Abstract
Effective treatment of epilepsy depends on medication compliance across a lifetime, and studies indicate that drug tolerability is a significant limiting factor in medication maintenance. Available antiepileptic drugs (AEDs) have the potential to exert detrimental effects on cognitive function and therefore compromise patient wellbeing. On the other hand, some agents may serve to enhance cognitive function. In this review paper, we highlight the range of effects on cognition linked to a variety of newer and older AEDs, encompassing key alterations in both specific executive abilities and broader neuropsychological functions. Importantly, the data reviewed suggest that the effects exerted by an AED could vary depending on both patient characteristics and drug-related variables. However, there are considerable difficulties in evaluating the available evidence. Many studies have failed to investigate the influence of patient and treatment variables on cognitive functioning. Other difficulties include variation across studies in relation to design, treatment group and assessment tools, poor reporting of methodology and poor specification of the cognitive abilities assessed. Focused and rigorous experimental designs including a range of cognitive measures assessing more precisely defined abilities are needed to fill the gaps in our knowledge and follow up reported patterns in the literature. Longitudinal studies are needed to improve our understanding of the influence of factors such as age, tolerance and the stability of cognitive effects. Future trials comparing the effects of commonly prescribed agents across patient subgroups will offer critical insight into the role of patient characteristics in determining the cognitive impact of particular AEDs.
Introduction
The incidence of adverse effects is an important issue when prescribing antiepileptic drugs (AEDs), as some of the most effective medications for seizures are associated with a considerable degree of toxicity. Studies indicate that drug tolerability is a significant limiting factor in treatment maintenance, and drug retention rates are often determined by side-effect profiles [Bootsma et al. 2009; Chung et al. 2007]. Older AEDs may still be prescribed, owing to advantages such as lower cost, wide availability and long-term experience, but often exhibit greater toxicity than newer drugs. Recently developed agents tend to differ in terms of mechanism of action and pharmacokinetic properties, and are often better tolerated than older drugs. However, all AEDs have the potential to exert detrimental effects on cognitive function. A thorough appreciation of the negative cognitive effects linked to a variety of AEDs makes a crucial contribution to therapeutic success.
There are currently more than 20 different agents which are licensed for use in treating seizures in the UK. These include phenobarbital and phenytoin, which were popular until the 1950s. Over the next couple of decades, sodium valproate and carbamazepine were developed. These drugs, in addition to primidone and ethosuximide, were the six agents of choice until the 1990s, when a newer generation of drugs became available. These include tiagabine, pregabalin, gabapentin, topiramate, clobazam, oxcarbazepine, lamotrigine and levetiracetam.
In terms of adverse cognitive effects the vulnerabilities of the individuals treated need to be carefully considered. Differing metabolic profiles in children, the elderly or the acutely unwell may lead to an increased chance of toxicity, and a further consideration is interactions with other medications the patient may be taking. Patients with existing cognitive problems may comprise one population at greater risk of the influence of agents with adverse cognitive effects.
Epilepsy can impair an individual’s functioning within work and educational domains [Bishop and Allen, 2003]. As well as adverse cognitive effects, some AEDs may have the advantage of improving cognitive performance. Such beneficial influences may simply occur as a result of seizure control, or in association with positive effects on mood or psychiatric profile. However, a number of agents may demonstrate efficacy in enhancing cognitive function in a more direct way, by improving alertness or cognitive capacity.
In this review of current literature, we highlight the range of effects on cognition linked to a variety of newer and older agents used to control seizures. These include key alterations in both executive and broader neuropsychological functions, which can exert a critical influence on patients’ quality of life and wellbeing. Because drug side-effect profiles are often related to patient characteristics, this review is followed by a critical discussion, which will offer recommendations for prescribing both in general and in relation to more specific clinical cases. Finally, we evaluate reviewed studies in terms of methodological limitations, providing essential guidance for insightful further research.
