Abstract
Multiple sclerosis (MS) is an autoimmune inflammatory demyelinating central nervous system disorder that is more common in women, with onset often during reproductive years. The female:male sex ratio of MS rose in several regions over the last century, suggesting a possible sex by environmental interaction increasing MS risk in women. Since many with MS are in their childbearing years, family planning, including contraceptive and disease-modifying therapy (DMT) counselling, are important aspects of MS care in women. While some DMTs are likely harmful to the developing fetus, others can be used shortly before or until pregnancy is confirmed. Overall, pregnancy decreases risk of MS relapses, whereas relapse risk may increase postpartum, although pregnancy does not appear to be harmful for long-term prognosis of MS. However, ovarian aging may contribute to disability progression in women with MS. Here, we review sex effects across the lifespan in women with MS, including the effect of sex on MS susceptibility, effects of pregnancy on MS disease activity, and management strategies around pregnancy, including risks associated with DMT use before and during pregnancy, and while breastfeeding. We also review reproductive aging and sexual dysfunction in women with MS.
Introduction
Multiple sclerosis (MS) is an autoimmune demyelinating disease of the central nervous system (CNS). Several factors implicate chromosomal sex and hormones in susceptibility and disease course in MS. MS is more common in women, with a female to male sex ratio of 3:1, 1 whereas before puberty and after menopause the sex ratio approaches 1:1.2–4 MS most commonly begins between 20 years and 40 years of age, and thus women of reproductive age are most commonly affected.
Whereas relapse rate decreases during pregnancy, there tends to be an increased relapse rate postpartum, 5 and relapse rate decreases after menopause. 6 Management of women with MS throughout their reproductive lifespan requires consideration of effects of pregnancy and breastfeeding, including understanding disease-modifying therapy (DMT) effects on children of women with MS. Hormonal factors may influence disability progression, as progression tends to occur earlier in men, 7 and later during the perimenopausal period in women.8,9
In this review, we discuss effects of sex on disease susceptibility, implications of MS on fertility and pregnancy, including peripartum DMT and other management considerations, the impact of pregnancy on the course of MS, the interaction between reproductive aging and MS, and sexual dysfunction in women with MS.
Susceptibility to MS
It has long been recognized that MS is more common in women, but recent observations suggest the sex ratio may be increasing due to a rise in cases in women over the last century. 10 An increasing sex ratio has been reported in several countries (e.g., in Canada the sex ratio increased from 1.9 to 3.2, and in Sweden from 1.7 to 2.7 for patients born in the 1930s compared with the 1980s).1,11 However, there appear to be regional differences in the changing sex ratio as this has not been observed in New Zealand, 12 and may differ by latitude. 13 More recently, the sex ratio was stable in Ontario, Canada for MS onset from 1996 to 2013. 14 A rise in incidence over the last century is too short for a genetic cause and suggests a sex by environmental factor interaction. While some question whether the rising incidence is confounded by better diagnostics, others urge searching for an environmental cause of the observed increased incidence in women. There have been many changes in women’s lifestyles in recent decades: a later age at first pregnancy, increased availability and use of hormonal contraception, a lower rate of childbirth, and higher rates of employment and smoking.
Many of these factors have been examined in the Danish MS Registry. 15 Having children reduced the risk of MS in women (but not in men) by about 46% during the following 5 years. This may be due to temporary immunosuppression during pregnancy, although reverse causation secondary to an “MS prodrome” resulting in fewer pregnancies in women with subclinical MS cannot be excluded. 16
It is likely that the increased susceptibility of women towards MS is influenced by genetic, hormonal, and environmental factors.
Genetics and epigenetics
The genetic predisposition of MS is approximately 25%, based on monozygotic twin studies. 17 With the success of genome wide association studies (GWAS) over the last decade, 48% of this heritability has been explained with 233 statistically independent loci (32 in the MHC region). 18 Carrying HLA-DRB1*15:01 accounts for 10.5% of the genetic risk for MS.
Leveraging the largest GWAS of nearly 50,000 MS cases, one single nucleotide polymorphism (SNP rs2807267, closest gene VGLL1) on the X chromosome has been associated with MS. 18 The SNP lies within an enhancer peak specific for T cells, and additional study is required to understand its functional consequence in MS. No susceptibility alleles have been identified on the Y chromosome.
Interaction between the genome and sex-specific biological and environmental factors may underlie at least part of the possible increase in MS incidence in women. One aspect of this may be sex-specific epigenetic changes. Maternal imprinting of the X chromosome or X dosage effects may contribute to autoimmunity in women. 19 Voskuhl et al. demonstrated differential methylation and expression of genes on the X chromosome in T lymphocytes from females versus males.20,21 In addition, an X chromosome gene (Kdm6a) that escapes X inactivation, with two copies expressed in females and one in males, is a histone demethylase that influences autosomal genome wide expression and is proinflammatory in T lymphocytes. 22 It is thus conceivable that a key environmental factor, which has changed over the last century, may interact with genes on either sex chromosomes or autosomes to create increased risk in women.
Hormones
The effects of puberty, pregnancy, and menopause – periods during which sex hormone levels change dramatically – have been the subject of several studies. Puberty represents a risk factor for MS; earlier age of menarche has been associated with increased risk of MS and younger onset of MS symptoms in women.23,24 In pediatric MS, girls largely present 2 years after menarche 25 ; the immune system may be stimulated by sex steroid hormones during puberty. 26 Additional work is needed to parse out the specific biological mechanism of the epidemiologic association of puberty with MS risk. As mentioned previously, nulliparous women may have higher risk of MS than those who had several pregnancies. 15 To reconcile this increased risk with female puberty onset and decreased risk with multiparity, estrogens have been shown to have a biphasic dose effect, being immunostimulatory at low levels consistent with menstrual cycling, while being immunosuppressive at high levels of pregnancy.27,28
Environmental
Environmental factors likely play a large role in MS risk given the 75% discordance rate amongst identical twins. 17 The most replicated environmental risk factors for MS include: active and passive smoking, Epstein Barr virus (EBV) seropositivity, low serum levels of vitamin D, and low sunlight exposure. Biological sex may interact with some of these factors to increase MS risk.
Smoking
Smokers of both sexes have increased risk of developing MS (odds ratio 1.4); risk increases with cumulative smoking dose.29,30 Data from The Swedish National Institute of Public Health showed that, at the beginning of the 21st century, 20–25% women smoked (compared with 15–17% men). United Kingdom (UK) smoking prevalence has been increasing in women throughout the 20th century, which has been hypothesized to contribute to increasing MS risk. 31
EBV
EBV is a ubiquitous gamma herpes virus. In adulthood, ~95% of the general population have evidence of prior EBV exposure; this proportion approaches 100% in MS. 32 While, on average, men seroconvert to positive EBV status at a slightly later age than women, there is no clear evidence that EBV plays a role in driving the unequal sex ratio of MS.
