Abstract
Background:
Hypertension is a significant risk factor for cardiovascular disease, and it is associated with dementia, including Alzheimer’s disease (AD). Although it may be correlated with AD in terms of symptoms, the link between hypertension and AD pathological biomarkers, and the potential underlying mechanism of hypertension with cognitive decline, are still not well understood.
Methods:
The Mini-Mental State Examination (MMSE) scores were used to evaluate cognitive function. Enzyme-linked immunosorbent assays were used to examine plasma amyloid-beta (Aβ)40, Aβ42, and tau concentration in hypertensive patients. Metabolomics and metagenomics were performed to identify the significantly changed circulating metabolites and microbiota between healthy individuals and hypertensive patients. Pearson’s correlation was used to examine the association between cognitive indicators and differential metabolites.
Results:
We found significantly decreased MMSE scores, elevated plasma Aβ40, and decreased Aβ42/Aβ40 ratio in hypertensive patients, which are critically associated with AD pathology. Based on metabolomics, we found that significantly altered metabolites in the plasma of hypertensive patients were enriched in the benzoate degradation and phenylpropanoid biosynthesis pathways, and they were also correlated with changes in MMSE scores and Aβ42/Aβ40 ratio. In addition, metabolomics signaling pathway analysis suggested that microbial metabolism was altered in hypertensive patients. We also identified altered blood microbiota in hypertensive patients compared with the controls.
Conclusions:
Our study provides a novel metabolic and microbial mechanism, which may underlie the cognitive impairment in hypertensive patients.
Introduction
Dementia is a global health problem that affects about 47 million people worldwide. This number is expected to double by 2030 reaching 75 million, and the number of dementia cases are estimated to almost triple by 2050 reaching 135.5 million. 1 Although the understanding of the relationship between cardiovascular dysfunction and brain health has significantly improved over the last decades, whether hypertension constitutes a potentially treatable risk factor for cognitive decline and dementia remains a subject of debate. Hypertension is highly prevalent and is a risk factor for cardiovascular disease as it can induce microvascular dysfunction and atherosclerosis, which can result in cerebral hypoperfusion, microhemorrhages, and cerebral small vessel disease. 2 Recently, several clinical trials, such as the Systolic Blood Pressure Intervention Trial (SPRINT) Memory and Cognition in Decreased Hypertension (SPRINT-MIND) supported by the National Institute of Health and Hypertension Cardiovascular Outcome Prevention and Evidence Asia Network, have tried to determine the association between blood pressure (BP) control and dementia.3,4 SPRINT-MIND study suggests that lowering BP actually has a measurable impact on mild cognitive impairment (MCI). 5 It has been reported that hypertension in patients with Alzheimer’s disease (AD) is associated with worse cognitive function, increased neuropsychiatric burden, and lower hippocampal glucose metabolism. 6 In addition, pulse pressure elevation is also related to increased phosphorylated tau and decreased amyloid-beta (Aβ)42, the most important pathological hallmarks of AD, in the cerebrospinal fluid (CSF) of cognitively normal older adults. 7 Nevertheless, the link between hypertension and AD biomarkers is still not well understood. Metabolomics is widely used to evaluate metabolic changes associated with disease and to explore disease biomarkers; it has also been utilized to characterize risk factors for cardiovascular diseases, including hypertension. 8 However, there is still no evidence showing whether metabolic changes contribute to hypertension with cognitive decline.
In this study, we evaluated the cognitive function and circulating metabolites and microbes in patients with hypertension. We found decreased Mini-Mental State Examination (MMSE) scores and Aβ42/Aβ40 ratio in hypertensive patients, which were correspondingly correlated with the significantly changed circulating metabolites. In addition, there appeared to be altered blood microbiota in hypertensive patients compared with the controls. Hence, our study provides a novel metabolic and microbial mechanism, which may underlie cognitive impairment in hypertensive patients.
Material and methods
This study complied with the principles of the Declaration of Helsinki and was approved by the ethics committees of the institutional review board at Shunde Hospital, Southern Medical University, China (approval number: 20190903). Written informed consent was obtained from the patients or their legal guardians, and all research was performed in accordance with the relevant guidelines and regulations.
Study populations
A total of 33 hypertensive patients and 36 age- and sex-matched healthy individuals (controls) were enrolled from Shunde Hospital, Southern Medical University (Guangzhou, China) (As shown in Table 1). Patients with heart failure, acute myocardial infarction, undergoing medical treatment for dementia, sepsis, history of malignancy, autoimmune disease, secondary hypertension, severe renal failure [estimated glomerular filtration rate (eGFR) < 15 ml/min/1.73 m2 or under renal replacement therapy], severe anemia (Hb <60 g/L), or severe hepatic disease [cirrhosis or bilirubin >3× upper limit of normal (ULN) or aspartate aminotransferase/alanine aminotransferase/alkaline phosphatase >5× ULN] were excluded.
Baseline characteristics in hypertension and control group.

Continuous variables are presented as median (inter-quartile range) or mean (standard deviation). Categorical variables are expressed as number (percentages).
ALB, serum albumin; ALT, alanine aminotransferase; BP, blood pressure; CK-MB, creatine kinase-MB; eGFR, estimated glomerular filtration rate; FPG, fasting plasma glucose; HbA1c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; hs-CRP, high sensitive C-reaction protein; LDL-C, low-density lipoprotein cholesterol; LVEF, left ventricular ejection fraction; TC, total cholesterol.
