Abstract
Aims:
To retrospectively assess factors associated with John Cunningham virus (JCV) seroconversion in natalizumab-treated patients.
Background:
Natalizumab is highly effective for the treatment of relapsing–remitting multiple sclerosis (RRMS), but its use is complicated by opportunistic JCV infection. This virus can result in progressive multifocal leukoencephalopathy (PML). Serial assessment of JCV serostatus is mandated during natalizumab treatment.
Methods:
Patients treated with natalizumab for RRMS at six tertiary hospitals in Melbourne, Australia (n = 865) and 11 MS treatment centres in Brazil (n = 136) were assessed for change in JCV serostatus, duration of exposure to natalizumab and prior immunosuppression. Sensitivity analyses examined whether sex, age, tertiary centre, prior immunosuppression or number of JCV tests affected time to seroconversion.
Results:
From a cohort of 1001 natalizumab-treated patients, durable positive seroconversion was observed in 83 of 345 initially JCV negative patients (24.1%; 7.3% per year). Conversely, 16 of 165 initially JCV positive patients experienced durable negative seroconversion (9.7%; 3.8% per year). Forty patients (3.9%) had fluctuating serostatus. Time-to-event analysis did not identify a relationship between JCV seroconversion and duration of natalizumab exposure. Prior exposure to immunosuppression was not associated with an increased hazard of positive JCV seroconversion. Male sex was associated with increased JCV seroconversion risk [adjusted hazard ratio 2.09 (95% confidence interval 1.17–3.71) p = 0.012].
Conclusion:
In this large international cohort of natalizumab-treated patients we observed an annual durable positive seroconversion rate of 7.3%. This rate exceeds that noted in registration and post-marketing studies for natalizumab. This rate also greatly exceeds that predicted by epidemiological studies of JCV seroconversion in healthy populations. Taken together, our findings support emerging evidence that natalizumab causes off-target immune changes that may be trophic for JCV seroconversion. In addition, male sex may be associated with increased positive JCV seroconversion.
Introduction
Natalizumab is a humanised monoclonal antibody that targets the cell adhesion molecule α4 integrin, thereby preventing migration of lymphocytes into the central nervous system (CNS). The subsequent amelioration of T-cell mediated inflammatory demyelination makes it highly effective in treating relapsing–remitting forms of multiple sclerosis (MS).1,2 However, by reducing immune surveillance in the CNS, natalizumab raises the risk of opportunistic infections. In particular, it increases the risk of progressive multifocal leukoencephalopathy (PML), 3 a potentially fatal and frequently catastrophic CNS infection caused by the John Cunningham polyomavirus (JCV).
JCV is ubiquitous and human hosts are common. In the absence of immunosuppression, the virus is not believed to be pathogenic. Annual seroconversion rates among healthy people are estimated at approximately 1% per year with 70% seroprevalence by the sixth decade of life.4,5 Accordingly, many patients commencing natalizumab are asymptomatic JCV carriers. It is important to assess JCV status prior to and during therapy with natalizumab as the risk of developing PML can be estimated as a function of JCV status and the duration of decreased CNS immune surveillance secondary to natalizumab exposure. 6
Epidemiological data from registration and post-marketing studies note annual JCV seroconversion rates of approximately 4% in patients treated with natalizumab.7,8 A recent review of natalizumab therapy by the European Medicines Agency identified a similar rate of positive seroconversion. 9 However, considerably higher rates of annualised JCV seroconversion have been reported in several European countries10–12 (cf. Kolasa et al. 13 ). In a large study, Schwab and colleagues recently reported annual rates of positive seroconversion between 8.4% and 10.3% in France and Germany, respectively (Supplemental material Table 1 online). 14 These findings raised questions regarding background rates of infection, potential JCV susceptibility in patients receiving CNS immunosuppression and accurate counselling of patients regarding cumulative PML risk.3,15 Accordingly, we undertook a retrospective longitudinal analysis of JCV seroconversion rates in patients treated with natalizumab at six tertiary hospitals in Melbourne, Australia and 11 MS treatment centres in Florianópolis, Joinville, Jundiaí, Manaus, Rio De Janeiro, Santos, São Paulo and Uberlândia, Brazil.
