considering the EEG findings (except for glioblastoma).
Treatment of late-onset seizures after surgery.10,73
For a single seizure, if the patient is still taking ASMs orally, the doses of ASMs may be increased or other ASMs may be added for treatment based on the type of seizure, drug and patient characteristics, and with considerations of the results of blood concentration monitoring (if feasible); if the patient is not taking oral ASMs, then the treatment with ASMs needs to be initiated according to the type of seizure, and drug and patient characteristics. If no seizures occur for 12 consecutive months, it is recommended to taper off and discontinue ASMs after considering EEG findings (except for glioblastoma).
When late-onset seizures repeatedly occur after taking ASMs, imaging, EEG examinations, and blood drug concentration testing should be performed to assess intracranial structural changes (such as recurrence or progression of brain tumors, whether there are related complications, etc.), epileptic discharge on EEG, neurologists or pediatric neurologists should be asked to conduct a consultation, and treatment with ASMs should be conducted according to the patient’s condition.
If it is in line with medically intractable epilepsy, a comprehensive evaluation should be conducted by the epilepsy center, and epilepsy surgery should be performed if necessary.
Application of ASMs in patients with seizures associated with lesions before surgery
Application of ASMs before surgery
Timing of use
If a seizure occurs before surgery and is associated with a craniocerebral disease that requires surgical treatment, the seizure can be diagnosed. 11 And ASM treatment should be performed immediately after the diagnosis. A 2-h video EEG examination is effective for the differentiation of epileptic from nonepileptic seizures. 10
Principles of drug selection
Drug treatment for seizures mainly entails selecting a sufficient dosage of a single ASM for treatment based on the type of seizures proposed by the International League Against Epilepsy (ILAE) in 2017. 7 Because the course of preoperative seizures is often short, it is recommended to choose ASMs that have a rapid onset of activity and do not require slow titration. Considering the need to continue to apply ASMs in the early postoperative period and for a considerable period, it is recommended to use ASMs that have full dosage forms (such as those equipped with injections, tablets or capsules, oral liquids, and many other dosage forms), are convenient to use, and have no obvious drug interactions with other anti-infective drugs, glucocorticoids, hemostatic drugs, etc. For patients with malignant tumors who need chemotherapy after surgery, ASMs (such as carbamazepine) that induce hepatic microsomal enzymes should be avoided. 71 Presently, the commonly used ASMs for patients with tumors after surgery are sodium valproate and levetiracetam.68,72 In addition, studies have confirmed that lacosamide also has good control over tumor-related seizures. 64
Discontinuation of ASMs before surgery
For the safety of surgery and anesthesia, fasting time of ASMs should be no less than s hr for tablets or capsules, and 6 hr for oral solutions before surgery. However, interrupt of ASMs should not be longer than 2 half-lives of the drugs taken before surgery.
Application of ASMs after surgery
Case selection
Patients with seizures before surgery need to use ASMs routinely after surgery.35,36,81–83
The principle of drug selection
Injectable ASMs should be used first on the day after surgery, and the same injectable ASMs or injectable ASMs with the same mechanism of action should be selected from the ASMs used before surgery; oral ASMs should be based on the type of preoperative drugs.
Commonly used drugs and application methods
Commonly used ASMs for injection and application methods are shown in Part III (II). The dosage and regimen for oral medication are the same as those before surgery.
Control of postoperative seizure
Application of ASMs during postoperative seizures
Refer to the treatment of seizure after the text in Part III (III).