Hypertension diagnosis
Hypertension was defined as systolic BP ⩾ 140 mmHg and/or diastolic BP ⩾ 90 mmHg, according to current Chinese guidelines for the management of hypertension. 9
MMSE evaluation
The MMSE scale was used to assess cognitive function. The MMSE scores ranged from 0 to 30, and higher scores indicated better cognitive function. Patients were asked to perform the MMSE on the day of enrollment, guided by a skilled nurse who had been trained for the task.
Human plasma collection
Fasting venous blood samples were collected to measure levels of glucose, total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), triglycerides (TGs), and creatinine using the Olympus AU2700 Automatic Biochemical Analyzer (Olympus, Tokyo, Japan). Low-density lipoprotein cholesterol (LDL-C) was calculated using Friedewald equation. High sensitivity C-reactive protein (hs-CRP) was determined by the high-sensitivity nephelometric method. After centrifugation at 1,500 g for 10 min at 4°C, plasma samples were stored at −80°C for future measurements of Aβ40 and Aβ42.
Metabolites extraction
The plasma samples were incubated on ice for 5 min and then were centrifuged at 15,000 rpm at 4°C for 5 min. Some of the supernatant was diluted to a final concentration containing 60% methanol by liquid chromatography–mass spectrometry (LC-MS) grade water. The samples were subsequently transferred to a fresh Eppendorf tube with a 0.22-μm filter and then centrifuged at 15,000 g at 4°C for 10 min. Finally, the filtrate was subjected to LC-MS/MS analysis.
Ultra-high-performance LC-tandem MS analysis
LC-MS/MS analyses were performed using the Vanquish ultra-high-performance LC (UHPLC) system (Thermo Fisher Scientific, Waltham, MA, USA) coupled to the Orbitrap Q Exactive HF-X mass spectrometer (Thermo Fisher Scientific). Samples were injected onto the Hyperil Gold column (100 × 2.1 mm, 1.9 μm) using the 16 min linear gradient at a flow rate of 0.2 ml/min. Eluents for the positive polarity mode were eluent A (0.1% Formic Acid in water) and eluent B (methanol). Eluents for the negative polarity mode were eluent A (5 mM ammonium acetate, pH 9.0) and eluent B (methanol). The solvent gradient was set as follows: 2% B, 1.5 min; 2–100% B, 12.0 min; 100% B, 14.0 min; 100–2% B, 14.1 min; 2% B, 16 min. The Q Exactive HF-X mass spectrometer was operated in the positive/negative polarity mode with a spray voltage of 3.2 kV, capillary temperature of 320°C, sheath gas flow rate of 35 arb, and aux gas flow rate of 10 arb.
Untargeted metabolomics data analysis
The raw data files generated by UHPLC-MS/MS were processed using the Compound Discoverer 3.0 (CD 3.0; Thermo Fisher Scientific) to perform peak alignment, peak selection, and quantitation for each metabolite. The main parameters were set as follows: retention time tolerance, 0.2 min; actual mass tolerance, 5 ppm; signal intensity tolerance, 30%; signal/noise ratio, 3; and minimum intensity, 1,00,000. Then the peak intensities were normalized to the total spectral intensity. The normalized data were used to predict the molecular formula based on additive ions, molecular ion peaks, and fragment ions. Finally, the peaks were matched with the mzCloud (https://www.mzcloud.org/) and ChemSpider (http://www.chemspider.com/) database to obtain accurate qualitative and relative quantitative results.
Circulating microbiota
A total of 41 hypertensive patients and 28 age- and sex-matched healthy individuals (controls) were enrolled from Shunde Hospital, Southern Medical University. The whole blood was collected, and microbiome sequencing was performed by Novogene, Inc. (Beijing, China). Microbial DNA was extracted using the cetyltrimethylammonium bromide/sodium dodecyl sulfate method. DNA concentration and purity were monitored on 1% agarose gels. Based on the content, DNA was diluted to 1 ng/μl using sterile water. Subsequently, 16S rRNA/18S rRNA/ITS genes of distinct regions (e.g. 16S V4/16S V3/16S V3–V4/16S V4–V5, 18S V4/18S V9, ITS1/ITS2, Arc V4) were amplified used specific primer with the barcode. All polymerase chain reactions (PCR) were performed in 30 μl reactions with 15 μl Phusion® High-Fidelity PCR Master Mix (New England Biolabs, Ipswich, MA, USA), 0.2 μM forward and reverse primers, and ~10 ng template DNA. Thermal cycling consisted of initial denaturation at 98°C for 1 min, followed by 30 cycles of denaturation at 98°C for 10 s, annealing at 50°C for 30 s, elongation at 72°C for 30 s, and finally, 72°C for 5 min. After sequencing, sequence libraries were generated using Ion Plus Fragment Library Kit 48 rxns (Thermo Fisher Scientific) based on the manufacturer’s recommendations, followed by bioinformatics analysis.
Enzyme-linked immunosorbent assay
Plasma Aβ and tau concentrations were measured with enzyme-linked immunoassay (ELISA) kits (Thermo Fisher Scientific), and ELISA was performed according to previous studies.10,11 The absorbance at 450 nm was measured with a microplate reader (Perkin-Elmer, Waltham, MA, USA). Data were obtained from three separate experiments, each performed in triplicate.
