Abstract
Chronic obstructive pulmonary disease (COPD) is a disease with increasing prevalence and burden for health systems worldwide. Every country collects its own epidemiological data regarding COPD prevalence, morbidity and mortality while taking steps to educate the population and medical community to improve early detection and treatment. The rising COPD prevalence creates a need for comprehensive guidelines. In 2012 and 2017–2018, the Romanian Society of Pneumology (SRP) organised national inquiries for COPD, while lung physicians in Romania began receiving education regarding the correct algorithms for COPD diagnosis and therapy. During 2019, a Romanian clinical guideline for diagnosis and treatment of COPD was published, and a condensed version of key points from this guideline are presented herein. COPD is diagnosed based on the presence of three major components: relevant exposure history, respiratory symptoms, and airway limitation that is not fully reversible. Clinical evaluation of patients diagnosed with COPD should include the level of symptoms, exacerbation rate, the presence of comorbidities and determination of phenotypes. The present abridged guideline is designed to be accessible and practical for assessing and managing patients with COPD. The application of up-to-date COPD guidelines may enhance the optimism of physicians and patients in managing this disease.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is recognised as a disease with increasing prevalence and burden for healthcare systems worldwide. Already the third leading cause of death worldwide, COPD led to 3.2 million deaths in 2017, with the toll expected to reach 4.4 million per year by 2040. 1
Every country endeavours to collect its own epidemiological data on COPD prevalence, morbidity and mortality, while taking steps to educate the general population and medical practitioners in order to achieve early detection and treatment. Guidelines and recommendations for COPD management from national respiratory societies are important in this context, as they are adapted to the requirements, possibilities and characteristics of medical policy in each respective country.
In 2012, an epidemiological study involving 1 543 participants revealed that the prevalence of COPD in Romania was 9.3%, with only 4% of patients receiving a diagnosis of COPD at the moment of evaluation. 2 In 2018, Blanco et al. 3 analysed 62 studies from 19 countries and observed that the prevalence of COPD in countries from Eastern Europe was between 10 and 15%. The lack of epidemiological studies and the rising prevalence of COPD has led to the need of comprehensive guidelines.
In 2012 and 2018, the Romanian Society of Pneumology (SRP) organised the national inquiries for COPD, and at the same time, a group of experts in COPD began educating Romanian lung physicians in the correct algorithms for use in COPD diagnosis and therapy. During this period, the need to renew SRP recommendations for the management of COPD in Romania became apparent, according to changes and evolution of the diagnostic and therapeutic approach towards patients with COPD over the previous two years. In 2019, a panel of experts in COPD produced new Romanian recommendations for the management of patients with COPD, in order to align Romanian recommendations with the new Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommendations, and with the medical policies of other European countries.
The aim of the present review was to provide a condensed, accessible version of the updated Romanian COPD guidelines, with a focus on key elements of the full Romanian clinical guideline for diagnosis and treatment of COPD.
Methods
The current paper represents a reduced format of the original version of the Romanian clinical guideline for diagnosis and treatment of COPD, that was written between January and September 2019, and officially published in Romanian in November 2019 (accessible at www.srp.ro). 4 The original guideline details publications that support the recommendations regarding COPD treatment, and is available to all physicians, as well as to the general public, in Romania.
The need to write and update the Romanian guideline for diagnosis and treatment of COPD was due to the increasing prevalence of COPD in Romanian patients, and dynamic changes to the diagnostic and therapeutic approach to patients with COPD over the previous few years. It is important to mention that the guideline is based on extensive literature research using the most widely used medical databases (Cochrane Database of Systematic Reviews; PubMed; Database of Abstracts of Reviews of Effects; and Health Technology Assessment Database). The Romanian COPD guideline owes greatly to the international GOLD report recommendations, 1 but is adapted for the Romanian health care system and for the income range of Romanian patients.
In the same year that the Romanian guideline was written and published, the SRP commissioned an expert group to draft a shorter version of the guideline covering only the most important parts of the original COPD guideline. In constructing the present report, particular attention was drawn to the algorithm of COPD diagnosis, extensive evaluation and phenotyping, differential diagnosis between COPD and asthma, and treatment sections, including a therapeutic scheme.