Vitamin D/sunlight exposure
The move away from outdoor-based lifestyles may be driving a reduction in serum vitamin D levels in the population. It is not known if women are more susceptible to downstream effects of low vitamin D, 33 but a study in an animal model demonstrated protection from experimental autoimmune encephalitis (EAE) with vitamin D only in female mice. 34 Low sunlight exposure is also associated with MS risk, with potential sex-specific effects of ultraviolet radiation exposure. 35
More data are needed to identify hormonal and environmental risk factors for MS, which act preferentially in women.
Fertility and contraception
Fertility and assisted reproductive technology
There are no studies that directly assess pregnancy success rates in MS,36,37 though some epidemiological studies have shown that women with MS may have fewer children than the general population.36,38,39 Potential underlying reasons for this could include the effect of autoimmune disease on fertility, the contributions of symptoms such as fatigue, sexual dysfunction, and bladder impairment on attempting pregnancy, or the individual’s decision to conceive being influenced by her disease.36,37,40 Certain older DMTs, particularly cyclophosphamide and mitoxantrone,41,42 may also impair fertility. Rigorous analyses for newer drugs are limited. More recent studies have assessed markers of ovarian reserve and function in women with MS, including levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), and anti-Mullerian hormone (AMH), and antral follicle count.9,43,44 In one study of 76 women with relapsing MS (most of whom were not taking DMT) and 58 controls, women with MS had reduced ovarian reserve (lower AMH level) compared with healthy controls. 43 Another study showed those with higher disease activity had lower AMH levels than those with lower disease activity. 44 However, in a study of 412 women with MS [mostly relapsing remitting (RRMS) and using injectable therapies] and 180 healthy controls, there was no difference in AMH level for women with MS compared with controls after adjustment for chronological age, birth control/hormonal therapy use, body mass index (BMI), and smoking. 9
Regardless of any potential impact of MS on fertility, 12% of women in the general population confront infertility and may turn to assisted reproductive technologies (ART). 45 ART involves administration of hormonal medications, and may include several procedures in vitro on oocytes and sperm, or on embryos, to establish a pregnancy. Artificial insemination (INSE) may be performed either with in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI). 46 Although the effect of ART on the immune system in women with MS requires further study, there are reports of increased MS activity after ART (Table 1).47–50 Hellwig et al. observed that 12 of 23 women with MS relapsed within 3 months after ART, and the difference in relapse rate pre- and post-ART was correlated with INSE procedure. 51 A small study of women treated with GnRH (gonadotropin releasing hormone) agonists and recombinant FSH observed increased clinical relapses and enhancing magnetic resonance imaging (MRI) lesions after ART. 52 Similarly, another study, wherein women with MS received GnRH agonist or antagonist, followed by FSH, found that the annualized relapse rate (ARR) increased during 3 months following ART, with correlation to GnRH agonist use and IVF failure. 53 A recent meta-analysis by Bove et al. 50 combined five published studies and reported a case series (n = 12).51–55 Whereas the Boston case series did not have higher ARR after ART compared with before, the overall meta-analysis including this cohort confirmed increased ARR after ART, with a mean ARR increase of 0.92 [95% confidence interval (CI) 0.33–1.51]. 50 On the other hand, Guzman-Soto et al. reported that leuprolide acetate, a synthetic analogue of GnRH used in IVF, has a neurotrophic effect on neurofilament, myelin basic protein expression, and axonal morphometry in EAE, thus opening horizons for studying protocols of ART in MS. 56
Summarized data from articles reporting on ART in women with MS.

ARR, annualized relapse rate; ART, assisted reproductive technology; GnRH, gonadotropin releasing hormone; IVF, in vitro fertilization; MS, multiple sclerosis.
In summary, ART, particularly the use of GnRH agonists, may increase MS disease activity in the short term, though further work is necessary to elucidate how induced hormonal changes may affect MS course.
Contraception
Contraception is an important topic in MS, particularly as women are often of childbearing age at disease onset and some DMTs are potential teratogens. 61 Multiple dimensions should be considered, including contraception effect on risk of MS and related disability, family planning, type of contraception available, and concurrent use with DMTs.
Prior studies have reported mixed effects of hormonal contraception on risk of developing MS.62–67 Different population-based, case-control, or cohort studies concluded a protective,65,66 neutral, 64 or even negative effect of oral contraceptive (OC) exposure on MS risk. 67 While the Nurses’ Health Study showed no effect of past or current use of OC on risk of MS, 64 a case-control study demonstrated decreased risk of MS in those using OC in the 3 years prior to MS onset, 65 and the Swedish MS registrar demonstrated that OC use before first MS symptoms was associated with an older age of MS onset. 66 On the other hand, a nested case-control study suggested a slightly increased risk of MS or clinically isolated syndrome (CIS) with former or current OC exposure, although this could have been due to an unmeasured confounder. 67 Limitations of most of these studies include observational design, small sample size, self-reported data on OC use, lack of information about OC hormonal composition and duration of exposure, and the potential for residual confounding. As such, definitive conclusions on the effect of OC on MS risk remains unclear.
There is scarce information about the effect of OC on long-term prognosis of MS, although, reassuringly, hormonal contraception does not seem to negatively affect disease progression or disability. 68 Two studies reported decreased risk of disability accumulation and conversion to secondary progressive MS (SPMS) in relapsing onset patients who had ever used OC.69,70 No significant differences in ARR between OC ever and never users were found. In contrast, D’Hooghe et al. described a shorter time from first symptom to reach Expanded Disability Status Scale (EDSS) 6.0 in OC users with primary progressive MS (PPMS). 71 The lack of consistency between studies could be partially explained by the influence of potential confounders that affect disease evolution and also determine the patient’s decision to take OC. Recently, a multivariable and time-dependent analysis applied to the Barcelona CIS cohort reported that OC use before or after CIS did not significantly influence the risk of MS or time to confirmed EDSS 3.0. 72 However, OC may have an impact in patients with established MS distinct to any effect on ARR due to transition from the more inflammatory early stage of disease to the more neurodegenerative stage, which may be more sensitive to neuroprotective effects of estrogens in OC. 73
Optimal contraceptive methods should be individualized, as women with MS may suffer from symptoms that make use of some methods difficult (such as using vaginal rings). 74 The US Medical Eligibility Criteria for Contraceptive Use, published by the United States (US) Centers for Disease Control and Prevention, outlines the safety of contraception in women with MS, and generally, the majority are felt to be safe in MS. Caution should be exercised in the use of combined hormonal contraceptives in individuals with prolonged immobility, due to increased thromboembolic risk. 75 Current DMTs do not appear to alter effectiveness of hormonal contraceptives,74,76 but there are limited formal drug–drug interaction studies, and symptomatic medications such as modafinil can decrease effectiveness of hormonal contraceptives. 74
