Abstract
The duration of action of oral levodopa becomes shorter as Parkinson’s disease (PD) progresses. Patients with advanced PD may develop potentially disabling motor fluctuations and abnormal involuntary movement (dyskinesia), which cannot be managed with optimized oral or transdermal PD medications. The progressively worsening symptoms can have a substantial impact on the patient quality of life (QoL). Levodopa–carbidopa intestinal gel (LCIG) is delivered continuously via a percutaneous endoscopic gastrostomy with a jejunal extension (PEG-J). LCIG is licensed for the treatment of levodopa-responsive advanced PD in individuals experiencing severe motor fluctuations and dyskinesia when available combinations of antiparkinsonian medications have not given satisfactory results. Initial evidence for the efficacy and tolerability of LCIG came from a number of small-scale studies, but recently, three prospective studies have provided higher quality evidence. A 12-week double-blind comparison of LCIG with standard levodopa therapy, a 52-week open-label study extension of the double-blind study, and a 54-week open-label safety study, demonstrated significant improvements in ‘off’ time and ‘on’ time without troublesome dyskinesia, and QoL measures that were maintained in the longer term. There are also observations that LCIG may be effective treatment for nonmotor symptoms (NMS) although the evidence is limited. There is a need for further research on the efficacy of LCIG in reducing NMS, dyskinesia and improving QoL. This review surveys the clinical evidence for the effectiveness and tolerability of LCIG in the management of advanced PD and highlights some practical considerations to help optimize treatment.
Keywords
Introduction
Parkinson’s disease (PD) is characterized by motor symptoms that include bradykinesia, rigidity, resting tremor and postural instability/gait disturbance [Olanow et al. 2009]. In addition to motor symptoms, people with PD experience a range of nonmotor symptoms (NMS) that include pain, drooling, dysphagia, constipation, bladder dysfunction, cognitive impairment, anxiety, depression, sexual dysfunction and insomnia [Chaudhuri and Schapira, 2009]. As motor symptoms and NMS worsen with disease progression they markedly interfere with daily activities and social interactions impacting upon the individual’s quality of life (QoL).
Depletion of striatal dopamine due to the degeneration of dopamine neurons in the substantia nigra pars compacta leads to the core motor features of PD. Oral levodopa, which is converted to dopamine following intestinal absorption, is the mainstay of treatment for PD. In the early stages of PD, it is hypothesized that dopamine loss due to neuronal degeneration is buffered by dopamine storage in the remaining neurons [Nyholm, 2007]. However, as PD advances, this buffering effect is lost and striatal dopamine levels become dependent on the peripheral availability of levodopa [Mouradian, 2012]. Intermittent oral dosing of levodopa provides fluctuating plasma levels of levodopa because of its short half-life, lack of controlled delivery and erratic absorption in the small intestine due to delayed gastric emptying [Nyholm et al. 2002; Olanow et al. 2009]. In advanced PD, this variability of levodopa plasma concentration leads to fluctuating striatal dopamine levels and intermittent or pulsatile stimulation of striatal dopamine receptors. Such intermittent stimulation leads to molecular and neurophysiological changes in the neurons, resulting in motor complications [Nyholm, 2007].
Continuous dopaminergic stimulation is a strategy aimed at decreasing motor fluctuations in advancing PD [Antonini et al. 2010b]. Three device-aided treatments are currently available that provide different kinds of continuous stimulation: levodopa–carbidopa intestinal gel (LCIG; Duodopa®, AbbVie, North Chicago, IL, USA) infusion [Nyholm, 2007; Olanow et al. 2014; AbbVie Ltd., 2016], subcutaneous (SC) apomorphine pump infusion, and deep brain stimulation (DBS) [Wolters, 2012]. LCIG is a carboxmethylcellulose aqueous gel containing 20 mg levodopa and 5 mg carbidopa monohydrate per ml [AbbVie Ltd., 2016]. LCIG is delivered directly to the proximal jejunum via a percutaneous endoscopic gastrostomy tube with a jejunal extension (PEG-J) connected to a portable, programmable infusion pump (CADD Legacy® Duodopa Pump, Smiths Medical, St Paul, MN, USA), including a cassette that contains the gel [Dam-Larsen et al. 2015]. Continuous infusion of LCIG bypasses gastric emptying, and thus avoids a potential cause of inadequate levodopa absorption [Nyholm et al. 2008]. The aim of LCIG infusion is to stabilize plasma levodopa concentrations and maintain them within a therapeutic window, avoiding the peaks and troughs that lead to motor fluctuations and dyskinesia [Nyholm, 2007].
LCIG infusion is currently licensed in Europe for the treatment of levodopa-responsive advanced PD in individuals experiencing severe motor fluctuations and hyper-/dyskinesia, although dyskinesia is not required in the US license, when available combinations of Parkinson medicinal products have not given satisfactory results [AbbVie Inc., 2016; AbbVie Ltd., 2016]. LCIG was approved in the EU in 2004, and has recently received approval in the USA following the publication of large-scale, high-quality clinical trials. Even with the publication of these large-scale studies, high-quality evidence on the use of LCIG is still limited; consequently, a review of the more recent literature on the management of motor fluctuations, NMS and QoL is necessary.
Literature search methods
A literature search was conducted on 30 May 2016 in PUBMED using the terms listed in Table 1.
Search terms used in PUBMED searches.