Diagnosis
Algorithm for diagnosis
The diagnosis of COPD is based on the presence of three major components: relevant exposure history, respiratory symptoms and airway limitations that are not fully reversible (Figure 1).

Three basic steps used in the diagnostic algorithm for chronic obstructive pulmonary disease (COPD). FEV1, forced expiratory volume in one second; FVC, forced vital capacity.
The diagnostic algorithm starts with clinical suspicion based on the following criteria:
adult with a significant smoking history (more than 10 pack-years) or exposure to other risk factors (e.g. occupational or environmental pollution, or passive smoking) family history of COPD, genetic factors, or growth or developmental disorders (e.g. low birth weight, or recurrent infections in childhood) the presence of chronic respiratory symptoms: progressive dyspnoea on exertion, chronic cough with or without expectoration, chest tightness, intermittent wheezing or history of recurrent respiratory infections. These symptoms are nonspecific, and can also occur in other thoracic or extrathoracic conditions.
5
Clinical examination is of limited use in definitively diagnosing COPD but may be suggestive in late-stage COPD. 6 Reduced chest expansion, hyperinflation of the chest, hyperresonance to percussion, prolonged expiratory phase and soft breath sounds or, occasionally, wheezing and bronchial rales, can be found on physical examination. Signs of right heart failure, increased respiratory rate at rest, cyanosis, and use of accessory respiratory muscles may be present in severe COPD. 7
Spirometry represents the main investigation for positively diagnosing COPD. In the presence of relevant exposure history and respiratory symptoms, the presence of airflow obstruction, defined by post-bronchodilator forced expiratory volume in one second (FEV1)/ forced vital capacity (FVC) < 0.70 confirms the diagnosis of COPD.1,8 However, this criteria may underestimate the obstruction in younger individuals, and over-diagnose in older people.1,7,9