Immunology of pregnancy: effects in MS
Immunological changes at the maternal–fetal interface
The maternal–fetal interface refers to the collocation between the uterus and extra-embryonic tissue. 77 On the fetal side, the blastocyst differentiates into an inner cell mass, the future fetus, and the outer extra-embryonic trophoblast. The trophoblast further separates into villous and non-villous cytotrophoblast and syncytiotrophoblast. 77 In preparation for potential conception, the endometrium undergoes a series of changes, or decidualization, that continue into pregnancy. 78 Decidualization requires a number of immune cells allowing trophoblast invasion (resulting in maternal–fetal interface), remodeling of spiral arteries, and placentation. 77 Decidualization is also important in the development of anti-microbial immunity. 79
Immunological changes in pregnancy and MS
Pregnancy affords protection from relapses in EAE and MS.5,80,81 Hormonal changes induced by pregnancy modulate immune response toward a state of tolerance to allow the semi-allogenic fetus to grow within the maternal uterus. Estrogen, progesterone, and human chorionic gonadotropin (hCG) modulate cells of the innate and adaptive immune system to adopt fetal-friendly phenotypes. 82 A shift from Th1 to Th2 response has been observed consistently in MS pregnancies,83–85 based on studies between 13 weeks and 27 weeks gestation. More recent data suggests active pro-inflammatory Th1 immunity before and after this period. 79 Regulatory T cells (Tregs) have been found to be increased,86,87 decreased, 88 or unchanged,89,90 probably due to different definitions of Tregs used across studies. The Th17 compartment seems unaffected by pregnancy, 88 whereas CD56bright natural killer (NK) cells were increased peripherally in one study. 88
In MS, pregnancy also alters the clonal composition of T cells toward a more uniformly distributed repertoire. 91 It induces a contraction of relapse-associated T cell clones, potentially contributing to reduced relapse rate from the first to third trimester. 91 Such clones re-expand after delivery in an individualized fashion. 91 Women gradually recover pre-pregnancy immunity along with decreased pregnancy hormones postpartum, which may lead to disease rebound, although immunological mechanisms are unclear. Tregs changed functionally in the early postpartum period in MS in one study, 89 and decreased numerically in another study. 88 Decline in CD4+, interferon-γ producing T cells, as well as in CD56bright NK cells, 89 has been correlated with postpartum MS relapses. 92 Although the immunopathogenic role of B cells is increasingly recognized, and these cells are sensitive to stimulation by female sex hormones, 93 there is a paucity of studies exploring whether they are modulated by pregnancy. Further studies investigating functional changes of immune cell subtypes are required to clarify complex relationships between pregnancy and immunomodulation. Indeed, the immune system during pregnancy is dynamic and responsive, promoting tolerance to fetal proteins and allowing fetal growth.79,89,94 Table 2 summarizes immunological changes by pregnancy trimester and effects on MS.
Immunological changes by pregnancy trimester and effects on MS.

APC, antigen presenting cells; ARR, annualized relapse rate; B cell, B lymphocyte; CD4 T lymphocytes, helper and regulatory type; CD8 T lymphocytes, cytotoxic type; GA, gestational age; MMP, matrix metalloproteinase; MP, macrophage; NF, nuclear factor; Tregs regulatory T lymphocytes; u-DC, uterine dendritic cells; u-NK, uterine natural killer cells.
Recommendations on planning a pregnancy in MS
Pregnancy planning is an important consideration for many women with MS. 103 Discussing DMT in the context of pregnancy and breastfeeding considerations is essential. It is generally recommended to establish pre-pregnancy baseline, through a clinical neurology visit and an MRI before pregnancy, and to choose a pregnancy-compatible DMT. Visits during the first and third trimester can be helpful, and, during the latter, breastfeeding and postpartum plans can be confirmed. Recommendations for postpartum management are outlined later in this review.
Ideally, women should aim for a period of disease and treatment stability prior to conception. When women receive maintenance DMT, care should be taken to proactively discuss future plans following conception – including whether or not to continue DMT during pregnancy and plans around breastfeeding. Such discussions are particularly pertinent in women with more active disease. DMTs with potential teratogenicity or contraindicated in pregnancy should be discontinued and replaced with acceptable alternatives prior to conception, or, in case of unintended pregnancy, changed as soon as able. In addition, the tendency for rebound activity after discontinuation of certain DMTs (fingolimod and natalizumab, as discussed later in the review) should be considered prior to initiating therapy in women with plans for pregnancy in the near future. The use of highly effective therapies without rebound risk, such as depleting antibodies, in women with more active disease prior to pregnancy may be preferable, as these may enable a balance between disease control and low potential exposure and risk to the fetus. In women with less active disease, continuing injectable therapies until conception, or even through pregnancy, appears safe, and may offer a favorable risk–benefit ratio. 104 Key recommendations are included in Box 1.
Key expert opinion recommendations for women planning pregnancy and postpartum.

Adequate vitamin D supplementation prior to conception and during pregnancy (up to 4000 IU/day) is important. 105 Timely commencement of folate-containing pre-natal vitamins, avoidance of active or passive smoking, pelvic floor exercises, and proactive diagnosis and treatment of urinary tract infections (UTIs) are also important. UTIs are associated with both worsening of MS symptoms and adverse pregnancy outcomes, and are of particular concern in this patient group. 106 Routine pre-natal and pregnancy care can be utilized, unless pregnancy is deemed to be high-risk due to specific obstetrical concerns. Women should be counselled about the risk of postpartum depression. Non-pharmacologic management of fatigue, insomnia, spasticity, and other symptoms during and after pregnancy should be pre-emptively discussed. Use of symptomatic therapies with potential fetal risk should be discussed with the neurologist and maternal fetal medicine specialist.
Pregnancy and DMTs
DMT safety before and during pregnancy
In the past decade, there has been an enormous increase in disease modifying treatment options in MS. Fortunately, most women with mild disease will remain relapse-free during pregnancy, and treatment can be safely stopped during pregnancy. At least 300 first-trimester pregnancy exposures, and preferably 1000 total exposures, are needed to assess the possible risk and safety of medication use during pregnancy. 107 However, rare events may only be captured with even more exposed pregnancies. Injectable therapies (glatiramer acetate and interferon-β) have the most comprehensive safety data available, and they are safe to continue at least up to conception.108–122 Many women stop treatment when they become aware of their pregnancy, most commonly during the first trimester. Therefore, very few data on entire pregnancy exposure exist, with the most data available for glatiramer acetate (Table 3). 123
DMT use in pregnancy and recommendations.