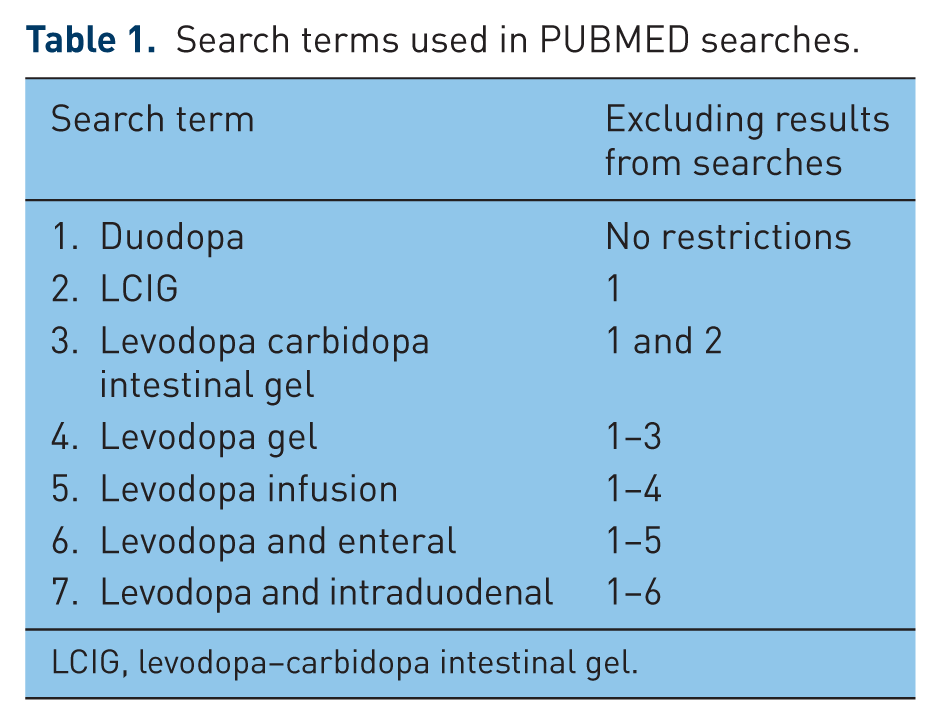
LCIG, levodopa–carbidopa intestinal gel.
From the search results, review articles (with a small number of exceptions), opinion pieces and publications that are not relevant have been removed. The process left 118 papers, which included (Figure 1):
Clinical trials and observational studies
Case studies/series
Selected review articles
Papers on specific issues (such as safety issues or QoL)
Pharmacokinetics (PK)
Non-English language that may be of particular interest

Flow diagram for article searches, exclusions and final included studies.
Of these 118 papers, those that duplicated information, or that were related to topics of minor clinical importance in the context of the current review were excluded from the final selection. Recent congress abstracts were also searched for clinical trial results not currently published in peer-review journals.
Levodopa–carbidopa intestinal gel dosing and cotherapies
The dosage of LCIG is individually titrated for the patient to achieve optimum control of motor fluctuations and reduce troublesome dyskinesia. The total daily dose (TDD) of LCIG comprises three individually adjusted doses: a morning bolus dose, the continuous maintenance dose administered over approximately 16 hours, and extra bolus doses that can be administered to regain control if there is a breakthrough of PD symptoms [AbbVie Ltd., 2016]. The initial dose on initiating LCIG treatment is based on the patient’s previous daily dose of oral levodopa (or levodopa dose equivalent), and is initially titrated over approximately 3–7 days to achieve an optimal response [AbbVie Ltd., 2016]. Dose titration may be conducted with a temporary nasojejunal tube, to ensure that the patient responds to levodopa and is able to manage the device, before a permanent PEG-J is established.
LCIG may be administered as monotherapy, or with adjunctive therapies. In centers where LCIG infusion is initiated, it is a common practice to stop previous adjunctive therapies before starting LCIG infusion. A retrospective review of two phase III studies found that stopping adjunctive therapy before initiating LCIG made little difference in the duration of dose titration, compared with continuing adjunctive therapy. The TDD of LCIG after titration in patients whose adjunctive therapy had been stopped tended to be higher than in patients whose adjunctive therapy was continued [Lew et al. 2015]. In the open-label study, where adjunctive therapy had been stopped, the mean TDD of LCIG after titration was 1572 ± 566 mg. In contrast, in the double-blind study, where patients could continue adjunctive therapy, the mean TDD was 1181 ± 480 mg. In the open-label study, the mean morning dose was 177 ± 68 mg and the mean continuous LCIG dosage 82 ± 33 mg/h. Patients self-administered a mean of approximately one extra dose per day (mean 43 ± 20 mg per dose) [Lew et al. 2015]. Mean TDD after the establishment of the PEG-J (1304 ± 62 mg) remained relatively stable after 6 months (1350 ± 624 mg) and 12 months (1412 ± 650 mg) in an ongoing global open-label study involving 375 patients in routine care [Antonini et al. 2015].
If patients require overnight treatment, one option is a combination of daytime LCIG infusion with slow-release oral levodopa taken at bedtime after stopping LCIG. In selected patients, overnight LCIG to improve quality of sleep can be used, however, this leads to a substantially increased TDD of levodopa, even though a lower infusion rate may be used during the night-time [Busk and Nyholm, 2012].
