Abstract
Background
In spite of the observed immunomodulatory properties of different sex hormones on Multiple Sclerosis (MS) in different investigations, to date, there has been no study to systematically review the documents to add more powerful data to the field.
Objectives
Therefore, in this paper we aim to systematically review clinical and randomized controlled trials (RCT) assessing the effect of sex hormone therapies on individuals with MS.
Design
A comprehensive search of electronic databases including PubMed, EMBASE, and Scopus was conducted. Clinical trials and RCTs that assessed the impact of sex hormones on individuals with MS were selected and included in the systematic review.
Data sources and methods
In the final phase of the search strategy, 9 papers reached the criteria for entering in the systematic review. Two independent reviewers extracted the relevant data from each article according to the standardized data extraction form. Two reviewers also assessed the quality of each study independently using PEDro scale.
Results
We categorized three different classifications of outcomes including clinical, MRI, and immune system findings and put each measured outcome in the category which matched best.
Conclusion
In conclusion, the existed investigations on the effect of sex hormones on inflammatory and neurodegenerative components of MS are promising particularly in relapsing-remitting MS (RRMS).
Plain language summary
Immunomodulatory properties of different sex hormones on Multiple Sclerosis (MS) have been proposed. Therefore, in this paper we aim to systematically review clinical and randomized controlled trials (RCT) assessing the effect of sex hormone therapies on individuals with MS. A comprehensive search of electronic databases was conducted. Clinical trials and RCTs that assessed the impact of sex hormones on individuals with MS were selected and included in the systematic review. In the final phase of the search strategy, 9 papers reached the criteria for entering in the systematic review. Two independent reviewers extracted the relevant data from each article according to the standardized data extraction form. We categorized three different classifications of outcomes including clinical, MRI, and immune system findings and put each measured outcome in the category which matched best. The existed investigations on the effect of sex hormones on inflammatory and neurodegenerative components of MS are promising particularly in relapsing-remitting MS (RRMS).
Highlights
• This systematic review highlights the existed findings reported in the clinical trials on the effect of sex hormones on multiple sclerosis (MS). • The effect of sex hormones on different aspects of MS have been discussed in this study.
Introduction
Multiple sclerosis (MS) is a demyelinating inflammatory disorder causing a wide range of potential symptoms including impairments in vision, motor ability, sensation and balance. MS has a significant impact on the affected individual, families and society due to presentation of the symptoms usually at a highly productive stage of the person’s life. According to estimates, the prevalence of MS has 30% increase in 2020 in comparison to 2013, affecting 2.8 million people worldwide. 1 The exact etiology of MS remains incompletely recognized and subsequently a definite cure is still lacking. 1
It has been described that the pathogenesis of MS has both inflammatory and neurodegenerative components. 2 The exact pathological mechanism of inflammatory component is not fully understood but peripheral activation of auto-reactive CD4+ cells which cross blood brain barrier (BBB) and attack neuronal myelin sheath in the central nervous system (CNS) has been theorized as one of the key mechanisms in this component. 3 Upon recognizing myelin antigens, a chronic inflammatory cascade initiate, resulting in demyelination of axons, formation of demyelinated white matter plaques and astrocytic scars. 4 In addition, diffuse axonal damage, long-term disease progression, and permanent disability is the hallmark of neurodegenerative component.2,5 However, whether neurodegeneration component is directly associated with inflammatory component is not clear yet. Axonal damage may occur separately from white matter inflammatory lesions and intense immunosuppressive medications is not usually sufficient to stop neurodegeneration.6,7
On the other side of story, over the last two decades, neuroimaging and neuropathological studies have indicated another pathological mechanism which can be attributed to neurodegeneration in MS.8-10 Several studies have represented gray matter atrophy to have stronger correlation with clinical disability and neurodegeneration than white matter lesions and inflammation.5,11,12 There are studies suggesting gray matter atrophy as a surrogate marker for neurodegeneration and progression of disability in patients with MS.13,14 Fisher et al, in a neuroimaging study have demonstrated that gray matter tissue lesions govern the pathological process as MS progresses underling neurological disability. 14 However, they represented that the mechanisms differ in relapsing-remitting MS (RRMS) and secondary progressive MS (SPMS). It is worth noting that while effective anti-inflammatory medications have the potential to improve white matter lesions, gray matter atrophy may benefit less form such medications.15,16 Given together, the need for shedding more light on novel treatment options combining anti-inflammatory and neuroprotective effects seems necessary.
