Abstract
Background and Aims:
Multiple sclerosis (MS) is the most common neuroimmunological disease of the central nervous system in young adults. Despite recommended contraception, unplanned pregnancies can occur in women of childbearing age with MS. MS- and comorbidities-related multimedication in these patients represents a potential risk. We aimed to raise awareness regarding the frequency of polypharmacy and drug–drug interactions (DDIs) in female MS patients of childbearing age.
Methods:
Sociodemographic, clinical and pharmaceutical data were collected through patient records, clinical investigations and structured patient interviews of 131 women with MS. The clinical decision support software MediQ was used to identify potential DDIs. A medication and DDI profile of the study population was created by statistical analysis of the recorded data.
Results:
Of the 131 female MS patients, 41.2% were affected by polypharmacy (concurrent use of ⩾5 drugs). Polypharmacy was associated with higher age, higher degree of disability, chronic progressive MS disease course and comorbidities. With an average intake of 4.2 drugs per patient, a total of 1033 potential DDIs were identified. Clinically relevant DDIs were significantly more frequent in patients with polypharmacy than in patients without polypharmacy (31.5%
Conclusion:
For the first time, a comprehensive range of potential DDIs in women of childbearing age with MS is presented. Polypharmacy is associated with the occurrence of clinically relevant DDIs. This shows the need for effective and regular screening for such interactions in order to prevent avoidable adverse effects.
Introduction
With more than 2 million people affected worldwide, multiple sclerosis (MS) is the most common immune-mediated neurological disease in young adults.1–3 The disease is associated with a variety of symptoms, such as paresis, spasticity, coordination disorders, gastrointestinal disorders, bladder afflictions, pain, sexual dysfunction, emotional as well as cognitive disorders, fatigue and dizziness,1,4–6 and can significantly reduce the quality of life of those patients.7–9
In general, women of childbearing age are the largest subgroup of people with MS. 10 Although contraception is recommended for female MS patients,11–13 unplanned pregnancies may still occur. Following estimations, the worldwide prevalence of unintended pregnancies was about 40% in 2012. 14 In Europe, estimates vary from 34% (Western Europe) to 54% (Eastern Europe). 15 There are few studies on MS patients, with rates of unplanned pregnancies ranging from 10% to 50%. 16 Prudent medication management is essential to protect potentially pregnant women with MS.
MS therapy is based on immunomodulating disease-modifying drugs (DMDs) and symptomatic drugs.13,17 Furthermore, comorbidity drugs and preparations of alternative or complementary medicine are an integral part of the medication profile of MS patients.18–20 It has been shown that polypharmacy in MS affects a considerable proportion of patients, ranging from 15% to 59%. 21 Multimorbidity and the associated risk of polypharmacy can have various consequences. On the one hand, there are immense costs for the healthcare system.22,23 On the other hand, certain measures of patient health, such as MS relapse frequency, hospitalisation rate, quality of life deficit, disability progression and cognitive impairment, are associated with polypharmacy.21,24,25 A further issue of polypharmacy is the risk of drug-drug interactions (DDIs). 26 In the United States (US), DDIs account for about 20% of adverse drug events, which cause costs between US $30 and $180 billion annually and are responsible for about 770,000 deaths. 27 Due to the frequency of unplanned pregnancies, particular vigilance must be exercised with regard to female MS patients of childbearing age.
The aim of this study was to raise awareness of the potential dangers of polypharmacy and DDIs in pre-menopausal women with MS. For this reason, we determined the prevalence and predictors of polypharmacy in this specific group of patients. Furthermore, for the first time, we provide an overview of the interaction potential of the drugs used in young women with MS.
Materials and methods
This cross-sectional study was conducted at the Department of Neurology (Section of Neuroimmunology) of the University Medical Centre of Rostock. This medical centre is the largest medical facility in the state of Mecklenburg-Vorpommern: MS patients with stable disease courses regularly get their routine check-ups at the outpatient ward, while patients with disease activity, severe disease courses and side effects are treated at the inpatient ward. Data collection took place from March 2017 to January 2020. Patients were included if they met the following criteria: women of childbearing age between 18 and 48 years with a confirmed diagnosis of MS or clinically isolated syndrome (CIS) according to the revised McDonald criteria. 28 The exclusion criteria were male sex or women over 48 years of age as menopause onset in most women worldwide is between 49 and 52 years. 29 Inpatients were asked to participate in this study during their clinic stay (usually several days), while outpatients were asked during the waiting period before their routine check-up. After patient education and informed consent, the study data of 131 participating women were gathered through anamnesis, medical records, clinical investigations and structured patient interviews.