Levodopa–carbidopa intestinal gel pharmacokinetics and pharmacodynamics
LCIG infusion reduces variability in plasma levodopa concentrations compared with oral administration. A crossover, PK study in patients with advanced PD reported that the coefficient of variation (CV) in plasma levodopa concentrations with LCIG was 14% compared with 34%, with a slow-release levodopa/carbidopa tablet, but mean levodopa concentrations were identical [Nyholm et al. 2003]. Subsequent studies confirmed that LCIG infusion results in very minor fluctuations in plasma levodopa levels [Nyholm et al. 2003; Antonini et al. 2010a; Westin et al. 2011; Nyholm et al. 2012a, 2013; Othman and Dutta, 2014; Othman et al. 2015]. The addition of oral catechol-O-methyltransferase inhibitors to LCIG regimens was assessed in a small-scale study which showed that the levodopa dose could be reduced by 20% without affecting motor fluctuations and levodopa concentration stability [Nyholm et al. 2012b]. Such a strategy could be useful to keep the daily levodopa dose lower, while maintaining effective levodopa concentrations in the plasma and CNS, for example, in patients who require a little more than one cassette of LCIG per day.
Motor symptoms
Several different rating scales have been used to assess the efficacy of LCIG; the most frequently used are ‘on’/‘off’ time from patient diaries and the Unified PD Rating Scale (UPDRS) (Table 2).
Clinical studies with 30 or more patients with advanced Parkinson’s disease treated with levodopa–carbidopa intestinal gel: motor score outcomes and UPDRS scores.

Change from baseline at last assessment.
Follow-up data available from 67 patients that received LCIG via PEG-J; §Of 37 and 34 randomized to LCIG and immediate-release levodopa/carbidopa, respectively; ¶Of 59 patients enrolled; #Of 72 patients enrolled.
UPDRS, Unified Parkinson’s disease Rating Scale, Apo, apomorphine; DB, double blind; DD, double dummy; dys, days; h, hour; LCIG levodopa–carbidopa intestinal gel; mo, month; PEG-J, percutaneous endoscopic gastrostomy with jejunal extension; SC, subcutaneous; SD, standard deviation; wks, weeks, CI, confidence interval; SD, standard deviation.
Early studies with LCIG demonstrated that continuous infusion reduced ‘off’ time without worsening of dyskinesia and increased ‘on’ time without troublesome dyskinesia [Nyholm et al. 2005; Antonini et al. 2007; Eggert et al. 2008; Devos, 2009]. For example, one study involving 75 patients with PD, who started LCIG treatment in French hospitals, from 2003 to 2007, reported that 96.0% experienced improvement in motor fluctuations and 94.7% experienced improvements in dyskinesia [Devos, 2009].
The efficacy of LCIG in reducing motor fluctuations is now supported by three prospective, phase III studies: a randomized double-blind, double-dummy study; a 52-week open-label extension to the double-blind study; and a 54-week open-label safety study, that also provided efficacy data [Fernandez et al. 2013, 2015; Olanow et al. 2014; Slevin et al. 2015]. Patients in the double-blind study were randomized to treatment with oral placebo and active LCIG infusion (n = 37), or active immediate-release oral levodopa/carbidopa and placebo intestinal gel (n = 34) following the establishment of PEG-J tube. After 12 weeks, mean ‘off’ time was reduced by 4.04 hours/day among patients who received LCIG, compared with a reduction of 2.14 hours/day in the control group, a difference of −1.9 hours/day (p = 0.0015). Mean ‘on’ time without troublesome dyskinesia was increased by 4.11 hours/day in the LCIG group and 2.24 hours/day in the control group (p = 0.0059) (Table 2).
Participants in the double-blind study could continue into a 52-week open label trial; those already receiving LCIG continued on the same regimen (n = 31) and those receiving oral levodopa-carbidopa were switched to LCIG (n = 29) [Slevin et al. 2015]. Patients continuing LCIG maintained their improved ‘off’ time (p = 0.377) over the 52 weeks of the study. Patients in the LCIG-naïve group experienced significant improvements in ‘off’ time from 4 weeks after switching (p < 0.001) until the final visit, when the mean change from baseline was −2.34 hours/day (p < 0.001). There were significant improvements from baseline in ‘on’ time without troublesome dyskinesia in both treatment groups (p < 0.05) (Table 2) [Slevin et al. 2015].
In the 54-week, open-label study, ‘off’ time was significantly decreased from baseline to final visit by 4.4 ± 2.9 hours/day (p < 0.001), and ‘on’ time without troublesome dyskinesia had increased by 4.8 ± 3.4 hours/day. Mean ‘off’ time and ‘on’ time without troublesome dyskinesia were significantly reduced at all time-points from the first assessment at week 4. At week 12, the mean reduction in ‘off’ time and increase in ‘on’ time without troublesome dyskinesia was similar to the changes seen in the LCIG group in the double-blind study [Fernandez et al. 2013, 2015].
A post hoc analysis of the double-blind and open-label trials, examined the effect of LCIG in patients who had ⩾1 hour of ‘on’ time with troublesome dyskinesia at baseline. LCIG treatment reduced ‘on’ time with troublesome dyskinesia in all studies compared with baseline, and an increased LCIG dose was not significantly correlated with increased ‘on’ time with troublesome dyskinesia in either study (double blind: r = −0.073, p = 0.842; open label: r = −0.001, p = 0.992) [Antonini et al. 2016].