Chest X-ray should be performed on the initial evaluation in order to exclude other causes of chronic respiratory symptoms. Chest X-rays may be normal or may show signs of pulmonary emphysema: hyperinflation (increase of ante-posterior diameter of the chest, increase of retrosternal airspace, flattening of the diaphragm, enlargement of intercostal spaces), amputation of peripheral branches of the pulmonary arteries or vertical position of the heart. 10
Computed tomography of the chest may be useful for a more detailed analysis of pulmonary emphysema lesions, to detect associated bronchiectasis and for evaluation for surgical treatment. 11
Extensive evaluation
Clinical evaluation of patients diagnosed with COPD should include the level of symptoms, the rate of exacerbation, the presence of comorbidities and determination of phenotypes (Figure 2). Using the modified medical research council (mMRC) dyspnoea scale and/or the COPD assessment test (CAT) questionnaire for the assessment of respiratory symptoms and the number of moderate and severe exacerbations per year, patients are classified into one of four categories, A, B, C or D, according to the GOLD 2019 recommendations. 1
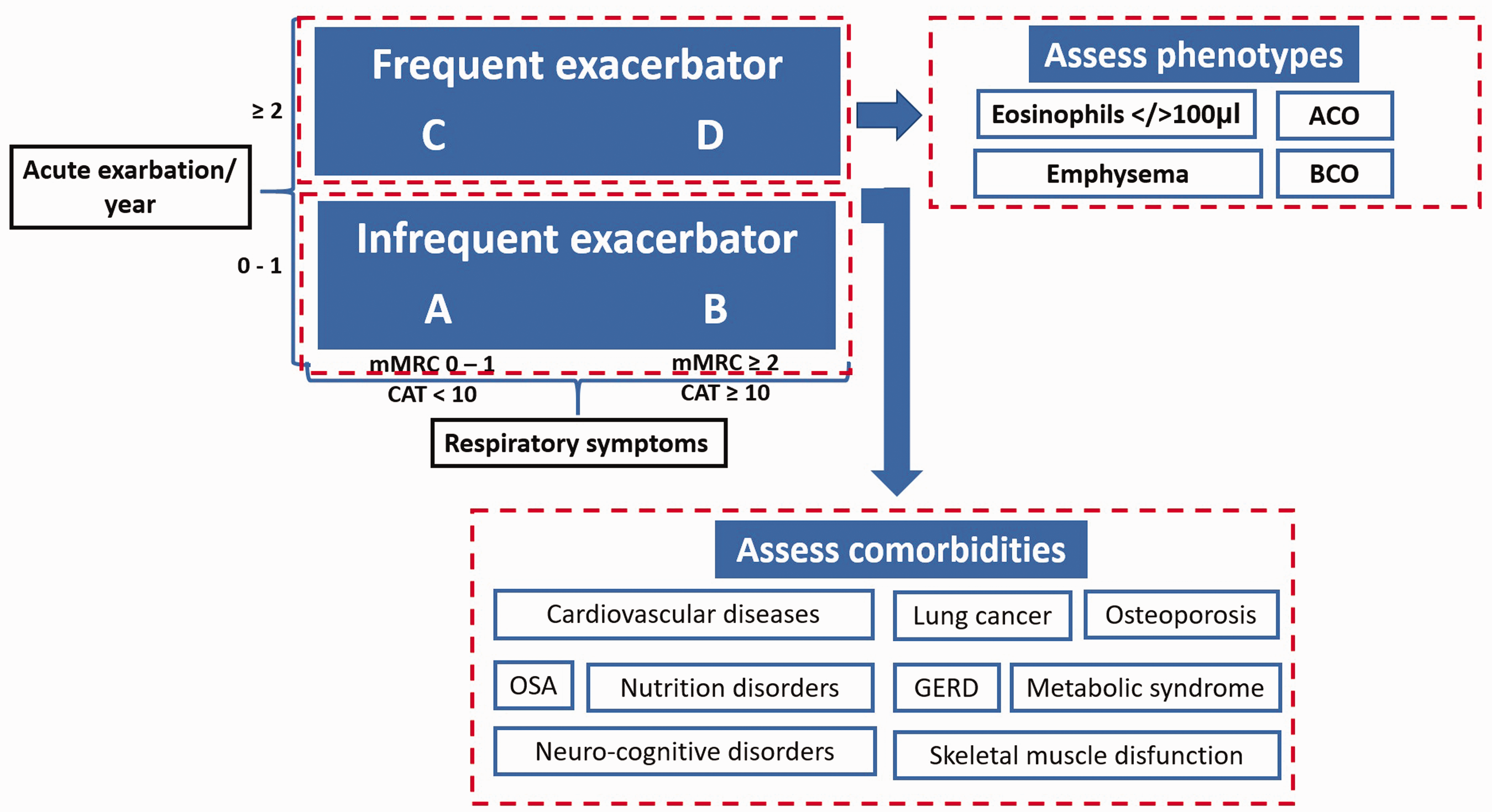
Assessment of patients with chronic obstructive pulmonary disease (COPD). ACO, asthma and COPD overlap; BCO, bronchiectasis and COPD overlap; CAT, COPD assessment test; GERD, gastroesophageal reflux disease; mMRC, modified medical research council dyspnoea scale; OSA, obstructive sleep apnoea.
Phenotype must be determined for every patient due to the large clinical variability of COPD, which is not covered only by A, B, C, or D classification, and the fact that in clinical practice, several phenotypes may occur concurrently within the same patient. The main criteria for defining phenotypes considered to be relevant for COPD management are summarised in Table 1.12–19
Chronic obstructive pulmonary disease (COPD) phenotypes relevant for therapeutic management.

BD, bronchodilator; DLco, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in one second; HRCT, high-resolution computed tomography; Kco, carbon monoxide uptake rate; LLN, lower limit of normal; RV, residual volume; TLC, total lung capacity; ULN, upper limit of normal.