Statistics
Continuous variables are presented as the mean ± standard deviation (SD) or median and interquartile range. Categorical variables are presented as the number and percentage. Baseline characteristics of hypertensive patients and controls were compared by the two-tailed t-test for normally distributed continuous variables, Wilcoxon rank-sum test for non-normally distributed continuous variables, and the chi-square test for categorical variables. The Pearson product-moment correlation coefficient (r) was calculated as a measure of linear association between MMSE and tau, and Aβ40 and Aβ42 in the controls and hypertensive patients, respectively, adjusting for age, smoking history, fasting plasma glucose (FPG), TG, TC, HDL-C, LDL-C, hs-CRP, and eGFR. Statistical analysis of the data was performed using GraphPad Prism 8.0 (GraphPad Software, La Jolla, CA, USA). All of the p-values were two-tailed and values of p < 0.05 were considered to be statistically significant.
Results
Baseline characteristics
To explore the metabolic change and its correlation with cognitive function, we included 36 healthy individuals (17 male and 19 female) and 33 hypertensive patients (17 male and 16 female) in this study (As shown in Table 1). Systolic BP was higher in hypertensive patients (142.4 ± 19.6 mm Hg) compared with healthy controls (125.5 ± 17.7 mmHg) (p < 0.001; Table 1). Patients had a lower eGFR (77.5 ± 21.6 ml/min/1.73 m2) and LDL-C (2.56 ± 0.60 mmol/L) than the healthy controls (eGFR: 89.4 ± 20.7 ml/min/1.73 m2; LDL-C: 2.92 ± 0.80 mmol/L, respectively) (p = 0.023 for eGFR and p = 0.039 for LDL-C, respectively; Table 1). Other baseline characteristics such as age, gender, HbA1c, and hs-CRP showed no obvious difference between these two groups.
Hypertension is associated with a worse cognitive function
Given that high BP is a risk factor for cognitive decline, we examined plasma Aβ40, Aβ42 and tau concentrations, and MMSE scores in patients with hypertension. We found elevated plasma Aβ40 and decreased Aβ42/Aβ40 ratio in subjects with hypertension compared with the controls (Aβ40: p = 0.0014, Aβ42/Aβ40 ratio: p = 0.002), whereas plasma Aβ42 and tau showed no obvious changes between the healthy controls and hypertensive patients (Aβ42: p = 0.2081, tau: p = 0.3145) (Figure 1A–C; Supplemental Figure 1A). In addition, we performed MMSE evaluation and found that MMSE scores were decreased in hypertensive patients compared with healthy controls (P = 0.0247; Figure 1D), which was consistent with our ELISA results showing a decreased Aβ42/Aβ40 ratio in hypertensive patients. Next, we performed Pearson’s correlation analysis to further evaluate the correlation among plasma Aβ40, Aβ42, tau concentrations, and MMSE evaluation. We found that plasma Aβ40 and tau concentration were negatively correlated with MMSE scores, while the Aβ42/Aβ40 ratio was positively correlated with MMSE scores in healthy controls (Figure 1E, I; Supplemental Figure 1B), suggesting that these parameters may be indicators for cognitive evaluation. Importantly, in hypertensive patients, we found a close correlation among plasma Aβ40, Aβ42/Aβ40 ratio, and MMSE scores (Figure 1F, J), which was consistent with altered Aβ40 concentration and Aβ42/Aβ40 ratio in patients with hypertension. However, we did not find a correlation between plasma Aβ42 concentration and MMSE scores in healthy controls (Figure 1G), or a correlation between plasma Aβ42, tau concentration and MMSE scores in hypertensive patients (Figure 1H; Supplemental Figure 1C). These results suggest that the increased Aβ40 concentration and decreased Aβ42/Aβ40 ratio were closely correlated with MMSE evaluation, and cognitive impairment existed in hypertensive patients in our study.

Metabolic landscape in hypertensive patients and controls
Because dysfunctional metabolism contributes to hypertension, in this study we performed LC-MS/MS to reveal the metabolic changes in hypertensive patients. A distinct separation of metabolites between controls and hypertensive patients was found by both three-dimensional principal components analysis and partial least squares-discriminant analysis (PLS-DA) score plots in the positive and negative ion mode, which indicated the significantly different metabolic profiles (Figure 2A–F). A total of 2604 compounds were identified in the hypertensive patients and healthy control individuals through metabolomics. Based on the variable importance in projection (VIP > 1.0) calculation through PLS-DA, we listed the significantly changed metabolites. In the positive ion mode, 28 compounds were increased and 231 compounds were decreased (Figure 2G); while in the negative the ion mode, 21 compounds were increased and 66 compounds were decreased (Figure 2H). Thus, a total of 49 and 297 compounds including metabolites, pollutants, medicines, and food additives had significantly increased or decreased abundance. To further evaluate the metabolic similarities and differences of each significantly biosynthesized metabolite, correlation analysis and hierarchical cluster analysis approaches were conducted. Correlation analysis of differential metabolites is shown in Figure 2I and J. Figure 3A shows that the metabolism markedly differed between the patients and the control group. Then the changed metabolites were subjected to the known metabolic relation network database, namely, Kyoto Encyclopedia of Genes and Genomes (KEGG). KEGG results suggested that the downregulated metabolic network primarily consisted of the benzoate degradation pathway, phenylalanine metabolism, tyrosine metabolism, phenylpropanoid biosynthesis, citrate cycle, and phospholipid metabolism (Figure 3B, C).

