Abstract
Background:
Therapeutic plasma exchange (TPE) is frequently used in glucocorticosteroid (GCS)-refractory multiple sclerosis (MS) relapses. Data regarding predictors of treatment response are scarce. The objective of this study was to analyze predictive factors for response to TPE in GCS-refractory MS patients.
Methods:
A total of 118 MS patients in two tertiary MS centers were analyzed. Primary outcome was TPE response defined as marked, mild, or no improvement. Secondary outcome was change in expanded disability status scale (ΔEDSS). ΔEDSS and relapse activity within 6 months after TPE were studied.
Results:
Marked or mild improvement was observed in 78.8% of patients. ΔEDSS correlated significantly inversely with time from relapse to start of TPE (τ = –0.239, p = 0.001), age (τ = 0.182, p = 0.009) and disease duration (τ = –0.167, p = 0.017). In multivariate analysis, TPE response was predicted by diagnosis of relapsing MS [odds ratio (OR): 3.1], gadolinum-enhancement on magnetic resonance imaging (OR 3.2), age (OR 0.5 per 5 years older) and time from relapse onset to TPE (OR 0.7 per 7 days longer).
Conclusion:
Patients with longer disease duration and higher EDSS pre and post-TPE were more likely to show further disability progression or relapses within 6 months after TPE. No sustained effects were observed during the follow-up period.
Keywords
Introduction
Multiple sclerosis (MS) is the leading cause of disability in young adults not attributable to trauma, 1 and has an unpredictable long-term prognosis. 2 MS can be classified into relapsing (RMS) and progressive forms (PMS). PMS can develop from an initial relapsing-remitting course (secondary PMS – SPMS) or persists from onset (primary PMS – PPMS). RMS accounts for up to 90% of newly diagnosed cases and is characterized by attacks (“relapses”) with new or deteriorating pre-existing symptoms and subsequent complete or incomplete remission. Superimposed relapses may also occur in SPMS or even rarely in PPMS. 3 In contrast to the increasing armamentarium of disease-modifying therapies (DMT) in MS, 4 the treatment of relapses itself remained unchanged over the last decades. 5 Glucocorticosteroids (GCS) are still the mainstay in the treatment of MS relapses.6,7 Current treatment guidelines recommend (i) high-dose (500–1000 mg per day) intravenous methylprednisolone (IVMP) for 3–5 days as a first-line treatment of MS relapses,5,8 (ii) a further high-dose IVMP course (up to 2000 mg per day) for 3–5 days in case of clinical non-responsiveness within 2 weeks after the first IVMP treatment,5,9 and (iii) therapeutic plasma exchange (TPE) or immunoadsorption as a therapeutic option for inadequate response to IVMP treatment.5,8,9
TPE has been used for decades in various immune-mediated diseases, 10 and was first established in acute inflammatory demyelinating disorders of the central nervous system (CNS) by Weinshenker et al. in 1999, 11 when 42.1% of patients who did not respond to GCS during a relapse showed improvement after TPE. Plasma exchange removes autoantibodies and immune complexes from the plasma with effects on the humoral immune system, whereas effects on cell-mediated immunity are not obvious. 10 Over the last decades, the importance of humoral immunity in the pathophysiology of MS has been increasingly recognized.12,13 Nevertheless, MS is a heterogeneous disorder both from a clinical and immunohistopathological point of view.2,3,12 This is mirrored by the observation that not all patients benefit from TPE, and response rates range from 40% to 90%.11,14 –16 Among other factors, such as small sample sizes, and retrospective designs, results are inconsistent due to the inclusion of various inflammatory demyelinating diseases of the CNS, such as MS, but also neuromyelitis optica spectrum disorders and rare cases of concentric sclerosis.
In light of this, we aimed to investigate response rates to TPE in a large, two-center, well-characterized study cohort of MS patients, and to identify possible predictive factors for a favorable response to TPE.