An interim 12-month analysis of an ongoing global, multicenter, long-term open-label study of 375 patients with advanced PD receiving LCIG reported reductions in mean daily ‘off’ time of −4.7 ± 3.4 h/day (p < 0.0001) and in ‘on’ time with dyskinesia of −1.7 ± 5.0 h/day (p = 0.0228) [Antonini et al. 2015].
Since the publication of the phase III studies, observational studies in Belgium and Romania (Table 2), a small-scale observational study in Australia, a small-scale pilot study in Japan, and a small-scale study in Japanese, Taiwanese and Korean patients with advanced PD have consistently demonstrated reductions in ‘off’ time and increased ‘on’ time without troublesome dyskinesia [Pickut et al. 2014; Othman et al. 2015; Bajenaru et al. 2016; Chang et al. 2016; Murata et al. 2016].
Significant improvements in the total UPDRS IV (motor complications) score were observed in most studies where this was reported [Antonini et al. 2008; Honig et al. 2009; Palhagen et al. 2012; Pickut et al. 2014; Zibetti et al. 2014; Martinez-Martin et al. 2015; Slevin et al. 2015].
Nonmotor symptoms
While there are considerable data on the effects of LCIG on motor fluctuations and dyskinesia, the data on NMS are more limited (Table 3). The first prospective study to assess the effects of LCIG on NMS was a multicenter observational study in 22 patients with advanced PD. After 6 months, the total score on the Non-Motor Symptom Scale (NMSS) showed a statistically significant 56% reduction (improvement). Statistically significant decreases ranging from 54% to 81% (p < 0.001 from baseline) were also observed in six of the nine NMSS domains [Honig et al. 2009].
Clinical studies with 30 or more patients with advanced PD treated with LCIG: in QoL and NMS outcomes.

Change from baseline at last assessment.
Of 37 and 34 randomized to LCIG and immediate-release levodopa/carbidopa, respectively.
Apo, apomorphine; DB, Double-blind; DD, double dummy; LCIG, levodopa–carbidopa intestinal gel; SC, subcutaneous; SD, standard deviation.
The effects of longer-term LCIG treatment on NMS were subsequently assessed in a 12-month interim analysis of the global, long-term open-label study of patients with advanced PD in a routine clinical setting. NMSS total scores were significantly reduced at 6 months (−20.5, p = 0.0001) and 12 months (−22.2, p = 0.0014). Three of the nine NMSS domains, sleep/fatigue, gastrointestinal tract and urinary, also showed significant improvements at 12 months [Antonini et al. 2015].
Improvements in various NMS domains have been reported in other observational and retrospective studies [Reddy et al. 2012; Cáceres-Redondo et al. 2014; Buongiorno et al. 2015; Martinez-Martin et al. 2015].
The sleep/fatigue, gastrointestinal tract and urinary domains of the NMSS appear to be consistently improved in these studies, but it is anticipated that further data on the effects of LCIG infusion on NMS will be provided by the ongoing 26-week interventional INSIGHTS study [ClinicalTrials.gov identifier: NCT02549092]. The INSIGHTS study will compare the effects of LCIG with those of optimized medical treatment on the NMSS total score and the Modified PD Sleep Scale (PDSS-2) total score. Other endpoints in the INSIGHTS study include the UPDRS, Clinical Global Impression (CGI) and Parkinson’s Disease Questionnaire-8 (PDQ-8) scores. An interim report on a recently completed open-label, multicenter study [ClinicalTrials.gov identifier: NCT01736176] has shown improvements in sleep parameters and gastrointestinal disturbances within 12 weeks of commencing LCIG treatment; full publication is awaited [Standaert et al. 2015].
Quality of life
In addition to symptom control, improvement in QoL measures, including the PDQ-39, have been reported with LCIG in small-scale studies and more recently, in prospective studies [Nyholm et al. 2005; Honig et al. 2009; Palhagen et al. 2012; Reddy et al. 2012; Fernandez et al. 2013, 2015; Bajenaru et al. 2016; Chang et al. 2016].
In the double-blind study, significant improvements in the PDQ-39 score were seen with LCIG compared with oral levodopa-carbidopa after 12 weeks of treatment (difference −7.0, p = 0.0155). There were also significant differences favoring LCIG in the UPDRS II (activities of daily living score) (p = 0.0086) [Olanow et al. 2014]. Similarly, significant improvements (p < 0.001) in measures associated with QoL and function were seen from week 4 in the 54-week open label study (Table 3) [Fernandez et al. 2013, 2015].
In the interim analysis of the 12-month ongoing open-label study in routine care, the PDQ-8 total score was significantly improved from 1 month after the start of treatment, and at 12 months showed a mean reduction of 8.6 ± 22.6 points (p = 0.01). At month 12 the UPDRS II at the ‘on’ state was −3.1 ± 8.7 points (p = 0.0107) [Antonini et al. 2015].
In a 12-month interim analysis of a prospective long-term observational study involving 27 patients, there was a 5-point improvement in the PDQ-39 score, which although it did not reach statistical significance, was greater than the minimum clinically important difference in PDQ-39 usually cited [Peto et al. 2001; Palhagen et al. 2012].
