Abstract
Background:
Diroximel fumarate (DRF) is a novel oral fumarate approved for relapsing forms of multiple sclerosis (MS). DRF demonstrated significantly improved gastrointestinal (GI) tolerability versus dimethyl fumarate (DMF) with fewer days of Individual Gastrointestinal Symptom and Impact Scale (IGISIS) scores ⩾2, GI adverse events (AEs), and treatment discontinuations due to GI AEs. Our aim was to evaluate the impact of GI tolerability events on quality of life (QoL) for patients with relapsing–remitting MS who received DRF or DMF in EVOLVE-MS-2.
Methods:
A post hoc analysis was conducted in patients who were enrolled in the randomized, blinded, 5-week, EVOLVE-MS-2 [ClinicalTrials.gov identifier: NCT03093324] study of DRF versus DMF. Patients completed daily IGISIS and Global GISIS (GGISIS) eDiary questionnaires to assess GI symptom intensity and interference with daily activities and work.
Results:
In total, 504 patients (DRF, n = 253; DMF, n = 251) received study drug and 502 (DRF, n = 253; DMF, n = 249) completed at least one post-baseline questionnaire. With DRF, GI symptoms were less likely to interfere ‘quite a bit’ or ‘extremely’ with regular daily activities [IGISIS: DRF, 9.5% (24/253) versus DMF, 28.9% (72/249)] or work productivity [GGISIS: DRF, 6.1% (10/165) versus DMF, 11.3% (18/159)]. DRF-treated patients had fewer days with ⩾1 h of missed work (DRF, 43 days, n = 20 versus DMF, 88 days, n = 26). DMF-treated patients reported highest GI symptom severity and missed work at week 2–3 shortly after completing the titration period, which coincided with the majority of GI-related treatment discontinuations [58.3% (7/12)]. GI tolerability AEs [DRF, 34.8% (88/253); DMF, 48.2% (121/251)], concomitant symptomatic medication use [DRF, 19.3% (17/88) versus DMF, 30.6% (37/121)], and GI-related discontinuations (DRF, 0.8% versus DMF, 4.8%) were lower with DRF versus DMF.
Conclusions:
The improved GI tolerability with DRF translated into clinically meaningful benefits to QoL, as patients experienced less impact on daily life and work and required less concomitant symptomatic medication use.
Trial registration:
[ClinicalTrials.gov identifier: NCT03093324]
Keywords

Introduction
The treatment landscape for patients with multiple sclerosis (MS) has rapidly evolved over the last decade. Patients and providers may now choose from nearly 20 available disease-modifying therapies (DMTs), including injectable, oral, and infused medications, each with varying efficacy, safety, and tolerability profiles.1–3 Patient-centric factors must be considered when selecting a treatment regimen, including the capacity for the treatment to be incorporated into the patient’s lifestyle with minimal treatment burden or interference with daily activities, in order to maintain or improve their quality of life (QoL). 4 Well-tolerated treatments are important for optimizing patient adherence and persistence on therapy, and therefore contributing to achieving maximum effectiveness.5–7
Diroximel fumarate (DRF) is a novel oral fumarate approved in the United States for patients with relapsing forms of MS. 8 Interim results from EVOLVE-MS-1, a long-term, open-label, safety, tolerability, and efficacy study of DRF, show significant reductions in annualized relapse rate and gadolinium-enhanced lesion counts, and an apparent low rate [<1% (5/696)] of treatment discontinuations due to gastrointestinal (GI) adverse events (AEs), suggesting disease control similar to that with dimethyl fumarate (DMF), but with a low treatment burden. 9
GI AEs are associated with DMF and typically emerge early in treatment, 10 potentially preventing patients and clinicians from achieving their treatment goals. The incidence of GI AEs with DMF ranges from 40% in the pivotal phase III clinical studies up to 88% in a real-world study in which patients self-assessed and recorded their GI symptoms.11,12 These GI AEs can lead to dose reductions, interruptions, or premature discontinuations, as has been observed in clinical and real-world studies with DMF in which GI AE-related dose interruption or reduction occurred in 7–21% of patients, and up to 19% of patients discontinued treatment due to GI AEs.11,13 These dosing interruptions or stoppages may prevent patients from maximally benefiting from the therapy due to reduced exposure to the treatment. From the patient perspective, GI AEs can negatively impact satisfaction with treatment. 14 Although the risk of GI AEs can be mitigated by taking DMF with food (specifically, high-fat food), 15 some patients may find this strategy challenging, as it may require a change to their daily routine or meal habits, or not be a sustainable, healthy lifestyle habit in the long term.