Methods
Participants
Patients treated with natalizumab for relapsing–remitting MS (RRMS) between March 2007 and February 2017 at the Royal Melbourne Hospital, Box Hill Hospital, the Alfred Hospital, Monash Medical Centre, St Vincent’s Hospital and the Austin Hospital were included in this study. In addition, natalizumab treated patients with RRMS at Universidade Metropolitana de Santos, Santa Casa de Misericórdia do Rio de Janeiro, Hospital de Caridade de Florianópolis, Hospital Beneficência Portuguesa de São Paulo e Hospital Paulistano, Universidade Estácio de Sá (Rio de Janeiro), Universidade da Região de Joinville, Universidade Federal do Rio de Janeiro, Pontifícia Universidade Católica de São Paulo, Universidade Metropolitana de Santos, Hospital Universitário Getúlio Vargas de Manaus, Universidade Federal de Uberlândia and Faculdade de Medicina de Jundiaí were included. All patient records were retrieved from MSBase and the following inclusion criteria were applied: (1) a diagnosis of RRMS; (2) at least one administration of natalizumab; (3) at least two determinations of JCV serostatus during the observation period. We collected data concerning demographics, date of MS diagnosis and duration of natalizumab therapy for all patients. We further recorded JCV status at the commencement of therapy and at the end of the observation period. Where detailed clinical histories were available, note was made of prior immunosuppressive exposure, including with high dose corticosteroids given for 3–5 days. Prior analyses have not identified a relationship between prior MS-specific disease modifying therapy use and JCV seroconversion rates.16–18 We also reviewed individual changes in JCV test values where multiple results were available. Reasons for exclusion from the study are set out in Figure 1.
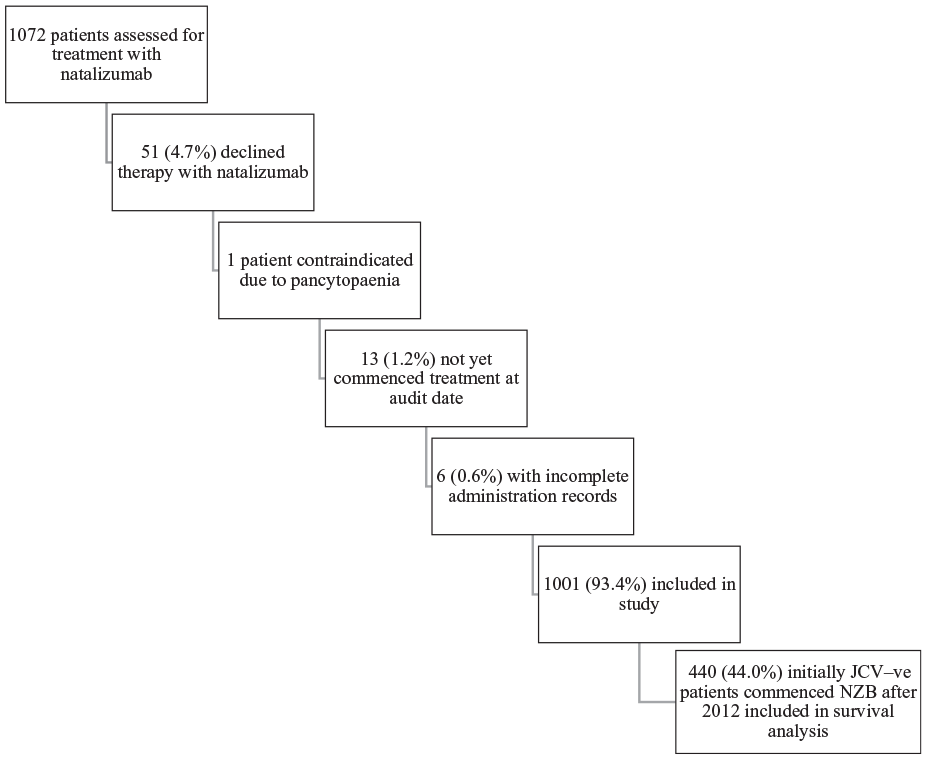
Reasons for exclusion from study cohort.