This study was reviewed by the ethics committee of the University of Rostock (permit numbers A 2014-0089 and A 2019-0048) and conducted according to the Declaration of Helsinki.
Data gathered
The data collection of sociodemographic, clinical and pharmaceutical data occurred in the same way for each study participant: review of the patient’s current medical records and clinical medical examination, followed by a structured patient interview.
Sociodemographic data were age, school years (not including training or higher education), educational level, employment status, partnership, place of residence (<5,000 residents: rural community, 5,000–19,999: provincial town, 20,000–99,999: medium-sized town, ⩾100,000: city), number of children and number of siblings.
Clinical data consisted of Kurtzke’s Expanded Disability Status Scale (EDSS; a measure of the patient’s degree of disability), 30 disease duration, disease course (relapsing-onset MS and chronic progressive MS) and the number of comorbidities.
Pharmaceutical data comprised the drug names, active ingredients, dosages and application types. A genuine overview of the medication spectrum of the 131 women was achieved by recording only those drugs that were actually taken.
Drug characterisation
For a more detailed assessment, the drugs were classified according to three criteria.
Interval of drug intake
Drugs were evaluated as long-term drugs or as drugs on demand [
Prescription
A distinction was made between prescription-only drugs (Rx) and over-the-counter (OTC) drugs.
Therapy goal
According to the therapy goal, the drugs were classified into DMDs, symptomatic drugs and comorbidity drugs. 31 The DMDs include immunomodulatory drugs for the therapy of MS. The symptomatic drugs do not belong to the immunomodulatory drugs but to the diverse treatment approaches to alleviate symptoms caused by MS (e.g. bladder dysfunction and limited walking ability). 3 All other drugs that do not pursue the goal of MS therapy are classified as comorbidity drugs.
For the pregnancy safety classification of the drugs involved in clinically relevant DDIs, we used the summary of medicinal product characteristics (SMPC; https://www.fachinfo.de/). The SMPC describes the medical characteristics and the indications of the drug preparation approved by the regulatory authorities. 32
Polypharmacy
In our study, polypharmacy was defined as the concurrent intake of at least five drugs. Out of over 20 numerical definitions of polypharmacy described in the literature, this was the most commonly used definition. 33
Comorbidities
On the basis of patient records, patient interviews and physician expertise, comorbidities were identified according to the recommendations of Laronie
Identification of DDIs
In order to identify potential DDIs, the medication plans of all included patients were checked with the clinical decision support software (CDSS) MediQ (https://mediq.ch). In our study, this CDSS was used because of consistent results regarding sensitivity and specificity in several CDSS comparison studies.36–39 Moreover, MediQ is used in the University Medical Centre of Rostock by physicians and pharmacists in the clinical daily routine. MediQ is a web-based CDSS with a database containing over 2000 active substances and over 20,000 comments on potential DDIs (drug–drug, drug–food, drug–alcohol, drug–polymorphism). Five categories of DDI are distinguished in MediQ: high danger (strong), average danger (clinically relevant), low danger (relevant in exceptional cases), no interaction and lack of evidence (possible but insufficiently documented interactions).
Statistics
Prior to data analysis with PASW Statistics 18 (IBM), the patient data were transferred anonymously to a database. Comparative analyses of clinical-demographic and drug-related data between patients with polypharmacy (PwP) and patients without polypharmacy (Pw/oP) were performed using two-sample two-tailed Student’s
Results
Demographic and clinical profile of the patients
The 131 women with MS included were 36.4 years old on average (Table 1). The majority of women were skilled workers (61.1%), were in a partnership (71.0%), had fewer than two children (71.8%) and had one sibling (67.9%). In addition, about half of the women were employed (48.1%), whereas more than a third were already receiving a disability pension (35.1%).
Sociodemographic, clinical and pharmaceutical data of female MS patients of childbearing age with and without polypharmacy.

DMD, disease-modifying drug; EDSS, expanded disability status scale; MS, multiple sclerosis;
Mean value (standard deviation).
Median.
Average number of drugs taken per patient (standard deviation).
Chi-squared test.
Fisher’s exact test.
Range.