Discontinuation of ASMs after surgery (Figure 1).10,73
The best time to discontinue ASMs in patients after craniocerebral surgery is not completely clear. Most studies suggest that the nature of the lesion, the degree of resection, preoperative seizure time, frequency, and epileptiform discharges on EEG, postoperative seizure control, and other factors should be considered when determining the postoperative withdrawal time, and ASMs generally start to be tapered off and discontinued after the patient has been seizure-free for 12–24 months; 84
If the surgery for benign lesions or low-grade malignant tumors has achieved total resection of the lesions, and the preoperative seizure time is < 6 months, the discharge on EEG is consistent with the lesion site, and there is no seizure after surgery, ASMs can be tapered off and discontinued after 6 months;
For benign lesions or low-grade malignant tumors, if the surgery has achieved total resection of the lesions but there were multiple preoperative seizures and the discharge on EEG is far away from the lesion site, or seizures occurred after surgery, ASMs should be tapered off and discontinued after the patient has been seizure-free for 24 consecutive months;
If surgery for WHO-grade III malignant tumors, or benign and low-grade lesions did not achieve total resection, and seizures occasionally occurred after surgery, ASMs should be tapered off and discontinued after the patient has been seizure-free for 24 consecutive months;
If repeated seizures occurred after surgery for WHO-grade IV or WHO-grade III malignant tumors, it is not recommended to discontinue ASMs.

Flow-chart of anti-seizure medications administration in perioperative period of patients with craniocerebral surgery.
Emergency treatment of postoperative seizures
Treatment of postoperative seizures
Although the risk of postoperative seizures could persist for several months, it is usually within 72 h after surgery. 85
Terminating single seizures and preventing accidental risks
Most seizures after craniocerebral surgery are either focal seizures or have focally progressed to general tonic-clonic seizures, which are transient, and it is not necessary to give benzodiazepines or other injectable ASMs immediately to terminate single seizures. 86 Take immediate steps to keep the patient’s airway unobstructed and prevent the patient from falls or injury; avoid irritating the patient, avoid pinching the philtrum, opening the mandibular joint, and pressing or shaking the patient to cause further injury; during the convulsions, the patient can be placed in a horizontal position with the head tilted to one side or placed in a lateral position, oral and nasal secretions should be cleaned up in time, and in particular, feeding oral ASMs should be avoided to prevent suffocation; at the same time, monitor vital signs, ensure normal cardiopulmonary function, inhale oxygen if necessary, and establish venous access. If the seizure lasts for >5 min, medication would be required to terminate it. 87 For specific treatment, see Part V (II).
Terminating multiple seizures and preventing accidental risks
If multiple seizures occur, with each seizure lasting for <5 min, and the patient’s consciousness returns to the baseline level during the seizures, it will be treated as a cluster seizure. Cluster seizures are likely to progress to status epilepticus. 88 Therefore, for cluster seizures, midazolam or other ASMs can be administered orally or intravenously to stop the seizures as soon as possible to prevent recurrences.
Identifying the cause of seizures
Seizures after craniocerebral surgery may be the continuation of preoperative seizures, or they may be caused by surgical trauma, electrolyte imbalances, hypoglycemia, and so on. There are also some non-epileptic symptoms (such as decortical rigidity, cardiogenic disturbance of consciousness, etc.) that need to be differentiated from seizures. Therefore, when patients develop seizures after surgery, electrocardiogram, blood glucose measurement, serum electrolyte measurement, brain imaging, and other examinations should be performed as soon as possible to determine the etiology and triggers of the seizures, and video EEG and ASM blood concentration tests should be performed when necessary.
Treatment of status epilepticus
Status epilepticus is a rare life-threatening complication after craniocerebral surgery with an incidence of less than 1%. According to whether there are symptoms of convulsion, status epilepticus can be divided into convulsive status epilepticus and non-convulsive status epilepticus (Supplementary Table 2). In 2015, the ILAE proposed two time points for status epilepticus (T1 and T2): T1 means that seizures hardly end on their own and treatment should be initiated; T2 means that irreversible neurological damage to the brain tissue occurs, and brain protection and intensive treatment should be initiated. For tonic-clonic status epilepticus, T1 is 5 min and T2 is 30 min; for focal status epilepticus with disturbance of consciousness, T1 is 10 min and T2 is > 60 min. 87
Treatment of convulsive status epilepticus
The overall principles for the treatment of convulsive status epilepticus during the perioperative period of craniocerebral surgery include the following: terminating seizures, maintaining vital signs, finding the cause, and so on.89,90 The specific recommended treatment process is as follows (Figure 2).