More a biomarker than clinical phenotype, blood eosinophilic level appears to be the most promising tool for assessing the risk/benefit ratio associated with use of inhaled corticosteroids (ICS) in patients with COPD. 20
As a systemic disease, COPD often coexists with other conditions. 21 Thus, every patient with COPD should be routinely assessed and appropriately treated for concomitant chronic diseases. Common comorbidities include cardiovascular diseases, metabolic syndrome, lung cancer, obstructive sleep apnoea (OSA), skeletal muscle disfunction, neuro-cognitive disorders, gastroesophageal reflux disease (GERD), osteoporosis, and nutrition disorders. 22
The six-minute walk test (6MWT) must be used to assess exercise tolerance, and for patients with severe disparity between the minimal symptoms reported and the severity of airflow obstruction. This test is also useful in monitoring the progress of disease and can be integrated into a multidimensional scoring system (e.g. BODE index) to predict long-term outcomes and risk of death for these patients. 23
Overnight polysomnography or portable devices for monitoring cardiorespiratory parameters should be addressed in patients with COPD who are susceptible to OSA. The existence of OSA in patients with COPD increases the severity of cardiac dysfunction and mortality rate.24,25
Differential diagnosis
Chronic obstructive pulmonary disease may be confounded by other respiratory or cardiac diseases that share common clinical features, for example, dyspnoea, wheezing, or productive chronic cough. Representative features of COPD include: occurrence over the age of 40 years, mostly smokers, and onset with progressive exertion dyspnoea. Unusual characteristics of COPD are: onset before the age of 35 years, paroxysmal dyspnoea, orthopnoea, and reversible airflow obstruction. The most common differential diagnoses of COPD include asthma, bronchiectasis, lower airway infections (including tuberculosis) and heart diseases.
Distinguishing between asthma and COPD represents a common diagnostic problem in clinical practice. A comprehensive approach comprising smoking/allergy history, symptoms, family history, associated conditions, along with pulmonary function tests is required but is not always conclusive: significant reversibility can be present in individuals with COPD, and may not always be present in patients with asthma. 26 If a definite diagnosis of asthma, COPD or asthma-COPD overlap is not clearly established in an individual patient, but they present reversibility in airflow obstruction and/or high levels of blood eosinophils, they may benefit from using ICS.8,27
Treatment strategy