Two-sample two-tailed Student’s
Mann–Whitney
The study population had a median EDSS score of 2.0 (range: 1.0–8.0) and a median disease duration of 7 years (range: 6 weeks to 30 years). More than 90% of the patients were diagnosed with relapsing-onset MS (91.6%) and the other women had a chronic progressive course of disease (8.4%). Notably, 57.3% of the examined women suffered from at least one comorbidity in addition to MS.
Polypharmacy
The definition of at least five drugs taken per patient resulted in a polypharmacy rate of 41.2% (

Number of drugs taken depending on age and degree of disability in female MS patients. The number of drugs taken by the 131 young women with MS is plotted against age. The size and colour of the points were visualised depending on the patients’ EDSS scores. In addition, a linear regression line (in red, slope: 0.115,

Correlation matrix of clinical-demographic variables associated with polypharmacy. The colours symbolise the degree of pairwise correlation according to Pearson’s correlation coefficient. The crosses mark the absence of significance (
Comorbidities
The most frequent comorbidities in our study were thyroid disorders (19.9%), followed by depression (13.0%), deficiency symptoms (12.2%; e.g. deficiency of vitamin B12, vitamin D, iron, folic acid or calcium), arterial hypertonia (11.5%) and migraine (10.7%). Considering the different groups of comorbidities, there was only one significant difference with regard to polypharmacy: psychiatric comorbidities occurred five times more frequently in PwP than in Pw/oP (27.8%

Impact of comorbidities on polypharmacy in relationship with clinical-demographic and pharmaceutical data. In this spider plot, the MS patients were divided into three groups: women without comorbidities (
Drug profile
On average, the women examined took 4.2 drugs (Table 1). The number of medications taken ranged from 0 to 15. The observed frequencies per drug group are shown in Table 2. DMDs were used by 89.3% of the patients examined. Considering the five most commonly used drug groups, PwP were significantly more likely to take dietary supplements (50.0%
Drug groups used by female MS patients of childbearing age with and without polypharmacy.

Antiepileptics, migraine drugs, neuroleptics, asthma drugs, diabetes drugs, anti-Parkinson drugs, eye drops, dermatics, hormone substituents, immunotherapeutic drugs for comorbidities and other drugs were each taken by less than 5% of the patients.
MS, multiple sclerosis; N, number of patients.
Fisher’s exact test.
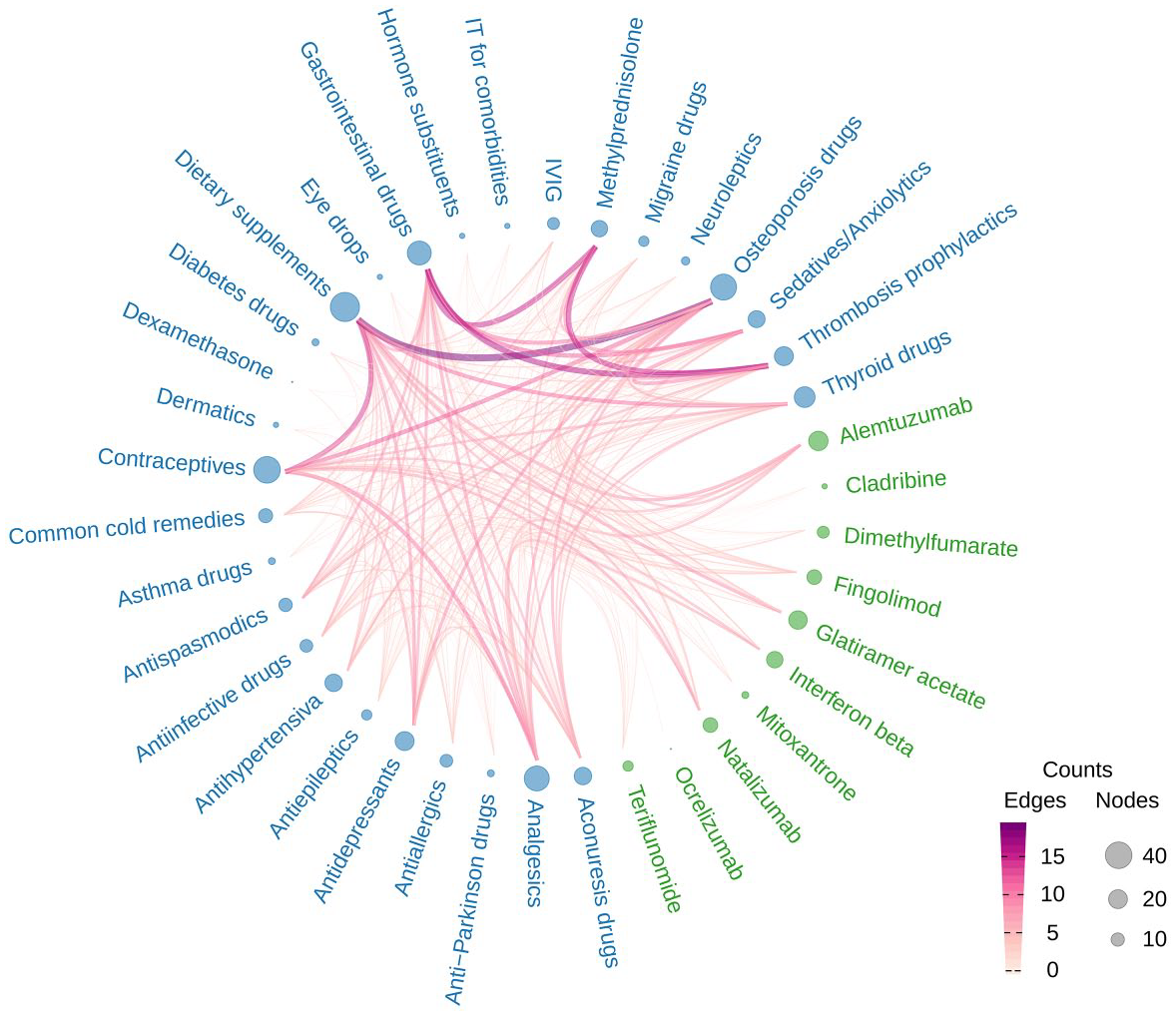
