Abstract
Immune-mediated necrotizing myopathy (IMNM) is a group of immune-related myopathies characterized by progressive proximal muscle weakness, extremely high serum creatine kinase (CK) levels, and necrotic muscle fibers with a relative lack of inflammation. Treatment of IMNM is challenging, with most cases refractory to high-dose steroids in combination with multiple immunotherapies. The role of rituximab (RTX) for IMNM has been explored in isolated case reports and small series. The aim of this article was to perform a literature review of patients with IMNM treated with RTX and to evaluate RTX efficacy and safety. A total of 34 patients with IMNM were reviewed: 52.9% (18/34) with anti-signal recognition particle (SRP) antibodies and 47.1% (16/34) with anti-3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR) antibodies. Patient age at onset varied from 11 years to 81 years (mean 41 years). The majority of patients presented as a severe proximal muscle weakness and the peak level of CK varied from 3900 IU/L to 56,000 IU/L (mean 18,440 IU/L). Prior to RTX administration, all patients were treated with high-dose steroids and most were treated with multiple immunotherapies. The reason for initiating RTX was that 64.7% (22/34) of patients showed no improvement after previous treatments, and 35.3% (12/34) of patients relapsed when attempting to wean steroids or other immunosuppressive agents. With regard to RTX efficacy, 61.8% (21/34) of patients presented a response to RTX.
Our data may support the use of RTX as an effective treatment strategy against IMNM resistant to steroids and multiple immunotherapies. Meanwhile, RTX as a first-line therapy could be a choice in IMNM, particularly in African Americans with anti-SRP antibody-positive subsets. ANA, antinuclear antibody; CK, creatine kinase; HMGCR, 3-hydroxy-3-methylglutaryl-CoA reductase; IMNM, immune-mediated necrotizing myopathy; MAC, membrane attack complex; MHC-I, major histocompatibility complex-I; RTX, rituximab; SRP, signal recognition particle.
Keywords
Introduction
Immune-mediated necrotizing myopathy (IMNM) is a group of immune-related myopathies characterized by progressive proximal muscle weakness, extremely high serum creatine kinase (CK) levels, and necrotic muscle fibers with a relative lack of inflammation.1–3 It is often associated with autoantibodies recognizing the signal recognition particle (SRP) or 3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR). 2 Many aspects of the pathophysiological mechanisms of IMNM remain unknown. However, increasing evidence suggests that B cells play an important pathogenic role in these diseases.4–7 Statin exposure, positive anti-HMGCR, positive anti-SRP, connective tissue diseases, HIV, and cancer may be the risk factors.2,3,8,9
Compared with other myopathies, IMNM is frequently associated with high risk of permanent muscle damage and disability. 1 Steroids with a combination of immunosuppressive agents remain the mainstay of treatment.2,10 However, most cases are refractory with poor muscle recovery even when treated with high-dose steroids and multiple immunotherapies.2,8 Thus, new therapeutic options with greater efficacy are clearly needed to manage IMNM properly.
Rituximab (RTX), a chimeric anti-CD20 monoclonal antibody, has been largely used for B-cell malignancies, 11 primary central nervous system lymphoma, 12 and some rheumatic immune diseases such as rheumatoid arthritis (RA), 13 systemic lupus erythematosus (SLE), 14 and antisynthetase syndrome. 15 The recommendation that using RTX as one of the treatment strategies for IMNM published by European Neuromuscular Centre (ENMC) 1 is not robust enough since it is derived from only one case series of eight patients. Recently, the role of RTX in IMNM had been increasingly assessed in isolated case reports and small series.7,10,16–25 Therefore, we performed a systematic review to evaluate the efficacy and safety of RTX in the treatment of IMNM.
Methods
Search strategy
We conducted a review by searching PubMed, Web of Science, Embase, and Cochrane articles with information on case reports and case series of patients with IMNM and RTX treatment until May 2020 for English-language sources. We used the following keywords: IMNM, anti-SRP, anti-HMGCR, RTX, and CD20 targeting. References of relevant articles retrieved from the initial search were manually identified and also reviewed. Figure 1 shows the flowchart for paper selection.

Flowchart for study selection.
Inclusion and exclusion criteria
Patients were included if they could be classified as IMNM after following the 119th ENMC or 224th ENMC classification criteria for IMNM.1,26 Subsequently, only cases with complete epidemiological data, clinical manifestations, and therapeutic information were selected. Exclusion criteria included a family history of muscle disease, muscular dystrophy, and a combination of other autoimmune diseases such as RA and SLE. Pediatric patients were considered as those younger than 18 years at disease diagnosis.