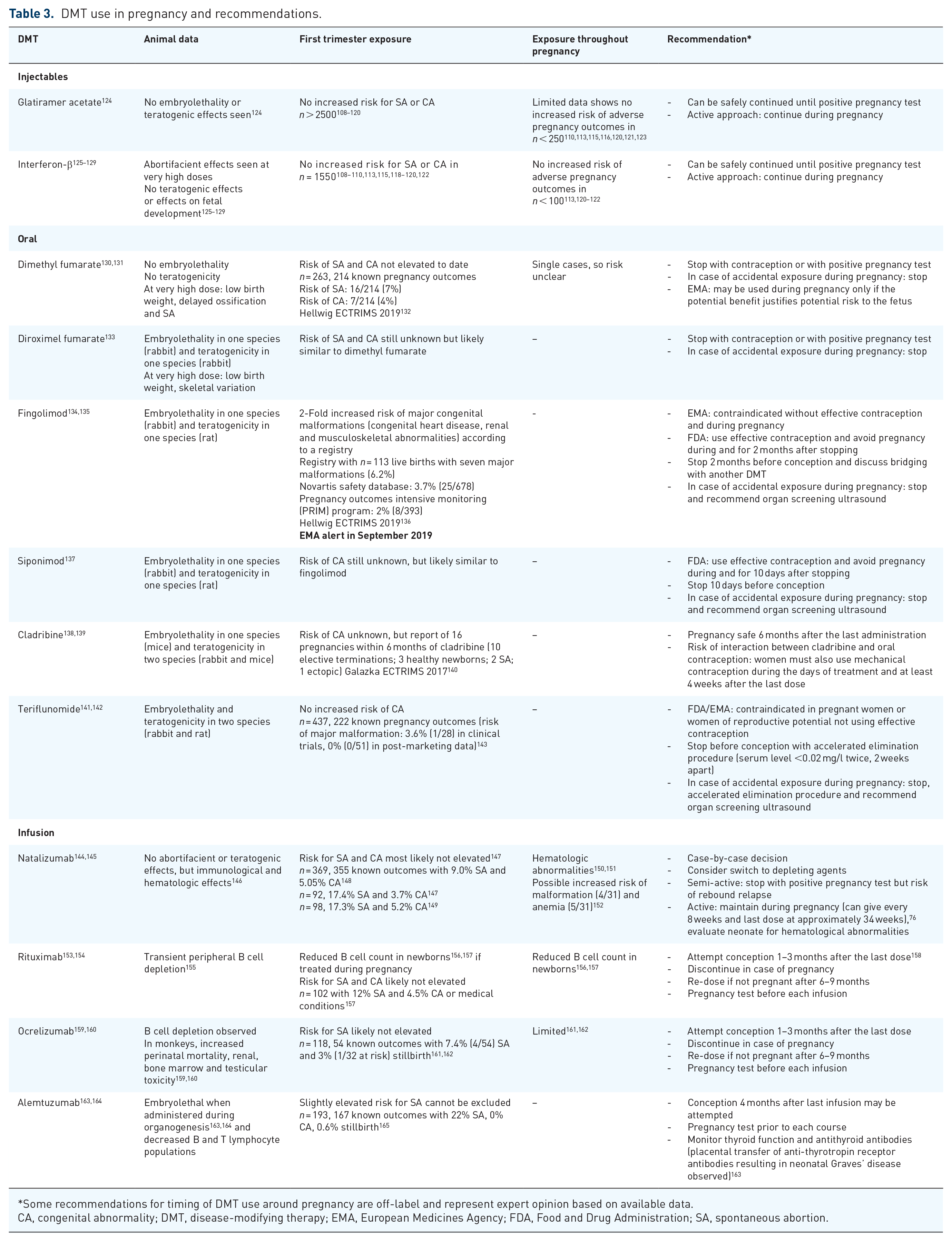
Some recommendations for timing of DMT use around pregnancy are off-label and represent expert opinion based on available data.
CA, congenital abnormality; DMT, disease-modifying therapy; EMA, European Medicines Agency; FDA, Food and Drug Administration; SA, spontaneous abortion.
Immunological changes during pregnancy may not be sufficient to protect women with active disease from relapses or rebound, especially after withdrawal from fingolimod or natalizumab.166–168 The continuation of natalizumab, or bridging with the use of depleting antibodies or cladribine prior to conception should be considered in these patients. Oral DMTs should not be continued in pregnancy, whereas depleting antibody therapies can potentially be used in women with active MS, ideally prior to pregnancy, but with biological effects that may persist after drug elimination.
More data are necessary to fully address this challenging clinical topic, especially for women with more aggressive MS who wish to have children. Current knowledge of the safety of MS DMTs during pregnancy is outlined in Table 3.
DMT safety in breastfeeding
Although breastfeeding may reduce risk, 169 individuals at high risk for postpartum relapse may require additional strategies to decrease relapse risk, such as restarting DMT. Interferon-beta preparations were recently approved by the European Medicines Association (EMA) for use while breastfeeding, 125 but the US Food and Drug Administration (FDA) has not done so. Mothers have historically faced a choice about whether to breastfeed – which has significant benefits to both the mother and infant – or treat their MS. 170 However, some DMTs are unlikely to pass in relevant or harmful quantifies to breastmilk, underscoring the importance of designing studies to support the ability for women to both safely breastfeed while treating their disease.
Transfer of drugs to breastmilk depends on several factors, including molecular weight, protein binding, lipid solubility, volume of distribution, and transport mechanisms, as well as the stage of breastmilk, with less transfer into mature milk than colostrum. 171 Lactation studies are required to determine breastmilk transfer, and a commonly used measure is the relative infant dose (RID), which represents the percent of the weight-adjusted maternal dose consumed in breastmilk over 24 h. RID of <10% is generally considered acceptable for breastfeeding, although the toxicity of each drug should be considered. 172 Based on these considerations, an overview of data on excretion of DMTs to breastmilk, and recommendations for DMT use while breastfeeding are listed in Table 4.
DMT use during lactation and recommendations.
