Abstract
We report a case of hyperammonemic encephalopathy due to extrahepatic portosystemic shunts in a noncirrhotic patient. A 79-year-old woman suffered from episodic confusion, disorientation, dysphasia and fluctuating level of consciousness. Electroencephalography (EEG) showed encephalopathic changes and serum levels of ammonia were elevated. Further investigation revealed mesenterorenal and mesenterocaval shunts, which had possibly evolved after pancreatic surgery 5 years ago. After shunt obliteration, the symptoms completely resolved, ammonia levels dropped to the normal range and EEG findings normalized. Clinicians should be aware of this rare but treatable cause of encephalopathy in noncirrhotic patients.
Case description
A 79-year-old woman developed recurrent episodes of confusion, disorientation, dysphasia and fluctuating level of consciousness. Medical history included fully-remitted marginal zone lymphoma (MZL) under previous treatment with rituximab and bendamustine. Furthermore, the patient had undergone curative pylorus-preserving pancreaticoduodenectomy 5 years ago due to intraductal papillary mucinous neoplasm (IPMN).
Over the past months, the patient had been hospitalized several times because of the abovementioned symptoms, including intensive care unit treatment. Nonconvulsive seizures and even nonconvulsive status epilepticus (NCSE) had been considered in the initial differential diagnosis, but despite regular administration of antiseizure drugs including levetiracetam and gabapentin, she continued to have intermittent episodes of confusion, reduced consciousness and/or severe neurocognitive deficits lasting for hours to several days. Treatment with intravenous valproic acid (VPA) even triggered a severe stuporous state, which completely resolved after withdrawal of VPA.
Approximately, 6 months after first onset, the patient was admitted again due to a prolonged episode of confusion. She was disoriented and dysphasic without motor or sensory deficits. Electroencephalography (EEG) showed generalized, irregular delta-theta activity as well as triphasic waves (see Figure 1) and laboratory testing revealed hyperammonemia of 121 μg/dl (reference range 19–55 μg/dl) despite normal liver and kidney function. Examination of cerebrospinal fluid (CSF) and magnetic resonance imaging (MRI) of the brain did not show any relevant abnormalities. Hyperammonemic encephalopathy was suspected and treatment with lactulose and rifaximin was initiated, supported by daily enemas. Serum levels of ammonia decreased (see Figure 2) and clinical improvement was observed under this treatment regimen. Abdominal ultrasound, including liver elastography and abdominal MRI showed only minor signs of chronic liver disease, however, without evidence of cirrhosis or significant portal hypertension. Finally, computed tomography (CT) angiography revealed a left abdominal venous convolute with portosystemic shunts between the inferior mesenteric vein and the left renal vein as well as the inferior vena cava (see Figure 3(a)). The patient underwent percutaneous transhepatic coil embolization and the shunts were successfully obliterated (Figure 3(b)–(g)), resulting in full and sustained clinical recovery with serum ammonia levels remaining within the normal range. EEG before discharge showed a posterior alpha-rhythm with no encephalopathic changes (see Figure 4). No further episodes of confusion and impaired consciousness occurred until a follow-up period of 12 months.