Ethics statement
This project was conducted as a MSBase Registry sub-study (registered with WHO ICTRP, ID ACTRN12605000455662). The MSBase Registry study was approved by the Melbourne Health Human Research Ethics Committee and by the local ethics committees in all participating centres. Written informed consent was obtained from all enrolled patients participating in the registry in accordance with the Declaration of Helsinki.
Anti-JCV antibody testing
Patients received JCV serology testing at the discretion of the treating physician, typically at six monthly intervals as suggested by natalizumab’s manufacturer. 19 Testing was performed centrally by Unilabs A/S (Copenhagen, Denmark). All samples sent to this laboratory prior to February 2012 were analysed using the STRATIFY JCV assay (first generation two-step JCV antibody enzyme-linked immunosorbent assay (ELISA)), as previously described by Gorelik et al. 6 After that date, the STRATIFY JCV DxSelect assay (second-generation two-step JCV antibody ELISA) was employed, as described by Plavina et al. 7
Statistical analysis
Categorical variables were summarised using frequencies and percentages. Continuous variables were summarised using means and ranges.
Positive seroconversion was defined as a positive JCV result in a patient who was JCV-negative at the time of starting natalizumab treatment. Negative seroconversion was defined as a negative JCV result in a patient who was JCV-positive at the time of starting natalizumab treatment. Seroconversions were considered durable when they reflected a consistent change from positive to negative serostatus (or vice versa) on subsequent tests, or if no subsequent tests were available. Conversion rates were estimated by calculating proportions of patients having conversions and dividing by the average duration of follow-up. Results in the study were based on both the STRATIFY I and STRATIFY-II assays. STRATIFY-I results were reported as either positive or negative. STRATIFY-II results were reported as positive or negative along with an index of sample optical density, considered a corollary of antibody titre. 7 For this analysis, the positive or negative assessment of JCV serostatus provided by UNILABS was adopted in all cases; no alternative assessments of serostatus on the basis of JCV index were undertaken.
Survival analysis was used to model time to positive anti-JCV antibody seroconversion, using the “survival” package in R. 20 Patients who commenced natalizumab prior to 2012 were excluded, as routine JCV serology was not performed before that time. Patients were excluded if a positive seroconversion was reported but no date of conversion was available. Data were censored at either natalizumab cessation or the date of data extraction. Cox proportional hazards regression was used to model associations with sex, age, year of starting treatment and prior corticosteroid exposure. The proportional hazards assumption was assessed through analysis of scaled Schoenfeld residuals using the cox.zph() command. 21 Kaplan–Meier survival curves were plotted, and differences between them assessed using the log-rank test. To test whether the seroconversion rate changes over time, a Weibull distribution was fitted to the survival dataset to determine whether the shape parameter differs significantly from 1.
Results
Longitudinal information was available for 1001 patients treated with natalizumab for RRMS between April 2007 and December 2017. The cohort represented 2829 patient-years of exposure to natalizumab and the mean length of exposure was 2.8 years. At the time of starting natalizumab treatment, patients ranged in age from 16 to 87 years. Female patients represented 74% of the cohort (742/1001).
Detailed clinical history regarding MS treatment prior to natalizumab exposure was available for 626 (63%) patients. Of those, 299 (48%) patients received short course (3–5 days) immunosuppressive therapy with corticosteroids prior to commencing natalizumab. Twelve (1.9%) patients received mitoxantrone prior to commencing natalizumab. Five (0.8%) patients received cladribine, two patients received rituximab and three patients received cyclophosphamide prior to natalizumab treatment (Table 1).
Patient demographics and natalizumab treatment details.

F, female; JCV, John Cunningham polyomavirus; M, male; NZB, natalizumab.
JCV status and seroconversion
Of the 1001 patients in the cohort, 865 were treated in Australia and 136 were treated in Brazil. Of the 865 Australian patients, 594 (68.7%) were JCV negative at commencement of therapy with natalizumab. Of the JCV seronegative patients (n = 594), 108 (18.2%) experienced positive JCV seroconversion. Positive JCV serology at treatment commencement was seen in 271 (31.3%) patients. At the end of the observation period 539 (62.3%) patients were JCV negative and 326 (37.7%) were JCV positive. Multiple JCV index results were available for 443 (51%) patients in the cohort (2195 individual JCV results for these 443 patients, average of 5.0 results per patient).
