Abstract
Lacosamide (LCM) is approved for anticonvulsive treatment in focal epilepsy and exhibits its function through the slow inactivation of voltage-gated sodium channels (VGSCs). LCM shows comparable efficacy with other antiepileptic drugs (AEDs) licensed in the last decade: in three randomized placebo-controlled trials, significant median seizure reduction rates of 35.2% for 200 mg/day, 36.4–39% for 400 mg/day and 37.8–40% for 600 mg/day were reported. Likewise, 50% responder rates were 38.3–41.1% for 400 mg/day and 38.1–41.2% for 600 mg/day. Similar rates were reported in post-marketing studies. The main adverse events (AEs) are dizziness, abnormal vision, diplopia and ataxia. Overall, LCM is well tolerated and has no clinically-relevant drug–drug interactions. Due to the drug’s intravenous availability, its use in status epilepticus (SE) is increasing, and the available data are promising.
Introduction
Epilepsy is a common and chronic neurological disorder that imposes a substantial burden on individuals, caregivers and society as a whole [Strzelczyk et al. 2008; Riechmann et al. 2015]. Antiepileptic drugs (AEDs) play a central and crucial role in treatment, as the majority of epilepsy patients require anticonvulsant treatment for an extended period of time. Because up to 30% of epilepsy patients are refractory to medical treatment [Kwan and Brodie, 2000; Kwan et al. 2011], the development of new therapeutic options is strongly warranted. Due to ongoing seizures, patients with drug-refractory epilepsy are affected by increased morbidity and mortality, social stigma, reduced employment opportunities and impaired quality of life for themselves and their caregivers [Smeets et al. 2007; Jacoby et al. 2011; Ryvlin et al. 2013; Riechmann et al. 2015]. Introduction of new AEDs provides an opportunity to achieve better seizure control for some of these patients [Luciano and Shorvon, 2007; Callaghan et al. 2011]. Furthermore, AEDs with an excellent efficacy and safety profile are needed for use as initial monotherapy as they might be continued for decades in patients [Glauser et al. 2013].
Lacosamide (LCM) was approved in 2008 in the European Union and in the USA as adjunctive therapy in the treatment of partial-onset seizures with or without secondary generalization in adults and adolescents with epilepsy. LCM is licensed for adolescents older than 16 years in the European Union and older than 17 years in the USA. Additionally, the US Food and Drug Administration has approved LCM as monotherapy in focal epilepsy in 2014.
LCM is a functionalized amino acid with a distinct mode of action: the drug enhances the slow inactivation of voltage-gated sodium channels (VGSCs) [Doty et al. 2007, 2013]. In contrast, traditional sodium channel blockers affect the fast inactivation of VGSCs. The slow inactivation of VGSCs results in the stabilization of hyperexcitable neuronal membranes, the inhibition of neuronal firing, and the reduction of long-term channel availability without affecting physiological function [Doty et al. 2013].
LCM is available as film-coated tablets (50 mg, 100 mg, 150 mg, 200 mg), syrup (10 mg/ml) and solution (10 mg/ml) for intravenous infusion. Bioequivalence among the three LCM formulations has been demonstrated, allowing for direct conversion without titration [Cawello et al. 2012]. LCM exhibits several favorable pharmacokinetic characteristics as rapid absorption, high oral bioavailability (100%) not affected by food, linear and dose-proportional pharmacokinetics with low interindividual and intraindividual variability [Cawello et al. 2012, 2014a]. LCM has low plasma protein binding of less than 15%. LCM is metabolized by CYP2C19, CYP2C9, and CYP3A4 into the pharmacologically inactive O-desmethyl-LCM and undergoes primarily renal elimination with less than 1% of the dose eliminated in the feces [Cawello et al. 2012, 2014b]. LCM has an elimination half-life of 13 h, which supports a twice-daily dosing regimen. Steady-state plasma concentrations after each dose adaptation are achieved after 3 days. In patients with severe renal impairment or end-stage renal disease, a maximum dose of LCM of 300 mg should not be exceeded. Hemodialysis significantly decreases systemic LCM exposure, by approximately 57% [Doty et al. 2013]. LCM dosage supplementation up to 50% of the divided daily dose should be considered directly after the end of hemodialysis. Also in patients with mild or moderate hepatic impairment a maximum dose of 300 mg/day should not be exceeded [Doty et al. 2013]. LCM exhibits low potential for clinically-relevant pharmacokinetic drug–drug interactions with AEDs as well as other common medications [Cawello et al. 2010, 2012, 2013; Stockis et al. 2013; Cawello et al. 2014a, 2014b; Cawello, 2015].
Clinical efficacy
Overview of double-blind randomized placebo-controlled trials
Results from three double-blind randomized controlled trials (RCTs) led to the approval of LCM as adjunctive treatment for partial-onset seizures in Europe and in the USA in 2008. These trials (referred to as ‘LCM-RCTs’ below) consist of:
– SP667 [Ben-Menachem et al. 2007]: a phase IIb study including 418 patients in the USA and Europe. The study was designed to compare placebo versus LCM at doses of 200 mg/day, 400 mg/day and 600 mg/day, respectively; the related long-term extension study was SP615 [Rosenfeld et al. 2014].
