Abstract
Background:
Levodopa-carbidopa intestinal gel (LCIG) is effective in the treatment of advanced Parkinson’s disease (PD). However, the patients’ profile that might benefit from treatment with LCIG has not been characterized.
Objective:
This retrospective study explored the influence of disease duration (DD) on the effectiveness of LCIG and identified factors associated with treatment discontinuation in a cohort of advanced PD patients.
Methods:
Patients initiating LCIG therapy between Jan-2006 and Dec-2011 in 18 Spanish centers were included. Effectiveness in treating motor symptoms (MSs), non-motor symptoms (NMSs), and adverse events (AEs) occurrence was compared in DD≥10 or <10 years and LCIG continuation/discontinuation groups. Factors associated with LCIG discontinuation were evaluated using univariate and multivariate analyses.
Results:
Overall, 177 PD patients were included (52.5% male; mean age 70.6±8.4 years; mean LCIG duration 35.6±18.6 months). Patients with DD≥10 years (n = 125) experienced less reduction in “off” time (–29%) than those with DD <10 years (–38%; n = 51; p = 0.021), and reported more severe AEs (32.8% vs. 17.6%; p = 0.043). DD did not significantly influence changes in NMSs or discontinuation rates. Fifty-four patients discontinued LCIG therapy, factors associated with discontinuation were higher percentages of waking day in the “off” state (OR, 1.028; 95% CI, 1.002–1.055; p = 0.0360) and in the “on” state with troublesome dyskinesia (OR, 1.032; 95% CI, 1.002–1.064; p = 0.0376) at baseline.
Conclusions:
Advanced PD patients with DD <10 years might benefit more from treatment with LCIG than patients with a longer DD. Although MSs severity at baseline was statistically associated with LCIG discontinuation, the probability was very low with little clinical significance.
Keywords
INTRODUCTION
Medical management of patients with advanced Parkinson’s disease (PD) is complex. As the disease progresses, a broad spectrum of signs and symptoms, both motor and non-motor in nature, combine with the complications of long-lasting treatment and the adverse events (AEs) of drug therapy [1, 2]. At present, dopamine replacement with levodopa is the gold standard for pharmacologic therapy [3]; however, the benefits of oral treatment are reduced in patients with advanced PD, partly due to altered gastric emptying [2, 4]. Treatment via direct infusion of levodopa-carbidopa intestinal gel (LCIG) to the proximal jejunum via a percutaneous endoscopic gastrojejunostomy (PEG) is undertaken to avoid variability in plasma levels of levodopa [5, 6], as pulsatile dopaminergic stimulation with oral levodopa seems to contribute to the development of late complications in patients with PD [7]. Several studies have shown that continuous infusion of LCIG provides consistent clinical benefits in patients with advanced PD [8–12], as shown by the main results of the eDuo study where a significant ”off” time reduction, an increase in ”on” time with troublesome dyskinesia and PGI-C improvement were found [13]. However, in practice, the patient profile of those who would benefit from treatment with LCIG has not been clearly defined. Younger age at the initiation of therapy has been described among the predicting factors of efficacy (67.1±7.2 vs 69.0±2.5; p = 0.039) [14], and several authors have already proposed earlier initiation of LCIG [14–16]. Further, recent data indicate that the risk of dropout from LCIG therapy is increased in patients aged≥70 years [17]. In view of these data, we hypothesized whether disease duration (DD) before the initiation of LCIG might have an impact on effectiveness. Therefore, the objectives of this subanalysis of the E-DUO study [13] were to explore the influence of DD from disease diagnosis on the effectiveness of LCIG treatment, and to analyze the rate and reasons for interruption and/or LCIG discontinuation in a cohort of Spanish patients with advanced PD.
METHODS
Study design and patients
E-DUO was a retrospective, observational, nationwide, multicenter study. Patient data were collected retrospectively from clinical records. All patients included in the cross-sectional evaluation provided informed consent for their participation in the study. It was approved by the institutional review boards of the 18 participating hospitals and was carried out in accordance with the principles of the Declaration of Helsinki [13].