PD can also negatively impact on the caregiver’s QoL. LCIG infusion has been shown to reduce the stress and burden felt by the caregiver and improve their QoL [Santos-Garcia et al. 2012; Ehlers et al. 2015].
Long-term efficacy
Although data are limited, the effectiveness of LCIG over periods longer than a year in controlling motor fluctuations and reducing dyskinesias has been reported in a number of studies. A study in specialized PD clinics and university hospitals reported on 73 patients with advanced PD with up to 2 years (mean 608 ± 292 days) of follow up. At final follow up, there were significant decreases in the UPDRS IV items duration of dyskinesia and disability due to dyskinesia, and improvements in the item duration of ‘off’ periods (p ⩽ 0.05 versus baseline) [Antonini et al. 2013].
In a Spanish study, 72 patients receiving LCIG, followed for up to 48 months, (mean 22 ± 14 months), showed significant reductions in mean ‘off’ time from 6.8 ± 2.8 hours/day before LCIG to 3.0 ± 3.5 hours/day at the final assessment (p < 0.0001) (Table 2) [Buongiorno et al. 2015].
In other smaller-scale studies in Spain, the Middle East and Italy, with mean follow ups of 32.2 ± 12.4 months, 48.5 ± 23.2 months and 25.8 ± 19.5 months, respectively, LCIG treatment produced long-term benefits in UPDRS III score, UPDRS IV total score, ‘off’ time duration, dyskinesia disability and ‘on’ time with dyskinesia duration [Zibetti et al. 2013; Cáceres-Redondo et al. 2014; Zibetti et al. 2014; Bohlega et al. 2015]. Significant improvements it QoL measures were observed at follow ups of 2 years and 25.8 ± 19.5 months in studies in Spain and Italy [Antonini et al. 2008; Cáceres-Redondo et al. 2014; Zibetti et al. 2014].
In a retrospective analysis of 150 patients who started treatment between January 1991 and June 2008 in centers in Sweden, the mean duration of treatment was 7.79 years [95% confidence interval (CI), 7.05–8.53] and the range 0–16 years. Currently, 16 years is the longest reported duration of LCIG treatment in any patient with advanced PD [Nyholm et al. 2012c]. LCIG treatment can be continued for as long as the patient gains benefit, but should be discontinued when side effects and disadvantages with the treatment are greater than the gain. For example, as the disease progresses there may come a time when even the maximum doses of LCIG are providing inadequate control of motor fluctuations, or dyskinesias during ‘on’ time are increasing. Treatment may also be withdrawn if the patient or their caregiver becomes unable to manage the medication or the device. In such circumstances, the PEG-J may be removed and treatment converted to oral levodopa therapy.
Safety profile
An integrated review of safety from four prospective multicenter phase III studies, three published and one ongoing [ClinicalTrials.gov identifier: NCT00360568] included a total of 412 patients with advanced PD who received LCIG and 395 with a PEG-J established [Olanow et al. 2014; Fernandez et al. 2015; Lang et al. 2015; Slevin et al. 2015]. This is the largest study evaluating the safety of LCIG infusion, covering a total of 963 years of exposure, with a median exposure to LCIG of 911 days (range 1–1980 days) [Lang et al. 2015].
Most patients [94% (387/412)] experienced an adverse event (AE); these were considered to be serious in 47% (194/412) [Lang et al. 2015]. A procedure or device-related AE occurred in 76% (300/412) of patients; 17% (68/395) of these events were considered to be serious (Table 4). Another analysis of the phase III safety data, which included 395 patients, reported similar incidences of serious AEs (SAEs) and serious device-related AEs [Epstein et al. 2016]. The prevalence of device or procedure-related events was highest within the 2 weeks after the PEG-J procedure and fell substantially afterwards, mostly resolving within the titration period, and then stabilized over time [Lang et al. 2015]. The second analysis of the safety data reported that 42% of all SAEs occurred within the first 4 weeks after establishment of the PEG-J [Epstein et al. 2016].
Incidence of adverse events (AEs) and serious AEs (SAEs) in the integrated safety review of clinical trials involving levodopa–carbidopa intestinal gel, Lang et al. [2015].

Occurred after end of dose titration; §re-emergence of Parkinson’s disease symptoms.
LCIG, levodopa–carbidopa intestinal gel; PEG-J, percutaneous endoscopic gastrostomy with jejunal extension, PD, Parkinson’s disease, AE, adverse event, SAE, serious adverse event.
AEs that were not procedure or device related occurred in 92% (379/412) of patients, and the most frequent that were probably or possibly related to LCIG were dyskinesia and weight decrease (Table 4) [Lang et al. 2015]
In this combined safety analysis, 17% (72/412) of patients discontinued LCIG treatment prematurely because of an AE: the most frequently reported events that led to discontinuation were complications of device insertion (2.4%), death (1.2%), abdominal pain (1%), pneumonia (1%), myocardial infarction (0.8%), and fall (0.7%). Of the patients who had the PEG-J established, 4.2% (19/395) discontinued due to a procedure or device-related AE [Lang et al. 2015].