Throughout the last two decades, sex hormones such as testosterone and estrogen (estriol or 17β-estradiol) have been administered as neuroprotective therapies to effectively target gray matter atrophy and prevent permanent disability based on two well-established clinical observations. 2 First, MS has a higher prevalence in females than males and second, during pregnancy particularly in the third trimester a decrease in disease activity occur. 2 Sicotte et al, in a clinical trial study treated non-pregnant women with MS with oral estriol (8 mg/day). Their results showed that the pregnancy hormone estriol significantly decreased delayed type hypersensitivity responses to tetanus, interferon-gamma levels in peripheral blood mononuclear cells, and gadolinium enhancing lesion numbers and volumes based on monthly cerebral imaging in RRMS patients. 17 Furthermore, Gold et al. treated ten men with MS with 10 g of gel containing 100 mg of testosterone in a cross over design. 18 They showed that testosterone has the potential to significantly reduce DTH recall responses inducing a shift in peripheral lymphocyte composition through decreasing CD4+ T cells and increasing NK cells. In addition testosterone administration produced BDNF and PDGF-BB supporting the potential neuroprotective and immunomodulatory effects of this sex hormone for central nervous system. 18
Although the immunomodulatory properties of testosterone, progesterone and estrogen (estriol or 17β-estradiol) have been well explored in the literature, to date, there has been no study to systematically review the existed data in order to add more powerful results to the field. Therefore, in this paper we aim to systematically review clinical and randomized control trials (RCT) assessing the effect of sex hormone therapies on MS. Undoubtedly, our results could better establish the strength of evidence and formulate suggestions for future researches in the field of interventional strategies for patients with MS.
Methods
Data source and searches
The search strategy, study selection, data extraction and analysis were conducted and reported according to standards of the preferred reporting items for systematic review and meta-analysis guidelines (PRISMA) in this study. 19 A comprehensive search of electronic databases including PubMed, EMBASE, and Scopus was conducted. Administrating the search strategy of population and intervention approach, the key terms and words were as follows: “Multiple Sclerosis” OR “Autoimmune Diseases of the Nervous System” OR “Demyelinating Autoimmune Diseases” OR “Chronic Progressive, Multiple Sclerosis” OR “Relapsing-Remitting, Multiple Sclerosis” OR “Primary Progressive, Multiple Sclerosis” OR “Secondary Progressive, Demyelinating Diseases” AND “Gonadal Hormones” OR “Sex hormones” OR “Corpus Luteum Hormones” OR “Gonadal Steroid Hormones” OR “Estradiol Congeners” OR “Progesterone Congeners” OR “Testosterone Congeners” OR Estr* OR Testosterone OR Progesterone AND therap* OR intervention OR effect AND “clinical trial” OR “randomized control trial” OR “open label study” OR “open label clinical trial” OR “open label trial” OR “clinical study”. A further manual search of references in review articles was conducted to find relevant investigations.
Eligibility criteria and study selection
Clinical trials and RCTs that assessed the impact of sex hormones on individuals with MS were selected and included in the systematic review. The inclusion criteria were to (1) be a clinical trial or RCT study (2) have MS as their exposure of interest (3) apply any type of sex hormones as their intervention of interest (4) be published between 2000-2022 and (5) be published in the English language. Animal studies, case reports, observational studies, letters, reviews, presentations or any reports without sufficient data were excluded from the study. Any inconsistencies or disagreements between two independent screeners who searched and selected the articles were resolved by discussion.
Data extraction
Two independent reviewers extracted the following data from each article according to the standardized data extraction form. Publication details (including the first author’s name, publication’s year, and the country in which the study conducted), study details (including study design, number of participants, gender of participants, age of participants, disease stage, disease type, and control group characteristics), intervention details (including type of sex hormone, duration of therapy, follow-ups, control interventions, and the reported outcomes), and moderator variables (including participant retention and dropouts, participant adherence, and adverse effects associated with sex hormones) were extracted from each selected study. A non-available (NA) or not-reported (NR) statement was assigned in cases in which supplementary methodological information was not provided either from the article or corresponding author’s contact. Disagreements between the reviewers on the extracted data were resolved by a third reviewer or consensus-based discussion.