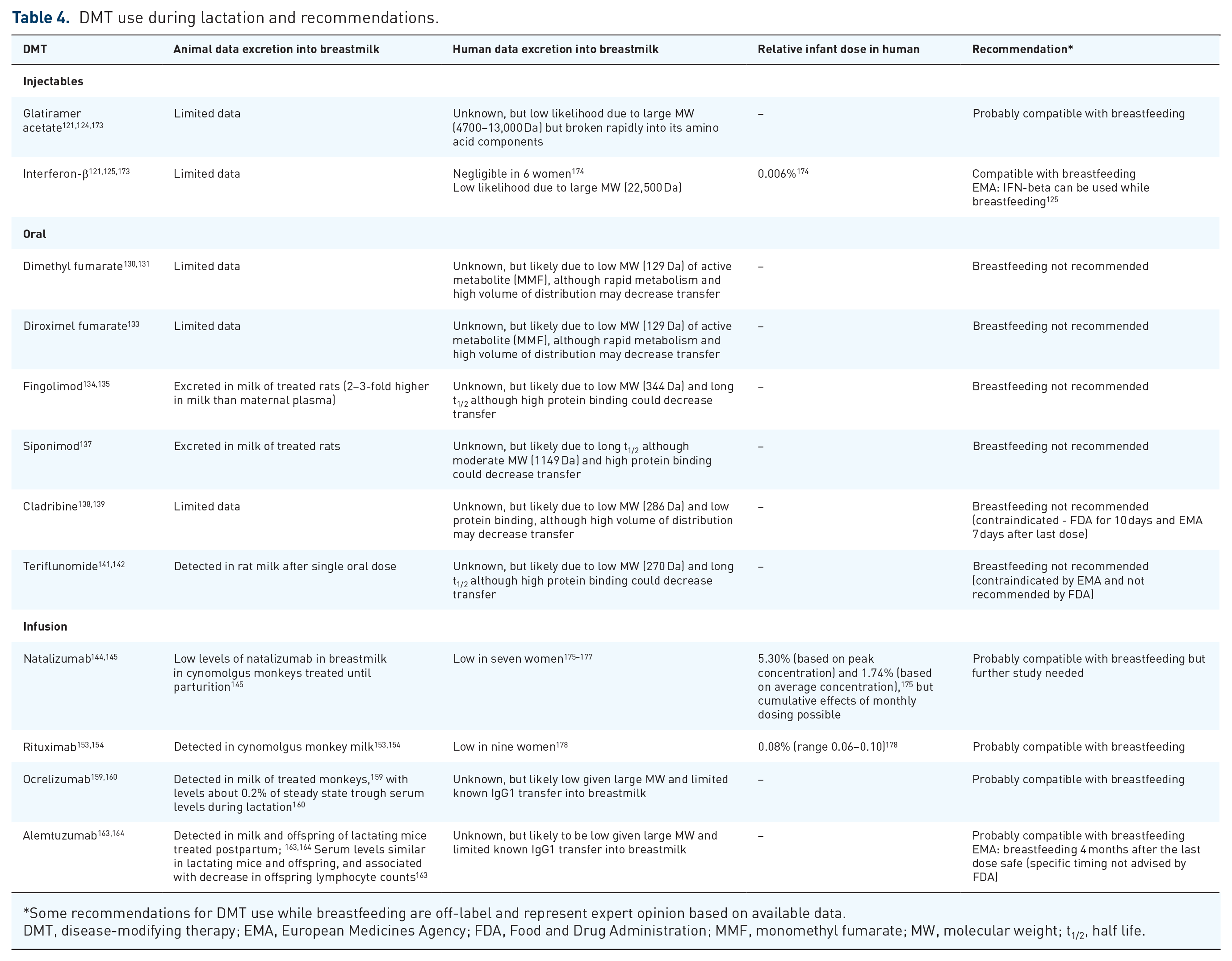
Some recommendations for DMT use while breastfeeding are off-label and represent expert opinion based on available data.
DMT, disease-modifying therapy; EMA, European Medicines Agency; FDA, Food and Drug Administration; MMF, monomethyl fumarate; MW, molecular weight; t1/2, half life.
While additional study of DMT use during lactation is required, when deciding whether to breastfeed while using DMTs, patients and clinicians should consider the risk of postpartum relapse balanced with potential adverse effects to the infant. In patients with high risk of postpartum disease activity, the benefits of breastfeeding despite DMT may outweigh risks for the injectable and monoclonal antibody therapies, while breastfeeding is not suggested while on oral DMTs. A recent review similarly suggested breastfeeding while on monoclonal antibody therapies can be considered in neuromyelitis optica spectrum disorders. 179 It is important to note that data are lacking regarding the long-term immunological and infectious profile of children of women with MS exposed to DMTs in breastmilk.
Obstetric management
Management of women with MS during labor and delivery is relevant to obstetricians, neurologists, anesthesiologists, and patients. Concern regarding associations between spinal anesthesia and MS relapses emerged almost 70 years ago. 180 Since then, prospective studies and a meta-analysis have demonstrated the safety of spinal anaesthesia in MS.5,81,181,182 The American Society of Regional Anesthesia and Pain Medicine states in its 2015 guidelines that epidural anesthesia is considered safer than spinal anesthesia because it does not deposit local anesthetic directly adjacent to the CNS. 183 However, choice of analgesia in labor is best left to discretion of the obstetrician and anesthesiologist in discussion with the woman.
Disease-related factors such as fatigue, lower limb weakness, and spasticity need to be considered when developing a birth plan, and should be discussed during prenatal care. 184 As commonly used symptomatic treatments such as baclofen and dalfampridine are contraindicated in pregnancy, a greater emphasis on physical therapy may be needed to manage symptoms. Clinicians’ apprehension may lead to an increase in cesarean section or instrumental interventions during delivery. However, reports of increased cesarean section deliveries in women with MS may be confounded by cultural and geographical influences on cesarean rates. 185 In many countries, obstetrical care during labor is managed by midwives, and it is important that education on management of women with MS during labor extends to all involved medical professionals.
A systematic review and meta-analysis of women with MS and their pregnancies concluded that women with MS do not have a significantly increased risk of obstetrical or neonatal complications such as prematurity or neonatal death. 186 Management of women with MS during labor and delivery is therefore generally left to the discretion of the obstetrician (or midwife) and anesthesiologist. Clear communication from the neurologist to outline the disease state of the woman with MS, any relevant functional impairments, as well as optimization of MS-related symptom management, remains an important part of holistic care during pregnancy and should be a key focus of the neurologist’s involvement.
Pregnancy and disease course: short-term outcomes and postpartum relapse risk
In the early 20th century, pregnancy was believed to promote poorer outcomes for women with MS, and was discouraged. This perception changed with the landmark Pregnancy in Multiple Sclerosis (PRIMS) study, published in 1998. 5 This prospective multicenter study, including data from 269 pregnancies across 12 European countries, revealed a ~70% decrease in relapse rate in the third trimester, relative to the 12 months pre-conception, and a postpartum relapse rate increase of ~170%. Subsequently, Vukusic showed that following the initial postpartum increase in relapses, 81 ARR returned to pre-pregnancy levels. The findings of the seminal PRIMS, 5 and its extension, 81 have been replicated across numerous cohorts in subsequent decades,39,187–191 and confirmed in a meta-analysis. 186 The seemingly high postpartum relapse rate is driven by a minority of women. Various studies estimate that relapses occur in 14–31% within the first 3 months postpartum.5,190,191 In women with mild MS, the trend for postpartum relapse has generally diminished in the two decades following the PRIMS study. 192 A contemporary population-based cohort of women with MS and CIS did not find rebound disease activity postpartum, attributed to inclusion of women with milder disease and high rates of exclusive breastfeeding. 192 However, recent studies have demonstrated that women treated with highly effective therapies, specifically fingolimod and natalizumab, are at increased risk of rebound relapse activity in pregnancy and the postpartum period once therapy is withdrawn, with rebound relapses associated with longer duration of wash-out.166–168,187,193–196
In terms of short-term MS outcomes beyond relapse activity, the PRIMS study reported a mean increase of 0.9 points on the Kurtzke disability scale over a 24-month period, without an apparent acceleration in disability worsening.5,81 Sixteen years later, a study of 338 women from an Italian multicenter cohort demonstrated that short-term increases in disability accrual (6-month confirmed disability progression) postpartum were driven primarily by relapse activity in the year after delivery. 197
Postpartum outcomes are not limited to women with live births. A recent Italian multicenter study of 188 abortions (17 elective) in RRMS reported that women were at increased risk of clinical and radiological inflammatory activity in the 12 months post-abortion compared with pre-abortion, and risk of inflammatory activity was higher in those with elective compared with spontaneous abortion. 99 Similarly, a smaller study reported a trend towards increased MS activity after pregnancy loss compared with before. 198 The relative risk of inflammatory activity in women who have had abortions relative to women with live births remains unknown.