The aim of treatment comprises two main strategies: controlling symptoms and reducing future risk, in order to prevent disease progression and decrease mortality (Figure 3). Extensive therapeutic intervention in patients with COPD includes both pharmacological and non-pharmacological means.
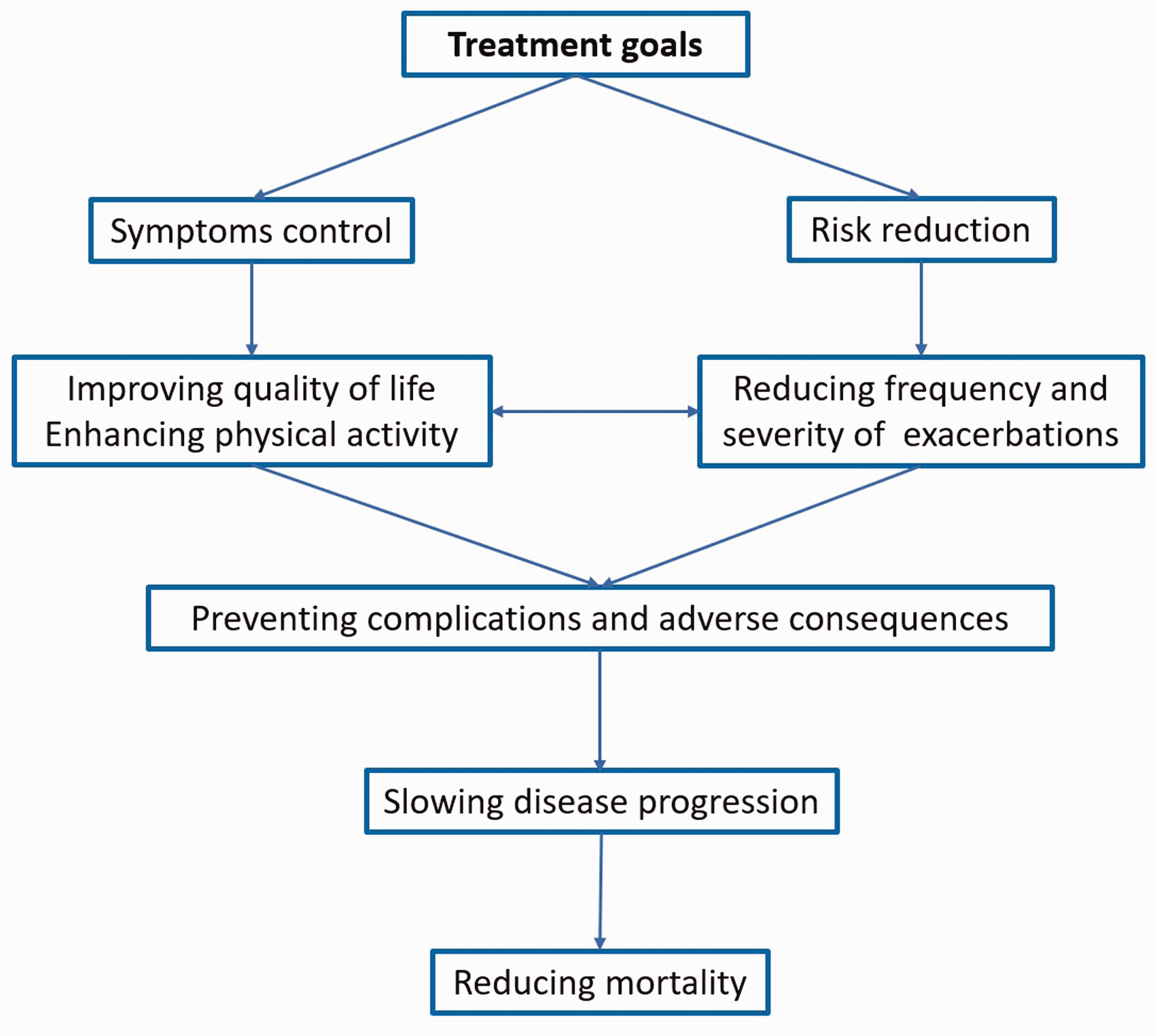
The main aspects in chronic obstructive pulmonary disease treatment.
Non-pharmacological treatment
Non-pharmacological treatment consists of a series of general measures, such as smoking cessation, regular physical activity adapted to the patient’s tolerability and pulmonary rehabilitation, regular assessment of inhaler technique, proper nutrition, oxygen therapy and ventilator support.
Smoking cessation is recommended for all patients with COPD who smoke, regardless of disease stage. 28 Smoking cessation has proved to be of great benefit in slowing disease progression, reducing exacerbation rate, and increasing the effectiveness of COPD therapy and quality of life. 29 Using both, intensive counselling and pharmacotherapy represents the most effective strategy for treating tobacco addiction in patients with COPD. Every tobacco user should benefit from at least a brief tobacco cessation counselling session at every contact with health care providers. 1
Pulmonary rehabilitation is demonstrated to be the most effective non-pharmacological intervention for improving health status and has become a standard of care for patients with COPD. 30 The benefits of pulmonary rehabilitation are well recognized: it decreases symptoms (dyspnoea and fatigue), improves muscle strength and endurance, resulting in improved exercise tolerance, increased quality of life and emotional functions, and it reduces health care utilization. 31 Education and training in inhalation techniques are major components of the rehabilitation program, and they should be assessed regularly.
Long-term oxygen therapy is indicated in patients with COPD who have severe resting chronic hypoxemia, while long-term non-invasive ventilation should be considered in patients with severe chronic hypercapnia who have been hospitalized for acute respiratory failure. 1
In selected patients, bronchoscopic interventions, lung volume reduction and lung transplant may be considered. 1
Pharmacological treatment
Pharmacological treatment of patients with stable COPD is generally based on long-acting bronchodilators, with further additions determined by the clinical phenotype(s). Possible additional therapies to bronchodilators include ICS (always used in combination with a long-acting bronchodilator), theophylline, mucolytics or antioxidants, and, in selected cases, prophylactic antibiotics.