Patients and methods
For this retrospective study, all MS patients who received TPE between 2001 and 2015 at two tertiary MS centers in Vienna, Austria, and Rostock, Germany, were enrolled. Inclusion criteria demanded for a definite diagnosis of MS according to the most recent diagnostic criteria, age >18 years, diagnosis of an acute relapse or worsening of pre-existing symptoms lasting for more than 24 h, lack of or incomplete improvement of relapse symptoms following intravenous methylprednisolone pulse therapy. 17 Patients who did not receive methylprednisolone for the same relapse for which TPE was performed have been excluded to prevent treatment bias. Additionally, patients with insufficient clinical data available to assess outcome have been excluded. The decision to initiate TPE was based on the lack of or incomplete improvement or further clinical deterioration of symptoms and was made by the treating physician and the number of TPE cycles was based on continuous clinical evaluation. The analysis of patient documentation included demographic data, relapse rate within 12 months before TPE, magnetic resonance imaging (MRI) findings, the degree of disability according to the expanded disability status scale (EDSS) immediately before TPE initiation and at discharge after last TPE session. 18 In addition, control visits and relapses within 6 months after TPE were recorded and assessed.
Response to TPE treatment was classified as marked, mild, or no improvement at discharge after TPE treatment. A marked response to therapy was defined as improvement in EDSS ⩾ 1.0 (if baseline EDSS before TPE ⩽5.5) or ⩾0.5 (if baseline EDSS > 5.5). A mild response was defined as an improvement in EDSS of 0.5 (if baseline EDSS baseline ⩽5.5) or an improvement in at least one functional system, but with no corresponding overall change in EDSS (ΔEDSS).
The study was approved by the local ethics committees of the Medical University of Vienna (EC No. 1660/2014) and the University of Rostock (A2015-0065). STROBE guidelines were followed for analysis and conduction of the study.
Statistics
Normal distribution of the data was tested with the Kolmogorov–Smirnov-test and could not be shown for any of the tested variables except age. The data are therefore reported as medians (md) and ranges or interquartile ranges (IQR) for continuous variables and as absolute numbers and percentages for categorical variables. Non-parametric tests, that is, Mann–Whitney U test and Kruskal–Wallis test, were used to compare the continuous data between groups. The distribution of nominal variables was analyzed using the chi-squared test. The correlations are presented as Kendall–Tau-b coefficients (τ).
To assess factors predicting response to TPE treatment, we performed multivariate multinomial logistic regression models with TPE response as the dependent variable and including all variables as covariates for which a univariate association with a p value < 0.1 was found.
Missing values were handled by multiple (20 times) imputation using the missing not at random (MNAR) approach with pooling of estimates according to Rubin’s rules. 19
A two-sided p value < 0.05 was considered statistically significant. All data analyses were performed using IBM SPSS Statistic 24.0 (SPSS Inc, Chicago, IL, USA) and R statistical software (version 4.0.0).
Results
A total of 158 patients treated with TPE between 2001 and 2015 have been retrospectively identified. Due to incomplete data, 25 had to be excluded from further analysis. In addition, 15 patients did not receive IVMP before TPE, resulting in the inclusion of 118 patients in our study, see Figure 1.

Flow chart of patients screened for eligibility of analysis.
A total of 76 patients were female (64.4%) and the median age was 37 years (IQR:15, min: 15, max: 73). RMS was diagnosed in 83.1% of the patients and 16.9% were classified as PMS with superimposed relapses. Of these 20 patients wiht PMS, five have been diagnosed with PPMS and 15 with SPMS; two and six patients, respectively, showed Gd-enhancement on MRI. The median disease duration was 2 years (IQR:8, min: 0, max: 24) and median EDSS was 3.5 (IQR: 3, min: 1, max: 8.5), see Table 1. Out of the 118 analyzed patients, 76 received a second course of IVMP treatment (64.4%), with a median dose of 15 g (IQR: 10). MRI was performed in 106 patients (89.8%), and, in 66 (62.2%) of these patients, gadolinium enhancement was detected.