Relevant studies were identified through searches of PubMed and ISI Web of Knowledge (1980–2011). PubMed was searched for articles containing ‘epilepsy’ and ‘cogniti*’ in the abstract/title, and ‘drug name’ (phenobarbital, primidone, phenytoin, carbamazepine, valpro*, ethosuximide, tiagabine, vigabatrin, clobazam, zonisamide, gabapentin, pregabalin, topiramate, lamotrigine, oxcarbazepine, levetiracetam) in the title. For the ISI search, we used ‘epilepsy’ and ‘cogniti*’ as topic, and the drug name as title. Only original studies on patients with epilepsy that were written in English were included. We excluded case studies, letters and editorials from our search. We also searched the reference lists of articles identified using this search strategy to increase the number of relevant randomized, blinded, controlled studies. Studies which failed to report findings related to cognitive effects were excluded.
Antiepileptic drugs and cognition
Phenobarbital and primidone
Some studies report few cognitive adverse effects (CAEs) with the use of phenobarbital [Wang et al. 2006]. However, studies involving children with epilepsy have linked this agent to lower IQ [Farwell et al. 1990; Camfield et al. 1979], and discontinuation of the drug can improve total IQ (mainly affecting nonverbal items) in children [Tonekaboni et al. 2006]. Phenobarbital is considered to have worse cognitive effects than valproate or carbamazepine [Calandre et al. 1990; Vining et al. 1987]. When compared with carbamazepine, primidone has been found to cause more adverse effects on motor performance and attention/concentration tests [Smith et al. 1987; Rodin et al. 1976]. One study reported attentional and memory difficulties in children [Riva and Devoti, 1996], but these effects were reversible after discontinuation. Another study [Manni et al. 1993] showed that when compared with controls, patients taking phenobarbital showed longer movement times, impaired attention and reduced processing speed, but no relationship was found with drug concentration.
Phenytoin
Phenytoin has been implicated in declines in concentration, memory, visuomotor functions and mental speed [Pulliainen and Jokelainen, 1995; Gillham et al. 1990; Andrewes et al. 1986]. These effects may be dose related [Gillham et al. 1990], although one study reported no such relationship for cognitive-motor performance [Aman et al. 1994]. Another study reported slowed performance on information processing tasks with phenytoin in comparison with carbamazepine, but no differences for memory or selective attention [Aldenkamp et al. 1994]. Some studies report more detrimental effects on memory than carbamazepine [Pulliainen and Jokelainen, 1995; Andrewes et al. 1986], although the opposite pattern has been seen in children [Forsythe et al. 1991]. A double-blind placebo-controlled study indicated attention and motor performance may improve after withdrawal [Duncan et al. 1990], and similar improvements in concentration and psychomotor performance were noted in another controlled study [May et al. 1992].
Despite reported CAEs, Pulliainen and Jokelainen concluded that the long-term effects of phenytoin on cognition are relatively few and restricted mainly to some visually guided motor functions [Pulliainen and Jokelainen, 1995]. More favourable findings include minimal cognitive effects in children [Forsythe et al. 1991] and elderly patients [Craig and Tallis, 1994], improvements in Stroop performance in partial epilepsy [Bittencourt et al. 1993], no differences on tasks assessing verbal and performance intelligence, memory and visuomotor function, before and after treatment in drug-naïve adult patients [Sudhir et al. 1995] and a similar cognitive profile to oxcarbazepine [Aikiä et al. 1992].
Carbamazepine
A number of cognitive and psychomotor effects have been linked to carbamazepine [Gillham et al. 1988]. A double-blind trial reported deterioration in measures of information processing speed and attention [Wesnes et al. 2009]. Other studies report poorer verbal fluency in adults with partial seizures than in control subjects [Aikiä et al. 2006], detrimental effects on memory [Shehata et al. 2009; Forsythe et al. 1991], worse arithmetic performance [Kang et al. 2007] and faster motor skills after discontinuation [Duncan et al. 1990]. Impaired movement time, reaction time, finger tapping rate and number cancellation were seen in one study, but these effects remitted by 1 month after treatment commencement [Larkin et al. 1992].
A randomized, double-blind, placebo-controlled study [Hessen et al. 2006] involving 150 epilepsy patients on AED monotherapy (mainly carbamazepine or valproate) found that drug discontinuation significantly improved performance in tests that required complex cognitive processing under time pressure, but not in more simple tasks of attention and reaction time. A later study reported similar findings, whereby performance improved in a verbal fluency task, a Stroop task, a language task and a reaction time task after discontinuation of carbamazepine [Hessen et al. 2009].