Common drug combinations in 131 female MS patients of childbearing age. Hierarchical edge bundling chart of medications taken together; 10 DMDs (green) and 28 other drug categories (blue) were considered. The frequency of intake of these 38 drug groups ranged from 1 (e.g. ocrelizumab) to 48 (dietary supplements). In the graph, thick dark lines indicate frequent combinations and large circles indicate the frequency per drug group. The most frequent drug combination was ‘dietary supplements and osteoporosis drugs’, which was found in 19 women.

Frequency of non-prescription drug intake in MS patients. A total of 73 different drugs were considered to be non-prescription drugs. OTC drugs (counted with repetitions) accounted for 25.3% (
Drug–drug interactions
DDI analysis revealed a total of 1033 potential DDIs (counted with repetitions) for the 131 examined female MS patients, i.e. an average of 7.9 interactions per patient. Of these 1033 potential DDIs, 3.0% were categorised by MediQ as average danger (clinically relevant), 17.0% as low danger (relevant in exceptional cases) and 80.0% as lack of evidence (possible but insufficiently documented interactions) (Figure 6). High danger DDIs were not recorded. Three average danger DDIs were found for two or more patients, namely the drug combinations ‘ethinylestradiol with levothyroxine’, ‘baclofen with oxybutynin’ and ‘citalopram with fingolimod’. Moreover, the results show that the prevalence of having at least one average danger DDI was significantly higher in PwP than in Pw/oP (31.5%

Potential drug interactions in women with MS. The distribution of potential DDIs according to the hazard levels is shown in a pie chart (a). Of 1033 potential DDIs (counted with repetitions) that were recognised in our patient cohort (
Discussion
In this study, we wanted to increase the awareness of potential dangers associated with polypharmacy and DDIs in female MS patients of fertile age. For this purpose, the frequency of polypharmacy and the clinical-demographic parameters associated with polypharmacy were examined in a large cohort of pre-menopausal women with MS. In addition, the spectrum of potential DDIs in this group of patients was comprehensively evaluated for the first time.
In our cohort of women of childbearing age, 41.2% were affected by polypharmacy. This rate is within the range of 15–59% of other studies on polypharmacy in MS.
21
Polypharmacy was significantly associated with higher age, higher degree of disability (EDSS), chronic progressive course of disease and comorbidities. With increasing age, higher multimorbidity rates occur,
41
which entails more complex drug therapies and can lead to polypharmacy. The situation is similar with regard to EDSS scores as more pronounced physical and cognitive impairments often require a more advanced therapy management. Depending on the symptoms that may develop in the course of MS (e.g. spasticity, pain, sexual dysfunction and bladder disorders), further medications can be used.