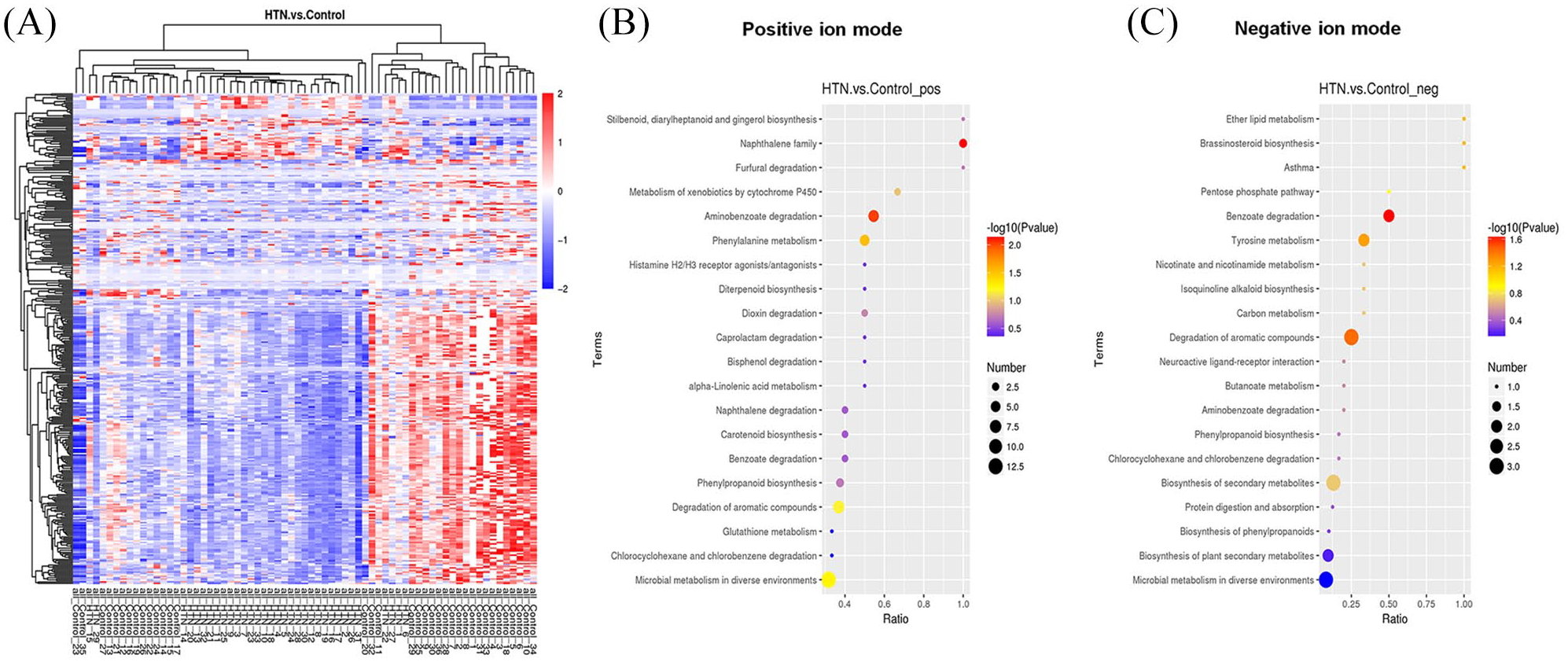
We showed the global metabolic network in which the changed metabolites in hypertensive patients compared with healthy individuals were involved (Figure 4). At the pathway and network levels, benzoate and acetyl-CoA were the key metabolites connected to different pathways. The identified compounds with a benzene ring were converted to benzoate before degradation through the citrate cycle. We showed the representative changed compounds enriched in the benzoate degradation pathway between controls and hypertensive individuals (Figure 5A–O). These compounds, containing phenylglyoxylic acid, benzoate, naphthalene, 1-naphthol, phenanthrene, phenol, 2,3-naphthalenediol, biphenyl, guaiacol, eugenol benzoate, cis,cis-muconic acid, 4-hydroxy-3-methylbenzoic acid, hydroquinone, 3-butylidenephthalide and naproxen, were significantly decreased in the plasma of hypertensive patients (Figure 5A–O). For phenylpropanoid biosynthesis, we listed the important metabolites involved in phenylpropanoid biosynthesis (Figure 6A–I). We found that caffeate (caffeic acid), caffeic acid phenethyl ester, vanillin, 4-methoxycinnamaldehyde, scopoletin, eugenol, 3,4-dimethoxycinnamic acid, ethyl methoxycinnamate, and cinnamaldehyde were significantly decreased in hypertensive patients compared with the controls (Figure 6A–I).