Quality assessment
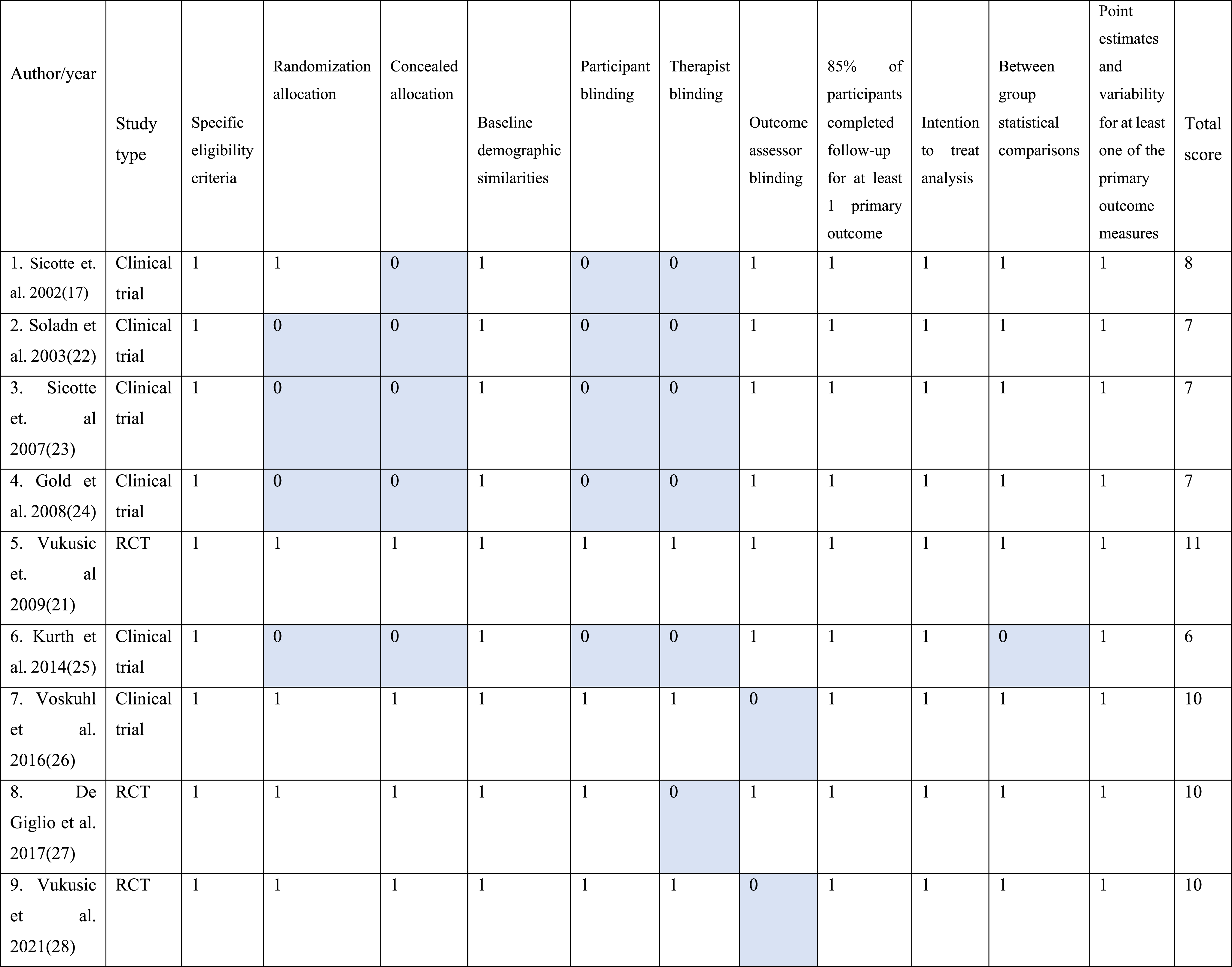
We administered PEDro scale in order to rate independently each article for quality by two reviewers. 20 The PEDro scale which is developed to evaluate the methodological quality of intervention studies is an 11 points scale evaluating the following aspects: specific eligibility criteria, randomization allocation, concealed allocation, baseline demographic similarities, participant blinding, therapist blinding, outcome assessor blinding, whether more than 85% of participants completed follow-up for at least 1 primary outcome, intention to treat analysis, between group statistical comparisons, and point estimates and variability for at least one of the primary outcome measures. Five of the included studies were RCT studies and the remaining four studies were clinical trials. The quality of all selected studies was assessed by two independent reviewers. Discrepancy between two reviewers’ opinion were resolved by discussion.