Baseline characteristics of final cohort.
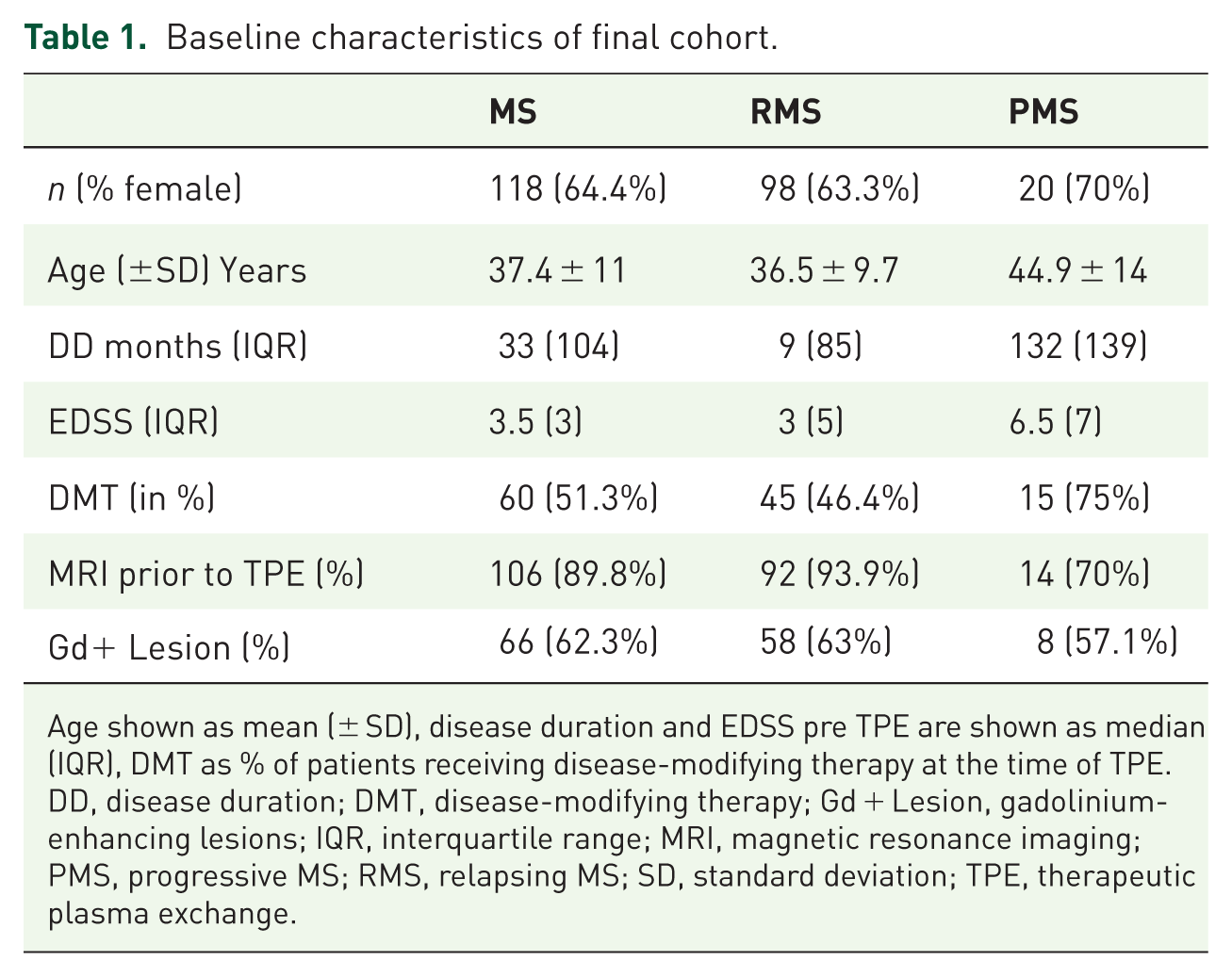
Age shown as mean (± SD), disease duration and EDSS pre TPE are shown as median (IQR), DMT as % of patients receiving disease-modifying therapy at the time of TPE.