– SP754 [Chung et al. 2010b]: a phase III study including 405 patients in the USA and comparing placebo versus LCM doses of 400 mg/day and 600 mg/day, respectively; the related long-term extension study was SP756 [Husain et al. 2012].
– SP755 [Halasz et al. 2009]: a phase III study including 485 patients in Europe and Australia was designed to compare placebo versus LCM doses of 200 mg/day and 400 mg/day, respectively; the related long-term extension study was SP774 [Rosenow et al. 2015].
Of about 1300 adult patients included in these three trials, 947 patients received LCM. There are several retrospective and prospective open-label studies of LCM treatment of children with epilepsy [Grosso et al. 2014b; Pasha et al. 2014; Yorns et al. 2014]; however, no children took part in any of the LCM-RCTs.
Efficacy data from RCTs
The efficacy data from LCM-RCTs are summarized in Table 1. LCM was effective in the reduction of the mean seizure frequency in doses of 400 mg/day and 600 mg/day. Median seizure reduction was also significant for 200 mg/day in SP755, but not in SP667. Furthermore, intake of 200 mg/day did not result in a significant increase of the 50% responder rate in either study, indicating the dose-dependency of LCM efficacy.
Efficacy data from phase II and phase III studies [Ben-Menachem et al. 2007; Halasz et al. 2009; Chung et al. 2010b] ITT population, ranges (min–max).

Statistically significant results are shown in
b, baseline period; db, double blind; ITT, intention-to-treat; m, maintenance period; pc, placebo-controlled; r, randomized; ti, titration period; tr, transition period.
Based on analysis of covariance (ANCOVA) models, the mean seizure reduction over placebo in the intention-to-treat set was 14% (200 mg/day), 15–28% (400 mg/day), and 21–25% (600 mg/day), respectively. In the per-protocol set (PPS), mean seizure reduction over placebo was consistently higher (200 mg/day: 22–35%; 400 mg/day: 21–45%, 600 mg/day: 32%).
Pooled analyses
Data from SP667, SP754 and SP755 are the basis for several pooled analyses. A Cochrane review [Weston et al. 2015] rated the three RCTs as adequate in terms of methodology and low risk for systematic bias, resulting in a high quality of evidence for 50% responder and withdrawal rates. Quality of evidence for seizure freedom was rated as moderate due to the small number of events. Risk ratios (RRs) for ⩾50% seizure reduction as compared with placebo were calculated as 1.7 [95% confidence interval (CI), 1.38–2.10] for all doses of LCM, while the RR for seizure freedom was 2.5 (95% CI, 0.85–7.34).
Median seizure reduction rates of 18% (placebo), 33% (LCM 200 mg/day; p < 0.01 compared with placebo) and 37% (LCM 400 mg/day; p < 0.001) were calculated [Beydoun et al. 2009]. Likewise, 50% responder rates calculated from the pooled data were significant in comparison with placebo (23%) for LCM 200 mg/day (34%; p < 0.05) and for LCM 400 mg/day (40%; p < 0.001).
Valuable post hoc analyses were performed by [Chung et al. 2010a]. Examining the onset of LCM efficacy, they found a significant difference in the median reduction of seizure frequency for LCM 100 mg/day versus placebo during the first week (33% versus 19%) and during the second week (34% versus 20%). Neither prior epilepsy surgery nor implantation of a vagus nerve stimulator was associated with efficacy differences. Also, there were no apparent differences in LCM efficacy between patients with different concomitant anticonvulsant drugs. Another post hoc analysis [Sake et al. 2010] confirmed the efficacy of add-on LCM in patients who were on a traditional sodium channel blocker (phenytoin, carbamazepine, oxcarbazepine, lamotrigine). Efficacy was higher in patients on non-sodium channel blockers with median seizure reduction of 38% (200 mg/day), 63% (400 mg/day) and 79% (600 mg/day) versus placebo (28%), but a direct comparison was not possible with the LCM-RCT database.
A meta-analysis that included all available RCT data of ‘new’ anticonvulsants (oxcarbazepine, lamotrigine, topiramate, gabapentin, pregabalin, levetiracetam, tiagabine, zonisamide, eslicarbazepine and LCM) confirmed efficacy of LCM over placebo but estimated a lower efficacy of LCM compared with the pooled efficacy of the other new anticonvulsants [Costa et al. 2011]. Because the differences disappeared after adjustment for the percentage of responders in the placebo group, this finding was attributed to the relatively high rate of seizure reduction in the placebo groups of the LCM-RCTs, which in turn may have been influenced by study location and year.