Among Australian patients with multiple JCV measurements, durable positive seroconversions were seen in 73/309 patients (23.6%) who were initially JCV negative. Given the mean observation period among these 309 patients of 3.31 years (40 months) we calculated an annual rate of durable positive seroconversion of 7.1%. This corresponds to a monthly rate of durable positive seroconversion of 0.6%. Durable negative seroconversions were seen in 16/134 patients (12%) who were initially JCV positive. The durable negative seroconversion was 4.5% annually and 0.4% monthly.
Of the 136 patients treated with natalizumab at centres in Brazil, 76 were JCV-positive at commencement of therapy (56%). Of the remaining 60 patients, 19 experienced positive JCV seroconversion. At the end of the observation period 43 (32%) patients were JCV negative and 93 (68%) were JCV positive. Multiple JCV index results were available for 67 (49%) patients in the cohort (201 individual JCV results for these 67 patients, average of 3.0 results per patient).
Among Brazilian patients with multiple JCV measurements, durable positive seroconversions were seen in 10/39 patients (26%) who were initially JCV negative. Given the mean observation period among these 39 Brazilian patients of 3.1 years (37 months) we calculated an annual rate of durable positive seroconversion of 8.4%. This corresponds to a monthly rate of durable positive seroconversion of 0.7%. No durable negative seroconversions were seen in 28 patients who were initially JCV positive.
Combining the Australian and Brazilian cohorts with multiple JCV measurements, 348 patients with multiple JCV measurements were JCV-negative at the start of the observation period. Among these initially JCV-negative patients, 83 experienced durable positive JCV seroconversion. The average observation period for this group was 3.3 years, resulting in an annual durable positive JCV seroconversion rate of 7.3%. Combining the Australian and Brazilian cohorts, 162 patients with multiple JCV measurements were JCV-positive at the start of the observation period. Among these initially JCV-positive patients, 16 experienced durable negative JCV seroconversion. The average observation period for this group was 2.6 years, resulting in an annual durable negative JCV seroconversion rate of 3.8% (Figure 2). No patients were diagnosed with PML during the observation period.

JCV serostatus of a longitudinally assessed cohort of 510 patients from Australia and Brazil. Period of observation for JCV-negative patients 3.3 years. Period of observation for JCV-positive patients 2.6 years. 348/510 JCV-negative patients reduced to 255/510 JCV-negative patients during observation period. The 162/510 JCV-positive patients increased to 219/510 JCV-positive patients during the observation period. There were eighty-three durable positive seroconversions during observation period and 16 durable negative seroconversions during the observation period.
No patient with prior cyclophosphamide, rituximab or mitoxantrone exposure experienced a seroconversion event. All remained anti-JCV antibody negative throughout the period of observation. One patient who received cladribine prior to natalizumab therapy experienced a durable positive seroconversion.
Survival analysis
Modelling time to positive seroconversion after starting natalizumab, we analysed data from 440 patients who started natalizumab treatment in 2012 or later and were JCV-negative at the time of starting treatment. This represented 440 patients with a total of 801 years of follow-up. There was considerable heterogeneity in seroconversion rates between sites (Table 2). Furthermore, there was evidence that the proportional hazards assumption does not hold across sites (global p = 0.006). Hence, all subsequent Cox survival analyses were stratified by site.
JCV serostatus by centre.

Av., average; f/up, follow-up; JCV, John Cunningham polyomavirus; NZB, natalizumab; pos., positive; RMH, Royal Melbourne Hospital.
Table 3 demonstrates the results of survival analyses examining associations between seroconversion and sex, age and year of starting natalizumab treatment. There were no associations with age or year of starting treatment. However, male sex was associated with a greater risk of JCV seroconversion in Australian and Brazilian cohorts [unadjusted hazard ratio (HR) 2.05, 95% confidence interval (CI) (1.17–3.60), p = 0.01] (Figure 3).
Results of Cox proportional hazards survival analyses looking at time to positive John Cunningham polyomavirus (JCV) seroconversion after starting natalizumab treatment, for 440 multiple sclerosis patients who were JCV negative at the time of starting treatment. Analyses stratified by site.