Data collection
Data recorded included age, sex, clinical manifestation (muscular involvement and extramuscular involvement), and laboratory data (peak of CK) at disease presentation. Serology results (positive for autoantibodies against SRP or HMGCR), muscle biopsy results, and treatment administered (e.g. prednisolone, immunosuppressive agents, intravenous immunoglobulin, or plasma exchange) before RTX administration was also recorded. Additional collected information included the RTX treatment schedule, RTX adverse effects or death (and cause of death), and clinical response to RTX.
Response criteria
Patients were considered responsive to RTX if: (a) the CK level was reduced to twice or less than the upper limit of normal (adjusted for ethnicity) and/or CK level decreased >50%; (b) the use of steroids could be reduced to ⩽15 mg/day and/or other immunotherapies could be weaned; (c) an improvement of Medical Research Council score ⩾1 grade or an improvement of Manual Muscle Testing 8 score >110% and improved ⩾30 grades, or muscle strength improved significantly.
Statistical analysis
Outcome parameters were summarized using simple descriptive statistics. Fisher’s exact test was used to compare the trends between groups of qualitative variables. All statistical analyses were performed using SPSS 25.0 software, and a two-sided p value of 0.05 or less was considered significant.
Results
Up to May 2020, the search for cases of IMNM treated with RTX yielded 496 articles, of which 484 were excluded as illustrated in Figure 1. Of the 12 articles selected, clinical data were extracted for 34 patients with IMNM meeting the inclusion criteria.7,10,16–25 The general features, clinical manifestations, laboratory data, treatments regimen, and outcomes are shown in Tables 1 and 2.
Clinical, laboratory, and histopathologic data of the 34 patients with IMNM treated with RTX.

ANA, antinuclear antibody; CK, creatine kinase; HMGCR, 3-hydroxy-3-methylglutaryl-CoA reductase; IMNM, immune-mediated necrotizing myopathy; MAC, membrane attack complex; MHC-I, major histocompatibility complex-I; RTX, rituximab; SRP, signal recognition particle.
Clinical course of IMNM patients using RTX.
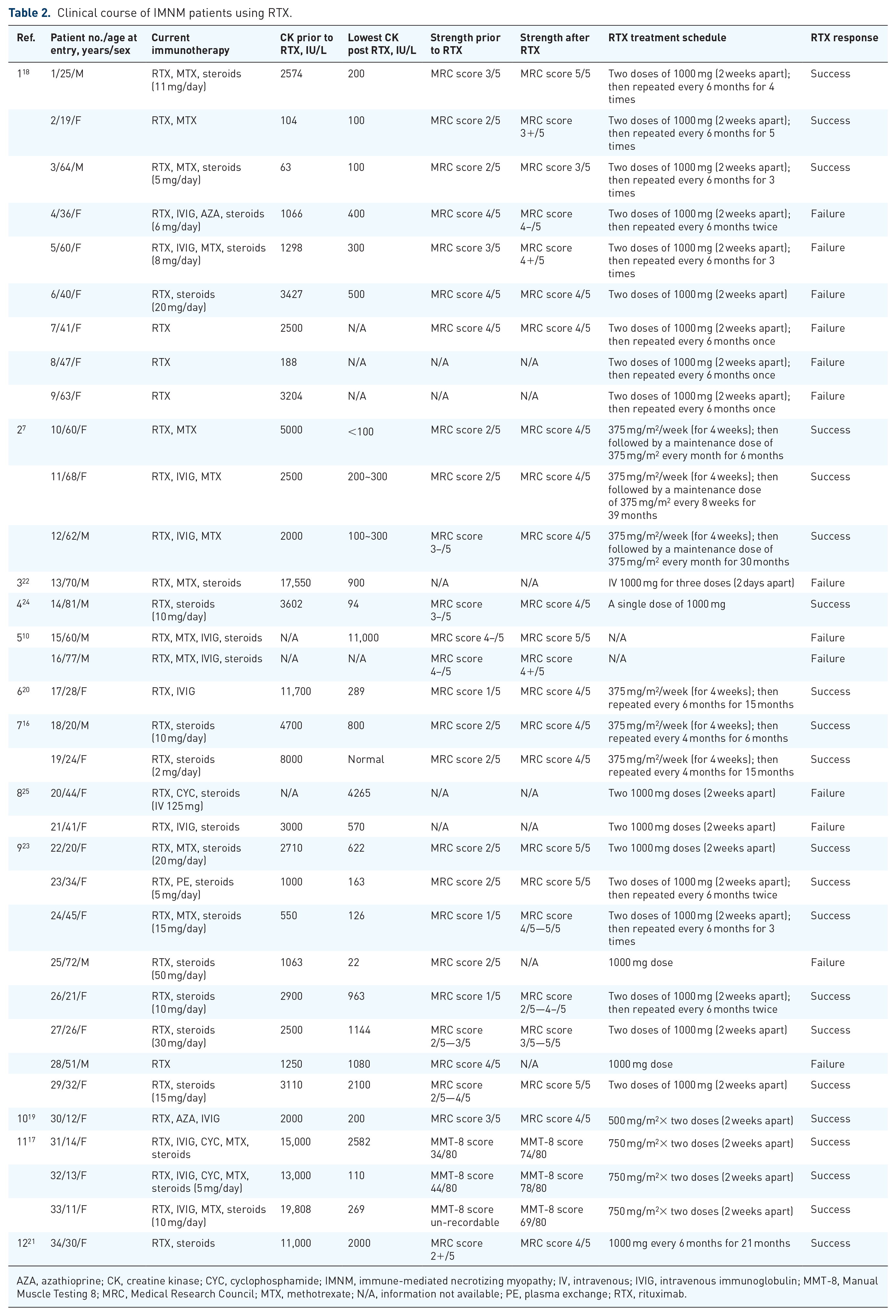
AZA, azathioprine; CK, creatine kinase; CYC, cyclophosphamide; IMNM, immune-mediated necrotizing myopathy; IV, intravenous; IVIG, intravenous immunoglobulin; MMT-8, Manual Muscle Testing 8; MRC, Medical Research Council; MTX, methotrexate; N/A, information not available; PE, plasma exchange; RTX, rituximab.
