Abstract
Objective:
In relapsing–remitting multiple sclerosis (RRMS), suboptimal adherence to injectable disease-modifying therapies (iDMTs; interferon β-1a/b, glatiramer acetate) is common, reducing their effectiveness. Patient retention on oral fingolimod and iDMTs was evaluated in PREFERMS, a randomized, parallel-group, active-controlled, open-label, 48-week study.
Methods:
Patients were included if they had RRMS, were aged 18–65 years and had Expanded Disability Status Scale score up to 6, enrolled at 117 US study sites, were treatment naïve or had received only one iDMT class. Patients were randomized 1:1 (fingolimod 0.5 mg/day; preselected iDMT) by interactive voice-and-web-response system without blinding, followed up quarterly, and allowed one study-approved treatment switch after 12 weeks, or earlier for efficacy or safety reasons. The primary outcome was patient retention on randomized treatment over 48 weeks. Secondary endpoints included patient-reported outcomes, brain volume loss (BVL), and cognitive function.
Results:
Analysis of 433/436 patients receiving fingolimod and 428/439 receiving iDMTs showed that patient retention rate was significantly higher with fingolimod than with iDMTs [352 (81.3%) versus 125 (29.2%); 95% confidence interval 46.4–57.8%; p < 0.0001]. The most common treatment switch was from iDMT to fingolimod for injection-related reasons. Patient satisfaction was greater and BVL less with fingolimod than with iDMTs, with no difference in cognitive function. Adverse events were consistent with established tolerability profiles for each treatment.
Conclusions:
In RRMS, fingolimod was associated with better treatment retention, patient satisfaction and BVL outcomes than iDMTs. Patients may persist with iDMTs, but many may switch treatment if permitted. Treatment satisfaction fosters adherence, a prerequisite for optimal outcomes.
Keywords
Introduction
In relapsing–remitting multiple sclerosis (RRMS), injectable disease-modifying therapies (iDMTs) interferon β-1a [Avonex (Biogen, Cambridge, MA, USA) or Rebif (EMD Serono, Rockland, MA, USA)], interferon β-1b [Betaseron (Bayer, Leverkusen, Germany) or Extavia (Novartis Pharma AG, Basel, Switzerland)], and glatiramer acetate [GA; Copaxone (Teva Pharmaceutical Industries, Petah Tikva, Israel)] are widely used first line,1–5 but suboptimal adherence is common (often for injection-related reasons) and reduces their effectiveness.1,2,4,6–8 Efficacy, safety, tolerability, and convenience influence adherence of patients to therapy,2,4,6,9,10 defined here as therapy retention rate. Retention is particularly important for effective therapy2,4,6,7,10,11 and is clinically meaningful when multiple treatment options are available.4,5
Switching between iDMTs is common,1,5 and newer high-efficacy oral therapies and infrequently administered intravenous therapies are alternatives to regular injections.4,5,12 Although often used second line, fingolimod [Gilenya (Novartis Pharma AG, Switzerland)] is approved first line in several countries (including the USA), 12 is efficacious in early multiple sclerosis (MS), 13 and shows a high rate of patient retention.9,14 Well-tolerated oral therapy could result in higher retention compared with iDMTs; however, no randomized controlled trials (RCTs) have demonstrated this. Prescription data from pharmacy registries and observational cohort studies suggest greater therapeutic retention with oral MS therapies than with iDMTs;14–16 however, interpretation of such data is limited by well-known biases associated with retrospective, nonrandomized data.
Here, we report findings from the 48-week Prospective, Randomized, active-controlled, open-label study to Evaluate patient retention on
Methods
Study participants
Patients aged 18–65 years, diagnosed with RRMS (2010 international panel criteria) 17 and with an Expanded Disability Status Scale (EDSS) score up to 6, were enrolled from 117 centers in the USA between June 2012 and June 2014; last patient last visit was in July 2015. Eligible patients were treatment naïve or previously treated with no more than one class of study-approved iDMT. If appropriate, a negative pregnancy test and effective birth control methods were required; study sites and eligibility criteria are given in supplementary data 1.
Standard protocol approvals, registrations and patient consents
The study followed the Declaration of Helsinki and Good Clinical Practice guidelines, with applicable local regulations.18,19 Patients provided written informed consent before any assessments. Protocol amendments are in supplementary table 1. The study protocol and all amendments were centrally approved by Quorum Review IRB, and were reviewed at each center’s Independent Ethics Committee or Institutional Review Board.