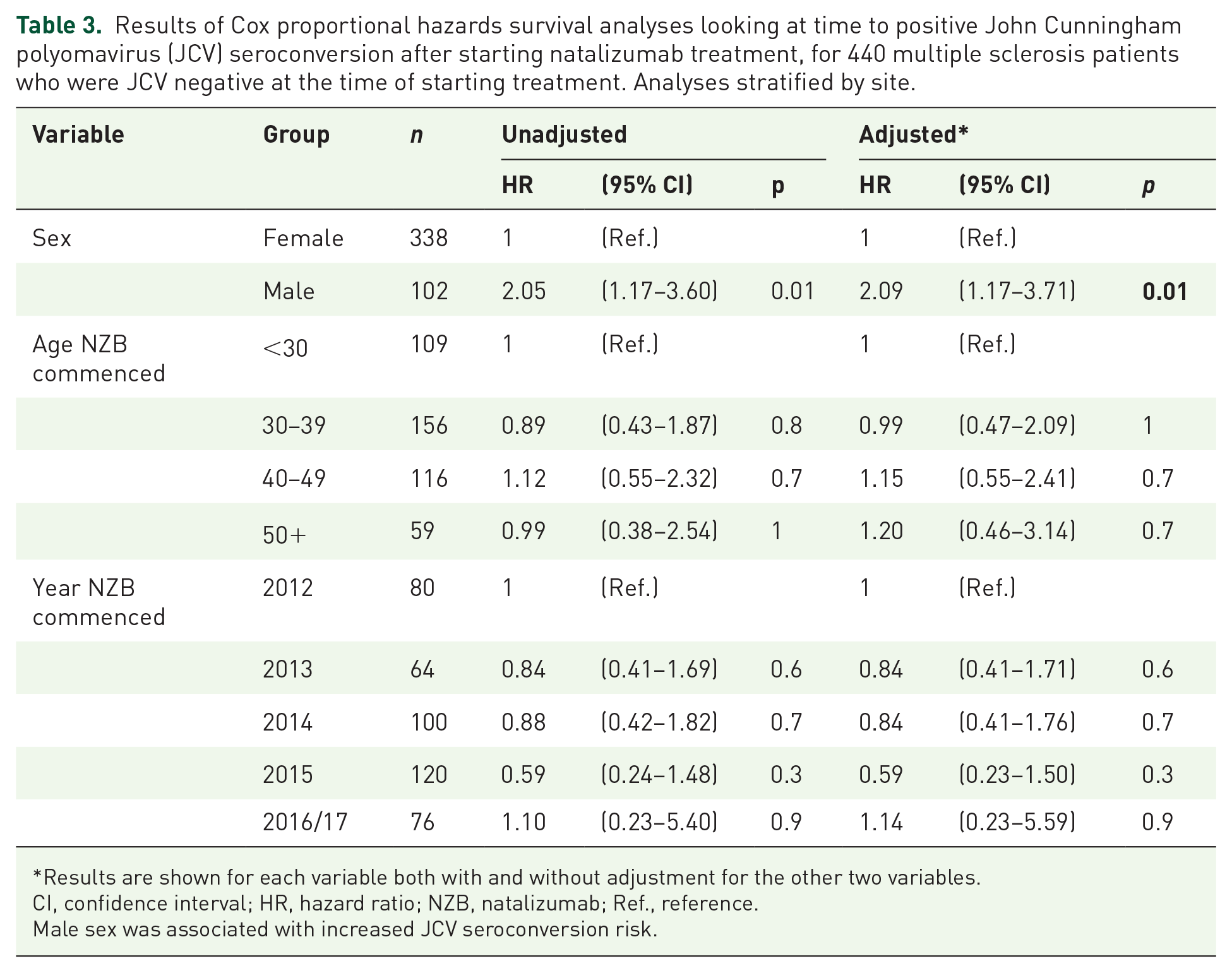
Results are shown for each variable both with and without adjustment for the other two variables.
CI, confidence interval; HR, hazard ratio; NZB, natalizumab; Ref., reference.
Male sex was associated with increased JCV seroconversion risk.

Kaplan–Meier plots showing probabilities of positive JCV conversion for Australia and Brazil, stratified by sex.