The incidence of discontinuations and AEs in other studies have been broadly in line with the findings from the integrated review outlined above [Nyholm et al. 2005; Klostermann et al. 2012a; Fernandez et al. 2013, 2015; Olanow et al. 2014; Pickut et al. 2014; Bohlega et al. 2015; Melgari et al. 2015]. Other reported reasons for discontinuation of LCIG treatment include stoma infection, dyskinesia, peritonitis, and duodenal perforation while replacing the PEG-J tube [Calandrella et al. 2015]. As in the combined safety analysis, other studies tended to show a peak in procedure or device-related AEs shortly after LCIG initiation, which then declined sharply. Subsequently, most AEs were related to the device or tubing, or were AEs consistent with patients receiving long-term oral levodopa treatment [Nilsson et al. 2001; Nyholm and Lennernas, 2008; Nyholm et al. 2012c; Antonini et al. 2013, 2015; Zibetti et al. 2013, 2014; Bohlega et al. 2015; Buongiorno et al. 2015; Calandrella et al. 2015; Slevin et al. 2015].
Peripheral neuropathy is associated with PD and levodopa therapy generally, but its causes are not fully understood [Toth et al. 2010; Grambalova et al. 2015]. Peripheral neuropathy may be related to a vitamin B12 (cobalamin) deficiency, homocysteine neurotoxicity or the neurotoxicity of the levodopa itself [Rajabally and Martey, 2011;Klostermann et al. 2012b]. A number of cases of neuropathy have been reported in patients receiving LCIG [Antonini et al. 2007; Manca et al. 2009; Urban et al. 2010; Muller et al. 2011; Klostermann et al. 2012b; Galazky et al. 2014; Lehnerer et al. 2014; Lang et al. 2015; Merola et al. 2015; Uncini et al. 2015]. The progression of peripheral neuropathy has been shown to be halted or reversed by vitamin B12 supplementation, vitamin B1 or B6 supplementation, and folate supplementation in some cases [Manca et al. 2009; Urban et al. 2010; Klostermann et al. 2012b; Uncini et al. 2015]. In a study that assessed PD patients for up to 2 years after commencing LCIG treatment, patients with peripheral neuropathy received vitamin B1 and B12 supplementation and showed clinical improvement and sometimes, stability at subsequent assessments [Merola et al. 2015]. Suggested preventative measures against peripheral neuropathy include laboratory testing for vitamin B12 and folate deficiency prior to starting LCIG, with supplementation before and during LCIG treatment for patients at risk of vitamin deficiency [Merola et al. 2015; Uncini et al. 2015]. Electrophysiological assessment before and at intervals during LCIG therapy has also been proposed to reduce the risk of peripheral neuropathy or identify its onset [Merola et al. 2015; Uncini et al. 2015]. In the phase III double-blind study, symptoms associated with neuropathy were reported in one patient that received LCIG and three patients that received oral levodopa-carbidopa [Olanow et al. 2014].
The dyskinesia-hyperpyrexia syndrome, comprising severe dyskinesia, hyperthermia, rhabdomyolysis and a confusional state, is a rare complication of PD treatment that may be associated with a high level of dopaminergic stimulation [Gil-Navarro and Grandas, 2010; Taguchi et al. 2015]. In a case study of a PD patient who had experienced episodes of dyskinesia-hyperpyrexia during three consecutive summers while receiving oral levodopa, commencement of LCIG therapy led to a complete cessation of further episodes [Herreros-Rodriguez and Sánchez-Ferro, 2016].
Due to the high percentage of AEs associated with device insertion, a gastroenterologist/surgeon experienced in the establishment of the PEG-J and an experienced post-procedural care team including specialist PD nurses could be expected to minimize the risk of complications of device insertion and tubing problems such as twisting, kinking or knotting [Negreanu et al. 2010; Krones et al. 2012; Pedersen et al. 2012; Stathis et al. 2014; Dam-Larsen et al. 2015; Del-Hoyo-Francisco et al. 2015].
Comparing levodopa–carbidopa intestinal gel infusion with other advanced therapies
There have been no prospective head-to-head trials with LCIG and continuous SC apomorphine infusion or DBS.
A 6-month open-label observational multicenter study comparing 44 patients receiving LCIG and 43 receiving SC apomorphine infusion showed similar changes in UPDRS III and IV scores, NMSS total score and PDQ-8 index with each treatment [Martinez-Martin et al. 2015]. The NMSS domains sleep/fatigue, gastrointestinal, urinary and sexual function all showed significantly better improvements with LCIG than with SC apomorphine, while SC apomorphine produced a significantly greater improvement on mood/apathy [Martinez-Martin et al. 2015].
A retrospective analysis assessed 40 patients with advanced PD with motor complications treated with LCIG (n = 20) or by DBS (n = 20). The patients in the LCIG group were over 70 years of age and had more impaired neuropsychological functions than in the DBS group. In both groups there were significant reductions in UPDRS II, UPDRS III, and UPDRS IV and reductions in the percentage of waking day as ‘off’ time, but only the DBS group showed a significant improvement in dyskinesia duration and disability (p < 0.001). DBS was associated with a significant drop in the phonemic verbal fluency score, whereas LCIG produced a milder worsening in this task [Merola et al. 2011]. The randomized INVEST study [ClinicalTrials.gov identifier: NCT02480803] is expected to report results of a comparison between LCIG and DBS in 2019 [Van Poppelen et al. 2016]. Although INVEST is primarily a cost-effectiveness study it will also provide data on the effect of the treatments on QoL measures and a number of neuropsychological parameters.
In addition to the existing therapies, a liquid levodopa-carbidopa formulation for SC administration is in development, but is not yet licensed [Giladi et al. 2015].