Consecutive patients with a diagnosis of idiopathic PD according to the UK Parkinson’s Disease Society Brain Bank Diagnostic Criteria [18] who initiated treatment with LCIG between January 2006 and December 2011 (continued or discontinued at enrollment) were included in the study. Participants had to have been evaluated for≥11 months by a neurologist in the same center, not related to the LCIG treatment duration. All centers were required to have some experience with LCIG (≥5 patients) to participate [13].
Study variables
This report includes 2 subanalyses that were conducted using data from the entire E-DUO population (Fig. 1). In order to analyze the influence of DD on LCIG effectiveness, the sample was dichotomized into 2 groups: DD≥10 years or DD < 10 years. This criteria was chosen per published LCIG studies that generally include patients in an advanced state and whose mean DDs were≥10 years (range of mean DDs, 12.5–17 years) [15, 19–24]. Further, a recent Spanish consensus on the definition of PD included a DD of >10 years as one of the characteristics that could be used to define advanced disease [25]. The analysis of the influence of DD on LCIG effectiveness in motor symptoms (MS) and non-motor symptoms (NMS), and safety variables was conducted using only those patients continuing LCIG at enrollment. Conversely, the analysis of factors associated with discontinuation was conducted using the entire patient cohort (i.e., including those patients who had discontinued; Fig. 1).
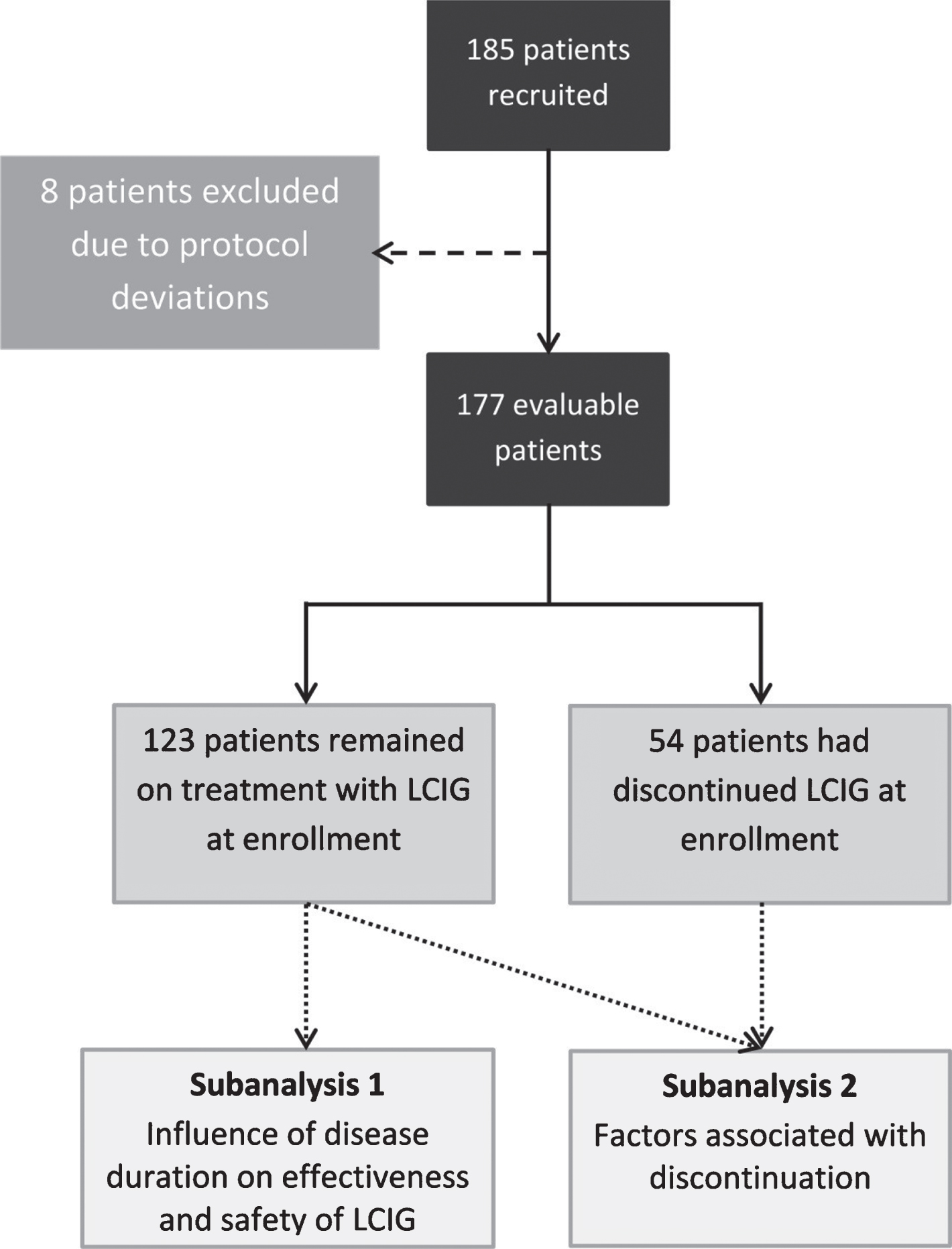
Study flowchart. This report includes two subanalyses performed using the patient population from the E-DUO study.
Baseline clinical data (i.e., onset of LCIG treatment) were retrieved from patients’ medical history based on clinical judgement after patient and caregiver interview and clinical examination of patient, and final data were collected during the enrollment visit. These variables included Unified Parkinson’s Disease Rating Scale part III (UPDRS III) scores and modified Hoehn and Yahr (H&Y) stage [26] during the “on” and “off” states, MS (tremor, freezing, dystonia, falls, hypophonia, and dysphagia), NMS (depression, cognitive impairment, and psychiatric and/or behavior disorders), motor complications (MC; i.e. the percentage of waking day [0%; 1% – 25%; 26% – 50%; 51% – 75%; 76% – 100%] in “off” time), “on” time without troublesome dyskinesia, and “on” time with troublesome dyskinesia (estimated by the neurologist according to the patient’s report). AEs were also recorded at the enrollment visit.
Sample size and statistical analyses
The sample size was based on clinical and logistic issues and was not calculated for power and precision. The main analyses consisted of the comparison of changes from baseline to the enrollment visit in percentage of mean daily “off” time, mean daily “on” time without troublesome dyskinesia, and mean daily “on” time with troublesome dyskinesia according to DD before the initiation of treatment with LCIG. Categorical and continuous variables were compared using the Fisher exact test and the Wilcoxon test, respectively. To estimate the median difference, it was assumed that the major categories had a value equivalent to their class center. Therefore, the category 0 received value of 0, category 1 (1% – 25%) received a value of 13%, category 2 (26% – 50%) received a value of 38%, and so on. Univariate analysis and logistic regression analyses were performed to identify demographic and clinical factors associated with discontinuation. All tests were 2-sided, and p < 0.05 was considered statistically significant. Results are expressed as frequencies and percentages for categorical variables and means±standard deviations (SDs) in the case of continuous variables, unless otherwise specified. All statistical analyses were carried out using SAS v9.2 (SAS Institute, Cary, NC, USA).
RESULTS
Patient baseline clinical and demographic characteristics are summarized in Table 1. At enrollment, the mean (SD) duration of treatment with LCIG was 35.6±18.6 months. Groups evaluated in the subanalyses (DD and discontinuation) were generally well balanced, without significant differences in baseline variables.