Study design
PREFERMS was a randomized, open-label, active-controlled, parallel-group, multicenter study that followed up patients for 48 weeks. Visits occurred at screening, at baseline (day 0), and at weeks 4, 12, 24, 36, and 48 (supplementary figure 1 and supplementary table 2). Patient medical history, medication, and current disease status were obtained at screening. For patients previously treated with GA, an interferon iDMT was preselected during consultation with their physician before randomization. Similarly, GA was preselected for patients previously treated with an interferon (supplementary table 3); iDMTs of either class were preselected for treatment-naïve patients. Similar numbers of patients were preselected for each iDMT class [interferon, n = 420 (48%); GA, n = 455 (52%)].
Patients were randomized 1:1 to fingolimod 0.5 mg/day or to an iDMT using an interactive voice-and-web-response system (IVRS/IWRS) that automated random assignment of patient numbers to different treatment arms. After baseline assessment and patient eligibility confirmation, study medication was allocated via IVRS/IWRS and administered according to US prescribing information (supplementary table 3). To foster and track medication adherence, at each visit study drug accountability logs were maintained and updated by investigators, and patients were asked to return any unused drug and packaging; noncompliance was recorded as a protocol violation. The adherence rate in each group was also determined (duration of exposure excluding interruptions as a percentage of the study duration). At each visit, clinical, radiographic, cognitive, and patient-reported outcomes, and safety information were also collected (supplementary figure 1 and supplementary table 2).
One on-study treatment switch was allowed. Before the week 12 visit, patients could switch from randomized treatment to another study drug only for efficacy or safety reasons. Thereafter, patients could switch treatment for any reason. The 12-week threshold was chosen to allow sufficient time for flu-like symptoms to abate and for patients to acclimatize to regular injections.4,20 Patients underwent ‘last assessment’ when they switched from randomized treatment and at the end of the study. New patients were not enrolled to replace those who withdrew. Patients were not excluded from analyses owing to protocol deviations according to the intent-to-treat principle (Figure 1).
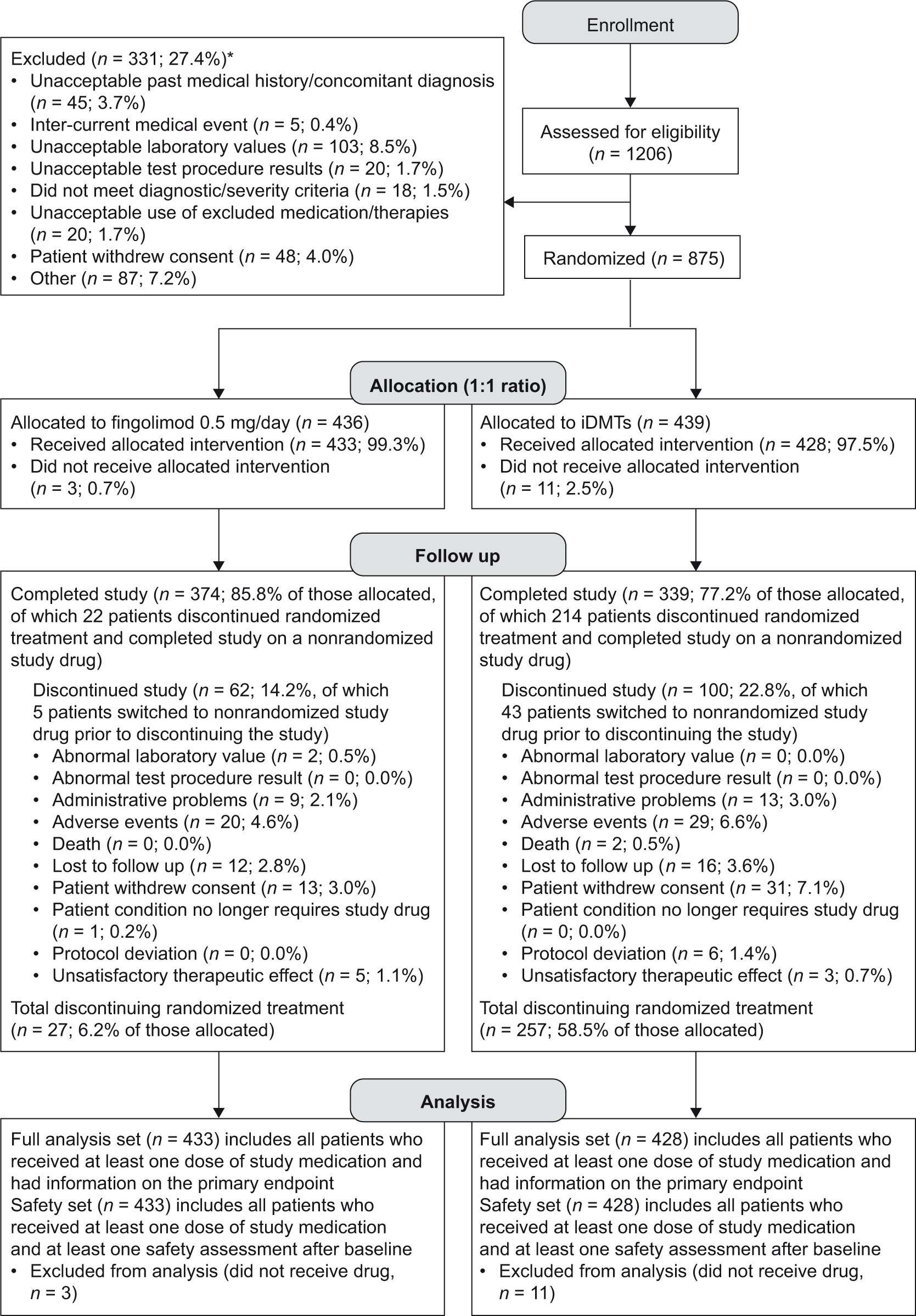
CONSORT patient flow diagram for PREFERMS.