Around 52.9% (18/34) of patients were positive for anti-SRP antibodies and 47.1% (16/34) of patients were positive for anti-HMGCR antibodies. Around 70.6% (24/34) were women; 29.4% (10/34) were men; 88.2% (30/34) were adults with the median age at onset of 42 years (varied from 19 years to 81 years), and 11.8% (4/34) were pediatrics with the median age at onset of 12 years (varied from 11 years to 14 years). The peak of CK varied from 3900 IU/L to 56,000 IU/L in the adult group, while it varied from 8826 IU/L to 25,937 IU/L in the pediatric group.
A total of 31 patients presented with severe proximal muscle weakness. Meanwhile, it was explicitly stated that 11 patients also had shoulder or hip girdle weakness, 12 patients had dysphagia, and 3 patients had dyspnea. Extramuscular involvement included cutaneous manifestation (n = 5) and Raynaud’s phenomenon (n = 6). Muscle biopsy was performed in 33 cases and 31 patients had evidence of necrotizing myopathy with no or minimal inflammation. Major histocompatibility complex-I-positive staining was seen in 11 cases and 10 of the biopsies were positive for membrane attack complex. Among the 15 patients for whom an electromyogram was performed, all presented an irritable myopathy.
Prior to RTX administration, all patients were treated with high-dose steroids and additional immunotherapies, including methotrexate (29/34, 85.3%), intravenous immunoglobulin (15/34, 44.1%), azathioprine (13/34, 38.2%), plasma exchange (10/34, 29.4%), cyclophosphamide (9/34, 26.5%), mycophenolate mofetil (7/34, 20.6%), and cyclosporine A (1/34, 2.9%). Around 88.2% (30/34) of patients were treated with two or more other immunotherapies and 11.8% (4/34) were treated with one immunotherapeutic agent. Notably, five treatment modalities were used in two patients.
The reason for initiating RTX was that 64.7% (22/34) of patients had no improvement after previous treatments and 35.3% (12/34) of patients relapsed when attempting to wean steroids or other immunosuppressive agents.
With regard to RTX efficacy, 61.8% (21/34) of patients presented a response to RTX. In the anti-HMGCR positive subgroup, 43.8% (7/16) presented a response to RTX. In the anti-SRP positive subgroup, 77.8% (14/18) of patients presented a response to RTX (p = 0.076). Moreover, among the anti-SRP-positive patients, the response rate of African American patients was 90.9% (10/11) and 50% (3/6) in Whites (p = 0.09).