Correlation analysis between the altered metabolites and cognitive impairment in hypertensive patients
Because we found significantly changed metabolites in hypertensive patients, and we also found a cognitive decline in these hypertensive patients, we examined the correlation of representatively changed metabolites with cognitive indicators in hypertensive patients using Pearson’s correlation. We found that the representative metabolites, benzoate, naphthalene, phenanthrene, phenol, 2,3-naphthalenediol, guaiacol, eugenol benzoate, 3-butylidenephthalide, and naproxen, which were enriched in the benzoate degradation pathway, were statistically significantly positively correlated with MMSE (Supplemental Figure 2B, C, E–G, I, J, N, O). These metabolites were decreased in hypertensive patients, consistent with the decreased MMSE score in these individuals, whereas other metabolites showed no obvious correlation with MMSE score (Supplemental Figure 2A, D, H, K, L, M). In addition, the representative metabolites, caffeate, caffeic acid phenethyl ester, vanillin, 4-methoxycinnamaldehyde, scopoletin, eugenol, ethyl methoxycinnamate, and cinnamaldehyde, which were enriched in the phenylpropanoid biosynthesis pathway, were statistically significantly positive correlated with MMSE (Supplemental Figure 3A–F, H, I), with the exception of 3,4-dimethoxycinnamic acid (Supplemental Figure 3G). Since Aβ42/Aβ40 ratio is a promising evaluation of cognitive function, we also correlate these representative metabolites with Aβ42/Aβ40 ratio. Here, our results showed that Phenylglyoxylic acid, Naphthalene, 1-Naphthol, Phenanthrene, 2,3-Naphthalenediol, Biphenyl, Guaiacol, Eugenol benzoate, and Naproxen, which were enriched in the benzoate degradation pathway, were statistically significant positive related with Aβ42/Aβ40 ratio (Figure 7A, C–E, G–J, O). While other compounds showed no significant correlation with Aβ42/Aβ40 ratio (Figure 7B, F, K–N). Regarding the phenylpropanoid biosynthesis pathway, Caffeic acid phenethyl ester, 4-Methoxycinnamaldehyde, Scopoletin, Eugenol, 3,4-Dimethoxycinnamic acid, Ethyl methoxycinnamate, and Cinnamaldehyde were statistically significant positively related with Aβ42/Aβ40 ratio (Figure 8B, D–I), except Caffeate and Vanillin (Figure 8A, C). Thus, our results confirmed that the abundance of key metabolites enriched in the benzoate degradation and phenylpropanoid biosynthesis pathways were significantly associated with MMSE score and Aβ42/Aβ40 ratio, which suggest that these key metabolites may contribute to the cognitive decline we observed in hypertensive patients.

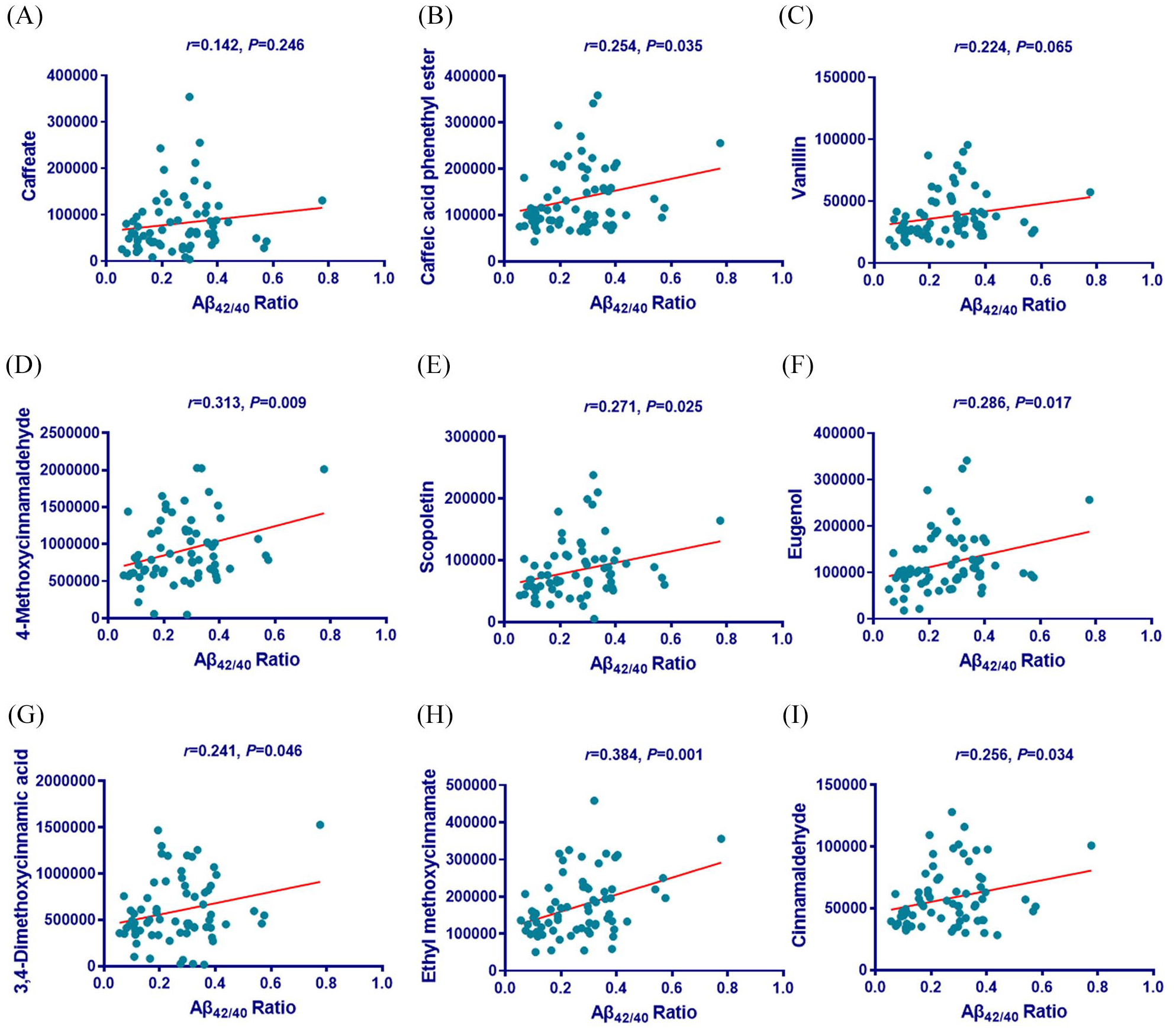
Correlations between the representative changed metabolites enriched in the phenylpropanoid biosynthesis pathway and Aβ42/Aβ40 ratio. (A–I) The correlations between the representative differential metabolites enriched in the phenylpropanoid biosynthesis pathway and Aβ42/Aβ40 ratio are shown. Correlation coefficients (r) and p-values from Pearson correlation analysis are shown.