Baseline demographic and clinical characteristics of the sample and subgroups
*Based on the time awake during the day; PD = Parkinson’s Disease, SD = standard deviation, NMS = non-motor symptoms, UPDRS III = Unified Parkinson’s disease rating scale part III. Continuous variables compared using Wilcoxon signed-rank test and frequencies compared with Chi-square test or Fisher test as appropriate.
Reasons for initiating LCIG
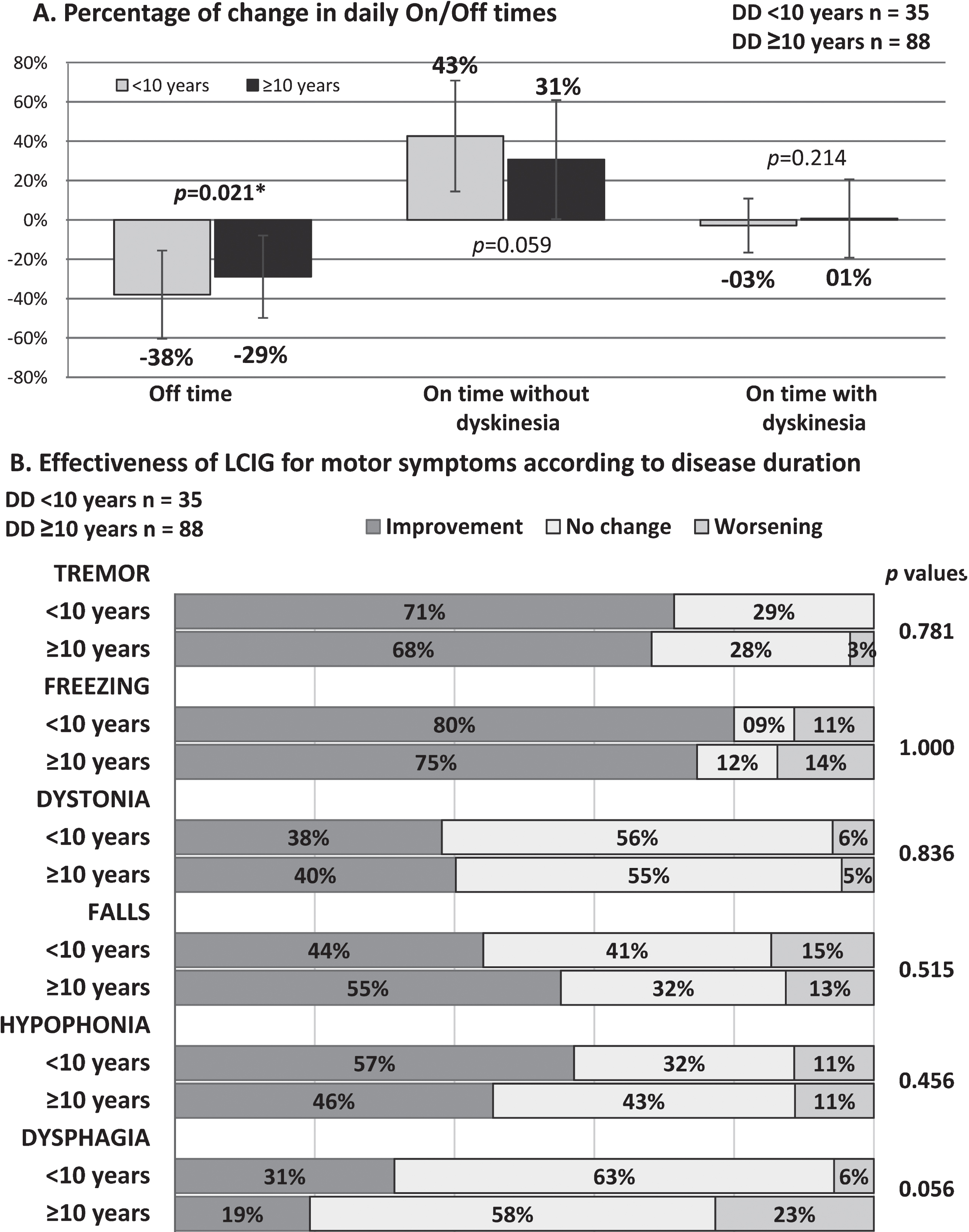
A) Influence of DD on the effectiveness of treatment with LCIG in patients with motor symptoms. B) Change in “on” and “off” times after LCIG treatment. DD, disease duration; LCIG, levodopa-carbidopa intestinal gel.