With regard to RTX safety, 23.5% (8/34) of patients developed infection after RTX and two patients died because of respiratory infection (Table 3).
Summary of patients with infections.

CMV, cytomegalovirus; CYC, cyclophosphamide; IVIG, intravenous immunoglobulin; MTX, methotrexate; RTX, rituximab.
RTX regimens were reported in 94.1% (32/34) of cases. Around 76.5% (26/34) of cases were treated with the lymphoma schedule or lymphoma-like schedule (a dose of 375 mg/m2 every week for 4 weeks) or the rheumatology schedule (two doses of 1000 mg given 2 weeks apart). Other schedules (two doses of 750 mg/m2 given 2 weeks apart in 8.8%; two doses of 500 mg given 2 weeks apart in 2.9%; 1000 mg every 6 months in 2.9%; 1000 mg for three doses, 2 days apart in 2.9%) were also reported.
Discussion
We present a systematic review of RTX effects on 34 patients suffering from IMNM (18 with anti-SRP antibodies and 16 with anti-HMGCR antibodies). In our review, we found that patients who were refractory or resistant to high-dose steroids and conventional immunosuppressive drugs were the ones who were prescribed RTX. The good clinical response after RTX use could be demonstrated by the decrease in CK levels, the improvement of muscle strength, the reduction in steroid doses, and the use of fewer additional immunosuppressive drugs. After RTX treatment, 61.8% (21/34) of patients responded with clinical and laboratory evidence of improvement, particularly in African Americans with anti-SRP antibody-positive subsets. Although the difference was not statistically significant (p = 0.076), we observed a trend that showed that patients with anti-SRP antibody-positive
From the safety perspective, 23.5% (8/34) of patients developed infection after RTX therapy and two patients died because of respiratory infection in our study. Interestingly, another systematic review by Hernández-Rodríguez et al., 27 which explored the efficacy and safety of RTX with Immunoglobulin A (IgA) vasculitis (also an immune-related disease), showed the rate of infections after RTX was only 8.6% (3/35). There was an obvious difference in the rate of RTX-associated infection between two studies, although both reported the treatment of immune-related diseases with RTX in a small size sample. When using RTX in the treatment of immune-related diseases, the occurrence of infection correlated poorly with the types of immune-related diseases and correlated more strongly with the depletion of B cells, as the use of RTX may cause CD20-expressing B-cell reduction and confer susceptibility to bacterial, viral, and fungal infections. 28 In addition, the limited number of cases in both studies can decrease statistical power, which may increase the likelihood of biases in the rate of RTX-associated infection. Recently, in a retrospective cohort study comprising a large sample size of 221 patients with immune-related diseases, 29 patients were treated with RTX and results showed that the rate of RTX-associated infection was 17.3%, which was similar to the rate of infections after RTX administration in IMNMs in our review.
It has been acknowledged that B cells play a critical role in immune response and the decrease of B-cell level can increase the risk of infection. 30 In the present study, the RTX regimen used in most patients with IMNM followed the lymphoma schedule or lymphoma-like schedule. In patients with lymphoma, lymphocyte populations increase in number and are monoclonal. 31 The lymphoma schedule of RTX may result in depletion of B cells in patients with IMNM, which could lead to life-threatening infection. Therefore, new therapeutic schedules of RTX administration with better safety profiles are clearly needed to manage properly patients with IMNM. When individualizing the protocol, knowledge of RTX pharmacokinetics and pharmacodynamics is needed: (a) a long elimination half-life of RTX of about 3 weeks; (b) a rapid depletion of circulating CD20+B cells after RTX administration.11,28,32 Furthermore, B-cell depletion induced by RTX and the reconstitution of B cells is heterogeneous in different people.11,28 Thus, we would recommend that a much safer protocol of RTX is maintaining B cells near the lower limit of the normal value instead of depletion in IMNM.
Owing to the high rate of infections after receiving RTX in our study, the infection events are of concern. We looked back and evaluated the possibility of ongoing immunosuppression from the concurrent regimen and found that patients with infections had one of the following characteristics (Table 3): (a) they received RTX in combination with a strong immunosuppressant such as cyclophosphamide; (b) they were treated with a more aggressive approach (1000 mg for three doses, 2 days apart) than the lymphoma schedule. They developed infection due to the depletion of B cells after induction of RTX therapy. In addition, two patients who died due to infection were of older age (>70 years). One patient required a tracheostomy and respiratory support with mechanical ventilation. Therefore, in using RTX to treat IMNM at high risk for infection, such as older age, history of diabetes, or history of cancer, in combination with mechanical ventilation and concomitant steroid therapy, 29 individualization is particularly important and we should be more cautious. Of note, RTX was weaned after infections as all infections occurred during the long dosing intervals (about 6 months). The use of steroids and other immunosuppressive agents included the following three conditions when developing infection: (a) weaned; (b) weaned and followed by intravenous immunoglobulin; (c) replaced by a less aggressive immunosuppressant.