DRF has a distinct chemical structure from DMF that is hypothesized to reduce GI irritation through previously described mechanisms. 16 DRF significantly improved GI tolerability compared with DMF, demonstrating a reduction in number of days that patients reported a score of ⩾2 [1.4 (95% confidence interval (CI) 1.1–1.9) days versus 2.6 (95% CI 2.0–3.3) days, respectively; 46% reduction; p = 0.0003] on the Individual Gastrointestinal Symptom and Impact Scale (IGISIS), lower incidence rate of GI AEs, and fewer treatment discontinuations due to GI AEs in the primary analysis of EVOLVE-MS-2. 17 Here, we report results of a post hoc analysis of EVOLVE-MS-2, which evaluates patient-centric measures of improved GI tolerability with DRF versus DMF to explore the impact of GI events on patient QoL.
Methods
Study design and patients
EVOLVE-MS-2 [ClinicalTrials.gov identifier: NCT03093324] was a phase III, randomized, double-blind, head-to-head study to compare GI tolerability of DRF versus DMF in adults with relapsing–remitting MS (RRMS). The study was conducted at 70 sites in the United States and Europe. The adaptive study design, patient population, and study endpoints have been reported previously. 17 In brief, the study consisted of a ⩽4-week screening period, a 5-week double-blind treatment period, and a 2-week follow-up period. The 5-week treatment period was selected based on previous studies of DMF and DRF demonstrating that the majority of GI AEs occur early in treatment, typically within 1 month after DMF or DRF initiation.9,11,12 Eligible patients were aged 18–65 years, with a confirmed diagnosis of RRMS, and neurologically stable, with no evidence of relapse within 30 days of randomization. Patients with a history of GI surgery (except appendectomy that occurred >6 months before screening), clinically significant recurring or active GI symptoms within 3 months of screening, chronic use (⩾7 days) of medical therapy to treat any GI symptoms within 1 month of screening, or prior fumarate use were not eligible.
Following randomization, patients received orally administered DRF (231 mg twice daily in week 1, 462 mg twice daily in weeks 2–5) or DMF (120 mg twice daily in week 1, 240 mg twice daily in weeks 2–5) at their approved dosing regimens over the 5-week double-blind treatment period [Figure 1(a)]. DMF was over-encapsulated to create a blinded study drug and all patients received four capsules of study medication per day [two capsules twice daily; Figure 1(b)] to maintain study blinding. Dose reductions were not permitted; patients who did not tolerate the study drug during the initial 1-week titration period or after the dose titration period were discontinued from the study.

EVOLVE-MS-2 (a) study design and (b) dosing regimen.
EVOLVE-MS-2 was conducted in accordance with local and central ethics committees and the International Conference on Harmonisation Guidelines for Good Clinical Practice. All patients provided written informed consent. CONSORT guidelines were followed for data reporting [Supplemental Figure 1(a) and (b)].
Assessments
IGISIS and Global Gastrointestinal Symptom and Impact Scale
IGISIS and Global Gastrointestinal Symptom and Impact Scale (GGISIS) are novel GI symptom scales that were administered as eDiary questionnaires, completed by patients once (GGISIS) and twice (IGISIS) per day. The IGISIS and GGISIS scales were developed for the EVOLVE-MS-2 study and used in the evaluation of primary and secondary endpoints. The development of these scales has been described previously. 17 In the IGISIS questionnaire, patients rated the intensity and duration of each of the five GI symptoms (nausea, vomiting, upper abdominal pain, lower abdominal pain, and diarrhea) on a scale of 0 (‘did not have’) to 10 (‘extreme’), and additionally assessed interference of GI symptoms with daily activities on a 5-point Likert scale [not at all < slightly < moderately < quite a bit < extremely; Supplemental Figure 2(a)]. In the GGISIS questionnaire, patients rated the overall intensity of any of the five GI symptoms (0–10 scale) and the level of interference of GI symptoms with daily activities and work productivity (5-point Likert scale) over the previous 24 h [Supplemental Figure 2(b)].
To assess the impact of GI symptoms on patient QoL, analyses included incidence of IGISIS scores ⩾1 over time, representing: the occurrence of any GI event, regardless of severity; incidence of consecutive days with GI symptoms; interference of GI symptoms with daily activities, collected using the IGISIS and GGISIS questionnaires; and impact of GI symptoms on work productivity and missed work hours, collected using the GGISIS questionnaire.
An additional analysis was conducted to determine whether an IGISIS score ⩾2, utilized in the primary endpoint of the study, was an appropriate threshold for comparing GI tolerability and detecting clinically meaningful QoL improvements in this study.
Patient-reported outcomes
EuroQoL Group Health Outcome Measure Five-Dimensions Five-Level version (EQ-5D-5L) 18 and the 12-Item Short Form Health Survey version 2 (SF-12) 19 were used to assess patient QoL. EQ-5D-5L is based on a descriptive system that defines health in terms of five dimensions: mobility; self-care; usual activities; pain/discomfort; and anxiety/depression. Each dimension has five response options corresponding with the level of severity (no problems < slight problems < moderate problems < severe problems < extreme problems). The SF-12 assesses general health status across eight health concepts that represent components of physical and mental health. Scores are evaluated in relation to those found in the general United States population [mean (standard deviation (SD)), 50 (10)]. Patients completed the EQ-5D-5L and SF-12 questionnaire at study drug initiation and again at the end of treatment.