DD, disease duration; DMT, disease-modifying therapy; Gd + Lesion, gadolinium-enhancing lesions; IQR, interquartile range; MRI, magnetic resonance imaging; PMS, progressive MS; RMS, relapsing MS; SD, standard deviation; TPE, therapeutic plasma exchange.
Response to TPE and predictive factors
Overall, 93 (78.8%) of the analyzed patients showed clinical improvement after TPE; 51 patients (43.2%) showed a marked improvement and 42 (35.6%) a mild improvement. In 25 (21.2%) patients, no clinical benefit could be observed after TPE.
Compared with patients without improvement after TPE, median age was lower in patients with marked improvement after TPE (33 versus 41 years, p = 0.024) and with mild improvement (37.5 versus 41 years, p = 0.06).
Disease duration was significantly shorter (p = 0.033) in patients with marked improvement and non-significantly shorter (p = 0.089) in patients with mild improvement compared with those without improvement. Both age and disease duration correlated inversely with the improvement of EDSS during TPE treatment (ΔEDSS) (age: τ = –0.182, p = 0.009, disease duration: τ = –0.167, p = 0.017). Patients with RMS had a higher proportion of TPE response than those with PMS (p = 0.014). Median duration from onset of relapse to start of TPE was significantly shorter in patients with marked response (39 days, IQR 36; min: 7, max: 154, p = 0.020) compared with the mild and no improvement groups (61 days, IQR: 37, min: 17, max: 152) and 61 days (IQR 63, min: 15 max: 163) days, respectively), see Table 2. Time from relapse to TPE correlated inversely with the improvement in EDSS score (τ = –0.239, p = 0.001).
Factors associated with response to TPE.

Number of RMS patients in the various response group (% as proportion of RMS patients in the various response group), age shown as mean (± SD), DD and EDSS pre TPE are shown as median (IQR), DMT as % of patients receiving disease modifying therapy at the time of TPE.
DD, disease duration; DMT, disease-modifying therapy; EDSS, ; IQR, interquartile range; NON, non-improvement; PMS, progressive MS; RMS, relapsing MS; SD, standard deviation; TPE, therapeutic plasma exchange.
Duration from relapse onset to start of IVMP treatment, cumulative doses of IVMP, as well as number of TPE sessions, body weight, relapse rate before TPE, and sex showed no association with response to TPE treatment (see Supplemental Table e-1), when analysed only for RMS patients, this trend was confirmed (see Supplemental Table e-2).
Sensitivity analyses showed no difference in TPE response between patients with one or two IVMP cycles or total doses of GCS prior to TPE, while the results were confirmed in terms of time to TPE and disease duration. Regarding the presence of gadolinium (Gd)-enhancing lesions in cerebral and/or spinal MRI before TPE, patients with improvement after TPE had a significantly higher proportion of Gd-enhancing lesions than the no improvement group. Investigating the effects of DMT on the outcome of TPE, we found that patients without DMT were more likely to benefit from TPE compared with patients with DMT, which might be explained by the fact that many patients were included with their first episode suggestive for MS.
In the multivariate regression model for prediction of TPE response, diagnosis of RMS [odds ratio (OR): 2.2], Gd-enhancement on MRI (OR 2.0) and age (OR 0.6 per 5 years older) were the only remaining significant predictors of mild improvement. Marked improvement was associated with a diagnosis of RMS (OR: 3.1), Gd-enhancement on MRI (OR 3.2) and age (OR 0.5 per 5 years older) as well, but additionally with delay from relapse onset to TPE (OR 0.7 per 7 days longer), see Table 3.
Prognostic factors of improvement after TPE.

Higher values indicate higher probability of mild/marked improvement (reference category: no improvement). calculated by multinomial logistic multivariate regression models.