The size, duration, and active comparator-controlled nature of PREFERMS, together with flexible treatment switching, were designed to determine whether starting fingolimod or an iDMT was associated with a higher therapeutic retention rate. A summary of the study design is presented in the PREFERMS video abstract (supplementary video 1).
Outcomes
The primary endpoint was patient retention on randomized treatment over 48 weeks. Secondary variables included reasons for discontinuation of randomized treatment; occurrence and persistence of drug-related adverse events (AEs); cognitive impairment [assessed by the Symbol Digit Modalities Test (SDMT)]; 21 changes in brain volume [measured as percentage change from baseline using magnetic resonance imaging (MRI)]; and patient-reported satisfaction [measured using the Medication Satisfaction Questionnaire (MSQ),22–24 a clinician-administered, single-item, seven-point Likert scale questionnaire, with a one-point change considered clinically meaningful]. All AEs and serious AEs (SAEs) were recorded by severity, duration, and relationship to study drug at each visit. Pre-existing medical conditions were classified as AEs only if they worsened after starting treatment. Exploratory endpoints included relapse (neurological abnormality present for ⩾24 h in the absence of fever or infection, occurring ⩾30 days after a preceding demyelinating event); cumulative number of newly active gadolinium-enhanced (Gd+) T1 lesions; number of new/enlarged T2 lesions; number of new active lesions; change in Gd+ lesion count; and changes in cortical gray-matter and thalamic volume measures.25–30 A central reader assessed all MRI data (NeuroRx, Montreal, Canada).
Statistics
Sample size and power calculations were based on retention rates from published studies;31–33 852 patients randomized 1:1 to fingolimod or iDMT had 84% power to detect an 8% difference in retention rates using a two-sided χ2 test with a significance level (α value) of 0.05. The study was powered only for the primary endpoint. For secondary and exploratory assessments, the last observation carried forward method was used to impute data missing at last assessment on randomized treatment. These analyses were used for hypothesis generation; no adjustments were made for multiple comparisons. Unless specified, all statistical tests assumed a two-sided alternative hypothesis, with a significance level (α value) of 0.05. All statistical analyses were performed under the direction of the corresponding author (BACC).
Retention rate over 48 weeks, and efficacy-related and patient-reported outcomes at last assessment on randomized treatment, were analyzed in the full analysis set (FAS; Figure 1). A Cochran–Mantel–Haenszel test assessed categorical variables, including the primary endpoint; a two-sample t test assessed continuous variables. Relapses and MRI lesion counts were analyzed using negative binomial regression, 34 which accounted for time on study drug, and SDMT scores and volumetric MRI measures using analysis of covariance. Sensitivity analyses and adjustments for treatment exposure and covariates are in supplementary table 4 and in the figure legends.
AEs and SAEs were analyzed in the safety set (Figure 1), with all events coded using the Medical Dictionary for Regulatory Activities. Following a protocol amendment (supplementary table 1 and supplementary table 4), AE and SAE counts were adjusted for treatment exposure and calculated per patient year (number of days on study drug for all patients in the group divided by 365.25). Exposure was from randomization to first occurrence of an event or, if a specific event was not reported, the entire study duration.
Results
Patient disposition
Of 1206 patients screened, 331 (27.4%) were excluded (Figure 1 and supplementary table 5), most frequently because of unacceptable laboratory test results, potentially signifying coexisting systemic disease [n = 103 (8.5%)]. In the fingolimod and iDMT groups, 436 and 439 patients were randomized, respectively, with 433 (99.3%) and 428 (97.5%) in the FAS [total, 861 (98.4%)]. Patient attrition was acceptable, with 713 patients (81.5%) overall completing the study including the switch phase, 477 (54.5%) on randomized treatment and 236 (27.0%) having switched treatment (Figure 1). On-study drugs to which patients switched are summarized in supplementary table 6. Study-drug adherence rates were 95.5% in the fingolimod group (n = 423) and 90.6% in the iDMT group (n = 427).