Statistical analysis
Since it was a systematic review, we did not need any definite statistical analysis.
Results
Study selection and characteristics
According to our primary search and after removing duplicates, 11503 articles were found. Title and abstract checking resulted in removal of 11450 papers and therefore 53 papers were entered to the phase of full text checking. Finally, nine articles meet the inclusion criteria for being systematically reviewed. Of the nine final studies, seven studies were clinical trials and the remaining three studies were RCTs. The main reasons for removing the articles were as follows: using a sample not comprised individuals with MS (11467), not assessing the impact of sex hormones (547), being of a non-empirical nature,
5
being a review study (4478) and assessing animals as their exposure of interests (534) Figure 1. Due to the fact that numerous articles had the combination of such categories, the number of each excluded group is approximate. The articles were published in three different countries including USA (6 studies), France (2 studies), and Italy (1 studies) between 2002 and 2021. More than half of the articles (6 studies) evaluated the effect of sex hormones on women with MS while the remaining (3 studies) assessed men with MS (no study evaluated both). In total 482 individuals with MS were included in the studies with a mean sample size of 43.81 (standard deviation (SD): 57.87 range: 4-150). In addition, the age range of individuals with MS was almost defined in all selected studies (M: 41.3, SD: 6.03) except in one study.
21
Six studies examined the effect of sex hormones on females with MS and the remaining three studies assessed on males. No study investigated both male and female. Tables 1–3 represents details and characteristics of the selected studies. Flowchart for representing the process of searching and selecting the bibliography. Basic and demographic characteristics of selected clinical trials and randomized controlled trials. M: mean, SD: standard deviation, RR: relapsing-remitting, SP: secondary progressive, IFN: interferon, NR: not reported, MS: multiple sclerosis, DMT: Disease Modifying Therapy. Therapeutic features and characteristics of selected clinical trials and RCTs. EDSS: Expanded Disability Status Scale, PASAT: Paced Auditory Serial Addition Test, IFN: interferon, NR: not reported, IL: interleukin, TNF: Tumor Necrosing Factor, BDNF: Brain-Derived Neurotrophic Factor, PDGF-BB: Platelet Derived Growth Factor BB, NGF: Nerve Growth Factor, CNTF: Ciliary Neurotrophic Factor. outcomes of selected clinical trials and RCTs based on three categories of immune system function, Imaging/MRI findings and clinical assessments. DTH: Delayed Type Hypersensitivity, RR: relapsing-remitting, SP: secondary progressive, EDSS: Expanded Disability Status Scale, IFN: interferon, TGF: Transforming Growth Factor, PBMCs: Peripheral Blood Mononuclear Cells, BDNF: Brain-Derived Neurotrophic Factor, PDGF-BB: Platelet Derived Growth Factor BB, PASAT: Paced Auditory Serial Addition Test, NR: not reported, TNF: Tumor Necrosing Factor, NK: natural killer.



Quality assessment
Quality score of selected clinical trial and RCT studies.

Dosage and type of sex hormones across selected studies
Three RCTs and three clinical trials used different forms of estrogen including estriol, estradiol, ethinylestradiol, and transdermal 17-beta-estradiol. In addition, two RCTs and two clinical trials administered synthetic forms of progesterone such as progestin, desogestrel, and NOMAc. The remaining three clinical trials used testosterone in order to investigate the effect of sex hormone on individuals with MS. Five out of nine studies administered the combination of two different sex hormones and the remaining only used a single hormone. Estriol was administered 8 mg-day in all three studies which assessed the impact of this hormone in individuals with MS. In contrast a wider spectrum of dosage was used across three studies using forms of estradiol including 75 micrograms weekly, 8 mg daily, and 10 mg daily. Moreover, synthetic forms of progesterone were also used in different dosages of 10 mg/daily, 100 mg daily, and 125 mg daily. However, the dosage for testosterone was fixed across three studies administrating the effect of testosterone on individuals with MS with 100 mg/day.