Various studies have proposed predictors of postpartum relapses,5,81,166,187,189–191,196,199–201 summarized in Table 5. Beyond withdrawal of highly effective therapies,166,193 these studies have consistently demonstrated that patients with higher relapse activity pre-conception and during pregnancy, as well as EDSS scores of >2.0 at conception are the key independent predictors of postpartum relapses.81,191,197 Potentially modifiable risk factors of postpartum relapse such as resuming DMT, vitamin D status, diet, smoking, alcohol, and stress have not been adequately studied. 202 On the other hand, breastfeeding has received substantial interest, and is discussed in detail in the following section.
Proposed predictors of postpartum relapses.

IV, intravenous; IVIG, intravenous immune globulin; MS, multiple sclerosis.
Breastfeeding and postpartum relapses
The effect of breastfeeding on postpartum relapse risk has been controversial, with some studies supporting a protective effect,192,200,201,203,204 while others have not.5,119,199,205–212 There is no evidence to suggest a harmful effect of breastfeeding on MS relapse risk, which is important given the many benefits of breastfeeding to the infant and mother. 170 A recent systematic review and meta-analysis included 24 studies evaluating the association between breastfeeding and postpartum MS relapses, of which 16 had data available to pool. 169 Overall, breastfeeding was associated with 37% lower odds of postpartum relapse compared with nonbreastfeeding. This association was stronger in studies of exclusive breastfeeding (no regular formula supplementation for ⩾2 months) with 48% lower odds of postpartum relapse, compared with 32% lower odds in studies of nonexclusive breastfeeding. One study reported that women who partially breastfed had similar relapse risk to those who did not breastfeed, 201 supporting benefit primarily of exclusive breastfeeding. Confounding and other sources of bias remain a concern given the observational design of these studies, although pooling four well-designed studies was supportive of a protective effect with 43% lower rate of postpartum relapse in breastfeeding compared with nonbreastfeeding groups. 169 The potentially protective effect of breastfeeding may be due to breastfeeding-associated hormonal changes including suppression of pulsatile release of GnRH and LH, as well as high prolactin. 213
Management of MS during the postpartum period should be individualized based on postpartum relapse risk. In those with particularly high risk of postpartum relapse, breastfeeding may be deferred to resume MS therapies. However, the majority of women with MS should be encouraged to breastfeed, and some therapies may be safe to use while breastfeeding. A better understanding of additional strategies to prevent postpartum relapses is urgently needed, including better understanding of the safety of breastfeeding during treatment with DMTs, to allow both the benefit of breastfeeding and treatment of MS.
Postpartum management
In the postpartum period, there are three treatment goals: to prevent inflammatory activity, to provide holistic care, and to optimize psychosocial functioning. Whenever possible, anticipatory guidance should be initiated prior to delivery to minimize delays in care. Care should be in collaboration with the mother, other family members, and, when necessary (e.g., concerns about maternal medications in breastmilk), other healthcare providers.
To prevent inflammatory activity, individualized decisions should be made regarding when to resume DMT, choice of DMT, breastfeeding plans, and use of bridge therapies if indicated. A surveillance MRI 4–6 weeks postpartum may assist in monitoring for subclinical disease activity, particularly in women who delay early DMT initiation. Most women with MS should be encouraged to breastfeed, but those who cannot, or do not wish to, breastfeed should be advised to resume DMT within 2–4 weeks postpartum. For those breastfeeding with higher risk of relapse, certain DMTs could be considered while breastfeeding, as outlined elsewhere in this review.
The second goal is to comprehensively evaluate the woman’s function. There are limited data to guide care. In our clinical experience, monitoring includes evaluating and treating the following functions and when warranted multidisciplinary referrals (e.g., psychologist, psychiatrist, physical therapist, pelvic floor therapist, and/or urologist).
Screening for peripartum depression (PPD), anxiety or milder “baby blues.” PPD is present in 7–19% of all women in the peripartum period (final weeks of pregnancy through 1 year postpartum), and a major risk factor is prior history of depression.214–216 For the general population, screening with the Edinburgh Postnatal Depression Scale with a cutoff of 13 is acceptable, and “screening should be implemented with adequate systems in place to ensure accurate diagnosis, effective treatment, and appropriate follow-up.” 217 PPD in MS is under-explored. 218
Fatigue may worsen postpartum as a result of sleep disruption (newborn needs, mood, or bladder changes) or hormonal changes. Contributing factors should be assessed prior to initiation of medications.
Strength and gait optimization including screening for weakness, loss of balance, or cardiopulmonary deconditioning.
Evaluation of bladder and bowel function, which could be disrupted from neurogenic and/or obstetrical causes but is understudied,219,220 and consideration of pre-emptive referral to pelvic floor physical therapy.
Screening for endocrine changes may include vitamin D level,211,221 which in some, but not all, studies has been associated with inflammatory activity in MS,211,222 as well as thyroid function. 223
The third goal is to optimize the patient’s psychosocial functioning, minimizing as possible disruption caused by postpartum recovery and care of the newborn, and optimizing support available to her. This support may include assistance (from the partner, other family members or a professional) with management of the newborn to enable periods of rest. A social worker may provide advice regarding short-term disability leave and/or financial resources when needed.
Pregnancy and disease course: long-term outcomes
Despite the potential increased relapse risk postpartum, the majority of individuals do not experience relapses in the postpartum period, and pregnancy does not appear to alter long-term relapse rate or disease progression.5,81,166 However, the impact of pregnancy on long-term outcomes remains less clear. Studies up to the mid-2010s demonstrated either a slower rate of disability progression in women becoming pregnant after MS onset,38,71,224–227 or failed to demonstrate an association between pregnancy and long-term disability.228–230 One notable exception found weak evidence for increased risk of converting to SPMS over 10-years in a parous cohort (on injectable or no DMT). 231
More recently, a protective effect of pregnancy on long-term outcomes was reported in a large real-world cohort (MSBase). Females with at least one pregnancy had lower EDSS scores over 10 years, after adjustment for relapse rate, therapy use, and other covariates. 232 Interestingly, when comparing proportion of time spent pregnant with proportion of time on first-line therapy, the protective effect of pregnancy was greater. While the possibility of reverse causality (women with milder disease more likely to attempt pregnancy) cannot be excluded, this is challenged by the fact that approximately half of pregnancies were conceived while on therapy. In the Barcelona CIS cohort, pregnancy after CIS was protective against risk of MS, and time to EDSS 3.0 if pregnancy was modelled as a baseline variable. However, these protective effects were lost when pregnancy was analyzed as a time-dependent variable. 233 Notably, 32% of this cohort did not fulfill Barkhof criteria, potentially limiting generalizability.