Patient selection
There are a number of relative and absolute contraindications to the use of LCIG in patients with advanced PD. These are summarized in Table 5. Factors that may predict the effectiveness of LCIG include the patient’s age and the absence of psychiatric/behavioral symptoms [Sensi et al. 2014]. In addition, a correlation analysis from two clinical trials showed that patients with the greatest motor disabilities, assessed by the UPDRS, gained the greatest benefits from LCIG infusion [Westin et al. 2006].
Relative contraindications, precautions, warnings and absolute contraindications to levodopa-carbidopa intestinal gel, Odin et al. [2015], AbbVie Ltd., [2016]. $

Based on Summary of Product Characteristics and published opinion.
CV, cardiovascular; MAO, monoamine oxidase.
In the Navigate PD program, a group of 103 experts in PD overseen by an International Steering Committee of 13 movement disorder specialists identified 10 critical questions related to device-aided management of PD. These questions were addressed through a review of the available published evidence synthesized with expert medical opinion at national and pan-European levels to develop information to supplement existing guidelines [Odin et al. 2015]. Consensus among the participants in the draft guidance from the Navigate PD program was that:
For patients aged under 70 years of age with motor fluctuations or dyskinesias who are otherwise healthy, any of the device-aided therapies may be considered;
For patients aged over 70 years, DBS surgery should be considered second line among the device-aided therapies (although patients can be operated on in the presence of a normal MRI and preserved cognitive function);
And for patients over 70 years of age with mildly or moderately impaired cognition (or other contraindications to DBS), LCIG infusion or SC apomorphine infusion may be considered with cessation or reduction in oral therapy [Odin et al. 2015].
In everyday clinical practice, and with the lack of definitive evidence, the timing and choice of device-aided therapy will be based on many individual factors. Among these, preference of the patient, their caregivers or family, and of the healthcare team at the center responsible for management will heavily influence the treatment choice. Not all treatments will be available to all eligible patients, and this will also limit the choice of treatments offered.
Limitations of levodopa–carbidopa intestinal gel therapy
The limitations of LCIG therapy mostly relate to the relative and absolute contraindications summarized in Table 5 [Odin et al. 2015; AbbVie Ltd., 2016]. The aforementioned problems with the tubing are the most common reasons for discontinuation of the therapy, so device-related issues are a limitation that needs to be addressed in future development. The cost of LCIG may also be regarded as a limitation on a societal level; this is discussed below.
Practical considerations for initiating levodopa–carbidopa intestinal gel therapy
A multidisciplinary team (MDT) approach is important for the management of patients with advanced PD receiving LCIG [Van Der Marck et al. 2014]. An MDT should contain individuals with a wide range of expertise and experience, such that the team is well equipped to deal with all aspects of caring for the patient with PD. The team will usually have a core comprising a movement-disorder specialist or neurologist with expertise in movement disorders, a specialist PD nurse, and depending on local practice, a gastroenterologist or surgeon and an interventional radiologist. A range of other healthcare professionals may form part of the team [Pedersen et al. 2012].
The movement disorder specialist has responsibilities for assessing the patient and calculating the individualized LCIG doses. Adjustments of concomitant PD medications may or may not be done at LCIG initiation; however, patients can be switched directly to LCIG from polytherapy without problems [Meiler et al. 2008; Pedersen et al. 2012].
Before establishment of the permanent PEG-J, the patient may undergo a test where the LCIG is administered via a nasojejunal tube, this allows the movement disorder specialist to determine whether the LCIG is effective and titrate the dose. The test phase also provides an opportunity for the PD nurse to train the patient on the use of the pump, and enables the patient to decide whether to proceed to a PEG-J. The PD nurse will also be involved in monitoring the patient’s response to LCIG.
The PEG-J is established by the gastroenterologist or surgeon, depending on local practice. The gastroenterologist, along with the PD nurse and other members of the MDT will monitor the patient for problems with the device and tubes, and to ensure the optimum dosage is being delivered, until discharge [Pedersen et al. 2012; Fasano et al. 2015]. Alternative homecare approaches to dose titration may help reduce the need for hospitalization [Willows et al. 2015].
Cost effectiveness
Costs associated with advanced PD include direct costs such as the costs of the medication, physician office visits, hospitalization and medical intervention, costs to the patient and caregiver, and indirect costs such as loss of productivity.
A recent review suggested that for patients with advanced PD, interventions such as LCIG or DBS are more cost-effective than standard treatment [Rodriguez-Blazquez et al. 2015]. This may be due to the patients’ improved ability to function, placing less of a burden on caregivers and health services. Healthcare costs arising from treating complications of PD might also be reduced.
A study in the UK concluded that LCIG was cost-effective compared with standard care, over an estimated duration of treatment of 5 years, in terms of cost for incremental increase in quality-adjusted life years (QALYs) [Lowin et al. 2011]. Likewise in Sweden, LCIG was judged to be a cost-effective in terms of cost for QALYs over the course of a year [Willis et al. 2010]. However, a study in Norway concluded that although LCIG improved function, it did not meet the threshold for cost-effectiveness in the Norwegian healthcare system [Lundqvist et al. 2014]. The randomized INVEST study [ClinicalTrials.gov identifier: NCT02480803] is expected to report the results of a cost-effectiveness comparison of LCIG and DBS in 2019 [Van Poppelen et al. 2016].