Bronchodilators
Bronchodilators are drugs that reduce bronchial smooth muscle contraction and dynamic hyperinflation, resulting in the relief of bronchial obstruction, improved FEV1 values, reduced dyspnoea and increased exercise performance and quality of life.1,32–34 The dose-response curves are relatively flat for all bronchodilators, meaning that increasing the dose is not expected to increase the bronchodilator effect.1,35–37 Conversely, adverse events may be dose-related. Increasing the dose of short-acting inhaled bronchodilators, particularly when administered by a nebulizer, may reduce dyspnoea in an acute episode, but may not be helpful as a long-term therapy.1,38 Generally, the use of short-acting inhaled bronchodilators is not recommended on a regular basis.1,8 Bronchodilators are grouped into short acting (3–6 h duration of bronchodilator effect), and long acting (12–24 h duration of bronchodilator effect). Based on their mechanism of action, bronchodilators can be divided into β2-agonists and anticholinergics. Both have short-acting and long-acting durations, and may be used in monotherapy or combined.
β2-agonists stimulate β2-adrenergic receptors and increase cyclic-AMP, which results in smooth muscle relaxation and bronchodilation. Short-acting β2-agonist (SABA) have a bronchodilator effect of 4–6 h, being effective in the rapid control of symptoms and improving exercise tolerance.1,39 They should usually be added to baseline therapy, preferably for as-needed treatment of symptoms, regardless of the level of disease severity. The duration of effect of long-acting β2-agonists (LABA) is 12 h (e.g. salmeterol, formoterol) or 24 h (e.g. indacaterol, olodaterol, vilanterol). LABA have been shown to exert better control of symptoms compared with SABA, to improve lung function and quality of life, and to reduce exacerbation rate.40–42 Generally, β2-agonists are well tolerated. Less than 1% of patients may report tachycardia, palpitation and tremor, however, in susceptible patients, β-agonists may precipitate cardiac rhythm disturbances.40,43,44 Anticholinergics target parasympathetic nervous system receptors in the airways (M1, M2 and M3) and inhibit their function, inducing bronchodilation and reduced bronchial secretions. Short-acting anticholinergics (SAMA; e.g. ipratropium) have a longer duration of bronchodilator effect (up to 8 h) than that of SABA. Furthermore, they provide small benefits over SABA in terms of lung function and health status.
45
Long-acting anticholinergics (LAMA) act for 12 h (e.g. aclidinium) or 24 h (e.g. tiotropium, glycopironium, umeclidinium). LAMA have been shown to reduce symptoms, improve lung function and quality of life, and reduce the need for rescue medication.46–48 The use of inhaled anticholinergics is generally safe, and adverse events rarely occur. Dry mouth, throat irritation, urinary retention, constipation, nausea, or increased ocular pressure, have occasionally been reported.
Recommendations:
If the patient is an infrequent exacerbator, either LAMA or LABA may be recommended, depending on the clinician or patient’s preference.
In the case of frequent exacerbations, treatment with a LAMA is the first recommendation, unless the patient has features of asthma and COPD overlap (ACO), in which case a combination of ICS and LABA may be used as a first option.
Dual bronchodilator therapy
Combining bronchodilators with a different mechanism or duration of action results in a better bronchodilator effect, and is associated with a lower risk of adverse events compared with increasing the dose of a single bronchodilator.1,49 A combination of SABA with SAMA improves symptoms and FEV1 compared with either medication alone. 45 Combining LAMA and LABA has shown an improved effect on respiratory function, symptoms and quality of life versus any single agent.50–53 The LABA/LAMA combination was shown to be more effective than an ICS/LABA combination in the prevention of exacerbations, 54 but another study reported reduced exacerbation rate in the group of patients with frequent exacerbations and high blood eosinophil concentration who used ICS/LABA compared with those treated with LAMA/LABA. 55
Recommendation:
LAMA/LABA combination is recommended in symptomatic patients and/or in patients with repeated exacerbation despite the use of a long acting bronchodilator in monotherapy.