Safety
Investigators monitored and collected AEs and performed safety assessments at each study visit. GI AEs were defined according to the Medical Dictionary for Regulatory Activities (MedDRA) system organ class for GI disorders. GI tolerability AEs were defined as those MedDRA preferred terms in the level 2 subordinate standardized MedDRA queries ‘GI nonspecific inflammations,’ ‘GI nonspecific symptoms and therapeutic procedures,’ or ‘GI nonspecific dysfunction’ (under the level 1 Standardized MedDRA Query ‘gastrointestinal nonspecific inflammation and dysfunctional conditions’). Use of concomitant symptomatic medication to treat GI AEs, which indicated GI AE severity and burden due to added health care resource utilization, and treatment discontinuation due to GI AEs were recorded by the investigator and served as markers for impact on QoL.
Statistical analyses
Patient-assessed GI tolerability analyses were performed in patients who completed at least one post-baseline GI tolerability assessment. All GI tolerability analyses based on IGISIS and GGISIS were summarized using descriptive statistics. A receiver operating curve analysis was used to choose a score of ⩾2 as an optimal cutoff value for IGISIS, which provided sufficient sensitivity and specificity to detect moderate or severe GI AEs (nausea, vomiting, diarrhea, upper and lower abdominal pain). Change from baseline to end of treatment for the SF-12 and EQ-5D-5L was summarized by treatment group. Safety analyses were performed in all patients who received at least one dose of study drug and data were summarized using descriptive statistics.
Results
Patients
A total of 506 patients enrolled in EVOLVE-MS-2 and 504 received at least one dose of study drug; 253 patients received DRF and 251 patients received DMF. Patient baseline demographics and disease characteristics in the two treatment groups were generally well balanced (Table 1).
Baseline demographics and disease characteristics in EVOLVE-MS-2.

DMF, dimethyl fumarate; DMT, disease-modifying therapy; DRF, diroximel fumarate; EDSS, Expanded Disability Status Scale; SD, standard deviation. Table adapted from “Diroximel Fumarate Demonstrates an Improved Gastrointestinal Tolerability Profile Compared With Dimethyl Fumarate in Patients With Relapsing-Remitting Multiple Sclerosis: Results From the Randomized, Double-Blind, Phase III EVOLVE-MS-2 Study” by Naismith RT et al. 17 is licensed under CC BY-NC-4.0.
Overall, 96.8% (245/253) of patients in the DRF group and 92.8% (233/251) of patients in the DMF group completed the study, and 3.2% (8/253) and 7.2% (18/251), respectively, discontinued treatment during the study period. Median (range) exposure to DRF and DMF was 36.0 (3–45) days and 35.0 (1–42) days, respectively. Compared with DMF, DRF-treated patients were less likely to discontinue treatment due to AEs (DRF, 1.6% versus DMF, 5.6%) or GI AEs (DRF, 0.8% versus DMF, 4.8%). Upon completion of EVOLVE-MS-2, 94.5% (239/253) of DRF-treated patients and 89.6% (225/251) of DMF-treated patients rolled over into the long-term, open-label EVOLVE-MS-1 study.
A total of 502 patients (DRF, n = 253; DMF, n = 249) completed at least one post-baseline IGISIS or GGISIS questionnaire and were included in the assessments of symptom intensity and interference with daily activity. Mean (SD) number of IGISIS/GGISIS exposure days (days on which the scales were used) for DRF and DMF was 35.2 (4.2) and 34.2 (5.9) days for IGISIS, and 34.2 (4.1) and 33.4 (5.1) days for GGISIS. Overall, 165 patients in the DRF group and 159 patients in the DMF group responded to the GGISIS question on whether GI symptoms affected work productivity. A total of 133 patients in both the DRF group and DMF group reported being employed during the study.
GI tolerability: IGISIS symptom intensity assessments
An IGISIS symptom intensity score of ⩾2 was utilized in the analysis of the primary endpoint. 17 Fewer DRF-treated patients reported an IGISIS score of ⩾2 at any time in the treatment period compared with DMF-treated patients [43.1% (109/253) versus 51.4% (128/249)]. Among those with a worst IGISIS score of ⩾2, DRF-treated patients were less likely than DMF-treated patients to have GI symptoms that interfered with daily activities ‘quite a bit’ or ‘extremely’ [DRF, 16.5% (18/109); DMF, 18.8% (24/128)], led to missed work [DRF, 16.5% (18/109); DMF, 18.8% (24/128)], or resulted in concomitant symptomatic medication use for GI AEs [DRF, 13.8% (15/109); DMF, 24.2% (31/128)]. In addition, DRF-treated patients were less likely to have two or more consecutive days with an IGISIS score ⩾2 compared with DMF-treated patients [17.4% (44/253) versus 29.3% (73/249)]. The mean (SD) duration of these consecutive day periods was also lower for DRF [4.8 (4.1) days] than DMF [6.1 (6.3) days]. Among patients with an IGISIS score of ⩾2, 50.5% (55/109) and 71.9% (92/128) of DRF- and DMF-treated patients, respectively, had GI tolerability AEs; 15.6% (17/109) and 32.0% (41/128), respectively, had any moderate or severe GI tolerability AEs; and 12.8% (14/109) and 31.3% (40/128), respectively, had moderate or severe GI AEs assessed in IGISIS (nausea, vomiting, diarrhea, and upper and lower abdominal pain).