Given IMNM is a rare disease, randomized controlled trials are going to be a challenging prospect. Although current adequate trials are insufficient, our review highlights the earlier use of RTX in IMNM. In our study, patients were given RTX after they were refractory or resistant to high-dose steroids and other immunotherapies for a long period (about 3 years on average). In this period, there might have already been irreversible damage and significant fatty replacement of muscle. 33 Therefore, normalization of strength may not be possible when we use RTX after a long period of failure of other therapeutic strategies. In addition, B cells can repopulate during the long dosing intervals, which can lead to disease recurrence. Based on the points above, the result from our review (61.8% of patients presented a response to RTX) may not be accurate (underestimated) since there are some limitations to evaluating the efficacy of RTX in IMNM.
With regard to the response criteria of IMNM, improvement in CK level alone does not indicate improvement, 34 as patients with immunotherapies, such as plasmapheresis, will also have a decrease in levels of CK but no response for RTX. 35 Meanwhile, the objective improvement of muscle strength is interpreted as a sign of improvement.34,35 Moreover, steroids and immunosuppressive agents are commonly used for the treatment of IMNM, hence the reduction of steroids and/or other immunotherapies, which was also adopted in the incorporated papers,7,10,16–25 can also be a sign of response for RTX. In conclusion, we chose the improvement of objective muscle strength and the reduction of steroids and CK levels as the response criteria.
Taken together, we would suggest use of RTX at the onset and the following three factors should be considered to improve the safety profile and clinical efficacy of RTX in IMNM: (a) B-cell level the next day after RTX infusion and before the second infusion (maintain the number of B cells near the lower limit of the normal value); (b) clinical data regarding changes in muscle strength and function (about 3 months) and CK levels (about 1 month) after RTX treatment; (c) whether the patient is high risk for infection. Overall, the points mentioned above need to be comprehensively evaluated to individualize the protocol for RTX in IMNM.
In addition, the recurrence of the disease may be a result of CD27+ memory B-cell response,36,37 and may therefore be more difficult to resolve than the primary immune response responsible for the induction of IMNM. B-cell clonal expansion and its positive selection may lead to a higher titer of autoreactive antibodies with a higher affinity for muscles and may be more difficult to combat with conventional RTX regimen therapies. Therefore, ambulatory CD27 monitoring is helpful to guide us on the follow-up infusion of RTX efficiently. 38 If there is a reappearance of CD27+ memory B cells throughout the treatment, it may indicate the higher risk of disease recurrence.36,37 Thus, the schedule for RTX, which is more aggressive than the one to maintain the number of B cells near the lower limit of the normal value, will be taken to kill CD27+ memory B cells and reduce the risk of recurrence, but the number of B cells will drop below the lower limit of the normal value and lead to a higher probability of infection. Hence, the antibodies designed to target the CD27 antigen might be a better choice to prevent the recurrence, as they can kill CD27+ memory B cells but not lead to B-cell depletion and reduce the risk of infection.
Conclusion
In summary, treatment of IMNM is notoriously challenging and there is no definitive effective treatment for IMNM to date. From the available data, RTX may be an effective treatment in IMNM resistant to high-dose steroids and multiple immunotherapies, particularly in African Americans with anti-SRP antibody-positive subsets. Better response criteria are needed to evaluate the efficacy of RTX in IMNM when RTX is administered as a first-line therapy. Meanwhile, the safety of RTX should be considered with caution and individualizing the protocol is a reasonable alternative.
Summary
The take-home points of this study are: (a) RTX could be a promising treatment modality for IMNM, particularly in African Americans with anti-SRP antibody-positive subsets; (b) RTX might have the advantage of a better safety profile and higher effectiveness in maintaining the number of B cells near the lower limit of the normal value when used in IMNM; (c) further prospective clinical investigation is needed to assess the exact position and optimal regimen of RTX in IMNM.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Government and School Cooperation Program of Nanchong, Sichuan, China (grant numbers 19SXHZ0148).