Outcome measures
Table 3 shows details on the main findings of selected studies in the three categories. Different outcomes were measured following sex-hormone therapy across nine selected studies. Generally, we categorized three different classifications of outcomes including clinical, MRI, and immune system findings and put each measured outcome in the category which matched best. In one study, Vukusic et al. reported no results in none of the categories since it is an on-going RCT project on the effect of progestin and estradiol in post-partum relapses. 21 Two studies measured outcomes of all three categories, one study assessed only one category and the remaining assessed a combination of two categories. Clinical assessments commonly consisted of batteries of cognitive function examination, disability (e.g., EDSS scores), date and onset of relapses, symptoms, level of severity, fatigue, depression, quality of life, and functions of daily living. In the category of MRI finding, outcomes of the number and volume of new and total gadolinium-enhancing lesions, T2 lesions, enhancing lesion activity, whole brain volumes, and focal gray matter loss. In the final category, measures of immune system modulation followed by sex-hormone therapy consisted of DTH responses to tetanus and candida as well as levels of IFN-alpha, Cytokine profiles of stimulated PBMCs determined by intracellular cytokine staining (IL-5, IL-10, IL-12 p40, TNF, and IFN), cytometric bead array (IL-2, IL-4, IL-5, IL-10, TNF, and IFN, lymphocyte subpopulation composition by flow cytometry, ex vivo protein production of cytokines (IL-2, IFN gamma, TNF alpha, IL-17, IL-10, IL-12p40, TGFbeta1) and growth factors (BDNF, PDGF-BB, NGF, and CNTF).
Clinical assessments
Six out of nine studies (four clinical trials two RCTs) evaluated outcomes relevant to clinical assessment category. In regard to frequency and duration of relapses, two studies (one RCT and one clinical trial) showed no significant changes,17,27 while one study showed that annualized confirmed relapse rate was significantly reduced in the estriol group in comparison to placebo group. 25 In addition, regarding cognitive function five studies reported a significant improvement in scores of different cognitive assessments. In this vein, three studies showed positive impact of sex hormone therapy on PASAT scores (one study showed positive impact only in RRMS and not in SPMS),17,18,23 one study on spatial memory test 23 while two studies showed no effect for nine-hole peg test.17,23 Moreover, two studies showed a significant improvement in symptoms of fatigue and mood imbalance following sex hormone therapy.25,26
MRI findings
Six out of nine studies (five clinical trial and one RCT) evaluated outcomes relevant to MRI findings category. In regard to number and volume of new gadolinium (GAD) enhancing lesions, two studies showed improvements (one study showed improvement only in RRMS group and not in SPMS group)17,22 while two other studies showed no significant difference before and after sex-hormone therapy.23,24 One study showed an improvement in brain volume loss 17 while another study showed no difference in whole brain volume before and after sex-hormone therapy. 23 Moreover, two studies showed improvements in gray matter volume following sex-hormone therapy.24,25
Immune system
Three out of nine studies (clinical trials) examined outcomes relevant to immune system category. Two studies showed decreased DTH responses to tetanus following sex hormone therapy.17,18 One study showed interferon-alpha levels in peripheral blood mononuclear cell in RRMS not in the SPMS group. 18 Another study also showed an increase in levels of IL-5 and IL-10 as well as a decrease in TNF-alpha following sex hormone therapy which were more prominent in RRMS than SPMS group. 22
Discussion
The effect of sex hormone therapy in individuals with MS have been explored in numerous investigations. However, to our knowledge this is the first study systematically reviewed the outcomes of clinical trials and RCTs in MS. Although single investigations reported such a possible association, a comprehensive and systematically conducted study can add more valuable insight to the field. We summarized findings with regards to clinical assessments, MRI findings, and immune system outcomes.
All identified studies in the current systematic review, examined RRMS while four out of nine selected studies investigated the effect of a particular type of sex hormones on both RRMS & SPMS females. Interestingly in such studies which assessed both SPMS and RRMS individuals, the outcomes were more robust in females with RRMS than SPMS. Among these four studies, two studies17,22 showed the positive impact of estriol on MRI findings and immune system outcomes only in RRMS group.
It is worth mentioning that there is a considerable heterogeneity among the existed trials assessing the effect of sex hormones on individuals with MS. There are different sex hormones, modes of administration, populations (male/female and SPMS/RRMS), measures (clinical, para-clinical, immunological), and findings across these nine selected trials. Such noticeable heterogeneity complicates the interpretation of the findings and the efficacy of sex hormone therapy in individuals with MS. Although the existed data and trials are so valuable in their own way, future studies are warranted to add further data in each specific category (either sex hormone type/mode of administration, sample type, or measures) in order to make a clearer interpretation.