Baseline demographics and characteristics
Patient demographic and baseline characteristics were similar between treatment groups (Table 1). Most randomized patients were women [n = 640 (73.1%)], and most were white [n = 710 (81.1%)]. Mean (SD) age was 41.7 (10.6) years; time since diagnosis was 4.3 (6.3) years; time since first symptoms was 7.25 (7.9) years; and EDSS score was 2.4 (1.5). Over half of patients had previously received iDMT treatment [fingolimod group, n = 223 (51.1%); iDMT group, n = 248 (56.5%), supplementary table 6].
Baseline patient demographics and characteristics in PREFERMS.
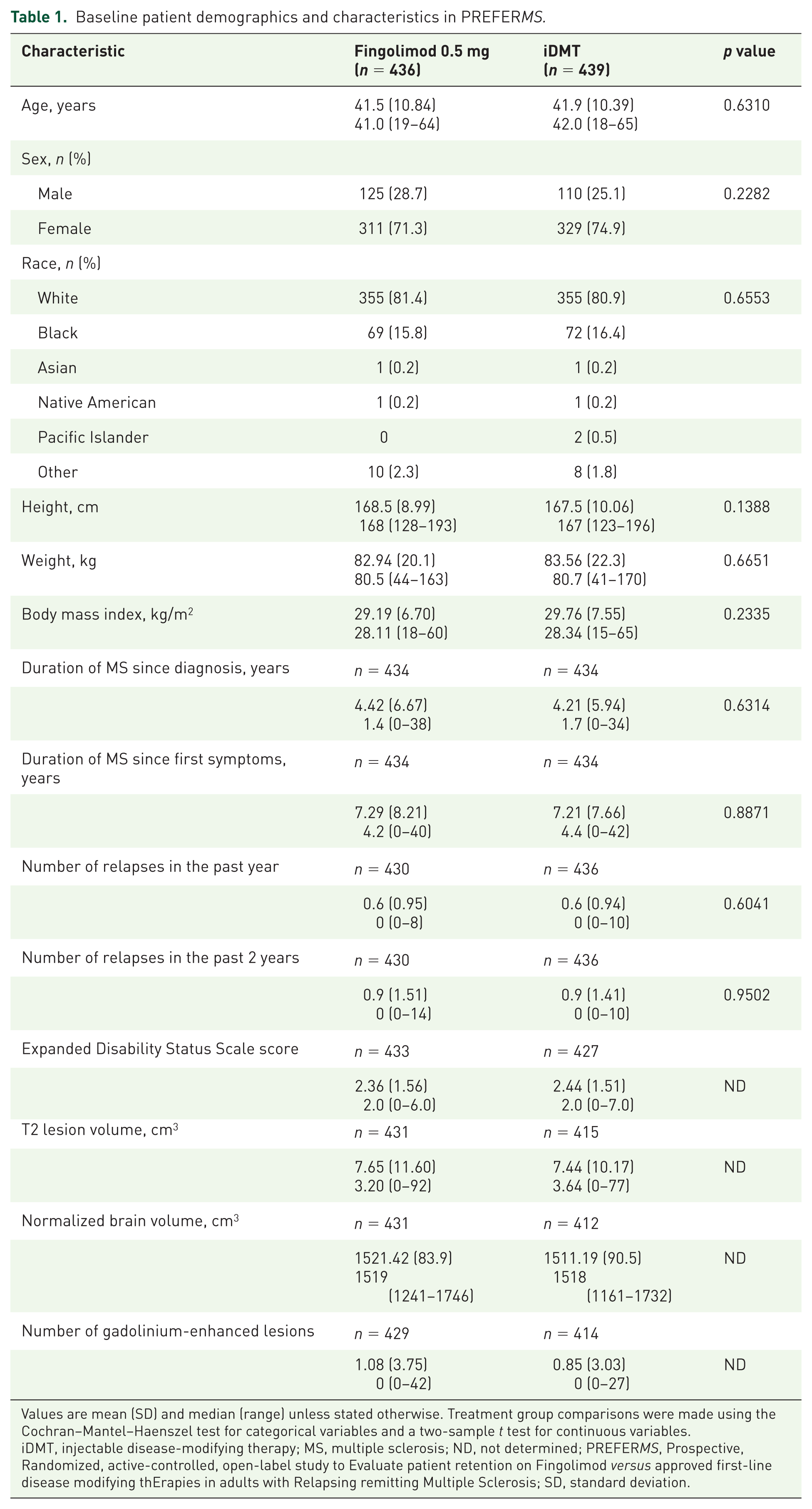
Values are mean (SD) and median (range) unless stated otherwise. Treatment group comparisons were made using the Cochran–Mantel–Haenszel test for categorical variables and a two-sample t test for continuous variables.
iDMT, injectable disease-modifying therapy; MS, multiple sclerosis; ND, not determined; PREFERMS, Prospective, Randomized, active-controlled, open-label study to Evaluate patient retention on Fingolimod versus approved first-line disease modifying thErapies in adults with Relapsing remitting Multiple Sclerosis; SD, standard deviation.