In general, DD had no impact on the percentages of patients that experienced improvement, no change, or worsening of their NMS (dichotomized; Fig. 3; p > 0.05). NMS related to the cardiovascular system (dizziness and falls), sleep/fatigue (daytime sleepiness, fatigue, insomnia, restless legs), and mood (disinterest, nervousness, sadness, flat mood, apathy) were the most improved domains of NMS, with approximately half of patients reporting improvement regardless of DD.
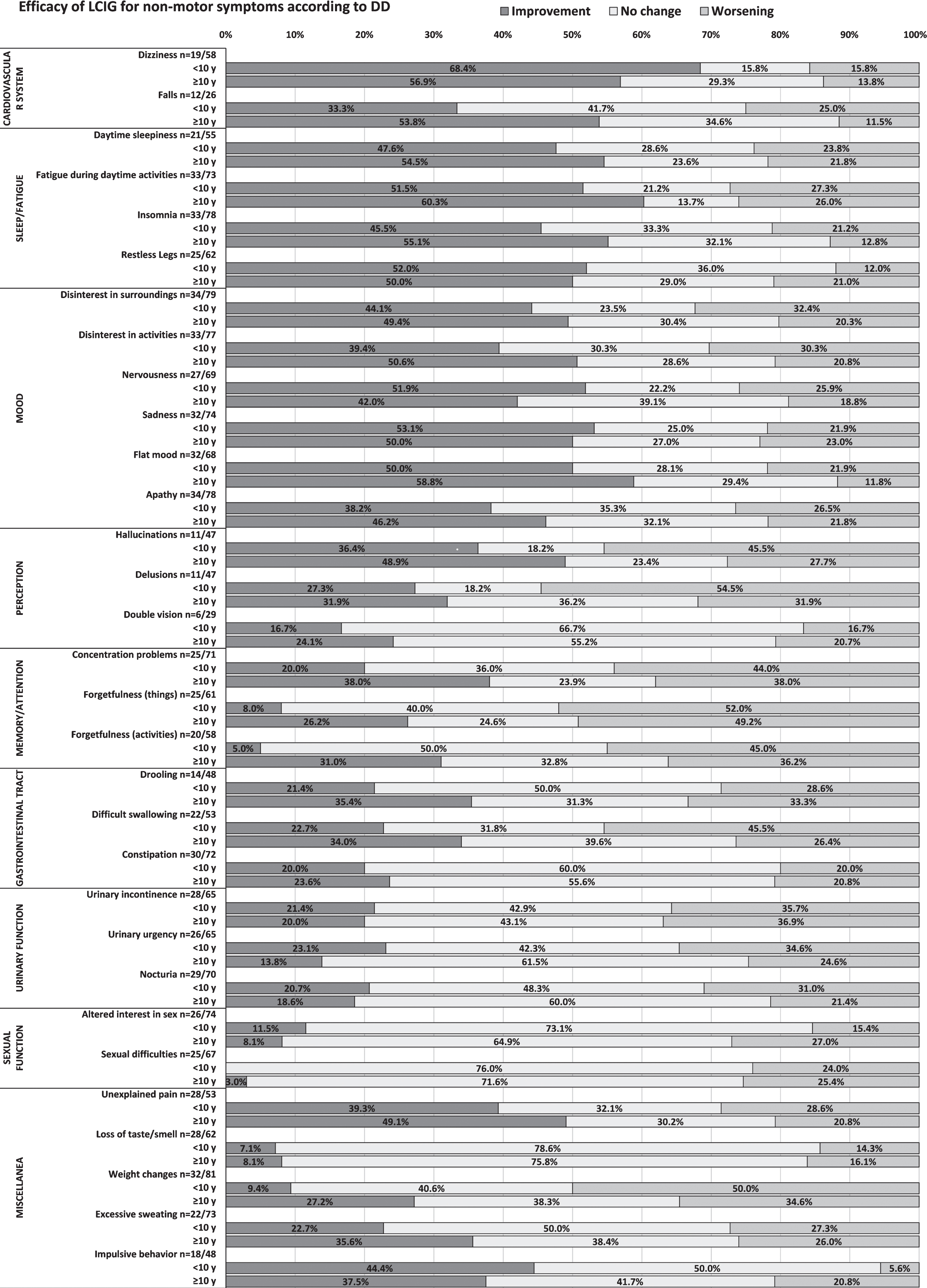
Influence of DD on the effectiveness of treatment with LCIG in patients with non-motor symptoms. DD, disease duration; LCIG, levodopa-carbidopa intestinal gel.
Multivariate analysis of baseline characteristics associated to discontinuation of treatment with levodopa-carbidopa intestinal gel
Variables highlighted in bold in the univariate analysis were included in the multivariate analysis and resulted significantly associated to discontinuation.
DISCUSSION
Determining which patients may benefit from device-aided therapies for advanced PD can be challenging. The invasiveness and cost of therapy [27, 28], including surgery, set-up, and training of patient and caregivers, has to be analyzed in light of the expected benefits of treatment. Our results suggest that longer DD at the initiation of LCIG (i.e.,>10 years) reduces its effectiveness in controlling MS in terms of number of ”off” time reduction and ”on” time with troublesome dyskinesia, and increases the occurrence of severe AEs, although this did not influence rates of discontinuation. These results also suggest that greater severity of MCs at baseline (i.e., higher percentages of daily “off” time and of “on” time with troublesome dyskinesia) is a factor statistically significantly associated with discontinuation; however, the proximity of OR values to 1 suggest a low influence and little clinical significance.