In an analysis using all IGISIS scores ⩾1, representing the occurrence of any event, the incidence and severity of GI events with a score of ⩾1 gradually declined over the 5-week treatment period for DRF-treated patients, whereas the incidence and severity of symptoms peaked during week 2–3 before declining for DMF-treated patients [Figure 2(a) and (b)]. Week 2–3, which occurs after the first week of full-dose treatment with DRF or DMF, was also associated with the majority of DMF discontinuations due to GI AEs [58.3% (7/12)].

Summary of severity of patient-assessed GI symptoms with (a) DRF and (b) DMF.
Interference of GI events with daily activities and work
Patients treated with DRF reported GI symptoms as less likely to interfere with regular daily activities and work compared with DMF-treated patients. Fewer DRF-treated patients characterized their GI events as interfering with regular daily activities ‘quite a bit’ and ‘extremely’ versus DMF-treated patients: nausea (2.4% versus 6.8%), vomiting (1.2% versus 5.6%), upper abdominal pain (1.2% versus 6.8%), lower abdominal pain (1.2% versus 3.2%), diarrhea (3.6% versus 6.4%), any GI event/IGISIS (9.5% versus 28.9%), and any GI event/GGISIS (7.9% versus 10.8%; Figure 3).

Worst interference of GI symptoms with regular daily activities ‘quite a bit’ or ‘extremely’ over the past 12 (IGISIS) or 24 h (GGISIS).
Among respondents to the GGISIS questionnaire of whether GI symptoms affected work productivity (DRF, n = 165; DMF, n = 159), DRF-treated patients were less likely to report that GI symptoms affected work productivity ‘quite a bit’ and ‘extremely’ compared with DMF-treated patients [6.1% (10/165) versus 11.3% (18/159)]. A greater number of DRF-treated patients compared with DMF-treated patients reported GI symptoms ‘not at all’ interfering with work productivity [DRF, 73.3% (121/165); DMF, 63.5% (101/159)].
Among patients who indicated they were employed (DRF, n = 133; DMF, n = 133), DRF-treated patients reported less missed time at work due to GI symptoms compared with DMF-treated patients. In the DRF group, 20 patients missed ⩾1 h of work on 43 separate days; eight patients missed ⩾4 h of work on 15 separate days; and six patients missed ⩾8 h of work on 11 separate days. By comparison, more patients missed time at work over a greater number of days with DMF: 26 patients missed ⩾1 h of work on 88 days; 13 missed ⩾4 h on 52 days; and 9 missed ⩾8 h on 35 days [Figure 4(a)]. In days with work hours missed, mean (SD) greatest number of missed work hours during these days was 4.3 (3.7) versus 5.5 (4.8) for DRF and DMF, respectively. Similar to the increase in GI symptom severity seen with DMF at week 2–3 that coincided with the majority of GI AE–related treatment discontinuations, a peak in mean number of missed work hours due to GI symptoms occurred with DMF at the ~3-week time point, compared with a gradual decline for DRF [Figure 4(b)].