Retention rates
Retention rate on randomized treatment in the FAS was significantly higher with fingolimod than with iDMTs [352 patients (81.3%) versus 125 (29.2%); absolute difference (AD), 52.1%; 95% confidence interval (CI) 46.4–57.8%; number needed to treat (NNT), 1.92; p < 0.0001; Figure 2(a)]. During the randomized phase, mean duration of exposure to fingolimod was nearly twice that observed with iDMTs (301 versus 163 days). The significance of the primary endpoint was sustained: when a small number of protocol-deviation switches (in the first 12 weeks, not for safety or efficacy; n = 17) were excluded from the iDMT group [n/N (% retained): fingolimod, 352/433 (81.3%); iDMTs, 125/411 (30.4%); AD, 50.9%; 95% Cl 45.1–56.7%; NNT, 1.96; p < 0.0001]; when patients who switched treatment during days 77–110 (around the time when switching treatment for any reason became permissible) were excluded [n/N (% retained): fingolimod, 352/429 (82.1%); iDMT, 125/302 (41.4%); AD, 40.7%; 95% CI 34.0–47.3%; NNT, 2.46; p < 0.0001]; and when both of these groups of patients were excluded [Figure 2(b)]. We performed a second sensitivity analysis calculating retention rates in the two groups for weeks 16–48. Retention for fingolimod was 83.8% (n = 352) and for iDMT was 54.1% (n = 125). The between-group difference was 29.7% (95% CI 22.4–37.0%; p < 0.0001).

Retention rates, patient satisfaction with treatment, and reasons for discontinuing randomized treatment in PREFERMS. (a) The primary outcome of retention rate in the two treatment groups as a Kaplan–Meier plot, statistically analyzed as a log-rank test adjusted for treatment. The data were also analyzed by a Cochran–Mantel–Haenszel test, a logistic regression and a Cox proportional hazard model, which were adjusted for treatment and treatment naïvety. The data were also measured by normal approximation performed using continuity correction. ****p < 0.0001 for all analyses. (b) Sensitivity analysis of the retention rate shown in panel (a) excluding patients who switched treatment for nonefficacy or safety reasons before week 12 in violation of the study protocol, and all patients switching treatment between days 77 and 110 in case any had enrolled with the intention of switching from iDMT to fingolimod as soon as it became permissible for any reason. (c) Patient-reported satisfaction as measured by the Medication Satisfaction Questionnaire at last assessment. The overall difference across categories between fingolimod 0.5 mg and iDMTs is significant (Cochran–Mantel–Haenszel test using modified ridit scores adjusted for treatment and treatment naïvety; p < 0.0001). (d) Primary reasons for discontinuing randomized treatment. Labels for each bar represent the number (percentage of the total number of patients discontinuing randomized treatment). *Injection-related reasons for discontinuation are listed on the left of the corresponding area of the graph. †Reasons for ‘Other’ stated in the fingolimod group include the following [n = 1 (0.35%), unless stated otherwise]: abnormal platelet counts; adverse drug reaction; AE of headache; AE of joint pain; AE of papilloma, hair loss, herpes simplex virus, and lymphopenia; anxiety; hypertension; lymphopenia [n = 2 (0.70%)]; mouth sores; possible diagnosis of macular edema; side effects; and tolerability. Reasons for ‘Other’ in the iDMT group include the following [n = 1 (0.35%), unless stated otherwise]: abdominal pain, chest pain, flushing; AE; AE of bilateral lower extremity edema; AE of headache; allergic reaction [n = 2 (0.70%)]; anxiety; anxiety and depression; anxiety and fatigue; arthralgia and myalgia; bruising; convenience; fatigue [n = 2 (0.70%)]; flu-like symptoms [n = 2 (0.70%)]; general body ache [n = 2 (0.70%)]; headache [n = 2 (0.70%)]; injection fatigue; injection pain [n = 8 (2.82%)]; injection reaction; intolerant; lack of efficacy; muscle aches; mood altering; needle phobia [n = 4 (1.41%)]; palpitations; panic and irritability; patient choice [n = 10 (3.52%)]; possible seizure exacerbation; rash; relapse [n = 3 (1.06%)]; stopped taking medication; and treatment dissatisfaction [n = 2 (0.70%)]. AE, adverse event; CI, confidence interval; iDMT, injectable disease-modifying treatment; MRI, magnetic resonance imaging; PREFERMS, Prospective, Randomized, active-controlled, open-label study to Evaluate patient retention on Fingolimod versus approved first-line disease modifying thErapies in adults with Relapsing remitting Multiple Sclerosis.