3
In our study, the association of progressive MS and polypharmacy was likely driven by the higher age (mean 43.1 years
Another point of interest is to consider drug groups that are associated with the presence of polypharmacy: ‘dietary supplements with osteoporosis drugs’ was the most common drug group combination in our analysis. In general, a considerable proportion of people with MS tend to take dietary supplements.
45
In the present study, more than a third of patients took at least one dietary supplement. Although these preparations can be used as a support, high-quality clinical trials are required to evaluate their actual benefit.
46
As expected, osteoporosis therapeutics, especially cholecalciferol, were taken by about 30% of the women studied. MS patients, especially women, have an increased risk of fractures and osteoporosis.47,48 In general, but also specifically for the osteoporosis drugs and dietary supplements, Rx drugs alone do not reflect the real medication profile of patients. OTC drugs accounted for 25.3% of the drugs in our study. The study by Barrenberg
About 1–5% of hospitalisations are caused by DDIs. 50 In Germany, about 200,000–1 million patients are affected by dangerous DDIs every year. 51 The probability of detecting at least one DDI in patients correlates with the number of drugs taken: it has been estimated to be 50% for people taking 5–9 drugs, 81% with 10–14 drugs, 92% with 15–19 drugs and 100% with at least 20 drugs. 52 In our patient cohort, a mean of 4.2 drugs per patient (range: 0–15 drugs) resulted in a huge spectrum of 1033 potential DDIs. To our knowledge, we are the first to provide here an overview of potential DDIs in MS patients. It is remarkable that the proportion of lack-of-evidence DDIs was 80.0%. Even though many of these DDIs are only theoretically possible and may have no practical relevance, their assessment should nevertheless be made on a patient-specific basis. Considering the most frequent average danger DDIs, the combination of ethinylestradiol and levothyroxine can lead to an increased requirement for levothyroxine.53,54 When starting or stopping an oral oestrogen while using levothyroxine, a monitoring of the thyroid function over several months and if necessary dose adjustment of levothyroxine might be useful. Combining baclofen with oxybutynin, side effects such as nausea, dry mouth and dizziness might increase.55,56 In addition, both drugs can lower the seizure threshold and lead to hallucinations. In the case of pregnancy, both the separate and combined use of baclofen and oxybutynin is not recommended unless the benefits outweigh the risks. Baclofen can pass the placental barrier and in newborns can lead to withdrawal symptoms and postnatal convulsions. 57 The available data on the effects of oxybutynin on pregnancy are insufficient and consequently its use is not recommended. 58 The last of the three most frequent average danger DDIs occurred in the combination of citalopram with fingolimod. The concurrent use of these drugs may increase the risk of QT prolongation, so exceeding the physiological intraventricular excitation time of the heart, and thus the risk of ventricular arrhythmias. 59 This should be observed especially during pregnancy. If the combination is necessary, electrocardiogram (ECG) monitoring should be performed initially and at dose increases. There is only insufficient data on citalopram in pregnancy, so it should only be used after careful risk–benefit analysis.60,61 Fingolimod is contraindicated in pregnancy and for women of fertile age without an effective contraception. There is an increased risk of severe congenital malformations of the fetus.12,62 In summary, the most frequent clinically relevant DDIs in our study involved not only comorbidity drugs but also MS-specific drugs: fingolimod (DMD), baclofen (antispasmodic), oxybutynin (parasympatholytic drug) and ethinylestradiol (recommended contraception) are used in MS-specific therapy.1,3
Considering especially the high rate of DMD use in our cohort, immunomodulatory therapies can pose a potential health risk to certain patient groups.
13
For instance, in the case of pregnancy (whether planned or unplanned), the use of biologicals such as alemtuzumab, natalizumab and ocrelizumab should take place only under careful risk–benefit consideration, because those monoclonal antibodies could cross the placenta and thus fetal damage cannot be ruled out.