Safety and tolerability
Safety and tolerability data from RCTs
The safety and tolerability data from SP667, SP754 and SP755 are summarized in Table 2. Overall, LCM tolerability was dose-dependent. Doses of 200 mg/day and 400 mg/day were fairly well tolerated, while treatment discontinuation increased to 30% in the 600 mg/day group.
Frequent and dose-dependent treatment-emergent adverse events (TEAEs). Frequency ranges min–max in % from LCM-RTCs [Ben-Menachem et al. 2007; Halasz et al. 2009; Chung et al. 2010b].

LCM, lacosamide; RTC, randomized controlled trial; TEAE, treatment-emergent adverse event.
Treatment-emergent adverse events (TEAEs) were usually of mild or moderate intensity. The most frequent and dose-dependent TEAEs (i.e. likely to be treatment related) were dizziness, abnormal vision, diplopia, nystagmus, fatigue, tremor, ataxia and other coordination abnormalities as well as nausea and vomiting. Another frequently reported TEAE was headache; however, frequency was not dose-related, and headache did not consistently appear more often in LCM-treated patients than in the placebo group. An additional seven RCTs conducted for other indications (diabetic neuropathic pain, migraine, fibromyalgia, knee osteoarthritis) [Zaccara et al. 2013] and one RCT that assessed cardiac side effects of LCM in healthy volunteers [Kropeit et al. 2015] reported very similar TEAE profiles in clinically different populations.
Serious adverse events (SAEs) were rare and comprised seizures, dizziness, nausea, vomiting, nystagmus, and psychiatric disorders. Overall, one patient died during the course of one of the LCM-RCTs. This patient, who was randomized to LCM 200 mg/day treatment, committed suicide. The death was considered to be unrelated to the trial medication [Biton et al. 2015].
Cardiac safety
Because LCM acts on VGSC, treatment-induced atrioventricular (AV) block is a possible concern. In SP667, SP754 and SP755, the PR interval increased in a dose-dependent manner (400 mg/day, 4.2–4.6 ms; 600 mg/day, 6.1 ms). This elevation remained clinically silent. A pooled analysis [Rudd et al. 2015] confirmed these findings: mean changes in PR interval from baseline to the end-of-maintenance period were −0.3 ms (placebo), 1.4 ms (LCM 200 mg/day), 4.4 ms (LCM 400 mg/day) and 6.6 ms (LCM 600 mg/day). The frequency of first-degree AV block differed between SP667 (placebo, 3.2%; LCM 200 mg/day, 4.7%; LCM 400 mg/day, 4.9%; LCM 600 mg/day, 4.9%) and SP754/755 (placebo: 2.4%; LCM 200 mg/day, 1.3%; LCM 400 mg/day, 2.3%; LCM 600 mg/day, 1.1%). The reason for this discrepancy remains unexplained, the more so as a higher threshold was defined for AV block in SP667 (220 ms) than in SP754/755 (209 ms). Patients with AV block were allowed to continue the study and apparently did not experience clinical side effects from the prolonged conduction time. No instance of second- or third-degree AV block was reported. Monitoring of electrocardiograms (ECGs) might be considered in patients with known conduction problems and in those treated by PR interval-prolonging drugs such as carbamazepine, lamotrigine, and pregabalin.
QTc intervals were not affected. A randomized, double-blind, positive- and placebo-controlled trial (SP640) was conducted in healthy volunteers to determine the effects of LCM (400 and 800 mg/day) on repolarization and AV conduction [Kropeit et al. 2015]. Moxifloxacin, which prolongs the QT interval, was used as a positive control. This trial confirmed the findings of the LCM-RCTs: QTc intervals remained unaffected, while LCM treatment led to a dose-dependent increase in PR intervals (400 mg/day: 7.3 ms over placebo; 800 mg/day: 11.9 ms over placebo). The cardiac-related TEAEs reported were palpitations (placebo, 3.2%; LCM 400 mg/day, 3.3%; LCM 800 mg/day, 4.2%) and sinus tachycardia (LCM 800 mg/day, 1.4%). Altogether clinical practice shows a good cardiac tolerability of LCM.
Pooled analyses
Combined evaluation of safety data from the three LCM-RTCs revealed similar results to those obtained for the single studies [Beydoun et al. 2009; Biton et al. 2015; Weston et al. 2015]. The RR for treatment withdrawal in LCM-treated patients as compared with placebo was 1.9 for all doses [Weston et al. 2015]. Abnormal coordination (RR 6.1), diplopia (RR 5.3), dizziness (RR 3.5) nausea (RR 2.4) and vomiting (RR 3.5) were significantly associated with LCM intake, while headache (RR 1.3), fatigue (RR 3.5), nystagmus (RR 1.5) and somnolence (RR 1.4) were not.
A thorough pooled safety and tolerability analysis was performed by [Biton et al. 2015]. In general, the incidence of TEAEs was higher during the titration phase than during the maintenance phase. Rare TEAEs observed included rash (incidence, 2.9% in the LCM group versus 3.0% in the placebo group), weight gain (1.2% versus 0.5%), weight loss (1.1% versus 0.8%), increased alanine aminotransferase (ALAT) levels ⩾3 times the upper limit of normal (0.5% versus 0%), psychosis (0.3% versus 0%) and depression (2.1% versus 0.5%).