CI, confidence interval; DMT, disease-modifying treatment; EDSS, expanded disability status scale; MRI, magnetic resonance imaging; OR, odds ratio; RMS, relapsing multiple sclerosis; TPE, therapeutic plasma exchange.
Side effects and tolerability
In all, 39 side effects of TPE treatment were reported. No specific side effects besides the expected ones, like hypotension, were observed. They had no effect on treatment response and were balanced across the groups that responded to treatment, see Supplemental Table e-3.
Follow-up period
To analyze whether the TPE had a prolonged effect on disease activity, follow-up examinations over the next 6 months were assessed. Of the patients analyzed, 100 (84.7%) had a follow up within 6 months after TPE. In 27 (27%) of them, a new relapse occurred during the follow-up period. Of the remaining 73 patients without relapse, 18 patients showed an EDSS improvement after discharge, and 14 patients without relapse experienced an EDSS increase. Patients who continued to improve clinically during the follow-up period had a shorter disease duration [median 2.5 months (IQR: 79)] compared with those with clinical stability [median 8 months (IQR: 104)] and those with EDSS progression [median 78 months (IQR: 127)] (p = 0.02). The higher the EDSS after TPE, the more likely there was a further deterioration in disability at the follow-up visit, while level of disability before TPE only showed a trend in this direction, see Supplemental Table e-4. In the follow-up period, 33 patients (28%) started with a new DMT, 24 (20.3%) continued to receive the same DMT, 26 (22%) switched to another DMT, and 14 patients (11.9%) remained without DMT.
Discussion
In this retrospective study, we analyzed 118 MS patients who were treated with TPE after inadequate response to IVMP treatment due to an acute relapse. A vast majority showed a marked or at least mild improvement in response to TPE. Factors that made clinical improvement more likely were diagnosis of RMS (2- to 3-fold more likely), Gd-enhancement on MRI (2- to 3-fold more likely), younger age (1.6-to 2-times more likely per 5 years) and shorter delay from relapse onset to TPE (1.4-times more likely per 7 days). The number of TPE sessions as well as exchange volume had no influence on efficacy (see Supplemental Table e-3).
Our study population is one of the largest reported so far and includes patients comprising a wide spectrum of MS patients ranging from very short course of disease. For example, after the first clinical event, to patients with a long disease duration and with a progressive disease course, but with the common denominator of an acute, steroid-unresponsive relapse. Patients after the first clinical event showed a similar response to TPE as relapsing patients, which is consistent with previous studies. 20 In consistency with literature a younger age, RMS disease course and Gd-enhancement on MRI are characteristics associated with a patient population with a larger inflammatory pathophysiological component being more responsive to TPE. Older patients with progressive MS, on the contrary, showed a comparatively limited response. 16
The delay between the onset of relapse and the initiation of TPE was crucial for the clinical response to TPE in our population, with a 7-day delay reducing the probability of TPE response by more than 30%. Thus, a delay of TPE by 14 or 21 days, which is common in clinical routine, corresponds to a respective two- and threefold decrease of the chance for clinically relevant improvement.
We could also demonstrate that this delay is significantly inversely correlated with the extent of clinical benefit expressed as the ΔEDSS. Hence, the shorter the delay of TPE, the more the patient will improve. The importance of early TPE initiation has also been reported in the literature. 16 In our cohort, we did not find evidence for the effectiveness of a second IVMP cycle as opposed to only one cycle before TPE initiation. Only the delay until the start of TPE was the decisive factor. Whether a second administration of IVMP should be avoided and TPE should be initiated has to be discussed on the basis of our data.
Of the 100 patients with a follow up of at least 6 months after TPE, 27% suffered a further relapse within this period. Interestingly, the proportion of patients who received DMT was higher in those who relapsed compared with those who did not, maybe mirroring a more active patient cohort. In those patients who did not experience a relapse, the number of patients who improved or remained stable with respect to EDSS was predominant. The patients who worsened without relapse were older, had a higher EDSS and a progressive course. In our patients, no prolonged effects beyond the acute relapse treatment were observed in relation to TPE.