Circulating microbiota in hypertensive patients
Because our metabolomic KEGG results revealed that the changed metabolites were enriched in microbial metabolism in diverse environments (Figure 3B, C) and circulating microbiota contributes to the pathogenesis of hypertension, 12 we further examined the microbial communities at the whole blood level using metagenomic approaches. We found that nearly 100 species of microbes were identified in the whole blood samples. At the phylum level, Firmicutes, Bacteroidetes, Proteobacteria, Cyanobacteria, and Actinobacteria were the highly abundant microbes, occupying about 90% of the total abundance in samples of both patients and the controls (Supplemental Figure 4). We also found that β-diversity was decreased in the hypertensive group compared with the controls (p = 0.018; Figure 9A), and the principal coordinates analysis plots of these two groups are shown in Figure 9B. However, α-diversity, including the observed species, Chao1, ACE, Shannon, Simpson, and Phylogenetic diversity (PD) whole tree showed no obvious changes between controls and hypertensive patients (Supplemental Figure 5A–F). Next, we used 16S rDNA gene sequencing to determine the alterations in blood microbiota composition between healthy and hypertensive patients. The taxon abundance of each sample was classified in terms of phylum, class, order, family, genus, and species levels, mainly using the small subunit ribosomal RNA reference database. The hierarchy cluster heat-map revealed the top 35 most abundant differentiated taxa (Figure 9C), and the abundance of top 10 species at the genus level in the blood is shown in Figure 9D. We also performed a t-test comparison to show the conspicuous group differences at the genus levels. According to KEGG pathway annotation, the number of different genes enriched in the different pathways is shown (Supplemental Figure 6A). We also revealed the relative abundance of top 10 functional pathways at the genus level between controls individuals and hypertensive patients (Supplemental Figure 6B). The functional prediction based on FAPROTAX analysis confirmed that the microbes accessed the patient circulating system primarily through gastroenteritis, diarrhea, pneumonia, and intracellular parasites (Supplemental Figure 6C). The reported datasets related to gut microbiota confirmed that Bacteroidetes and Firmicutes dominated the adult gut microbiota followed by Actinobacteria, Verrucomicrobia, and Proteobacteria. 13 Those results were consistent with the abundance of microbes in the current study, which confirmed that the blood microbiota primarily stemmed from the gut. We found representative changes in six blood microbiota at the genus levels, and found that Stenotrophomonas and Turicibacter were significantly decreased in the whole blood of hypertensive patients compared with the controls (Figure 9E, F), whereas Streptococcus, Lactococcus, Parabacteroides, and Helicobacter were significantly increased in hypertensive patients compared with the controls (Figure 9G–J).

Discussion
Hypertension is associated with cognitive decline and pathological characteristics in AD
Recently, some groups reported that dementia was highly associated with cardiovascular disease, such as coronary heart disease and congestive heart failure.14,15 Hypertension is a significant risk factor for cardiovascular disease, and they share aging as the most important risk factors. Thus, hypertension is closely correlated with cognitive decline, and vice versa, cognitive impairment is related to hypertension severity.16,17 Moreover, antihypertensive approaches show potential benefits in improving cognitive function in elderly hypertensive patients. 18 Importantly, several studies have explored the underlying mechanisms between high blood pressure and cognitive impairment, and the evidence may lie on the disruption of the neurovascular unit, age-related insulin-like growth factor 1 deficiency, neurovascular oxidative stress, and neuroinflammation.19–21 Intriguingly, during aging, high blood pressure reduced cerebral hemodynamics and connectivity and led to cognitive impairments in a genetic AD mouse model. 22 In addition, Nazarian et al. also reported the genetic association between AD and hypertension. 23 However, the metabolic mechanisms in the relationship between hypertension and cognitive impairment have yet to be fully elucidated.