LCM tolerability seemed lower when the drug was combined with ‘traditional’ sodium channel blockers, such as carbamazepine, oxcarbazepine, lamotrigine and phenytoin [Sake et al. 2010].
Regarding LCM influence on mood and anxiety, one study [Moseley et al. 2015] showed significant decrease in depressive symptoms in patients on LCM. Overall, NDDI-E and GAD-7 scores prior and after initiation of LCM were not significantly affected by a history of mood disorders, concomitant psychiatric medications, or concomitant AEDs with mood-stabilizing effects [Moseley et al. 2015].
Post-marketing surveillance
Further information on the efficacy and tolerability of LCM as an adjunctive AED can be drawn from mono- and multicenter reports on the use of LCM in daily practice. For details regarding efficacy, please refer to Table 3. A multitude of clinical trials with a prospective or descriptive study design and a total number of more than 1500 enrolled patients showed a >50% reduction in seizure frequency in 23–48.2% of patients [Wehner et al. 2009; Garcia-Morales et al. 2011; Stephen et al. 2011; Husain et al. 2012; Miro et al. 2013; Novy et al. 2013; Verrotti et al. 2013; Villanueva et al. 2013; Legros et al. 2014; Borzi et al. 2016; Lang et al. 2016]. Seizure freedom under adjunctive therapy with LCM of up to 33% and a reduction of seizure frequency >90% in approximately 26% has been shown [Harden et al. 2012; Steinhoff et al. 2016]. In addition, several retrospective, descriptive or comparative trials confirmed these results, reporting a reduction in seizure frequency of >50% in up to 69% and an overall seizure freedom of up to 33% under treatment with LCM [Flores et al. 2012; Harden et al. 2012; Kamel et al. 2013; Villanueva et al. 2013]. In addition to its efficacy in adults, LCM has been shown to achieve a >50% responder rate in 33.3% of children suffering from generalized seizures (n = 19) and 36.8% of pediatric patients with focal seizures (n = 12, mean age 10.8 years) [Verrotti et al. 2013]. Aside from oral application, adjunctive intravenous administration of LCM (n = 130, mean dosage 300 mg/day, mean duration of treatment 2d) in adults and children suffering from generalized and focal seizures was recently shown to be highly effective and well tolerated in a multicenter non-interventional study [Lang et al. 2016].
Efficacy data from post-marketing studies (ranges min–max).

AE, adverse event; AED, antiepileptic drug; IV, intravenous; LCM, lacosamide; LGS, Lennox–Gastaut syndrome; MRI, magnetic resonance imaging; SE, status epilepticus; TEAE, treatment-emergent adverse event; TLE, temporal lobe epilepsy.
Moreover, several case reports have proven the efficacy of adjunctive LCM treatment in various epilepsy syndromes or chronic diseases accompanied by epileptic seizures, such as post-hypoxic myoclonus syndrome (Lance–Adams syndrome) [Galldiks et al. 2010] and Lennox–Gastaut syndrome (LGS). Regarding the efficiency of LCM in LGS, contradictory reports have been published. In a multicenter retrospective open-label treatment study of children suffering from LGS (n = 18, mean age 12.3 years), a responder rate of 33% was reported [Grosso et al. 2014a]. Conversely, several case reports have shown an increase in seizure frequency in LGS [Cuzzola et al. 2010; Andrade-Machado et al. 2012, 2015]. One case report showed de novo continuous epileptic negative myoclonus triggered by the addition of LCM to carbamazepine, that promptly reverted after carbamazepine dosage reduction [Belcastro et al. 2011]. These discrepancies show the difficulty of an anticonvulsive treatment of LGS but prove LCM to be an alternative AED in the therapy of LGS.
Aside from its robust efficacy, LCM has also proven acceptable in terms of its tolerability and side-effect profile. In summary, the most common reported adverse effects (AEs) in post-marketing studies were similar to those reported in the RCTs. These included sedation, somnolence, fatigue, dizziness, nausea, unsteadiness, ataxia, headache and double vision [Flores et al. 2012; Husain et al. 2012; Kamel et al. 2013; Novy et al. 2013; Verrotti et al. 2013; Villanueva et al. 2013; Steinhoff et al. 2016]. For details on common AEs, please refer to Table 4. Several case reports have documented life-threatening AEs, such as higher-degree AV blockage, atrial flutter/fibrillation and sinus node dysfunction [Degiorgio, 2010; Nizam et al. 2011; Chatzistefanidis et al. 2013; Chinnasami et al. 2013; Degiorgio et al. 2013]. Another case report showed reversible suicidal ideation after several months of exposure to 400 mg/day LCM in a 47-year-old man suffering from partial epilepsy since adolescence [Kellinghaus, 2013]. In a report of an attempted suicide with 12 g of LCM, a transient increase in PR interval was shown, while complete physical recovery occurred after several days of supportive treatment [Bauer et al. 2010]. A recent international noninterventional multicenter study focused on the safety and tolerability of LCM in patients with uncontrolled partial-onset seizures. The study’s findings revealed an incidence of cardiovascular TEAEs in the LCM-treated cohort of 0.8% (n = 487, <65 years), which is comparable with the incidence observed in the cohort treated with other AEDs (0.9%, n = 448, <65 years) [Steinhoff et al. 2016]. Likewise, the incidence of psychological TEAEs, such as depression or suicidal ideas/attempts, has been shown to be indistinguishable (LCM 4.3%, other AEDs 5.1%).