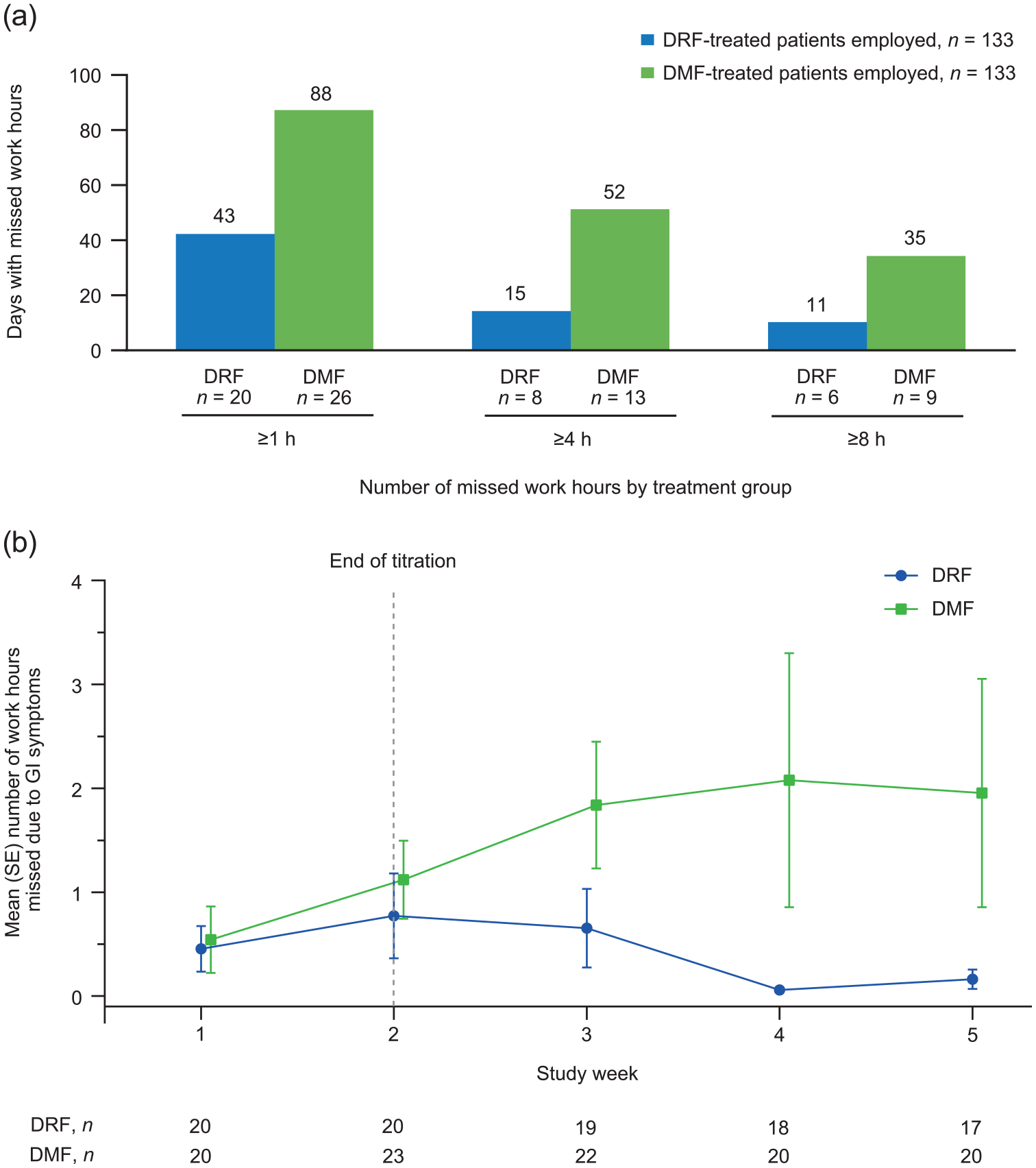
(a) Total number of days with missed work hours and (b) mean (SE) number of missed work hours by study week among patients reporting missed work due to GI symptoms.
Sensitivity and specificity analysis of IGISIS
An analysis was conducted to determine whether an IGISIS score ⩾2 was an appropriate threshold for comparing GI tolerability. An IGISIS score ⩾2 detected moderate or severe GI AEs of IGISIS with a high degree of sensitivity (90%) and specificity [59%; Supplemental Figure 3(a) and (b)]. Mean (SD) worst IGISIS score among patients with moderate or severe GI AEs of IGISIS was 4.7 (3.0; n = 16) with DRF and 5.0 (2.9; n = 44) with DMF. Overall, 87% (59/68) of patients in the study with moderate or severe GI AEs of IGISIS had a worst IGISIS score ⩾2. Similarly, mean (SD) worst IGISIS score among those who discontinued treatment due to GI AEs was 5.5 (2.1; n = 2) with DRF and 4.9 (3.6; n = 12) with DMF. Overall, 86% (12/14) of patients in the study with GI-related discontinuation had a worst IGISIS score ⩾2. The majority (71%; 10/14) of patients who discontinued due to GI AEs had a worst IGISIS score of ⩾4.
In addition, of patients who reported GI symptoms leading to ‘quite a bit’ or ‘extreme’ interference with daily activities [DRF, 7.9% (20/253); DMF, 10.8% (27/249)], missed work [DRF, 7.9% (20/253); DMF, 10.4% (26/249)], or concomitant symptomatic GI medication use [DRF, 6.7% (17/253); DMF, 14.7% (37/251)], the majority [89.4% (42/47); 91.3% (42/46); 85.2% (46/54), respectively] also had a worst IGISIS score ⩾2, further suggesting a cutoff score of 2 on IGISIS was likely to be associated with increased measures of treatment burden.
PROs
As may be expected given the short-term duration of the study, EQ-5D-5L visual analog scale scores in all five dimensions remained stable over the course of the study. Mean (SD) changes from baseline to end of treatment for DRF and DMF were stable: 1.5 (11.2) and 1.3 (12.2), respectively. In the overall population, most patients were likely to maintain ‘no problems’ or ‘slight problems’ on the usual activity dimension (DRF, 91% versus DMF, 88%) from baseline to end of treatment. When stratifying patients with no or slight problems in usual activities by those who had a GI AE (DRF, n = 88 versus DMF, n = 122; 91% versus 82%) or reported any IGISIS score ⩾2 (DRF, n = 109 versus DMF, n = 128; 88% versus 84%), scores were generally stable, although a moderate improvement was seen in patients treated with DRF. The proportion of patients maintaining this same status on the mobility and pain/discomfort dimensions were also similar. SF-12 physical and mental component scores (PCS and MCS, respectively) remained similarly stable over the course of treatment [mean (SD) change from baseline to end of treatment for DRF, n = 244 versus DMF, n = 231: PCS, −0.2 (6.8) and −0.3 (6.5); MCS, 1.0 (6.7) and 1.0 (7.0)].