Inhaled corticosteroids
The use of ICS as single long-term treatment of COPD should be avoided. ICS use is associated with higher prevalence of oral candidiasis, skin bruising, hoarse voice, and pneumonia, 56 and possibly with poor control of diabetes, cataract and tuberculosis. 1 In patients with moderate to very severe COPD, ICS combined with LABA is shown to be more effective than placebo, or either component alone, in improving lung function, health status and reducing exacerbation rate.57–60 However, clinical trials have not proved a statistically significant effect on survival.61,62
A positive correlation has been shown between the level of blood eosinophils and the efficacy of therapy that contains ICS in preventing future exacerbations,55,63,64 but a definite threshold of blood eosinophil level that can predict this response could not be established. However, if blood eosinophils are below 100 cells/mm3, the probability of a beneficial preventive response to an ICS-containing regimen is weak. 1 Discontinuation of ICS therapy may be safely accomplished in the majority of patients to decrease the risk associated with long-term side effects, but it may precipitate COPD exacerbation in some patients (patients with ACO or with eosinophilic inflammation).65,66
Recommendation:
ICS combined with LABA may be recommended as first line therapy only in patients who present ACO features, or as an alternative option for patients with eosinophilic inflammation (eosinophils >100 cells/mm3) and recurrent exacerbations despite receiving long acting bronchodilators.
If patients continue to present frequent exacerbations despite dual bronchodilator treatment or ICS/LABA, then triple therapy (LAMA/LABA/ICS) may be administered.
Theophylline
Theophylline exhibits a modest bronchodilator effect in COPD, but the exact mechanism of action remains unknown, 67 however, theophylline may have a positive effect on the function of inspiratory muscles, increase the strength of the diaphragm, reduce air trapping and improve mucociliary clearance. 68 The toxicity of theophylline is dose related, and the most serious problems include atrial and ventricular arrythmias and grand mal convulsions. 1 Other reported adverse effects include nausea, vomiting, diarrhoea, and signs of central nervous system stimulation (e.g. headache or agitation).69,70 For this reason, when theophylline is indicated on a long-term basis, plasma concentrations should be regularly checked, in addition to possible interactions with other medication, such as erythromycin, ciprofloxacin, alopurinol, digitalis, cimetidine or benzodiazepines, among others. 1
Recommendation:
Because of the widespread toxicity and frequent drug interactions, the use of theophylline should be limited. It may be used only as an additive drug to long acting bronchodilators in patients with severe symptoms or unless other long acting bronchodilators are unavailable or unaffordable.
Antibiotics
A meta-analysis of nine randomized controlled trials showed that 6–12 months of macrolides (erythromycin or azithromycin) therapy, with either a daily or intermittent dosing regimen, could significantly decrease the exacerbation rate in patients with COPD. 71 However, no significant difference was reported between the macrolide therapy and control groups in the overall number of hospitalizations or in the all-cause rate of death. Long-term macrolide therapy may increase the rate of adverse events (gastrointestinal reactions, prolongation of QTc interval, hearing impairment) and the incidence of bacterial resistance. 1 Only macrolides are mentioned here due to the fact that many studies have demonstrated their protective effect against COPD exacerbations, while this has not been shown with other antibiotics (e.g. quinolones).71–73 The recommendations here are for protecting against exacerbations, and not for treating exacerbations, for which other antibiotics may be used.
Recommendation:
Potential benefit from long-term use of macrolides may be considered in patients with severe COPD who present recurrent exacerbations with or without hospitalization despite optimal pharmacologic and non-pharmacologic therapy. The recommended regimen includes azithromycin (250 mg/day or 500 mg/day, three times per week) or erythromycin (500 mg two times per day) for one year, repeated during periods with low seasonal temperatures (cold winter and spring).
Mucolytics and antioxidant agents
Mucolytics are well-tolerated oral drugs that decrease sputum viscosity, improve mucociliary clearance and modulate the inflammatory response. Furthermore, given their antioxidant properties, mucolytics, particularly N-acetylcysteine (NAC), are important for the long-term management of patients with COPD, in reducing the rate of exacerbations.74,75 Erdosteine is a more recently developed mucolytic drug with antioxidant and anti-inflammatory properties, that also modulates bacterial adhesiveness, all of which are valuable properties in the treatment or prevention of COPD exacerbations. 76
A recent meta-analysis demonstrated that mucolytic/antioxidant agents significantly reduced the risk of acute exacerbation of COPD, with the following rank of effectiveness: erdosteine > carbocysteine > NAC. 77 Erdosteine and NAC both significantly reduced the duration of exacerbation, while only erdosteine decreased the risk of hospitalization due to COPD exacerbation. Also, it has been shown that erdosteine is associated with increased quality of life, decreased respiratory symptoms and preservation of pulmonary lung function. 78
Recommendation:
In patients with COPD who have frequent exacerbations despite an adequate inhaled therapy, administration of mucolytics and antioxidant agents may be considered.