There was information for 369 of the 440 patients who received corticosteroid therapy prior to the commencement of natalizumab. In a survival analysis adjusted for sex, there was no increased hazard of positive JCV seroconversion for the 245/369 patients (66%) who received prior corticosteroids [HR 1.08, 95% CI (0.50–2.34), p = 0.8].
There was no evidence that the seroconversion rate varied by time after commencement of natalizumab (p = 0.2).
Variable seroconversion status
When experiencing positive JCV seroconversion, most patients returned a strongly positive test result (average index 1.6). However, in keeping with other studies,14,22,23 we noted a subgroup of patients with variable seroconversion status where results fluctuated between positive and negative serostatus. Among 510 patients with multiple JCV index results, 45 patients (8.8%) returned variable positive and negative anti-JCV Ab status. As set out in Figure 4, of those 45 patients, 26 (58%) experienced transient positive anti-JCV Ab status before returning to a negative baseline. Ten patients (22%) experienced variable anti-JCV Ab status from a negative baseline before settling on positive serostatus. Five patients (11%) experienced transient negative anti-JCV Ab status before returning to a positive baseline. Four patients (9%) experienced variable anti-JCV Ab status from a positive baseline before settling on negative serostatus.

Patients with variable JCV serostatus.
Fifteen patients (33%) experienced multiple positive and negative serostatus results during the period of observation. Among patients with variable serostatus, only one experienced transition from variable negative status to strongly positive (index >0.9) serostatus. No patient with variable positive serostatus experienced a transition to a strongly negative value (index <0.4).
STRATIFY-II sub-group analysis
A sub-analysis was performed using results from the STRATIFY-II assay only. There were 738 patients in this sub-cohort, of whom 608 were treated in Australia and 130 were treated in Brazil.
Among Australian and Brazilian patients with multiple JCV measurements, 246 patients with multiple JCV measurements were JCV negative at the start of the observation period. Among these initially JCV-negative patients, 48 experienced durable positive JCV seroconversion. The average observation period for this group was 2.3 years, resulting in an annual durable positive JCV seroconversion rate of 8.5%. Combining the Australian and Brazilian cohorts, 131 patients with multiple JCV measurements were JCV positive at the start of the observation period. Among these initially JCV-positive patients, 12 experienced durable negative JCV seroconversion. The average observation period for this group was 2.1 years, resulting in an annual durable negative JCV seroconversion rate of 4.4%.
Discussion
We report longitudinal JCV seroconversion data from a large international cohort of natalizumab-treated patients. In particular, we identified an annualised durable positive seroconversion rate of 7.3% from the combined cohorts of Australian and Brazilian patients. We also found that durable positive JCV seroconversion was more common in male patients receiving natalizumab therapy. An analysis of immunomodulation prior to commencement of natalizumab did not identify a correlation with later seroconversion for the JC virus.
The reported annual positive seroconversion rate from this study of 7.3% is notably higher than that reported in registration and post-marketing studies for natalizumab.6,7 This rate of positive seroconversion is also higher than recent European Medicines Agency assessments 9 and is more in keeping with elevated rates of JCV seroconversion reported by Schwab and colleagues in large French and German cohorts of natalizumab treated patients (Supplemental Table 1 online). 14 While some of the variability in reported seroconversion rates may reflect geographical differences in JC virus genomes or differing reported observation periods, all of these studies confirm that the rate of JCV seroconversion in patients receiving natalizumab is higher than expected background rates of seroconversion, typically assumed at 1% per year.4,5
It has been established that natalizumab administration causes changes in T- and B-cell subset composition in peripheral24,25 and CNS compartments. 26 In particular, natalizumab drives immature CD34+ and pre-B-cells into circulation. 27 More recent work by Meira et al. 28 suggests a causal link between changes in mRNA expression in B-cells and peripheral blood mononuclear cells caused by natalizumab administration and the activation of regulatory regions of JCV DNA that may facilitate the emergence of viral variants with greater pathogenicity. Importantly, the implicated transcription factors identified in that study decreased with cessation of natalizumab.