In relation to other AEDs, it has been suggested that carbamazepine has a cognitive profile that is worse than levetiracetam [Lee et al. 2011] and lamotrigine [Gillham et al. 2000] but better than phenytoin [Pulliainen and Jokelainen, 1994; Andrewes et al. 1986]. Forsythe and colleagues reported that carbamazepine-treated children performed worse than valproate-treated children on memory tasks [Forsythe et al. 1991]. However, another study [Coenen et al. 1995] reported that the cognitive profiles of valproate and carbamazepine were similar except for some aspects of attention and memory, in which individuals taking valproate scored better, and for some motor tests, in which individuals taking carbamazepine performed faster. These effects appear mild when compared with those of phenytoin and phenobarbital.
Detrimental effects on cognition are unlikely to be widespread. One study reported significantly worse performance on the Digit Symbol Substitution Test (DSST), but no difference for tracking tasks and visual analogue scales [Pieters et al. 2003]. Another study of patients with partial epilepsy showed no impairments in selective attention, memory or executive functions compared with controls, although slower information processing speed was seen with monotherapy [Engelberts et al. 2002]. Despite no decline in coordination, memory, concentration or mental flexibility, a lack of practice effects on tasks appeared to suggest subtle changes in cognitive function in one study [Prevey et al. 1996]. Similar findings were reported by Pulliainen and Jokelainen [Pulliainen and Jokelainen, 1994]. Another report indicated that for children with partial epilepsy, carbamazepine did not have significant negative effects on memory and attention tasks, although performance improved slightly after withdrawal [Riva and Devoti, 1999]. Other studies report little evidence of CAEs [Donati et al. 2007; Sudhir et al. 1995].
Some positive effects on cognition have been reported, consisting mainly of improvements in memory. For example, one study showed that story recall improved in children with benign rolandic epilepsy when treated with carbamazepine [Seidel and Mitchell, 1999]. Other beneficial effects on memory include improved immediate memory and late recall [Bittencourt et al. 1993] and better retrieval from episodic and semantic memory in adults and adolescents [Kälviäinen et al. 1995]. There may be a mild beneficial effect on hand–eye coordination in children with complex partial epilepsy in addition to improvements in memory [O’Dougherty et al. 1987]. Memory and visual information processing may benefit most from the use of controlled-release medication [Aldenkamp et al. 1987]. Despite these encouraging findings, some studies suggest carbamazepine is more likely to lead to cognitive deterioration than improvement [Helmstaedter and Witt, 2010]. Factors that may be related to a greater incidence of cognitive effects include higher dose [Gillham et al. 1988; O’Dougherty et al. 1987], longer duration of intake [Shehata et al. 2009] and polytherapy [Gillham et al. 1988].
Sodium valproate and ethosuximide
Studies are listed in Table 1. A number of studies have indicated that valproate exerts little detrimental impact on cognitive function [Sun et al. 2008; Donati et al. 2007; McKee et al. 1992; Gillham et al. 1991]. However, a small minority of patients (5 of 364 adults in one study) can develop parkinsonism with associated memory problems and psychomotor slowing [Ristic et al. 2006]. One study reported no decline in tasks assessing coordination, memory, concentration or mental flexibility, although no practice effects were seen that were evident in controls, perhaps indicating very subtle changes in cognitive function [Prevey et al. 1996]. Minor changes in cognitive function have been reported in elderly patients [Craig and Tallis, 1994], adults [Spitz and Deasy, 1991] and children [Stores et al. 1992]. However, in the latter study, modest adverse cognitive effects were seen early in treatment, which could have been the result of seizure discharge. Another study showed no relationship between plasma concentration and cognitive performance in children [Brouwer et al. 1992]. One double-blind, placebo-controlled study reported more convincing evidence of improved motor skills after discontinuation [Duncan et al. 1990]. Fortunately, the CAEs associated with valproate are likely to be reversible [Galassi et al. 1990].
Studies on the cognitive effects of valproate in patients with epilepsy.