Patient satisfaction on randomized treatment
Patient satisfaction with treatment was greater with fingolimod than with iDMTs throughout the study: the distribution of responses in the fingolimod arm was more heavily weighted towards ‘very satisfied’ and ‘extremely satisfied’ than in the iDMT arm [p < 0.0001 at last assessment, Figure 2(c)].
Discontinuation of randomized treatment
Of 284 patients who discontinued randomized treatment, 27 (9.5%) switched from fingolimod (6.2% of patients randomized) and 257 (90.5%) switched from an iDMT (58.5% of patients randomized). Figure 2(d) summarizes the reasons for discontinuation. Side effects of iDMTs and their mode of administration were the primary reasons for discontinuation reported by 66.1% of patients who discontinued an iDMT, accounting for 59.9% of all discontinuations. Of 257 patients who discontinued an iDMT, 255 switched to fingolimod, one to GA and one to intramuscular interferon β-1a. Most of the 27 patients switching from fingolimod did so owing to the occurrence of relapse or AEs. Of those individuals who discontinued fingolimod, 16 switched to GA (59.3%) and 11 to interferon (40.7%). A breakdown of the reasons for patients reporting ‘other’ is included in the figure legend. The likelihood of discontinuation by discontinuation category (efficacy, safety, tolerability, or convenience) is shown in supplementary figure 2; the likelihood of discontinuing iDMTs versus fingolimod was significantly more likely in all of these categories except safety, for which the between-group difference was nonsignificant.
Annualized relapse rates
There was a nonsignificant trend towards lower annualized relapse rate (ARR) among patients on fingolimod, with a rate 30% lower than those on iDMTs [fingolimod ARR, 0.22; iDMT ARR, 0.31; ARR ratio (95% CI), 0.70 (0.47–1.05); p = 0.084; Figure 3(a)].

ARR, key MRI measures, and volumetric brain changes at last assessment in PREFERMS.
MRI lesion activity
On randomized treatment at last assessment, the new Gd+ lesion count was significantly lower with fingolimod than with iDMTs [mean (SD) fingolimod, 0.16 (0.82); iDMT, 0.39 (1.45); p < 0.0001; Figure 3(b)]. The new/enlarged T2 lesion count was significantly lower with fingolimod than with iDMTs [mean (SD) fingolimod, 1.76 (4.82); iDMT, 2.46 (6.24); p < 0.0001; Figure 3(c)], as was mean cumulative number of newly active lesions [mean (SD) fingolimod, 0.58 (2.06); iDMT, 1.55 (5.22); p < 0.0001; Figure 3(d)]. There was a significantly greater reduction from baseline in Gd+ lesion count with fingolimod than with iDMTs [mean (SD) fingolimod, −0.86 (3.56); iDMT, −0.41 (2.82); p < 0.0001; Figure 3(e)].
Changes in brain volume, cortical gray-matter volume and thalamic volume
Because of differences in exposure to fingolimod and iDMTs, analyses of brain volumetric changes were adjusted for treatment exposure. At last assessment, there was less brain volume loss (BVL) and cortical gray-matter volume loss from baseline with fingolimod than with iDMTs [Figure 3(f)–(g)]. Fingolimod did not significantly reduce thalamic volume loss [Figure 3(h)]. Volumetric MRI outcomes unadjusted for treatment exposure are presented in supplementary figure 3.
Changes in cognitive function
Small increases in SDMT score from baseline were seen on randomized treatment in both groups, with higher values in the oral test (up to three-point increases from baseline) than in the written version (all less than one-point increase from baseline). Increases were numerically greater with fingolimod than with iDMTs at all assessments, but the between-group differences were nonsignificant except at last assessment among patients taking the oral test [fingolimod (n = 73); iDMT (n = 65); least-squares mean difference (standard error), 3.1 (1.5); p = 0.033]. Sensitivity analyses accounting for upper limb impairment and visual acuity yielded similar results, with oral test results no longer significant (p = 0.051; supplementary figure 4).