Common AEs from post-marketing studies (studies including more than 100 patients were included).

GI, gastrointestinal; CNS, central nervous system.
Monotherapy in focal epilepsies
The efficacy and safety of LCM monotherapy in focal epilepsy patients with partial-onset seizures have been evaluated in prospective, historical-controlled, head-to-head, and retrospective studies.
A historical-controlled, multicenter, double-blind, conversion to monotherapy study by Wechsler and colleagues enrolled 425 patients with partial-onset seizures, a mean age of 40.6 years (range 16–70 years), and 1–2 concurrent anticonvulsive drugs [Wechsler et al. 2014]. Patients were randomized to two subgroups and treated with a LCM dose of 400 mg/day (n = 319 patients) or 300 mg/day (n = 106 patients). Starting at 200 mg/day the LCM dose was titrated over 3 weeks. A 6-week withdrawal of background AEDs was followed by a 10-week LCM monotherapy maintenance period [Wechsler et al. 2014].
In patients with a LCM dose of 400 mg/day and completing the monotherapy phase (n = 201), seizure freedom was observed in 30 (14.9%) patients. A reduction in seizure frequency >50% compared with the baseline was observed in 122 (60.7%) patients. A total of 72 (16.9%) patients withdrew due to TEAEs. The most common reported TEAEs were dizziness (24.0%), headache (14.4%), nausea (13.4%), convulsion (11.5%), somnolence (10.4%), and fatigue (10.1%) [Wechsler et al. 2014].
In order to achieve monotherapy approval by the European health authorities, a prospective controlled non-inferiority trial was conducted in patients aged ⩾16 years with newly diagnosed focal epilepsy [Baulac et al. 2016]. LCM was compared with controlled release carbamazepine (CBZ-CR). Stepwise up-titration to the target dose (LCM: 200, 400, 600 mg/day; CBZ-CR: 400, 800, 1200 mg/day) was based on seizure control, reflecting clinical practice. Overall, 886 patients with mean ages of 41.9 (LCM) and 41.8 years (CBZ-CR) were included in the full analysis set (FAS) (LCM: 444; CBZ-CR: 442) and 805 (408; 397) in the PPS. Kaplan–Meier estimates for the proportion of patients in FAS remaining seizure-free for 6 months were 89.8% with LCM versus 91.1% with CBZ-CR; similar results were obtained for the PPS. Therefore, LCM was considered non-inferior to CBZ-CR [Baulac et al. 2016]. In the FAS, 327 (73.6%) LCM-treated and 308 (69.7%) carbamazepine-CR–treated patients completed 6 months of treatment on the last evaluated dose without having a seizure. AEs were reported by 328 (73.9%) LCM-treated and 332 (75.1%) CBZ-CR-treated patients and were the most common reason for withdrawal in 10.8% versus 15.6% of cases. The AEs occurring in >5% of either group were headache (13.7; 12.9%), dizziness (11.7; 8.6%), fatigue (7.2; 10.4%), nasopharyngitis (6.3; 6.6%), nausea (5.9; 5.0%), somnolence (5.4; 9.3%) and an increase in gamma glutamyl transferase (1.6; 8.1%).
There have been two uncontrolled long-term studies published. In a prospective study by Lattanzi and colleagues, 58 patients with partial-onset seizures with or without generalization and a median age of 40.5 (range, 28–47) years were converted to LCM monotherapy after 1 year of freedom from seizures on LCM add-on therapy by withdrawal of the additional AEDs [Lattanzi et al. 2015]. Assessments followed every 3 months up to 1 year [Lattanzi et al. 2015]. After 1 year, LCM monotherapy was maintained in 37 (63.8%) patients. More than half of the patients (32 patients, 55.2%) remained seizure-free during the entire follow up. Mild to moderate AEs were reported in 12 (20.8%) patients, including drowsiness (n = 7), dizziness (n = 3), and headache (n = 2). Fewer than three AEDs during lifelong anticonvulsive treatment appeared to be a significant predictor of seizure freedom [Lattanzi et al. 2015].
A retrospective study by Giraldez and colleagues examined the charts of 66 patients with a mean age of 49.4 (range, 16–92) years with partial-onset seizures with or without generalization who had been treated with LCM monotherapy [Giraldez et al. 2015]. The subgroups included 18 patients naïve to AEDs (Group 1) and 48 patients who had previously received another AED treatment (Group 2). Follow up comprised 0.5–54 (mean 15.5) months [Giraldez et al. 2015].