GI tolerability AEs
Overall, GI AEs were reported in 34.8% (88/253) of DRF-treated patients and 49.0% (123/251) of DMF-treated patients. GI tolerability AEs were reported in 34.8% (88/253) of DRF-treated patients and 48.2% (121/251) of DMF-treated patients (Table 2). GI tolerability AEs were reported as moderate or severe for 7.9% (20/253) of DRF-treated patients and 18.7% (47/251) of DMF-treated patients. Among patients who experienced GI tolerability AEs, a lower proportion of DRF-treated patients [19.3% (17/88)] compared with DMF-treated patients [30.6% (37/121)] used concomitant symptomatic medication to treat GI symptoms. Mean (SD) duration of concomitant GI medication use was lower with DRF versus DMF [2.4 (8.8) days versus 4.0 (9.4) days]. The most common types of concomitant medication used to treat GI tolerability AEs were proton-pump inhibitors [DRF, 29% (5/17); DMF, 24% (9/37)] and anticholinergics/antispasmodics [DRF, 47% (8/17); DMF, 68% (25/37)].
Incidence of GI tolerability AEs and use of concomitant medication to treat GI symptoms.

Two patients in the DMF group had a GI AE that was considered not related to tolerability (toothache, n = 1; dry mouth, n = 1).
If patients took more than one concomitant GI medication, the total durations from all these concomitant medications were summarized.
The majority of concomitant medications used to treat GI symptoms were proton-pump inhibitors [overall cohort, 26% (14/54); DRF, 29% (5/17); DMF, 24% (9/37)] and anticholinergics/antispasmodics [overall cohort, 61% (33/54); DRF, 47% (8/17); DMF, 68% (25/37)].
AE, adverse event; DMF, dimethyl fumarate; DRF, diroximel fumarate; GI, gastrointestinal; SD, standard deviation.
Summary of safety
AEs were reported in 78.3% (198/253) of patients in the DRF group and 83.7% (210/251) of patients in the DMF group. The majority of AEs were mild or moderate in severity [DRF, 97.5% (193/198); DMF, 93.3% (196/210)]. The most common AEs (occurring in ⩾10% of patients in either group) for DRF- versus DMF-treated patients were flushing (32.8% versus 40.6%), diarrhea (15.4% versus 22.3%), nausea (14.6% versus 20.7%), and upper abdominal pain (6.7% versus 15.5%). Serious AEs occurred in 1.6% (4/253) and 1.2% (3/251) of DRF- and DMF-treated patients, respectively. There were no deaths in either treatment group.
Discussion
In this post hoc analysis of EVOLVE-MS-2, the improved GI tolerability experienced by DRF-treated patients translated into clinically meaningful benefits to QoL, with less interference of GI symptoms with daily activities and work productivity, fewer hours of missed work, fewer reported GI tolerability AEs, and less use of concomitant symptomatic GI medications compared with DMF-treated patients. DRF-treated patients were less likely to self-report GI symptoms of any severity, and among those who did, symptoms were generally less severe than those reported by DMF-treated patients. Importantly, GI symptom severity generally declined over the study period with DRF, suggesting that patients may better tolerate the transition from titrated to full dose (231–462 mg twice daily), enabling patients to initiate DRF as per the approved instructions and achieve the full efficacy benefit sooner. In contrast, DMF-treated patients experienced a transient increase in overall number and severity of patient-assessed GI symptoms shortly after the twice-daily dose of DMF increased from 120 mg to 240 mg, which corresponded with the time period at which most DMF treatment discontinuations occurred [Figure 2(b)]. The observed lower treatment burden with DRF as measured in this study is clinically meaningful and relevant to patients with MS. Patients value their overall well-being and minimal treatment burden; previous studies involving patient focus groups tasked with ranking attributes most likely to affect MS treatment decisions found that impact of a DMT on QoL was more important or nearly as important as that therapy’s effect on disease progression.4,20
GI AEs were less likely to impact a patient’s ability to work in those receiving DRF compared with DMF. The difference in number of patients reporting missing work due to GI symptoms between the DRF and DMF groups was not large; however, the total number of days with ⩾1 h missed work for the 26 DMF-treated patients was approximately two times larger than the 20 DRF-treated patients (88 days versus 43 days), translating into approximately 3.4 days with ⩾1 h missed/patient with DMF, compared with 2.2 days with ⩾1 h missed/patient with DRF among those who missed work. Notably, the proportion of patients missing ⩾4 h or ⩾8 h of work, representing a half or full standard 8 h shift, respectively, was lower for DRF compared with DMF. Missed work is a primary contributor to MS disease-related costs and can result in voluntary or involuntary termination of employment.21,22 Ability to work can impact a patient’s QoL, as demonstrated by data from a real-world study showing a positive association between improvements in work productivity and health-related QoL in DMF-treated patients compared with those who received β-interferons or glatiramer acetate. 23 Likewise, data from a cohort of 377 working patients with MS from the CLIMB study, a longitudinal observational study of patients with MS in the era of current treatment, showed that nearly half of the patients reported any overall work impairment and that presenteeism (impairment while at work) was associated with reduced QoL. 24
Conclusions made in the primary analysis of EVOLVE-MS-2 were based on patient self-assessed GI symptoms with a severity score of ⩾2 on the IGISIS and GGISIS scales. In the present analysis, it was determined that an IGISIS score ⩾2 detected moderate or severe GI AEs with high sensitivity and specificity and was able to capture the majority (~90%) of patients with moderate/severe GI AEs and those who discontinued treatment due to GI AEs. An IGISIS score of ⩾2 also captured the majority (~90%) of patients who reported interference of GI symptoms on daily activities, missed work, and concomitant symptomatic medication use. Together, these data suggest that an IGISIS score ⩾2 represents an appropriate threshold for comparing GI tolerability with DRF versus DMF.