Overall, there is little-to-no evidence that pregnancy has a negative impact on long-term outcomes at the group level, with most studies demonstrating a neutral or protective effect. MS does not preclude parenthood or pregnancy. 234 Nonetheless, the effect of pregnancy on disease outcomes, a woman’s ability to care for her child, and future financial stress are key concerns of women with MS considering family planning.235–238 Relapses are the greatest independent driver of long-term disability accrual. 232 Therefore, careful pregnancy planning and monitoring including appropriate use/withdrawal of DMTs is paramount to ensuring positive long-term outcomes for women with MS.239,240
Further studies are needed to evaluate effects of pregnancy in PPMS, address issues of reverse causality, and understand the long-term impact of pregnancy in heterogeneous cohorts to allow pregnancy planning advice tailored to the individual.
Stigma around family planning
Stigma means mark of inferiority.241–243 People with MS or their partners who want to procreate may be questioned about wishing to have children despite a neurodegenerative condition. They may be judged by their family, society, the healthcare team, and even their partners. Moreover, they may feel guilty and fearful of unforeseen consequences,244,245 even though MS has negligible impact on pregnancy and vice versa. 240 They may conceal their desires and their condition,242,246 or discontinue treatment. This fear and stigma may have greater impact on quality of life of prospective parents than the actual disease.240,247
While it is not implied that every couple should procreate, expert support and multidisciplinary guidance could help patients structure their lives and manage their condition, to allow them to have a positive reproductive experience.241,248
Exogenous hormones as a DMT with impact on long-term prognosis of MS
High doses of estrogen seem to be protective by decreasing MS disease activity in the EAE animal model and women during pregnancy. 249 Observational studies of potential effects of oral contraceptives and ART are discussed earlier in this review. To solve the inherent limitations of retrospective and observational studies, two randomized-controlled, phase II, clinical trials have assessed the impact of exogenous hormones on disease course.249,250 They suggest that the addition of ethinyl estradiol 40 µg and desogestrel 125 µg to interferon-β-1a, 250 and estriol to glatiramer acetate, 249 are associated with fewer lesions on brain MRI (26.5% reduction of cumulative number of combined unique active lesions, p = 0.04), and lower ARR (0.25 versus 0.48, p = 0.016 at 12 months), respectively. In addition, both studies showed promising effects of estrogen on cognitive disability as exploratory outcomes. Estriol treatment-induced cognitive improvement was correlated with less cerebral cortex gray matter atrophy, 249 which was mapped to sparing of frontal cerebral cortex. 251 Further research is needed to understand the effect of exogenous hormones on long-term MS prognosis in women.
Menopause and reproductive aging
Like puberty and pregnancy, perimenopause leads to widespread changes in biology. Changes in the immune and nervous systems, fluctuations in gonadal hormones, and symptoms that overlap with those caused by MS may contribute to changes in clinical phenotype to a progressive disease course over the fifth decade. 252 Challenges in understanding the role of perimenopause in MS include distinguishing effects of somatic versus reproductive aging and the potential confounding from comorbid illnesses that become more frequent with older age.
Ovarian aging and disease course
The mean age of onset of secondary progressive MS, characterized by a change from a relapsing remitting phenotype to a continuously progressive form of disease, conspicuously occurs for most women during the perimenopausal period. Several studies have aimed to determine if the menopausal transition affects disease course. Some have reported clinical worsening when patients were asked about their disease perception during menopausal transition,253–255 possibly due to the additive effect of menopause and MS overlapping symptoms. On the other hand, others have focused on the search for an inflection point in MS course centered on menopause. Bove et al. described an inflection point in EDSS worsening at menopause (difference of 0.076 units; 95% CI 0.010–0.14, p = 0.024) in 124 women followed longitudinally (mean follow up 10.4 years). 8 This was replicated by Baroncini et al., who also reported a significant annual EDSS increase in the post-menopausal period (3 years) as compared with the pre-menopausal period (3 years) (0.4 ± 0.7 versus 0.2 ± 0.6 points, p = 0.014) in 108 women. 256 In a smaller cohort (n = 37), Ladeira et al. detected stable EDSS variation across the menopausal transition. 6 Ladeira et al. and Baroncini et al. also reported significant decrease in annual relapse rate after menopause.6,256 On the other hand, Otero et al. found that menopause did not influence the risk of disability accumulation when trajectories of EDSS over the complete disease course (from CIS through menopause) were accounted for, and once adjusting for age and disease duration. 72
Much of the ovarian aging biology accumulates years before the final menstrual cycle, highlighting a continuum of physiological changes, rather than any abrupt change arising at the final menstrual period that could influence MS course. To study the prolonged period of ovarian decline, Graves et al. used AMH as a marker of ovarian function. 9 AMH is produced by ovarian follicles, and levels decline exponentially during the peri-menopausal period. Within a 10-year longitudinal cohort, declines in AMH level were associated with clinically and statistically significant increases in disability and gray matter atrophy, even with adjustment for chronological age, BMI, and disease duration. 9
Mechanistically, the loss of ovarian estrogen around menopause could potentiate several aging-associated phenomena, including decrease in brain repair mechanisms, decrease in immune activation, and, ultimately, a loss of neuro-homeostasis leading to accelerated neurodegeneration and subsequent disease progression.257–259 The subsequent results of these potential changes include increased levels of senescent immune cells and neurons as well as increased CNS atrophy and disability. Notably, cognition is one disability in MS that may be particularly sensitive to loss of estrogens during menopause in light of the well-documented “brain fog” described in otherwise healthy women during natural menopause with aging and surgical menopause not due to aging.260–265 However, estrogen may not be the only culprit as there are also substantial changes in androgen production, and it is difficult to tease apart the effects of somatic aging processes that are linked with perimenopause in women.
Symptom management in perimenopause
Few studies have addressed management of MS symptoms at menopause. Symptom management and choice of DMT should be tailored to account for fatigue and pseudo-exacerbations triggered by hot flashes, which can become more prominent at menopause.266,267 Co-management with primary or women’s health providers may be beneficial. Bladder symptoms may also worsen and urology assessment may be needed.
Prospective, randomized studies on the effect of hormone therapy in patients with MS during menopause are needed, as prior studies reported improved quality of life in post-menopausal women with MS. 266 Wellness approaches, attention to co-morbidities, as well as adding a neuroprotective agent may be appropriate when these are substantiated and become available for women living with MS at menopause and beyond.
Female sexual dysfunction in MS
Sexual dysfunction affects up to 95% of women with MS,268,269 but it is rarely discussed in the office setting. Many patients are embarrassed to bring up the topic, while many physicians feel that they have little to offer and do not ask. Nevertheless, sexual satisfaction is heavily linked to quality of life for women with MS, 270 and it is important for care providers to help manage this.
Female sexuality is multifaceted, and MS can impact it at every level. Direct damage to the brain and spinal cord can impede desire, decrease vaginal sensation and lubrication, and impair orgasm, as well as contribute to pain with sex. Indirect factors impacting sexual function can include physical problems like bladder/bowel dysfunction, fatigue, weakness, and spasticity, as well as emotional problems like cognitive dysfunction or depression. 271
“Invisible” psychologic and emotional factors may also negatively impact sexuality. Desire, for women, is heavily correlated with stress levels, fatigue, relationship quality, and many other intangibles that are vulnerable in settings of chronic neurologic disease. Women with MS often struggle with body image. Many couples struggle as they cope with the physical, emotional, and financial stressors imposed by the disease, and subsequently experience deterioration of their sexual relationship.