A total of 42 (63.6%) patients stayed seizure-free throughout follow up. After 6 and 12 months the seizure-free rates amounted to 77.6% and 72.3%. Mild-to-moderate AEs were reported in 15 patients (22.7%), the most common being somnolence/fatigue (7.5% of patients) and dizziness (6.1% of patients). Overall, 10 (15%) patients withdrew LCM therapy due to side effects (3 patients), lack of efficacy (6 patients) and other reasons (1 patient). Group differences regarding efficacy or tolerability were not significant [Giraldez et al. 2015].
Use in status epilepticus
Status epilepticus (SE) is a life-threatening condition and a medical emergency imposing a considerable burden on the affected person and on society in general [Kortland et al. 2015]. The refractory and super-refractory SEs that fail to respond to first and second line agents or even to anesthesia are associated with a high number of fatalities and high morbidity. Therefore, additional treatment options are of great interest. The efficacy and tolerability of LCM in SE have been the focus of numerous case reports and several clinical studies, most of which have employed a retrospective design. Overall, data from more than 400 patients including case studies and reports with adult SE patients and an additional pediatric SE patients are available, for details please refer to Table 5. The overall success rate for SE termination suggests a favorable role in SE treatment, with rates ranging from 81% [Hofler et al. 2011] to 33% [Kellinghaus et al. 2014] of patients. In pediatric patients, success rates in terminating SE of 45–78% are described. LCM is currently not approved for use in SE. Concurrently, most studies used LCM as an adjunctive therapy in refractory SE patients. This impacts the evaluation of LCM efficacy and may have led to an underestimation of the efficacy of LCM in SE treatment. AEDs are commonly less effective in terminating SE if used at a later position in the succession of anticonvulsive drugs [Chen and Wasterlain, 2006], which is reflected in the findings from several studies. The efficacy with later positioning decreased from 60 to 20% [Kellinghaus et al. 2011], 84.6–55.6% [Santamarina et al. 2013], and 72.2– 56.3% [Miro et al. 2013], respectively. A longer time from seizure onset to LCM administration also significantly reduced treatment success [Garces et al. 2014]. The efficacy of LCM with regard to different SE manifestations [nonconvulsive status epilepticus (NCSE), generalized convulsive status epilepticus (GCSE), focal SE] was not accounted for in most of the included studies, probably due to small sample sizes. Varying termination rates were reported among seizure types, ranging from 91% for convulsive status epilepticus (CSE) to 70% for NCSE [Hofler et al. 2011].
Safety and efficacy data from studies on LCM in SE.
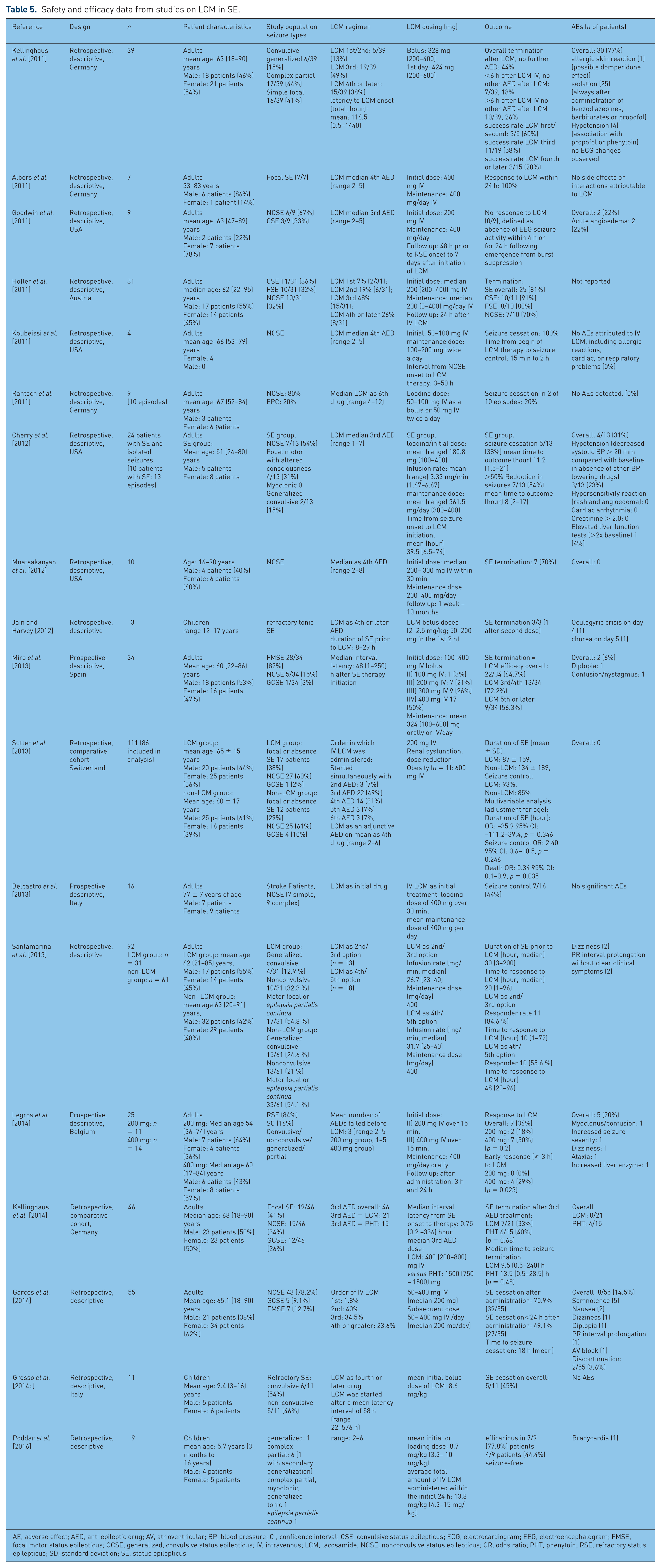
AE, adverse effect; AED, anti epileptic drug; AV, atrioventricular; BP, blood pressure; CI, confidence interval; CSE, convulsive status epilepticus; ECG, electrocardiogram; EEG, electroencephalogram; FMSE, focal motor status epilepticus; GCSE, generalized, convulsive status epilepticus; IV, intravenous; LCM, lacosamide; NCSE, nonconvulsive status epilepticus; OR, odds ratio; PHT, phenytoin; RSE, refractory status epilepticus; SD, standard deviation; SE, status epilepticus
Höfler and Trinker reviewed the literature on LCM in SE published during the period from January 2009 to May 2012 [Hofler and Trinka, 2013]. A total of 10 single case reports were included, representing 10 seizures. An additional 126 seizures were extracted from nine retrospective case series. The success rate of LCM treatment was 100% in the case reports, possibly owing to publication bias. Among the two case series with 30 or more patients, success rates were 44% and 81%, respectively. The overall success rate of LCM was 56% (70/136 episodes of SE).