Adverse events
In total, 91.0% of patients on fingolimod (394/433) and 82.9% of patients on iDMTs (355/428) experienced AEs during the randomized treatment phase. Most AEs were mild or moderate in severity, with no severe AEs reported in over 2.8% of patients. Safety outcomes for all treatments were consistent with the respective prescribing information. Macular edema (ME) was confirmed by optical coherence tomography in seven patients while on fingolimod (1.1%) and one patient on GA (0.2%). There were pre-existing risk factors in four of the seven fingolimod cases (uveitis in both eyes with inactive macular thickening; retinitis pigmentosa with macular degeneration and a legally blind left eye; astigmatism with intermittent blurred vision; controlled diabetes), and ME was bilateral in five cases. All seven patients discontinued fingolimod, and all recovered. Two patients on fingolimod (0.3%) experienced mild symptomatic bradycardia (dizziness; headache; confused state), one during first-dose monitoring, and one on study who had a history of cardiac murmur. Neither patient required treatment or discontinued fingolimod. To account for differences in exposure, rates of AEs were calculated per patient year. Thus, the proportions of patients who experienced AEs during the randomized-treatment phase on fingolimod (91.0%) and on iDMTs (82.9%) equated to rates of 4.008 and 7.011 AEs per patient year, respectively (Table 2). In total, 9.2% of patients on fingolimod and 23.4% on iDMTs experienced AEs leading to discontinuation of randomized treatment [0.112 and 0.540 per patient year, respectively (Table 2)].
Safety assessment summary in PREFERMS: rates of AEs, SAEs, and AEs causing discontinuation of randomized treatment.

AEs causing study drug discontinuation are reported for events affecting at least 1% of patients in either treatment group. Rates of serious AEs are reported for events affecting at least two patients in either treatment group. Most of the between-group difference was attributable to injection-related reactions, fatigue, and influenzalike symptoms in the iDMT group. AE, adverse event; iDMT, injectable disease-modifying therapy; MS, multiple sclerosis; PREFERMS, Prospective, Randomized, active-controlled, open-label study to Evaluate patient retention on Fingolimod versus approved first-line disease modifying thErapies in adults with Relapsing remitting Multiple Sclerosis; SAE, serious adverse event.
The rates of SAEs per patient year were similar in the treatment groups (fingolimod, 0.083; iDMT, 0.076). SAEs were reported in 6.7% of patients on fingolimod and in 3.5% on iDMTs. Except for MS relapse (five patients in each group), no SAE was reported for more than two patients in either group. Pneumonia, dehydration, and suicidal ideation were each reported as SAEs for two patients in the fingolimod group. Anxiety was reported as an SAE for one patient in the fingolimod group and two patients in the iDMT group (Table 2). Serious opportunistic infections, such as progressive multifocal leukoencephalopathy, were not observed. Three patients died, one during screening (myocardial infarction), one from metastatic small-cell lung carcinoma (started on iDMT and switched to fingolimod), and one from cardiopulmonary arrest (iDMTs group). Deaths were unrelated to study medications.
A summary of the results is presented in the PREFERMS video abstract (supplementary video 1).
Discussion
A significantly higher retention rate over 48 weeks was observed in patients treated with fingolimod than among those on iDMTs. Between-group differences were anticipated,2,6,8,9,14–16,32,33 and retention rates of 86% for fingolimod and 78% for iDMTs were expected over 48 weeks, calculated from 6-month discontinuation rates reported in the EPOC study. 32 Similar retention rates over 1–2 years have been reported elsewhere.15,16,33 Most patients switching therapy during randomized treatment switched to fingolimod rather than to an alternate iDMT. Moreover, treatment satisfaction was higher with fingolimod than with iDMTs. These data indicate a generally greater tolerance and acceptance of fingolimod than of injectable therapies. Other studies, including meta-analyses and registry publications, support these findings.2,15,16,27,32
The proportion of screening failure in PREFERMS was relatively high at 27%, although this was lower than was seen in the phase III TRANSFORMS trial of fingolimod that used similar eligibility criteria. 27 This level of exclusion may hinder generalization of the findings to all clinical settings. Also, compared with routine clinical practice, an iDMT retention rate of 29.2% at 1 year is low. Owing to prescribing restrictions during the enrollment period (June 2012–June 2014), it is likely that some patients would not have been able to start or switch to fingolimod in a routine practice setting. Moreover, patients were aware at enrollment that they could switch to fingolimod after 12 weeks when switching for any reason became permissible. This may have biased recruitment in terms of preferred treatments or routes of administration. To address this, a sensitivity analysis excluded patients who switched treatment between weeks 11 and 16, and found that the retention rate with iDMT increased to 41.4%, but the between-group difference in retention relative to fingolimod remained highly significant. A second sensitivity analysis comparing retention rates during weeks 16–48 found similar highly significant results. Had we surveyed study participants regarding their preference for iDMT or fingolimod prior to randomization, such information could have been helpful in interpreting the decision to switch treatment. Nonetheless, we found that factors related to tolerability and convenience appeared to have the most influence on treatment retention. Injection-related issues dominated the reasons for discontinuing iDMTs in PREFERMS; thus, the preference for oral over injectable therapy might be expected. Nonetheless, the scale of this preference in PREFERMS was striking, and suggests that many patients receiving iDMTs would choose to switch to another therapy if given the opportunity.