Previous studies have indicated that advanced age could be a potential issue when initiating LCIG therapy, as younger age at PEG procedure was found to be one of the factors predicting efficacy [14], and the risk of dropout from LCIG treatment was increased in patients aged≥70 years [17]. In our study, effectiveness was reduced in patients with a DD≥10 years; however, DD was not related to patient age, as this group had a similar mean age to those patients with a shorter DD. Studies that explored and have demonstrated the efficacy of LCIG in advanced PD generally included patients in an advanced state, including DD. This is the case of the analysis published by Standaert et al. [29] where they tried to identify the baseline characteristics predictive of treatment response, measured by improvement in Off state (the magnitude of the OFF-time reduction). The extend of the improvement was not related to DD or patient’s ages at PEG placement. But certainly, the mean DD was in all the subgroups≥10 years. Mean DDs are usually≥10 years and baseline characteristics testify the high severity of symptoms [15, 19–24]. Antonini et al. commented that the relatively high rate of discontinuation in the literature reflected that LCIG was being used in patients with advanced disease [20]. DD did influence motor outcomes, and treatment with LCIG was significantly less effective in patients with a DD≥10 years. In other words, LCIG may be more useful if it is introduced earlier in the treatment paradigm, though this recommendation has been usually also linked to patient age [15, 16].