Since then, several additional studies on SE treatment with LCM have been published: in a prospective multicenter study using intravenous LCM as an add-on AED in refractory SE, LCM was effective in terminating SE in 64.7%. Overall, 50% of SE ended within the first 12 h after LCM application. Two mild AEs were seen, both resolving with reduced doses of LCM [Miro et al. 2013]. Sutter and colleagues reported results from a retrospective comparative cohort study in patients with refractory SE employing historical cohorts. No significant differences in age, SE severity or etiology were found between the groups [Sutter et al. 2013]. Using univariable analyses, SE duration and seizure control were altered favorably, but nonsignificantly. After adjustment for age, mortality was significantly lower in patients treated with LCM. The authors concede that the reduction in mortality was confounded by the implementation of continuous electroencephalogram (EEG) monitoring 1 year prior to the cohort change. No AEs were reported. In another retrospective study, LCM terminated seizures in 67.7% of cases. Among a total of 92 patients, 31 received LCM. When LCM was used earlier in the course of treatment (as second or third AED), the termination rate was significantly higher with LCM than without (84.6% versus 47.8%). No significant difference in termination rate was seen when LCM was positioned later in the AED succession. Overall, efficacy was higher when LCM was administered earlier during the course of treatment. Belcastro and colleagues reported data from 16 stroke patients with post-stroke NCSE diagnosed by video-EEG [Belcastro et al. 2013]. LCM was used as the first AED and ended SE in 7 of 16 cases (44%), with no significant AEs noted. The mean time from stroke onset to NCSE diagnosis was not reported. Kellinghaus and colleagues retrospectively compared the use of intravenous phenytoin to intravenous LCM in patients with refractory SE (n = 46), defined as failure of the first and second anticonvulsants to stop SE [Kellinghaus et al. 2014]. Pretreatment using benzodiazepines and levetiracetam achieved similar results. SE ended in 40% of phenytoin patients and 33% of LCM patients. The patients in the phenytoin group had a higher rate of generalized convulsive seizures. Significant AEs were seen in 27% of the phenytoin group, and no AEs were reported in the LCM group. The authors conclude that LCM showed similar success rates to phenytoin in refractory SE with less risk of AEs. In a retrospective multicenter observational study (LACO-IV) [Garces et al. 2014], data from 55 patients were reviewed. LCM ended 70.9% of SE episodes, while cessation after <24 h from administration ended 49.1% of SE episodes. The treatment response was significantly better if LCM was administered earlier. AEs were reported in 8 patients (14.5%), leading to the discontinuation of LCM administration in 2 patients. Legros and colleagues compared the efficacy of 200–400 mg loading doses of LCM in SE and seizure clusters (SC) in a prospective observational study that included 25 patients [Legros et al. 2014]. The higher initial dose was nonsignificantly-associated with a higher degree of early SE termination and a higher response rate. Overall, 36% (9/25 patients) responded to LCM treatment. There were 4 AEs (16% of patients), all in the 200 mg group. Lang and colleagues describe an overall efficacy of 70% in patients with SE and seizure clusters upon intravenous LCM treatment. In addition to 19 patients with seizure clusters (SC), 51 patients with SE were included, but efficacy and AEs were not reported separately for these patients [Lang et al. 2016].