Sexual problems may develop early in the course of MS, and tend to persist or worsen over time.268,272 Nevertheless, many factors contributing to sexual dysfunction in MS can be effectively modified (Table 6). 273
Contributors to sexual dysfunction in MS and management options.

Limited evidence exists to support these treatment options; these medications are not FDA-approved for hypoactive sexual desire in women.
Safety is incompletely understood in this population so risks and benefits should be considered.
CBT, cognitive behavioral therapy; DHEA, dehydroepiandrosterone; MS, multiple sclerosis.
Conclusion
Sex hormones play a significant role in the risk and course of MS. Dramatic hormonal fluctuations can influence clinical, radiographic, and disability-related disease parameters. The role of sex chromosomes on sex differences in MS risk and disease progression represents a new frontier for exploration. More research efforts are needed to fully understand unique questions related to MS and fertility, contraception, pregnancy, and reproductive aging.
Footnotes
Acknowledgements
We would like to thank the Executive Committee and members of the International Women in MS (iWiMS) group.
Conflict of interest statement
Kristen M. Krysko is funded by a Sylvia Lawry Physician Fellowship through the National Multiple Sclerosis Society [FP-1605-08753 (Krysko)]. She also had fellowship funding through Biogen.
Jennifer S. Graves has received recent grant and clinical trial support from the National MS Society, Race to Erase MS, UCSF CTSI RAP program, Biogen, and Genentech. She has received honoraria from Biogen and Genzyme for non-promotional trainee education events. She has received personal fees from Novartis and Celgene.
Ruth Dobson works within the PNU, which is funded by Barts Charity. She receives grant support from the UK MS Society, Horne Family Charitable Trust, Biogen, Celgene, and Merck. She has received honoraria for Advisory boards and/or educational actives from Biogen, Teva, Sanofi, Merck, and Roche.
Ayse Altintas has received speaker honoraria for non-promotional education events and travel grants from Merck, TEVA, and Novartis.
Maria Pia Amato has received research grants and honoraria as a speaker and member of advisory boards by Biogen, Merck, Roche, Teva, Sanofi Genzyme, and Novartis.
Jacqueline Bernard has served as a Consultant for Biogen.
Simona Bonavita has received speaker honoraria and/or Advisory Board fees from Novartis, Teva, Sanofi-Genzyme, Biogen-Idec, and Merck-Serono.
Riley Bove is funded by a Harry Weaver Scholarship through the National Multiple Sclerosis Society. She has received recent research support from the National Multiple Sclerosis Society, the California Initiative to Advance Precision Medicine, the Hilton Foundation, the Sherak Foundation, the Weill Innovation Fund, and the UCSF CTSI RAP program. She has received consulting fees from Alexion, Biogen, EMD Serono, Genzyme Sanofi, Novartis, and Roche Genentech.
Paola Cavalla has received recent grants and clinical trial support from Biogen and Sanofi-Genzyme. She has received honoraria from Almirall, Biogen, Merck-Serono, Novartis, Roche, Sanofi-Genzyme, and Teva for non-promotional trainee education events.
Marinella Clerico has received personal compensation for speaking/advising/consulting from Merck, Sanofi-Genzyme, Biogen, Novartis, and Teva; was supported in travelling expenses for congresses from Merck, Sanofi-Genzyme, Biogen, Novartis, Teva, and Almirall; has received research grants from Italian MS Foundation (FISM), Italian Ministry of Research, Merck, Sanofi-Genzyme, Biogen, and Novartis.
Teresa Corona declares no conflicts of interest.
Anisha Doshi declares no conflicts of interest.
Yara Fragoso declares no conflicts of interest.
Dina Jacobs has clinical research trial support from Genentech/Roche, Biogen, Novartis, and Medimmune, and has received consulting/advisory board honoraria from Celgene, Biogen, Novartis, EMD Serono, and Sanofi Genzyme.
Vilija Jokubaitis is funded by an MS Research Australia Fellowship (16-0206). She receives project grant support from the National Health and Medical Research Council (NHMRC) of Australia (GNT1156519) and MS Research Australia (18-0424; 19-0665). She has received honoraria from Biogen and Roche for non-promotional educational activities, and conference travel support from Merck and Roche.
Doriana Landi has received travel funding from Roche, Biogen, Merck-Serono, Sanofi-Genzyme, and Teva; speaking or consultations fees from Bayer-Schering, Sanofi-Genzyme, Merck-Serono, Teva, Biogen, Roche, Mylan, and Almirall.
Gloria Llamosa declares no conflicts of interest.
Erin E. Longbrake has received honoraria from Genentech, Genzyme, Alexion, Biogen, EMD Serono, and Celgene. She has grant support from NIH K23NS107624 and Race to Erase MS.
Elisabeth Maillart has received grant support from Biogen, Novartis, and Roche. She has also received consultant honoraria from Biogen, Celgene, Merck-Serono, Novartis, Roche, Sanofi Genzyme, and Teva.
Monica Marta has received honoraria and travel costs from Genzyme, AbbVie, Roche, and Novartis.
Luciana Midaglia declares no conflicts of interest.
Suma Shah has received honoraria from Biogen.
Mar Tintoré has received compensation for consulting services and speaking honoraria from Bayer Schering Pharma, Merck-Serono, Biogen Idec, Teva Pharmaceuticals, Sanofi-Aventis, Novartis, Almirall, Genzyme, Viela Bio, and Roche.
Anneke van der Walt has served on scientific advisory boards, received travel support and speaker’s honoraria from Biogen, Merck, Novartis, and Roche. She is supported by funding from the National Health and Medical Research Council of Australia.
Rhonda Voskuhl declares no conflicts of interest.
Yujie Wang declares no conflicts of interest.
Rana K. Zabad has been a site investigator or site PI for clinical trials funded by Adamas, Biogen, Genentech, Novartis, Sunpharma, and PCORI. In the last 5 years, she has served as a consultant for Bayer, Biogen, Genentech, Celgene, Genzyme, TEVA Neuroscience, and TG therapeutics, and has given unbranded lectures sponsored by TEVA, Novartis, and Genentech. She is a member of the Adjudication Committee for a clinical trial of biotin in primary and secondary progressive multiple sclerosis sponsored by PAREXEL and medDay pharmaceutical.
Burcu Zeydan reports funding from NIH (NIA: U54 AG44170).
Maria Houtchens has received grant support from Biogen, Serono, Sanofi Genzyme, and Genentech/Roche. She also received consultant honoraria from Biogen, Serono, Sanofi Genzyme, Roche, and Celgene.
Kerstin Hellwig receives grant support from the Innovation Fund of the Federal Joint Committee. She has also received consultant and speaker honoraria and grant support from Bayer, Biogen, Merck, Novartis, Sanofi Genzyme, Roche, and Teva.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Kristen M. Krysko is funded by a Sylvia Lawry Physician Fellowship through the National Multiple Sclerosis Society [FP-1605-08753 (Krysko)].