In this study, we found that plasma Aβ40 was increased and Aβ42/Aβ40 ratio was decreased in subjects with hypertension. It is well established that extracellular deposition of amyloid plaques and intracellular neurofibrillary tangles (NFTs) are the two major hallmarks of AD. Aβ40 and Aβ42, the main components of the amyloid plaque, are derived from the amyloid precursor protein, which is cleaved by β-secretase and γ-secretase. In physiological conditions, Aβ40 is the most abundant species, while Aβ42 is the most toxic due to its high hydrophobicity; Aβ42 has a high tendency for aggregation. Usually, Aβ is cleared through the blood–brain barrier (BBB), and plasma Aβ40 level is much higher than Aβ42. In this study, we found elevated plasma Aβ40 rather than Aβ42 in subjects with hypertension compared with the controls, consistent with a previous report. 24 Although Aβ deposition in the brain is the pathological hallmark of AD, a lower plasma Aβ42/Aβ40 ratio was recently found to be highly associated with AD dementia.25–28 Circulating Aβ42/Aβ40 ratio (plasma or CSF) could reflect the function of the BBB, which is responsible for Aβ clearance. However, as a more easily detectable method, circulating Aβ42/Aβ40 ratio could predict the MCI and prevent the aggravation of AD dementia. In this study, we also found that the plasma Aβ42/Aβ40 ratio was decreased in patients with hypertension, which was closely correlated with decreased MMSE scores in these patients. Thus, our results indicate that cognitive decline may occur in hypertensive patients, consistent with other reports that hypertension induces cognitive impairment and memory deterioration.29–31
Regarding the potential mechanism, we conclude that, on one hand, high BP causes several acute effects, and the most established in the brain is stroke. In this case, high BP disrupts the neurovascular units via breakdown of cerebral vasculature, leading to brain damage and cerebral degeneration. 2 In addition, hypertension damages the BBB, impairs microvascular deposition and clearance of Aβ and NFTs through the BBB, and induces the production of Aβ and NFTs.32,33 Moreover, dietary factors also contribute to cognitive decline and vascular disease, which may involve hypertension via gut–brain communication. 34
Benzoate degradation and phenylpropanoid biosynthesis are the most altered pathways in hypertensive patients
In this study, we found that the key metabolites enriched in benzoate degradation and phenylpropanoid biosynthesis were decreased in hypertensive patients. Pollutants with an aromatic ring were converted in the pathway of benzoate degradation connected citrate cycle through salicylate and acetyl-CoA. Among these aromatic compounds, polycyclic aromatic hydrocarbons phenanthrene, anthracene, and naphthalene are regarded as the priority control pollutants by both the U.S. Environmental Protection Agency and the European Environment Agency because of their toxicity and persistence. The primary sources of these compounds’ exposure to humans are polluted air, from ambient air and indoor air, and foodstuffs, especially those prepared through cooking methods of baking, frying, and grilling. 35 Because of the similar quality of ambient air for people living in the same district, the low abundance of polycyclic aromatic hydrocarbons and their metabolites 1-naphthol and 2,3-naphthalenediol in the samples of hypertensive patients was primarily due to the low intake of grilled foods, followed by low inhalation exposure to tobacco smoking. Other aromatic compounds, including 4-hydroxy-3-methylbenzoic acid, 4-methoxybenzoate, and eugenol benzoate, were converted to benzoate by the catalysis of oxygenases or through anaerobic respiratory chains using Fe(III) or sulphate as terminal electron acceptors. 36
For compounds in the pathway of phenylpropanoid biosynthesis, caffeate is a major phenolic phytochemical in coffee with anti-inflammatory properties, the ability to suppress DNA methylation in cancer cells, and inactivation of several ways participating in tumorigenic processes. Caffeate significantly inhibits intracellular superoxide anion generation and cell proliferation induced by angiotensin II, which stimulates proliferative and hypertrophic growth in vascular smooth muscle cells. 37 Furthermore, caffeate inhibits tyrosine phosphorylation. The decreased abundance of this compound leads to the high risk of damage in the DNA and target proteins of hypertensive patients. This inference was supported by the direct evidence that the abundance of its phenethyl derivatives, such as phenethyl caffeate and caffeic acid phenethyl ester, with similar function also decreased. Caffeic acid phenethyl ester, an active polyphenolic component of honeybee propolis, has been demonstrated to have many medicinal properties, such as antitumor and neuroprotection. Phenethyl caffeate has a role as an antineoplastic agent, anti-inflammatory agent, immunomodulator, antioxidant, neuroprotective agent, antiviral agent, and antibacterial agent. Coffee is a mixture of compounds containing flavonoids, chlorogenic acid, melanoidins, and various lipid-soluble compounds. It is difficult to make comparisons between studies of coffee consumption from different regions due to different preparation methods and components of coffees. Some studies have suggested that coffee consumption increases BP, whereas the SUN Project confirmed that consumption of regular coffee can reduce the risk of hypertension among women. 38
The aromatic amino acid biosynthesis pathway is one of the core metabolic pathways that lead to the production of aromatic compounds, including phenylalanine and tyrosine. 39 Phenylalanine is the key metabolite connected with tyrosine metabolism, the benzoate degradation pathway, and ethyl methoxycinnamate transformation. The abundance of phenylalanine is stable and that of its intermediates phenylethyl alcohol and phenylglyoxylic acid was decreased in the current study, suggesting that the generated phenylalanine was prioritized for protein biosynthesis. In this study, based on circulating metabolomics, we identified the differential metabolites enriched in the pathway of tyrosine metabolism, and also found that some medicinal compounds and food additives were changed in hypertensive patients; these are listed in the Extended Data.
Cognitive decline in hypertensive patients may be associated with metabolic changes
We also found that some metabolites enriched in the benzoate degradation and phenylpropanoid biosynthesis pathways were correlated with MMSE and Aβ42/Aβ40 ratio. Several compounds reportedly regulate cognitive function; for example, benzoate has been shown to improve cognitive function in patients with chronic schizophrenia. 40 Because the phenylpropanoid pathway is suggested to be involved in modulating cognitive function, 41 caffeate and caffeic acid phenethyl ester have been proven to protect cognitive deficit in the animal model of AD and cerebral ischemia by regulating neuroinflammation and inhibiting lipid peroxidation and nitric oxide production.42–45 In addition, vanillin has been shown to attenuate scopolamine-induced cognitive impairment via modulating the DNA-binding protein inhibitor ID-1 and neuronal differentiation.46,47 Ethyl methoxycinnamate reportedly improves cognitive impairment independent of direct acetylcholinesterase and glutamate receptor inhibition in an aluminum-induced memory deficit rat model. 41 Moreover, cinnamaldehyde attenuates cognitive deficits via regulating extracellular signal-regulated kinase signaling in the prefrontal cortex. 48
Taken together, most of the metabolites enriched in the phenylpropanoid pathway were involved in improving cognitive performance, and according to its significant correlation with MMSE and Aβ42/Aβ40 ratio, we conclude that this pathway may contribute to the cognitive decline in hypertensive patients. Though little is known about the role of metabolites in the benzoate degradation pathway in regulating cognitive function, considering their distinct correlation with MMSE and Aβ42/Aβ40 ratio, further studies are needed to clarify these compounds in regulating cognitive performance.