There were trends towards better efficacy outcomes and treatment satisfaction when initiating and remaining on fingolimod than on iDMTs (numerically fewer brain lesions, reduced BVL and cortical gray-matter loss, and greater treatment satisfaction), and such treatment effects on cortical gray-matter volume and overall BVL are clinically relevant because they correlate with effects on disability progression and cognitive impairment.28,29,35,36 It is unexpected that cognition showed signs of improvement with fingolimod while thalamic volume on average decreased, but data were missing from both analyses so the two effects may not relate to the same subgroup of patients. Comparisons of therapeutic efficacy must acknowledge the greater treatment exposure to fingolimod than to iDMTs, and, although this did not affect the primary outcome or the adjusted safety assessments, the secondary and exploratory efficacy variables were affected. Owing to the large number of patients switching treatment, the iDMT group lost statistical power as the study proceeded, and had a shorter disease duration than the fingolimod group at last assessment. When adjusted for differences in treatment exposure, all volumetric MRI measures (total brain, gray matter, and thalamic volume loss) had decreased less with fingolimod than with iDMTs at the last assessment.
The study was open label owing to the different routes of administration in the treatment arms, and high rates of injection-related reactions complicated direct comparison of AE frequencies between treatments. Nevertheless, this design is informative in the setting wherein physicians may consider a switch to oral therapy for patients who poorly tolerate or dislike injectable medication and where payer policy allows.4,5,37 In patients at a relatively early stage of RRMS, PREFERMS showed that fingolimod is associated with a higher therapy retention rate than iDMTs, and revealed trends towards greater patient satisfaction and improved clinical and MRI outcomes with fingolimod.
Conclusion
Our findings indicate that, given the choice, many patients receiving iDMTs would consider switching to fingolimod. Whether this reflects a preference for oral therapy in general or for fingolimod in particular was not investigated. Patients with MS will benefit most in the long term if they adhere to therapy from an early stage of disease. There are nonmedical obstacles to switching MS therapy, but clinical practice might serve patients better if regular reviews to check treatment adherence, as well as safety and effectiveness, were routinely conducted.
Supplementary Material
Supplementary Material, Supplementary_files_final_version_PREFERMS – Phase IV study of retention on fingolimod versus injectable multiple sclerosis therapies: a randomized clinical trial
Supplementary Material, Supplementary_files_final_version_PREFERMS for Phase IV study of retention on fingolimod versus injectable multiple sclerosis therapies: a randomized clinical trial by Bruce A.C. Cree, Douglas L. Arnold, Mark Cascione, Edward J. Fox, Ian M. Williams, Xiangyi Meng, Lesley Schofield and Nadia Tenenbaum in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors would like to thank the patients and study site staff who participated in PREFERMS and Dr Jeremy R. Bright at Oxford PharmaGenesis for editorial assistance in preparing the manuscript. The authors would also like to thank Dr Augusto Grinspan, formerly an employee of Novartis Pharmaceuticals Corporation, for his contribution to protocol development.
Author contributions
BACC (corresponding author): study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. DLA: study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. MC: study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. EJF: study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. IMW: data interpretation, drafted and revised the manuscript for intellectual content. XM (statistical analysis): study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. LS: study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. NT (principal investigator): study design and conceptualization, data analysis and interpretation, drafted and revised the manuscript for intellectual content. All authors approved the final version of the manuscript for publication. A list of co-investigators and contributors is given in Appendix 1 in the Supplement. Trial Registration: ![]() identifier: NCT01623596.
identifier: NCT01623596.
Funding
PREFERMS was supported by Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA. Oxford PharmaGenesis, Oxford, UK provided editorial support, which was funded by Novartis Pharmaceuticals Corporation.
Conflict of interest statement
BACC has received personal compensation for consulting from AbbVie, Biogen, EMD Serono, GeNEuro, Novartis and Sanofi Genzyme. DLA has an equity interest in NeuroRx Research, and has received personal fees from Acorda Therapeutics, Biogen, EMD Serono, Genentech, Genzyme, Hoffmann-La Roche, MedImmune, Mitsubishi, Novartis, Receptos, and Sanofi-Aventis, and grants from Biogen and Novartis. MC has received research support or consulting fees from Acorda Therapeutics, Bayer HealthCare, Biogen Idec, EMD Serono, Genzyme, Genentech, Novartis, Roche, and Sanofi Aventis. EJF has received consultancy fees, honoraria, and travel or research support from Acorda Therapeutics, Bayer, Biogen, Chugai, Eli Lilly, EMD Serono, Genzyme, Novartis, Ono, Opexa Therapeutics, Roche, Sanofi, and Teva Neuroscience. IMW is an employee of Oxford PharmaGenesis. XM, LS, and NT are employees of Novartis Pharmaceuticals Corporation.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