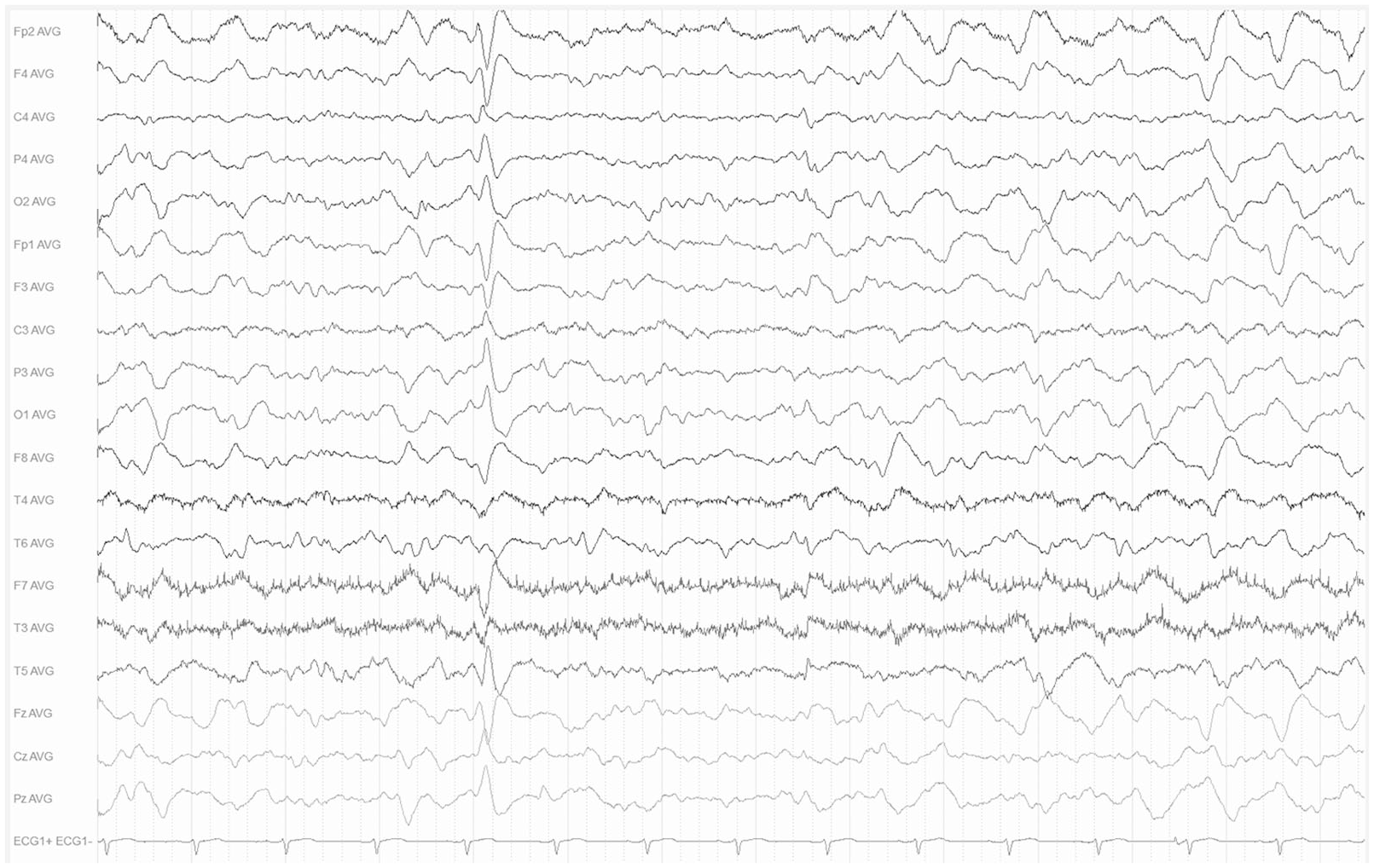
Electroencephalography on admission showing generalized, irregular delta-theta activity as well as triphasic waves in average montage. EEG was recorded by surface electrodes following the 10/20 system using the following parameters: sensitivity 7 µV/mm, time constant 0.3 s, 70 Hz low-pass filter and 50 Hz notch filter.

Serum levels of ammonia over time during hospitalization.