Flow-chart of management for generalized convulsive status epilepticus after craniocerebral surgery.
If intravenous access has not been established, intramuscular injections of midazolam 0.3 mg/kg (⩽10 mg/time) could be administered. For those who have established intravenous access, the first choice is to slowly inject intravenous diazepam at a dose of 0.3–0.5 mg/kg (⩽10 mg/time) at a rate of 1–2 mg/min. If the seizure stops during the injection, stop the bolus injection. If the seizure is not controlled after 5 min or it recurs after control, the administration can be repeated once. If it still cannot be controlled, treat it as refractory convulsive status epilepticus, and it is recommended to use high-dose valproic acid injection or levetiracetam (>30 mg/kg) for treatment.91,92 For refractory status epilepticus that cannot be controlled after more than 40 min, the patient should be transferred to the intensive care unit for intubation for respiratory management, and then anesthetics such as propofol and midazolam should be continuously administered by intravenous pumping for treatment.
Treatment of non-convulsive status epilepticus
Non-convulsive status epilepticus after craniocerebral surgery is common. 93 This guideline recommends that in case a patient has clinical manifestations of suspected non-convulsive status epilepticus (acute onset of mental and behavioral abnormalities, cognitive abnormalities, changes in the state of consciousness, language disorders, various sensory and autonomic abnormalities, etc.), which cannot be fully explained by the lesions, the operation itself, or other known causes, the neurological function has not recovered after treatment of convulsive status epilepticus, or if the symptoms persist for more than 10 min, the possibility of non-convulsive status epilepticus should be considered, and video EEG monitoring (video EEG monitoring under conditions permission) should be put in place as soon as possible. If either of the following is met, the diagnosis can be made: (1) the patient’s EEG shows sustained epileptic discharge at >2.5 Hz and (2) the epileptic discharge is ⩽ 2.5 Hz or shows rhythmic δ/θ activity (>0.5 Hz), and the EEG and clinical results improved after intravenous injection of ASMs, there are subtle clinical seizures or there is a typical spatiotemporal evolution of the EEG (voltage, frequency, and distribution). 94
Once the diagnosis of nonconvulsive status epilepticus is established, ASMs should be given as soon as possible. The longer the duration of non-convulsive status epilepticus before treatment, the more difficult it is to terminate the seizure. The first choice for treatment is oral or intramuscular injections of midazolam or oral or intravenous injections of other available benzodiazepines and so on; as a second choice, intravenous administration of valproic acid or levetiracetam may also be performed; oral lacosamide, topiramate, and so on may also be considered; ultra-refractory non-convulsive status epilepticus can also be treated with ketogenic diets, mild hypothermia, and other therapies. 95
Recommendations
The recommendations were given according to relevant domestic and foreign literatures, which were evaluated and graded based on the Oxford Center for Evidence-based Medicine Levels of Evidence (2009). See Table 2.
Recommendations of this guideline.