Nevertheless, in the current systematic review, we have discussed the findings of existed trials base on the type of sex hormone as well as the measures.
In regard to the impact of a particular type of sex hormone and the gender difference, we have classified and discussed the results attributed to each sex hormone in two categories of testosterone and steroids in the following. Evidence shows that women represents earlier onset, more frequent relapses, and more inflammatory lesions. 28 In contrast, men experienced worse outcomes, faster progression, and greater gray matter atrophy in spite of a stable prevalence. Moreover, there are considerable differences regarding disease progression and inflammatory activity between male and female with MS. Thus, hormonal and genetic factors (in particular sex hormones) maybe involved in managing the disease course in women and men. Therefore, although both men and women with MS can benefit from sex hormone therapy, different female’s hormone-related physiological conditions such as pregnancy, puerperium, and menopause can considerably affect the frequency, prevalence, and disease course in females.
Testosterone
Previous estimates showed that approximately 40% of men with MS have low levels of testosterone which is correlated with their physical, and cognitive disability as well as worse clinical outcomes. 29 In the current systematic review, three out of nine studies assessed only males.18,23,24 The sex hormone which was evaluated in these three studies was testosterone and the type of MS was RRMS. Regarding MRI findings, their cumulative results showed that while testosterone could significantly improve brain atrophy, brain volume loss, and gray matter volume, no effect was observed in GAD lesions. In addition, Sicotte et al 23 represented positive effect of testosterone on PASAT scores, and spatial memory tests, while there was no significant improvement regarding EDSS and Hole-peg test scores on males with RRMS. Therefore, one can hypothesize a potential neuroprotective effect based on the increase in BDNF and platelet-derived growth factors following testosterone intervention in men with MS.
Steroid combinations
Five out of nine studies investigated the effect of steroid combinations (i.e., estrogen and progesterone) on females with both SPMS and RRMS. There was no study assessing the effect of steroids on men with MS. The cumulative results of these studies showed the positive impact of steroid combinations on immune system outcomes. In addition, two of these studies showed that steroid combinations could exert positive effect on reducing the number and volume of new enhancing brain lesions. It is worthy of note that the aforementioned findings were more robust in females with RRMS than SPMS. However, among the studies in this group, Vukusic et al 27 failed to show any prominent difference before and after steroid therapy in females with both RRMS and SPMS. In addition, in terms of clinical outcomes, studies in this group showed improvements in mood and fatigue symptoms, and PASAT scores which were also more prominent in females with RRMS. However, in term of EDSS scores, nine-hole peg test and relapses, these studies failed to represent any positive effect of steroid combinations on females with both RRMS and SPMS. Of the possible explanations for such positive effects of estrogen and progesterone is their neuroprotective, pro-myelinating and immunosuppressive mechanisms. 30
In addition, as clinical relevance of immunological findings such as reduced “DTH recall” or “higher IL-5” level on outcomes of individuals with MS is still unclear, in regard to the impact of sex hormones on different outcomes, we classified and discussed the findings attributed to two categories of clinical and para-clinical outcomes in the following.
Clinical findings
The clinical measures among 9 selected studies are generally categorized to measures of relapses, level of disability, motor dexterity, mood/fatigue, menses irregularities, and cognitive assessments such as auditory neuro-psychological tasks and spatial memory test. cognitive assessments have been the most common clinical assessment among the studies (five out of nine studies).17,18,23,25,26 The cumulative findings on different cognitive assessment showed a general positive effect of striol, testosterone, and estradiol on measures of PASAT scores, spatial memory, and spatial recall. In addition, three out of 9 studies assessed the effect of sex hormone therapy on relapse rates. While two studies represented no significant change in relapse rate following sex hormone therapy in females with MS (estriol and NOMAC),17,27 Voskuhl et al showed that the annualized confirmed relapse rate was significantly reduced in females with MS following estriol administration. 25 In addition, two studies assessed the effect of sex hormone therapy on level of disability (EDSS scores) and motor dexterity. Both studies showed no prominent changes in disability scores as well as motor dexterity following estriol and testosterone administration in females and males with MS respectively.17,23 At last, findings on fatigue and mood improvement following estriol/estradiol administration showed no consistent results in two studies.17,23 Although the existed evidence on the effect of sex hormone on clinical aspects of MS is so valuable, however, further studies and trial are needed to more clarify the effect of the specific sex hormones on particular aspects of cognition and clinical features in individuals with MS.