The range of DMTs used in the patients enrolled in the study varied widely, and due to the inclusion of patients between 2001 and 2015, many of the now widely used DMTs were not available at that time, which precludes further conclusions regarding the role of DMTs in TPE treatment. The relatively large proportion of progressive patients with DMT may be due, on the one hand, to the small number of included patients with PMS, but on the other hand they are active and thus correspond to a specific patient cohort.
The retrospective design of the analysis is a limitation. There were also no pre-standardized MRI protocols, and not all patients received an MRI. Furthermore, documentation of adverse events might be subject to reporting bias as no standardized recording or grading was used. Lastly no immunology or quantitative imaging analysis was performed to explore as predictive factors in response to TPE. However, our study is one of the largest in the field of TPE and MS. Together with the careful characterization of our cohort, which covers the entire spectrum of MS, this strengthens the validity of this study.
In conclusion, predictors of favorable response to TPE are RMS, younger age, Gd-enhancement on MRI. Importantly, the period of time from relapse onset until TPE plays a significant role. TPE response will be more likely the earlier it is initiated.
Supplemental Material
sj-pdf-1-tan-10.1177_1756286420975642 – Supplemental material for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study
Supplemental material, sj-pdf-1-tan-10.1177_1756286420975642 for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study by Stephan Blechinger, Johannes Ehler, Gabriel Bsteh, Alexander Winkelmann, Fritz Leutmezer, Stefanie Meister, Agnes Santer, Michael Hecker, Thomas Berger, Paulus Rommer and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-2-tan-10.1177_1756286420975642 – Supplemental material for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study
Supplemental material, sj-pdf-2-tan-10.1177_1756286420975642 for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study by Stephan Blechinger, Johannes Ehler, Gabriel Bsteh, Alexander Winkelmann, Fritz Leutmezer, Stefanie Meister, Agnes Santer, Michael Hecker, Thomas Berger, Paulus Rommer and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-3-tan-10.1177_1756286420975642 – Supplemental material for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study
Supplemental material, sj-pdf-3-tan-10.1177_1756286420975642 for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study by Stephan Blechinger, Johannes Ehler, Gabriel Bsteh, Alexander Winkelmann, Fritz Leutmezer, Stefanie Meister, Agnes Santer, Michael Hecker, Thomas Berger, Paulus Rommer and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-4-tan-10.1177_1756286420975642 – Supplemental material for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study
Supplemental material, sj-pdf-4-tan-10.1177_1756286420975642 for Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study by Stephan Blechinger, Johannes Ehler, Gabriel Bsteh, Alexander Winkelmann, Fritz Leutmezer, Stefanie Meister, Agnes Santer, Michael Hecker, Thomas Berger, Paulus Rommer and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We acknowledge all our patients. Statistical analysis was conducted by Michael Hecker, University of Rostock, Rostock, Germany and Gabriel Bsteh, Medical University of Vienna, Vienna, Austria
Authorship contributions
All authors whose names appear on the submission made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; drafted the work or revised it critically for important intellectual content; approved the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
BS, EJ, MS and SA declare that there is no conflict of interest.
BG has participated in meetings sponsored by, received speaker honoraria or travel funding from Biogen, Celgene, Merck, Novartis, Sanofi-Genzyme and Teva, and received honoraria for consulting Biogen, Roche and Teva. None of them resulted in a conflict of interest regarding the submitted manuscript.
WA, LF, HM and BT declare that there is no conflict of interest with regard to this work.
PSR received research grants from Biogen, Merck, Roche; consultancy or speaker fees from Allmiral, Biogen, Cellgene, Merck, Roche, Novartis, Sanofi Genzyme, Sandoz. None of them resulted in a conflict of interest regarding the submitted work. ZUK received speaker fees from Alexion, Allmiral, Bayer, Biogen, Merck, Novartis, Roche, Sanofi Genzyme and Teva. None of them resulted in a conflict of interest regarding the submitted manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