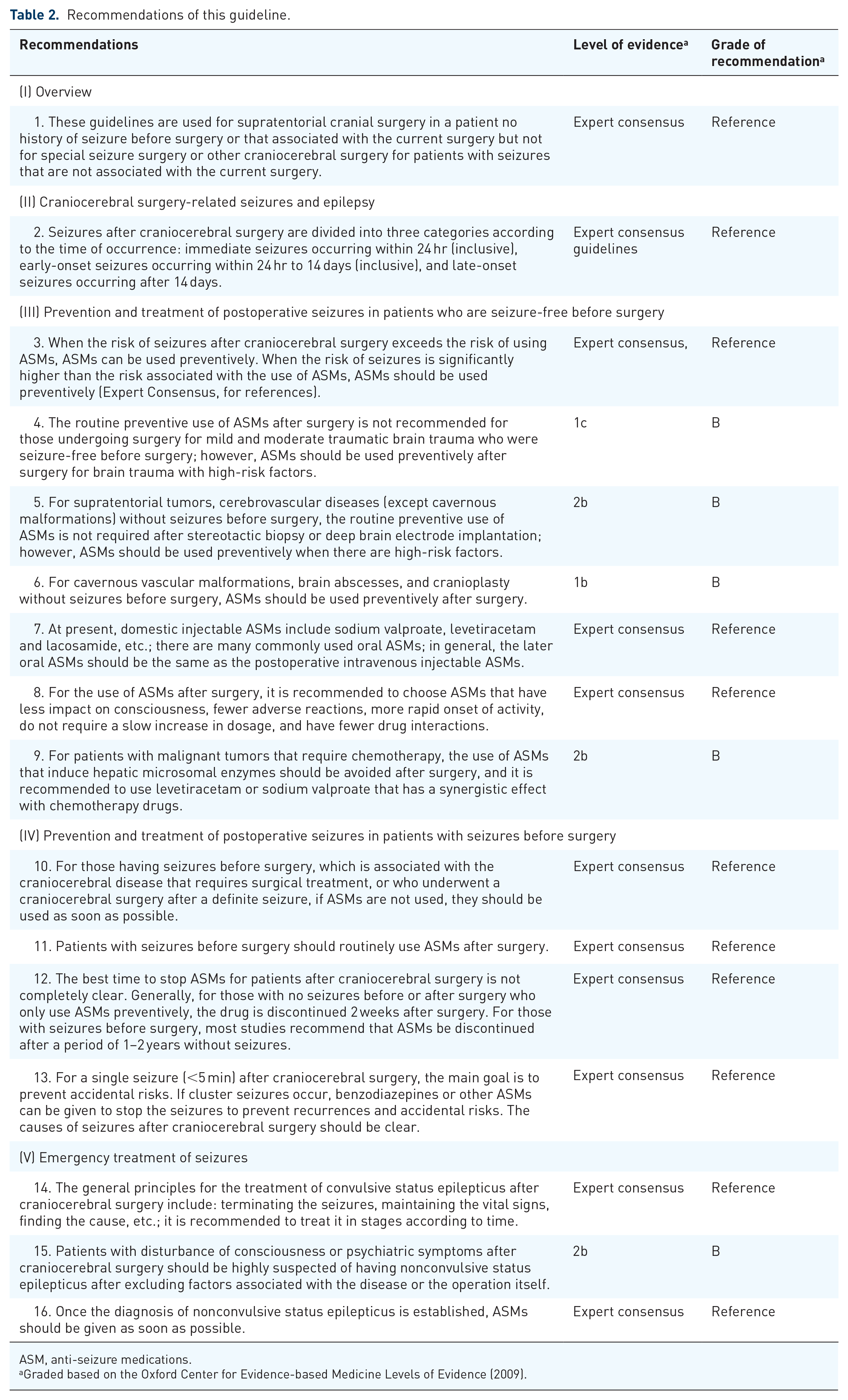
ASM, anti-seizure medications.
Graded based on the Oxford Center for Evidence-based Medicine Levels of Evidence (2009).