Reasons for discontinuation in this cohort were mainly related to the treating neurologist’s decision or drug-related problems. In addition, the possibility of unsuccessful treatment of MC should be considered when selecting candidates for LCIG therapy. Buongiorno et al. recently reported that dyskinesia periods significantly increased after 3 months of treatment in patients with baseline dyskinesia lasting <50% of the day before LCIG, whereas in patients with≥50% of time with dyskinesia at baseline, periods of troublesome dyskinesia were significantly reduced [30]. In light of this, baseline MC are related to effectiveness, and our results suggest that they are also slightly related to discontinuation. In this cohort, long durations of “off” time and “on” time with troublesome dyskinesia at baseline were factors associated with discontinuing treatment with LCIG. The ORs were close to 1 indicating a small effect, but these clinical features might be taken into account when evaluating patients for LCIG treatment.
This study has limitations inherent to its retrospective nature, including the recruitment procedures, the inability to calculate a sample size for power and homogeneous distribution into groups, and the absence of a control group. On the other hand, there could be some other factors like clinical characteristics (e.g. cognitive status) that could impact the LCIG response (clinical benefit) not solely DD that have not been considered in this subanalysis. These limitations should be taken into account when interpreting the results. However, this sample represents the greatest number of patients with advanced PD treated with LCIG evaluated in a non-interventional study from a single country and provides novel effectiveness data in a subgroup of patients with DD that was not previously analyzed.
As shown in the previous paper [13], all PD patients treated with LCIG benefit from the treatment independently of their baseline characteristics. However, in this post hoc analysis our results suggest that the benefits of treatment with LCIG might be greater in patients with less advanced disease in terms of DD and severity of baseline MC. Although the latter has a low influence, baseline MC might be taken into account when considering probability of discontinuation. Determining the patients most likely to benefit from treatment with LCIG should be further explored in specially designed prospective studies.
CONFLICTS OF INTEREST
I. Regidor has occasionally received reimbursement for travel expenses and honoraria for speaking at meetings from Medtronic, Boston Scientific, and AbbVie.
D. Santos-García has received honoraria during the past year for educational presentations from AbbVie, UCB Pharma, Zambon, KRKA and Lundbeck and has also received honoraria from AbbVie for participation in clinical trial coordination.
M.J. Catalán has received honoraria for consulting, advisory services, speaking services, and research from AbbVie Laboratories and Merz.
V. Puente has honoraria for lecturing from AbbVie, UCB Pharma, and Lundbeck and for participating in advisory boards from AbbVie.
F. Valldeoriola received honoraria for advice and lectures from AbbVie, Medtronic, and Boston Scientific.
F. Grandas has received honoraria for lecturing from Merz, AbbVie, UCB, and GE Healthcare and for participating in advisory boards from AbbVie.
P. Mir has received honoraria for lecturing or advisory boards from AbbVie, UCB, Allergan, and Merz and has received research grants from the Ministerio de Economía y Competitividad de España [SAF2007-60700], the Instituto de Salud Carlos III [PI10/01674, CP08/00174, PI13/01461], the Consejería de Economía, Innovación, Ciencia y Empleo de la Junta de Andalucía [CVI-02526, CTS-7685], the Consejería de Salud y Bienestar Social de la Junta de Andalucía [PI-0377/2007, PI-0741/2010, PI-0437-2012], the Sociedad Andaluza de Neurología, the Jacques and Gloria Gossweiler Foundation, and the Fundación Alicia Koplowitz.
J. C. Parra is an employee of Abbvie and owns shares or options of Abbvie.
J.M. Arbelo has received honoraria for lecturing or advisory boards from AbbVie and UCB Pharma.
Footnotes
ACKNOWLEDGMENTS
The authors thank Dr. Blanca Piedrafita at Medical Statistics Consulting S.L. (Valencia, Spain) for the support in manuscript preparation. Statistical analysis was performed by Ana Drozdowskyj at PIVOTAL S.L. (Madrid, Spain).
This study was sponsored by AbbVie Spain S.L.U., Madrid, Spain. Data collection was coordinated by the sponsor and designates, who also conducted the study. AbbVie also supported the reporting of study results and reviewed the final manuscript; final content was at discretion of authors. All authors had full access to all of the data and contributed to the revision of the manuscript and approval of the final version.