Poddar and colleagues retrospectively analyzed the efficacy and safety of LCM as an add-on treatment in nine pediatric patients (mean age 5.7 years) [Poddar et al. 2016]. LCM was successful in 77.8% (7/8) cases in ending SE. Overall, four patients (44%) remained seizure-free. An AE of bradycardia was observed in one child. Grosso and colleagues retrospectively analyzed data from 11 pediatric patients with a mean age of 9.4 years and SE [Grosso et al. 2014a]. LCM was effective in ending seizures in five patients (45%). No SAEs were reported.
Additional case reports hint at a positive effect of LCM in focal motor SE: Spalletti and colleagues describe a 39-year-old male with focal motor SE responsive to LCM as a fifth AED [Spalletti et al. 2013]. Hawkes and colleagues describe two additional patients with focal motor SE responsive to oral LCM as adjunctive AED [Hawkes et al. 2013]. A single case report suggests that LCM is effective in absence SE [Sodemann et al. 2014], although this is contested [d’Orsi et al. 2014].
The rate of AEs, including mild ones, ranged from 0–77% of patients in a review by [Hofler and Trinka, 2013]. Overall, the most common side effects are sedation, dizziness, nausea and diplopia. In studies including >15 patients treated with LCM, the rate of AEs ranged 0–77% [Kellinghaus et al. 2011]. Of note, Kellinghaus and colleagues suggest possible effects of other drugs given concomitantly or in rapid succession with LCM prior to the onset of AEs [Kellinghaus et al. 2011]. The rate of significant AEs was low in all studies. Allergic skin reactions [Kellinghaus et al. 2011], angioedema [Goodwin et al. 2011], hypotension [Kellinghaus et al. 2011; Cherry et al. 2012] as well as PR interval prolongation [Santamarina et al. 2013; Garces et al. 2014], AV block [Garces et al. 2014] and elevation of liver enzymes [Cherry et al. 2012; Legros et al. 2014] are described. Psychobehavioral AEs were not reported, probably due to patient characteristics. Rarely, AEs led to discontinuation of LCM therapy [Garces et al. 2014]. Of note, AEs are difficult to attribute to a single drug because the patients included in the study received several drugs in succession for SE treatment. No SAEs occurred in the studies in pediatric patients [Grosso et al. 2014c; Poddar et al. 2016], a smaller case series of three patients described two patients with delayed oculogyric crisis and chorea, respectively [Jain and Harvey, 2012]. LCM seems to be a safe and efficacious treatment option in SE. Data on comparative efficacy regarding other new AEDs or the choice of synergistic combinations remains elusive, only one study showed similar efficacy of LCM as compared with phenytoin or levetiracetam [Kellinghaus et al. 2014]. Data from the 6-Hz-induced seizure model in mice showed synergistic combinations with carbamazepine, lamotrigine, topiramate, gabapentin and levetiracetam [Shandra et al. 2013]. The transferability to humans is questionable. Overall, the lack of RCTs precludes further interpretation of LCM efficacy. In particular, a direct comparison of LCM with other well-tolerated intravenous drugs such as levetiracetam or valproate is missing, resulting in difficulties in clinical decision-making over which drug to use first or second.
Conclusion
In summary, LCM shows comparable efficacy with other AEDs such as zonisamide [Cox et al. 2014], eslicarbazepine [Rocamora, 2015], perampanel [Strzelczyk et al. 2015] or brivaracetam [Strzelczyk et al. 2016] licensed in the last decade for the treatment of partial-onset seizures. Head-to-head comparisons with other new anticonvulsants are desirable to clarify if there are any real differences in efficacy. The strength of LCM is the lack of interaction potential and the option for intravenous use in emergency situations requiring rapid up-titration. Due to its overall good tolerability, LCM use in SE is increasing. So far, the data are promising.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Bauer, E. Paule, C. Petschow and J.P. Zöllner have nothing to disclose.
Dr Willems reports travel support from Eisai, outside the submitted work.
Dr Rosenow reports personal fees from Eisai, grants and personal fees from UCB, grants and personal fees from Desitin Pharma, personal fees and other from Novartis, personal fees from Medtronic, personal fees from Cerbomed, personal fees from ViroPharma and Shire, grants from European Union, grants from Deutsche Forschungsgemeinschaft, outside the submitted work.
Dr Strzelczyk reports personal fees and grants from Bayer HealthCare, Boehringer Ingelheim, Desitin Arzneimittel, Eisai, Pfizer, Sage Therapeutics, and UCB Pharma, outside the submitted work.
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