Prospects
Although substantive progress has been made in the management of perioperative seizures associated with supratentorial craniocerebral surgery in recent decades, it remains difficult to derive conclusive knowledge for some issues. For instance, ASM prophylaxis and discontinuation are still elusive and evidences included here may documented contradictory findings. On these issues, the guideline can only provide current recommendations with high feasibility, future high-quality clinical evidences are needed.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221114357 – Supplemental material for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery
Supplemental material, sj-docx-1-tan-10.1177_17562864221114357 for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery by Shuli Liang, Xing Fan, Feng Chen, Yonghong Liu, Binghui Qiu, Kai Zhang, Songtao Qi, Guojun Zhang, Jinfang Liu, Jianguo Zhang, Jun Wang, Xiu Wang, Ziyang Song, Guoming Luan, Xuejun Yang, Rongcai Jiang, Hua Zhang, Lei Wang, Yongping You, Kai Shu, Xiaojie Lu, Guoyi Gao, Bo Zhang, Jian Zhou, Hai Jin, Kaiwei Han, Yiming Li, Junji Wei, Kun Yang, Gan You, Hongming Ji, Yuwu Jiang, Yi Wang, Zhiguo Lin, Yan Li, Xuewu Liu, Jie Hu, Junming Zhu, Wenling Li, Yongxin Wang, Dezhi Kang, Hua Feng, Tinghong Liu, Xin Chen, Yawen Pan, Zhixiong Liu, Gang Li, Yunqian Li, Ming Ge, Xianming Fu, Yuping Wang, Dong Zhou, Shichuo Li, Tao Jiang, Lijun Hou and Zhen Hong in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864221114357 – Supplemental material for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery
Supplemental material, sj-docx-2-tan-10.1177_17562864221114357 for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery by Shuli Liang, Xing Fan, Feng Chen, Yonghong Liu, Binghui Qiu, Kai Zhang, Songtao Qi, Guojun Zhang, Jinfang Liu, Jianguo Zhang, Jun Wang, Xiu Wang, Ziyang Song, Guoming Luan, Xuejun Yang, Rongcai Jiang, Hua Zhang, Lei Wang, Yongping You, Kai Shu, Xiaojie Lu, Guoyi Gao, Bo Zhang, Jian Zhou, Hai Jin, Kaiwei Han, Yiming Li, Junji Wei, Kun Yang, Gan You, Hongming Ji, Yuwu Jiang, Yi Wang, Zhiguo Lin, Yan Li, Xuewu Liu, Jie Hu, Junming Zhu, Wenling Li, Yongxin Wang, Dezhi Kang, Hua Feng, Tinghong Liu, Xin Chen, Yawen Pan, Zhixiong Liu, Gang Li, Yunqian Li, Ming Ge, Xianming Fu, Yuping Wang, Dong Zhou, Shichuo Li, Tao Jiang, Lijun Hou and Zhen Hong in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864221114357 – Supplemental material for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery
Supplemental material, sj-docx-3-tan-10.1177_17562864221114357 for Chinese guideline on the application of anti-seizure medications in the perioperative period of supratentorial craniocerebral surgery by Shuli Liang, Xing Fan, Feng Chen, Yonghong Liu, Binghui Qiu, Kai Zhang, Songtao Qi, Guojun Zhang, Jinfang Liu, Jianguo Zhang, Jun Wang, Xiu Wang, Ziyang Song, Guoming Luan, Xuejun Yang, Rongcai Jiang, Hua Zhang, Lei Wang, Yongping You, Kai Shu, Xiaojie Lu, Guoyi Gao, Bo Zhang, Jian Zhou, Hai Jin, Kaiwei Han, Yiming Li, Junji Wei, Kun Yang, Gan You, Hongming Ji, Yuwu Jiang, Yi Wang, Zhiguo Lin, Yan Li, Xuewu Liu, Jie Hu, Junming Zhu, Wenling Li, Yongxin Wang, Dezhi Kang, Hua Feng, Tinghong Liu, Xin Chen, Yawen Pan, Zhixiong Liu, Gang Li, Yunqian Li, Ming Ge, Xianming Fu, Yuping Wang, Dong Zhou, Shichuo Li, Tao Jiang, Lijun Hou and Zhen Hong in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We are grateful to Secretary General Mrs. Hui Zhang, Deputy Secretary General Mr. Lirong Duan, and Secretary Mrs. Xueya Hao, and all staff of CAAE Secretariat for their organization and support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