Para-clinical findings
The para-clinical measures in the 9 selected studies generally were total brain volume, gray/white matter volume, and number of enhancing lesions. Four out of nine studies measured either total brain volume or regional (gray/white matter) volume following testosterone as well as progesterone/estriol in males and females with MS respectively.17,23-25 The cumulative findings point to a potential positive effect of the mentioned sex hormones on improving total and regional brain volume at different time points after hormone administration. In addition, while two studies showed the positive effect of estriol on reducing the size and number of new enhancing lesions of females with MS,17,22 another three studies showed no improvement in the number of new enhancing lesions following testosterone administration in males23,24 and estriol administration in females with MS. 25 This is somehow interesting and can point to the potential therapeutic effects of different sex hormones on different outcomes. Ignoring the gender of participants, although this is driven from a small number of studies but it implies that estriol can be more effective in reducing new enhancing lesions in comparison to testosterone. It is worthy of note that while FDA-approved medications and treatments for MS diminish relapse and inflammation rate, but have not strong effects on re-myelination, gray matter atrophy and disability.24,31 However, interestingly based on several MRI findings in the current systematic review, sex steroids may achieve re-myelination, and reduce gray matter atrophy. Several mechanisms may underlie such effect of sex hormones such as sex hormones’ interaction with astroglia, insulin-like growth factor −1 and recruiting oligodendrocytes. 32 In addition, sex steroids modulate chemokine expression and signaling which is associated with demyelinating diseases. 33 Similar to clinical parts, further studies are also required to add information to the specific roles (if any) of different sex hormones on various para-clinical findings.
Conclusion
In the current systematic review, we represented a substantial heterogeneity regarding different variables in the existed clinical and randomized trials investigating the effect of sex hormones on individuals with MS. For example, more future studies are required to have a more comprehensive view on the role of a specific sex hormone such as estriol or testosterone on a specific outcome such as enhancing lesions or relapse rate in individuals with MS. Such heterogeneity in the existed trials complicate the interpretation of outcomes. In addition, in spite of possible valuable effect of sex hormones on pathological mechanisms of MS, many mechanisms and effects remain undefined yet in different pathological components and different types of MS. As represented in the current systematic review, the existed clinical trials and RCTs have shown a more robust impact of sex hormones on RRMS than SPMS. However, the exact justification of such observation is not debated yet. Besides, there was no clinical trial or RCT investigating the effect of sex hormone on Primary Progressive MS (PPMS). In addition, gender-specific responses to different sex hormone therapies are required more investigations due to gender-specific differences in responses to inflammation within CNS. For example, brain lesions in females with MS, the activation of 3β-hydroxysteroid-dehydrogenase, a precursor of progesterone and the progesterone receptor has been observed.
Moreover, regarding the appropriate combination of sex hormones and their effect on MS, there is also still poor information and remains to be studied. For example, previous evidence has shown that a high estrogen to progesterone ratio could give rise to a significantly greater number of active MRI lesions than a low ratio. Nevertheless, there is a paucity of data on sex hormone combinations in demyelinating diseases such as MS. Moreover, there are three estrogens with different strength including Estrone (E1), Estradiol (E2), and Estriol (E3) and the optimal efficacy of each one is yet to be defined for MS. However, based on the current systematic review, the general findings on the effect of estriol on different clinical and para-clinical outcomes of females with MS seem somehow promising. In particular, immune-regulatory effect of estriol may be much more than estradiol. Besides, there are few studies investigating the effect of Estrone on different types of MS.
Given together, the existed investigations on the effect of sex steroids on inflammatory and neurodegenerative components of MS are promising. Different types of MS require therapeutic agents targeting improvement of neurodegenerative component and re-myelination as much as inflammatory component. However, further and more exact investigations are warranted to study such possibility of sex hormones either alone or in combination with other to induce re-myelination.
Footnotes
Author contributions
Monir Shayestefar: Investigation, Project administration, Writing – original draft, Mehri Salari: Resources, Validation, Visualization, Shahedeh Karimi: Writing – review & editing, Massoud Vosough: Supervision, Writing – review & editing, Amirhossein Memari: Formal analysis, Investigation, Writing – original draft, Seyedmassood Nabavi: Conceptualization, Data curation, Supervision, Visualization. CreditValidation Error Authors: Monir Shayestefar, please check and link manually.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.