Direct acquisition costs of SC apomorphine may be lower than for LCIG in many countries [Valldeoriola et al. 2013], however, other daily costs (such as infusion sets with cannulas for SC apomorphine that need replaced twice daily) also influence direct medical costs. Currently there are no studies that compare the cost-effectiveness of SC apomorphine infusion with that of LCIG infusion for the treatment of advanced PD, and therefore, no conclusions can be made on the relative societal costs of these treatments. A model analysis based on healthcare costs from the UK and Germany concluded that SC apomorphine infusion is cost-effective compared with standard of care [Walter and Odin, 2015].
Conclusion
Device-aided therapies such as LCIG infusion potentially provide a wide variety of benefits for patients with advancing PD, and therefore, there is a need for the patient’s physician to recognize motor fluctuations and refer patients to a movement-disorders specialist. A number of guidelines exist for the management of advanced PD with LCIG; these focus on control of the main motor symptoms in PD, and guidance on the use of LCIG for relief of NMS is limited. The Navigate PD program was a pan-European education program that aimed to reach consensus on unresolved issues in PD management and involved contributions from 103 experts in 13 countries [Odin et al. 2015]. There is strong evidence in published studies for the efficacy of these device-aided treatments in control of the main motor symptoms of PD, reviewed here, but questions only partially addressed are the effect of these treatments on dyskinesia and NMS.
The recent 12-week double-blind study, 52-week open-label follow-up study and 54-week open label study demonstrated that optimizing levodopa delivery with LCIG reduces motor symptoms without increasing, and sometimes decreasing, troublesome dyskinesia in patients with advanced PD whose motor symptoms are not effectively controlled by oral therapies [Fernandez et al. 2013, 2015; Olanow et al. 2014; Slevin et al. 2015]. These high-quality studies have measured the efficacy and safety of LCIG for up to 1 year, but other studies have indicated sustained improvements in ‘off’ time for up to 7 years in selected patients. In addition, a number of observational studies have shown a reduction in dyskinesias with short- and long-term LCIG infusion [Antonini et al. 2007; 2015; Eggert et al. 2008; Devos, 2009; Pickut et al. 2014; Othman et al. 2015; Bajenaru et al. 2016; Chang et al. 2016]. There have also been some reports of improvement in QoL measures with LCIG in a variety of studies in both the short and long term [Nyholm et al. 2005; Honig et al. 2009; Palhagen et al. 2012; Reddy et al. 2012; Fernandez et al. 2013, 2015; Chang et al. 2016]. The effect of LCIG on NMS comes from a few observational studies and a single open-label, small-scale prospective study [Honig et al. 2009; Reddy et al. 2012; Cáceres-Redondo et al. 2014; Antonini et al. 2015; Buongiorno et al. 2015; Martinez-Martin et al. 2015]. Further studies are required to investigate LCIG as a treatment for NMS.
There are currently no published prospective randomized clinical trials comparing LCIG with SC apomorphine infusion or DBS; limited data suggest some differences in response on the motor scales and NMSS [Merola et al. 2011; Martinez-Martin et al. 2015]. However, patient selection for a particular device-aided therapy may depend on many factors that would be difficult to assess in the clinical trial setting. Some contraindications for LCIG therapy have been defined, but many practical considerations and patient preference are crucial in defining individualized treatments for patients with advanced PD. Future data may help define the impact of LCIG on NMS, which in turn, may refine patient selection. In addition, more long-term experience may indicate whether earlier LCIG therapy initiation could have benefits [Antonini et al. 2015], similar to earlier DBS [Schuepbach et al. 2013].
Footnotes
Acknowledgements
The authors thank Martin Gilmour and Tim Latham of ESP Bioscience (Crowthorne, UK) for editorial assistance with the preparation of this manuscript, and AbbVie for funding this editorial support. The terms of the literature search were defined by both JV and DN and editorial support was provided to implement these searches. Both authors were substantially involved in developing the outline and structure of the review, and editorial assistance was provided in drafting and copy-editing this outline. Both authors were substantially involved in drafting and critically reviewing all drafts of the manuscript, editorial support comprised assisting in the drafting, copy-editing and incorporation of amendments. The final draft of the manuscript for submission was approved by both authors, and administrative assistance was provided in the submission process.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The editorial support for development of this manuscript work was funded by AbbVie. The company provided no input into the content of this article. The authors did not receive any honoraria from AbbVie or any other source for the preparation of this manuscript.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Johan Virhammar has received an educational lectures honoraria from Medtronic, receives remuneration from the website internetmedicin.se and has been supported with a research grant from Selanders Foundation. Dag Nyholm has received lecture fees from AbbVie; has received research support from AbbVie and has, until 2012, received compensation from AbbVie for serving as a consultant; has received royalties from Liber AB; has served as a consultant to Sensidose AB and OrbiMed Advisors LLC; has received honoraria from H. Lundbeck AB, Movement Disorders Society, NordicInfu Care and The National Board of Health and Welfare; NordicInfu Care; Ipsen, Selanders Foundation, Swedish Knowledge Foundation, Swedish PD Foundation, Swedish Research Council and VINNOVA Sweden’s innovation agency; is cofounder and stock owner in Jemardator AB; receives remuneration from the website netdoktor.se for participation in an expert panel; and received institutional support from Uppsala University Hospital.