Portocaval shunt from inferior mesenteric vein (IMV) to left renal vein and inferior vena cava (IVC), respectively, on coronally reformatted computed tomography (CT, a) and digital subtraction angiography (b–g). (a) Initial CT imaging showed a vascular convolute left of the aorta (bracket), connecting the IMV (arrowhead) to the left renal vein (arrow). After percutaneous puncture of the right portal vein (b, arrowhead) selective catheterization of the superior mesenteric vein (c, arrowhead) showed main drainage of blood through retrogradely perfused IMV (c, curved arrow). Diagnostic contrast injection into IMV visualized feeding portal vein (d, arrowhead) and draining vessels into left renal vein (e, arrow) and IVC (e, arrowhead). Stationary contrast agent (f, *) after coil embolization of draining vessels with IDC and microplex coils (f, arrowheads) proved efficient occlusion. Final diagnostic contrast injection confirms change of perfusion direction of IMV toward the liver (g, curved arrow).

Electroencephalography after portocaval shunt obliteration showing a posterior alpha rhythm with no encephalopathic changes in average montage. EEG was recorded by surface electrodes following the 10/20 system using the following parameters: sensitivity 7 µV/mm, time constant 0.3 s, 70 Hz low-pass filter and 50 Hz notch filter.
Discussion
Although hyperammonemia is rare in noncirrhotic patients, there are well-known nonhepatic causes of increased ammonia production or decreased elimination that can lead to noncirrhotic hyperammonemic encephalopathy.1–3 These include disorders of the urea cycle, which usually occur in children but might manifest in adult patients as well, hematological diseases, such as multiple myeloma or leukemia, increased muscle catabolism and refeeding syndrome.4–6 Urease-producing microorganisms should also be considered in the differential diagnosis, especially in patients with anatomical alterations of the urinary tract that predispose for bacterial infections.7,8 In addition, a recent case report highlighted cryptococcal infection as a rare cause of hyperammonemia in an immunosuppressed patient, which underlines the need for a broad microbiological workup, especially when patients are at particular risk for infection. 9 Hyperammonemic encephalopathy has also been described as a complication of bariatric surgery.10,11 Furthermore, exposure to various substances has been associated with hyperammonemia, including carbamazepine, ribavirin, methamphetamine and chemotherapeutic agents, such as 5-FU.12–16 While the underlying mechanisms are not well understood in most of these agents, a clearer concept exists for the etiopathogenesis of VPA-induced hyperammonemia. Among many other interactions, VPA disrupts the urea cycle by inhibiting the rate-limiting enzyme (carbamoylphosphate synthetase I) through one of its metabolites (propionic acid).17,18 Notably, intravenous administration of VPA was followed by severe clinical worsening in the reported case, which in retrospect could have been due to a further, VPA-associated increase in serum ammonia levels.
In the present case report, the underlying cause of hyperammonemic encephalopathy was a portosystemic shunt. For this rare but important differential diagnosis of noncirrhotic hyperammonemic encephalopathy, special attention should be given to patients with a history of abdominal surgery or trauma, although congenital types and shunts of uncertain etiology have been described.19,20 Noncirrhotic hyperammonemic encephalopathy due to portosystemic shunting has also been observed in patients with portal vein thrombosis. 21 In the reported case, postoperative shunt formation after pancreaticoduodenectomy was the most likely cause, an etiology that has been described elsewhere. 22 Although therapeutic approaches must address each case individually with regard to the vascular anatomy, interventional treatment seems feasible and effective in most patients. 23
Conclusion
Serum levels of ammonia should be assessed in all patients with unexplained alterations of consciousness or mental status, regardless of liver function, to not miss cases of noncirrhotic hyperammonemic encephalopathy. Investigating causes of this rare but well-described clinical entity can be challenging and requires a holistic workup: Thorough examination of patient history with an emphasis on trauma, surgery and drug exposure should be performed, as well as abdominal imaging and a broad infectious workup. Clinicians should be aware of extrahepatic portosystemic shunts as an unusual but treatable cause of hyperammonemic encephalopathy in noncirrhotic patients.
Footnotes
Consent for publication
The patient provided written, informed consent to publication of images and medical information. The need for ethical board approval was waived in compliance with local regulatory requirements.
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