Patient-reported outcomes (PROs) generally remained stable on EQ-5D-5L and SF-12, with advantages noted in favor of DRF. More DRF-treated patients maintained stable scores on the usual activity dimension on EQ-5D-5L, indicating a lesser patient-perceived impact of treatment on daily life in the overall DRF/DMF population and a subgroup analysis in those who experienced GI AEs or reported any IGISIS score ⩾2. However, because the study duration was only 5 weeks, the magnitude of changes in these PRO scales were small, and longer follow up is needed to assess the true impact on these measures. Longer-term assessment of PROs with DRF treatment is currently underway in the ongoing, open-label, 96-week EVOLVE-MS-1 study.
As MS treatment options continue to evolve, considering the impact of treatment on MS disease activity is important, as are patient treatment preferences and factors that influence the patient’s ability to take the drug as prescribed. 4 Suboptimal treatment tolerability due to side effects can prevent the treatment from conferring maximum effectiveness due to dose interruption, reduction, or discontinuation of treatment, in addition to having a negative impact on QoL and/or medication adherence. 5 Patients randomized to DRF in EVOLVE-MS-2 had lower rates of treatment discontinuation due to AEs (DRF 1.6% versus DMF 5.6%) and GI AEs (DRF 0.8% versus DMF 4.8%) compared with DMF, and the observed rates with DRF are supported by interim findings from the ongoing 96-week EVOLVE-MS-1 study.9,17 Medication adherence remains a challenge in MS, with a population-based study in Canada finding that nearly 20% of patients with MS discontinued their first oral DMT within 1 year. 25 AEs are the leading reason for treatment discontinuation with DMF, and GI AEs make up the largest percentage of AE-related discontinuations.12,26,27 A real-world, open-label, single-arm study of GI tolerability found that of the 14.7% (31/211) of patients who discontinued treatment, 10% (21/211) discontinued due to AEs and 6.6% (14/211) discontinued due to GI AEs, despite the use of symptomatic medication. 26 Use of symptomatic medication, although defined as a mitigation strategy for DMF-treated patients, 15 may add to overall treatment burden and additional out-of-pocket medication costs. Additionally, some patients who rely on taking DMF with a high-fat food to mitigate the risk of GI AEs 15 may find it challenging to maintain this habit and lifestyle over the long term.
Together, these data suggest that MS treatments that reduce incidence of GI AEs, such as DRF, may improve persistence to medication and minimize overall treatment burden. Improved tolerability could have different meanings to individual patients and healthcare providers. Therefore, adopting a patient-centric approach that considers the treatment’s tolerability profile, treatment burden, and potential impact on patient QoL is important when making an individualized choice of DMT.
General limitations of the EVOLVE-MS-2 study have been described previously. 17 However, limitations to this analysis should be noted. The 5-week study period for EVOLVE-MS-2, although optimized for the analysis of early-onset GI tolerability events, limits the ability to draw conclusions on outcomes requiring longer-term observations such as EQ-5D-5L and SF-12. Statistical significance (i.e. p values) were not reported for the DRF versus DMF comparisons reported in this secondary analysis because these endpoints were descriptive and the study was not powered to detect significant differences on these outcomes. The findings from this study reflect outcomes in patients who have been screened for history of GI conditions and active/recurring GI symptoms per the entry criteria in order to reduce confounders and determine the true effect of study drug on GI tolerability. Additionally, although IGISIS and GGISIS are non-validated scales, they are adapted from GI symptom scales previously used in other DMF trials that were based on a validated questionnaire for flushing12,17,26,28 and provide the ability to assess perspectives of patients with MS on the interference of GI symptoms with daily activities and work.