Accordingly, changes in gene expression induced by natalizumab may potentiate JCV seroconversion in susceptible patients and herald nascent JCV infection. While it may appear plausible that trophic changes induced by natalizumab in susceptible hosts that are permissive of JCV seroconversion are more likely to occur soon after commencement of therapy, we did not note any weighting of JCV seroconversion events towards commencement of therapy.
Our finding that male patients are more susceptible to JCV seroconversion is intriguing and is in keeping with analyses from Germany, 16 Canada 29 and the Middle East. 18 While the result must be interpreted with caution in our data set given the heavy contribution of two centres to the outcome, there is an increasingly diverse array of literature suggesting fundamental differences in the performance of male and female immune systems.30,31 This is reflected not only in human subjects32,33 but also in animal models of central demyelination where the administration of female sex hormones alters the outcomes of experimental autoimmune encephalomyelitis (EAE) and cuprizone induced demyelination.34,35
Prior studies have identified immunosuppression as a risk factor for the development of PML, along with positive anti-JCV antibody serology and duration of exposure to natalizumab. 36 We also note prior research examining the use of natalizumab in the treatment of Crohn’s Disease that found an association between positive JCV seroconversion and thiopurine exposure. 37 In our study, we did not identify an association between prior immunosuppression and JCV seroconversion. Prior studies have not identified a relationship between serological concentrations of natalizumab and associated JCV seroconversion rates. 38 Serological data was not available for patients in the current cohort.
As in prior studies, we found a substantial proportion of patients with variable anti-JCV antibody status. All patients with fluctuating results remained close to the pre-determined threshold for JCV positivity by index calculation. These patients continue to present a clinical challenge. It is difficult to accurately appraise the cumulative risk of developing PML in this context. However, we note that the STRATIFY JCV DxSelect second generation assay offers a high degree of diagnostic precision 39 and accordingly we counsel a conservative approach to the assessment of PML risk in these patients.
Our study is subject to several limitations. This longitudinal analysis is retrospective in nature. In addition, detailed pre-natalizumab treatment history and multiple antibody index results were available for different subsets of patients. Lower JCV antibody positivity at the commencement of therapy compared with some European cohorts may limit the applicability of these findings to a broader context. The data sources used in this analysis report duration of therapy, rather than number of doses of natalizumab. It is possible that interruptions in consistent natalizumab administration (for example, due to intercurrent pregnancy) could influence seroconversion rates. It was not possible to interrogate that potential effect with the data available. Finally, given the long period of observation, the data presented here reflect results from different JCV assays. We note, however, a high degree of positive and negative correlation between first and second generation JCV assays. 40 Our study benefits from a large and ethnically diverse patient population representing over 2800 patient-years of natalizumab exposure.
Conclusion
Natalizumab remains one of the most effective medications for the treatment of the inflammatory stage of MS. However, in this large international cohort of natalizumab-treated patients we observed an annual durable positive seroconversion rate of 7.3%. This rate exceeds that noted in registration and post-marketing studies for natalizumab and is consistent with increased rates of seroconversion reported recently by European centres. This rate also greatly exceeds that predicted by epidemiological studies of JCV seroconversion in healthy populations. This finding supports the contention that this otherwise highly effective agent causes important off-target changes in the immune system that may be trophic for JCV seroconversion. This may limit natalizumab’s ability to provide long-term control of the inflammatory aspects of RRMS.
Supplemental Material
sj-docx-1-tan-10.1177_1756286421998915 – Supplemental material for High rates of JCV seroconversion in a large international cohort of natalizumab-treated patients
Supplemental material, sj-docx-1-tan-10.1177_1756286421998915 for High rates of JCV seroconversion in a large international cohort of natalizumab-treated patients by Christopher M. Dwyer, Vilija G. Jokubaitis, Jim Stankovich, Josephine Baker, Jodi Haartsen, Helmut Butzkueven, Adriana Cartwright, Neil Shuey, Yara Dadalti Fragoso, Louise Rath, Olga Skibina, Kylie Fryer, Ernest Butler, Jennifer Coleman, Jennifer MacIntrye, Richard Macdonell and Anneke van der Walt in Therapeutic Advances in Neurological Disorders
Footnotes
Availability of data
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to restrictions regarding the presence of information that could compromise the privacy of research participants.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