13
A recent pharmacovigilance data analysis by Hellwig
The result that 30.5% of our study cohort used contraceptive drugs is consistent with data on women in fertile age in Germany. 64 However, we expected a somewhat higher proportion in MS because women receiving DMD are usually advised to use effective contraception.11–13 Although non-drug methods of contraception also exist, oral contraceptives are by far the most common method of contraception. 64 Therefore, unintended pregnancies are theoretically more likely in the subgroup of female MS patients not using hormonal contraception. Furthermore, our analysis revealed that 61.9% of the patients with at least one average danger DDI used no contraceptive drugs. Consequently, in the case of an unplanned pregnancy, drug combinations or individual drugs could have a negative effect on the pregnancy. For example, ibuprofen, as one of the most commonly used drugs in our cohort, carries an increased risk of miscarriage and fetal cardiac malformations in early pregnancy and is contraindicated in the third trimester of pregnancy. 65 Therefore, DDIs and drug utilization in MS patients are an issue of high interest.
It is worth noting that there are also some limitations of this study. Due to the cross-sectional study design, there is an absence of explanatory power with regard to causality. Longitudinal studies on the individual medication profiles of MS patients may provide new insights into the causal dependencies between polypharmacy and clinical-demographic parameters. Contrasting our findings against other polypharmacy and DDI studies in other autoimmune disease patients as well as in the general population would be an interesting point. Despite MediQ’s comprehensive drug databases, some compounds could not be studied. Either the corresponding compounds were not yet in the database or the medication plans collected from the patients were incomplete with regard to these compounds. In general, there is a lack of evidence on DDIs and side effects of relatively newly approved drugs. Information in databases grows over time with documented case reports.
Conclusion
In summary, our study showed that polypharmacy is present in almost half of female MS patients of fertile age. In addition to the known associations of polypharmacy with higher age, higher degree of disability, chronic progressive course of MS and comorbidities, we were also able to show that clinically relevant DDIs occur significantly more frequently in PwP than in Pw/oP. For the first time, a comprehensive overview of the spectrum of DDIs in MS patients is presented to create awareness for potential risks of multimedication in the light of possible unintended pregnancies. Especially for women of employable age, the topic of polypharmacy requires a high level of attention in everyday clinical practice. Further investigation into the risks of commonly used drugs in MS patients of childbearing age with regard to potential fetal damages would be useful.
Supplemental Material
sj-xlsx-1-tan-10.1177_1756286420969501 – Supplemental material for The risk of polypharmacy, comorbidities and drug–drug interactions in women of childbearing age with multiple sclerosis
Supplemental material, sj-xlsx-1-tan-10.1177_1756286420969501 for The risk of polypharmacy, comorbidities and drug–drug interactions in women of childbearing age with multiple sclerosis by Niklas Frahm, Michael Hecker, Silvan Elias Langhorst, Pegah Mashhadiakbar, Marie-Celine Haker and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-xlsx-2-tan-10.1177_1756286420969501 – Supplemental material for The risk of polypharmacy, comorbidities and drug–drug interactions in women of childbearing age with multiple sclerosis
Supplemental material, sj-xlsx-2-tan-10.1177_1756286420969501 for The risk of polypharmacy, comorbidities and drug–drug interactions in women of childbearing age with multiple sclerosis by Niklas Frahm, Michael Hecker, Silvan Elias Langhorst, Pegah Mashhadiakbar, Marie-Celine Haker and Uwe Klaus Zettl in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors would like to express their special thanks to the nurses and physicians of the outpatient and inpatient MS ward of the Department of Neurology at the University of Rostock for providing help with data acquisition.
Author contributions
NF, MH and UKZ conceptualised and designed this paper. NF, SEL and PM designed the questionnaires, conducted patient interviews and gathered the clinical-demographic data. NF analysed and interpreted the data, drafted the manuscript and prepared the tables. MH helped with the data analysis and prepared the figures. MCH was involved in the data analysis, including classification of drugs and drug interactions. UKZ and MH contributed to the interpretation of the results and provided important intellectual content. All authors read and approved the final version of the manuscript.
Conflict of interest statement
NF received travel funds for research meetings from Novartis. MH received speaking fees and travel funds from Bayer HealthCare, Biogen, Merck Serono, Novartis and Teva. SEL declares no competing interests. PM declares no competing interests. MCH declares no competing interests. UKZ received research support and lecture fees or travel funds from Alexion, Almirall, Bayer HealthCare, Biogen, Merck Serono, Novartis, Roche, Sanofi and Teva.
Data availability
The datasets generated and analysed in the current study are available from the corresponding author on reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly funded by Novartis Pharma GmbH, Nürnberg, Germany. The funder had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