Vaccination
Vaccination is an important component of treatment in patients with COPD. For example, influenza vaccination has been shown to reduce exacerbations and death in this patient population. 79
Pneumococcal vaccination, PCV13 and PPSV23, reduces the risk of community acquired pneumonia in adults aged over 65 years, this population also including the majority of patients with COPD. 80 The PPSV23 vaccine has been shown to reduce the risk of community acquired pneumonia in patients with COPD aged below 65 years, who have severe airflow limitation (FEV1 <40%) or comorbidities. 81
Recommendation:
Annual influenza vaccination is recommended for all patients with COPD.
Pneumococcal vaccination is recommended for patients with COPD aged over 65 years and for younger individuals with severe airflow limitation and/or significant comorbidities.
Phenotype-specific treatment
In patients presenting with frequent exacerbations (≥2 moderate or >1 with hospitalization), assessment of phenotype should be considered to facilitate the possibility of administering phenotype-specific treatment. This therapy may include, beside a long acting bronchodilator, adding ICS in the case of ACO features or eosinophilic inflammation; or adding mucolytics and antioxidant agents in patients with chronic bronchitis, and/or macrolides in patients who present with recurrent infective exacerbations and associated bronchiectasis. Roflumilast, which may be administered as add-on therapy in the case of severe airflow limitation, chronic bronchitis, and previous hospitalization, is not available in Romania.
Taking into account the medical evidence, Romanian policy and available medication, Romanian pulmonology experts have developed the following scheme (Figure 4). This is a staged scheme for use in the diagnosis and treatment of patients with COPD. The Romanian national guideline complements existing European national guidelines (e.g. France, Finland).

Therapeutic scheme proposed by Romanian experts for treating chronic obstructive pulmonary disease (COPD). ACO, asthma and COPD overlap; BCO, bronchiectasis and COPD overlap; CAT, COPD assessment test; Eo, eosinophil count; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; ICS, inhaled corticosteroids; LAMA, long-acting anticholinergics; LABA, long-acting β2-agonists; mMRC, modified medical research council dyspnoea scale; 6MWT, six-minute walk test; N-ACC, N-acetylcysteine; NIV, non-invasive ventilation; SABA, short-acting β2-agonist.
Conclusions
The present condensed guideline is designed to be accessible and practical for the assessment and management of Romanian patients with COPD. The application of recent guidelines in the management of COPD may enhance the physician’s and patient’s approach in managing this disease. The present guideline complements the ‘bigger picture’ of COPD treatment in Europe.
Expert opinion
Chronic obstructive pulmonary disease is an important disease that affects a very large category of people worldwide. In addition to the known classical factors, such as smoking, the impact of biomass and pollution factors are constantly increasing. Due to the fact that dyspnoea is a slow onset symptom, the vast majority of diagnosed cases are in moderate to severe form. Exacerbations aggravate the disease leading to disease progression and death. The management of comorbidities due to this disease should be carefully monitored. At international level, there are some strategies, such as GOLD, which give certain recommendations for diagnosis and treatment. Following these recommendations, many countries have created their own guidelines that take into account local health policies, as well as the diagnostic and treatment possibilities available. The present work is primarily based on the main pillars of the GOLD strategy and the Spanish COPD treatment guideline, but other national guidelines were also taken into account. In addition, the specificity of the Romanian population, available diagnostic tools, the existing therapy area, adherence to treatment and the opinions of Romanian pulmonologists over the previous few years were taken into consideration for the present paper. Analysis of these strategies and consensus among Romanian pulmonologists has resulted in a treatment scheme that is more adapted to the needs of the Romanian population. This Romanian guideline represents a condensed article that complements existing European guidelines.