Conclusion
Improved GI tolerability was demonstrated for DRF compared with DMF as indicated by a reduction in number of days with patient self-assessed GI symptoms, lower incidence and severity of GI symptoms, decreased use of concomitant medication to treat GI AEs, and decreased interference of GI symptoms with daily activities and work productivity. These consistent, clinically meaningful improvements on GI tolerability translated to a lesser impact of treatment on QoL and a low rate of GI AEs leading to treatment discontinuation.
Supplemental Material
sj-pdf-1-tan-10.1177_1756286421993999 – Supplemental material for Improved gastrointestinal profile with diroximel fumarate is associated with a positive impact on quality of life compared with dimethyl fumarate: results from the randomized, double-blind, phase III EVOLVE-MS-2 study
Supplemental material, sj-pdf-1-tan-10.1177_1756286421993999 for Improved gastrointestinal profile with diroximel fumarate is associated with a positive impact on quality of life compared with dimethyl fumarate: results from the randomized, double-blind, phase III EVOLVE-MS-2 study by Annette Wundes, Sibyl Wray, Ralf Gold, Barry A. Singer, Elzbieta Jasinska, Tjalf Ziemssen, Jerome de Seze, Pavle Repovic, Hailu Chen, Jerome Hanna, Jordan Messer, Catherine Miller and Robert T. Naismith in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-2-tan-10.1177_1756286421993999 – Supplemental material for Improved gastrointestinal profile with diroximel fumarate is associated with a positive impact on quality of life compared with dimethyl fumarate: results from the randomized, double-blind, phase III EVOLVE-MS-2 study
Supplemental material, sj-pdf-2-tan-10.1177_1756286421993999 for Improved gastrointestinal profile with diroximel fumarate is associated with a positive impact on quality of life compared with dimethyl fumarate: results from the randomized, double-blind, phase III EVOLVE-MS-2 study by Annette Wundes, Sibyl Wray, Ralf Gold, Barry A. Singer, Elzbieta Jasinska, Tjalf Ziemssen, Jerome de Seze, Pavle Repovic, Hailu Chen, Jerome Hanna, Jordan Messer, Catherine Miller and Robert T. Naismith in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Biogen provided funding for medical writing support in the development of this manuscript; Susan Chow, PhD, from Excel Scientific Solutions, wrote the first draft of the manuscript based on input from authors, and Miranda Dixon from Excel Scientific Solutions copyedited and styled the manuscript per journal requirements. The authors had full editorial control of the manuscript and provided their final approval of all content.
Conflict of interest statement
AW reports advisor fees from Biogen and AbbVie; and research support from AbbVie, Alkermes, and Biogen. SW reports consulting fees from Biogen, EMD Serono, Genentech-Roche, Sanofi-Genzyme; speaker bureaus for Biogen, EMD Serono, Genentech/Roche, and Sanofi-Genzyme; and research support from Alkermes, Biogen, Celgene, Genentech/Roche, Novartis, Sanofi-Genzyme, and TG. RG reports honoraria from Bayer, Biogen, Merck Serono, Novartis, and Teva Neuroscience; and research support from Bayer, Biogen, Merck Serono, Novartis, and Teva Neuroscience. BAS reports speaker/consulting fees from AbbVie, Alexion, Bayer, Biogen, Bristol Myers Squibb, EMD Serono, Genentech, Novartis, Roche, Sanofi-Genzyme, Teva, and TG; and research support from AbbVie, Alkermes, Biogen, MedImmune, Novartis, Roche, and Sanofi-Genzyme. EJ reports advisory boards for Biogen; and speaker fees from Adamed, Allergan, Novartis, Polpharma, Roche, and Teva. TZ reports advisory boards for Bayer, Biogen, Genzyme, Merck Serono, Novartis, Synthon, and Teva; speaker fees from Almirall, Bayer, Biogen, Genzyme, GlaxoSmithKline, Merck Sharp & Dohme, Novartis, Sanofi, and Teva; and research support from Bayer, Biogen, Genzyme, Novartis, Sanofi, and Teva. JDS reports honoraria from Biogen for advisory boards and consulting. PR reports speaker/consulting fees from Alexion, Biogen, Celgene, EMD Serono, Medison, Novartis, Roche, Sanofi-Genzyme, and Viela Bio. HC, JH, JM, and CM are employees of and hold stock/stock options in Biogen. RTN reports advisor/speaker/consulting fees from Alexion, Alkermes, Bayer, Biogen, Celgene, EMD Serono, Genentech, Lundbeck, NervGen, Novartis, Sanofi-Genzyme, Third Rock, and Viela Bio. The Editor-in-Chief of Therapeutic Advances in Neurological Disorders is an author of this paper, therefore, the peer review process was managed by alternative members of the Board and the Editor was not involved in the decision-making process.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Biogen, Cambridge, MA, USA.
Data sharing
EVOLVE-MS-2 was registered with ClinicalTrials.gov [ClinicalTrials.gov identifier: NCT03093324]. Study data will be shared in accordance with applicable regulations and laws.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