Circulating microbiota reveals a novel underlying mechanism in hypertension
The relative abundance of microbes with the function of aromatic compound degradation was significantly higher in the hypertension group. This result is partially responsible for the lower abundance of aromatic compounds in patient blood. For sulfur transformation, the function of microbes, including Geobacter, Thiobacillus, Desulfovibrio, Ferruginibacter, and Acidibacter, in patients and healthy individuals is related to the respiration of sulfur compounds and respiration of sulfate. Therefore, the metabolism of organic compounds containing sulfur is enhanced in hypertensive patients, which is partially responsible for the decrease in abundance of methylsulfonylmethane, methionylserine, 3-thiotolene, 3-mercaptopropane-1,2-diol, S-3-oxodecanoyl cysteamine, laurilsulfate, 2-ethylhexyl hydrogen sulfate, 3,4-dehydrothiomorpholine-3-carboxylic acid, and S-3-oxodecanoyl cysteamine. In the control samples, microbes for sulfur respiration primarily used sulfate as an electron acceptor to oxidize organic carbon compounds or hydrogen under anaerobic conditions with the production of aqueous sulfide. 49
Bioconversions of nitrogen by nitrogen-transforming microorganisms are often depicted as a cycle consisting of distinct processes that proceed in an orderly fashion. 50 In the current study, microbes in the blood of patients for nitrogen metabolism were primarily involved in nitrite ammonification and nitrogen fixation. In the nitrite ammonification processes, nitrite was converted to ammonia through the reaction equations 1–4.




Dinitrogen in blood is further reduced into ammonia via N2 fixation (equation 4). Based on these reactions, the abundance of nitrite in the patient’s blood would decrease. In the control samples, the functions of microbes were primarily associated with nitrate reduction, nitrogen respiration, and nitrate respiration. Nitrate reduction is the reaction catalyzed by nitrate reductase with the conversion of NO3− to NO2−. 51 Nitrate respiration processes proceed by using nitrate as an electron acceptor at low oxygen concentrations resulting in the production of dinitrogen. The absence of the microbes with ammonia oxidation and nitrite oxidation functions confirms that these microbes dwelled in anaerobic conditions, for example, in the gut. Some microbes that trigger gastroenteritis and diarrhea are the direct evidence that verified the above inference. An analysis clarified that a nitrate-rich diet is effective in normalizing BP values. 52 Modulating the nitrate/nitrite-reducing bacterial communities to increase nitrite production is a potential solution to treat hypertension. Therefore, a lower nitrite/nitrate ratio in the patient’s blood in the current study is harmful to the patient.
Conclusion
In this study, we showed cognitive impairment in hypertensive patients, and based on the metabolomics and microbiome, we identified the differential circulating metabolites and microbiota between controls and hypertensive patients. Collectively, our results provide a novel metabolic and microbial mechanism that may underlie cognitive impairment in hypertension, and the altered metabolites and microbiota may be potential therapeutic targets in treating hypertension.
Supplemental Material
Extended_Data – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Extended_Data for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Supplemental Material
Supplemental_Table_1-Positive_ion_model-All_metabolites – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Supplemental_Table_1-Positive_ion_model-All_metabolites for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Supplemental Material
Supplemental_Table_2-Positive_ion_model-Differential_metabolites – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Supplemental_Table_2-Positive_ion_model-Differential_metabolites for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Supplemental Material
Supplemental_Table_3-Negative_ion_model-All_metabolites – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Supplemental_Table_3-Negative_ion_model-All_metabolites for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Supplemental Material
Supplemental_Table_4-Negative_ion_model-Differential_metabolites – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Supplemental_Table_4-Negative_ion_model-Differential_metabolites for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Supplemental Material
Supplementary_Figures – Supplemental material for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension
Supplemental material, Supplementary_Figures for Circulating metabolomics profiling reveals novel pathways associated with cognitive decline in patients with hypertension by Yuli Huang, Haoxiao Zheng, Kuan Tan, Xiangdong Sun, Jinshao Ye and Yunlong Zhang in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Yuli Huang, Haoxiao Zheng, and Kuan Tan performed the data collection. Yuli Huang and Xiangdong Sun completed the data analysis. Yuli Huang, Jinshao Ye, and Yunlong Zhang reviewed articles, and wrote the manuscript. Yunlong Zhang designed the study and revised the manuscript. All the authors read and approved the final version of the manuscript.
Data statement
The raw datasets generated and analyzed for this study can be viewed on request from the Authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81704130 to YZ, No. 21876064 to JY), the Science and Technology Planning Project of Guangzhou (No. 201904010238 to YZ), the Natural Science Foundation of Guangdong Province of China (No. 2017A030310643 to YZ), the Science and Technology Innovation Project from Foshan, Guangdong (No. FS0AA-KJ218-1301-0006 to YH), the Clinical Research Startup Program of Shunde Hospital, Southern Medical University (No. CRSP2019001 to YH), and the Startup Research Fund of Guangzhou Medical University (No. B195002002045 to YZ).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