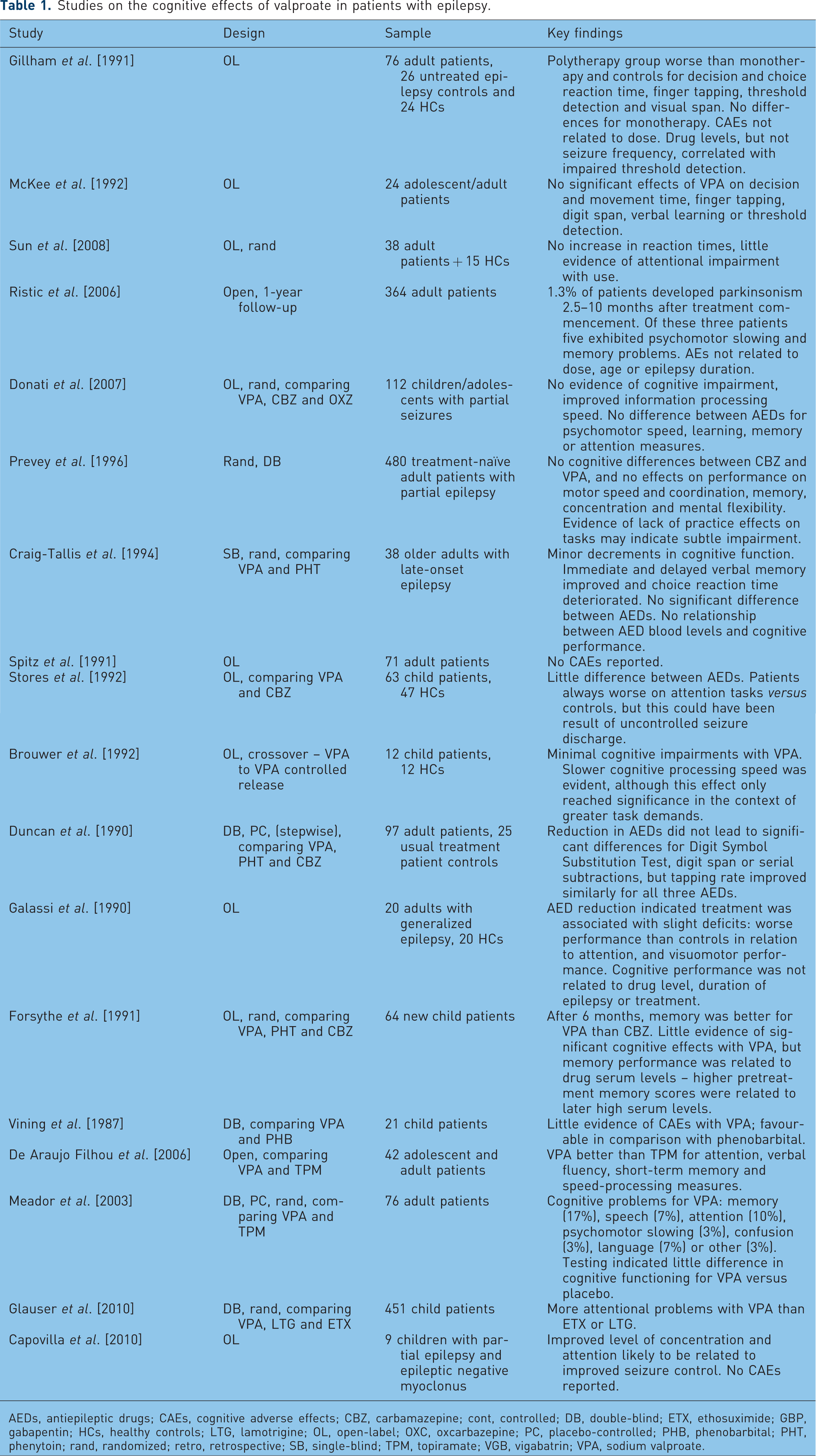
AEDs, antiepileptic drugs; CAEs, cognitive adverse effects; CBZ, carbamazepine; cont, controlled; DB, double-blind; ETX, ethosuximide; GBP, gabapentin; HCs, healthy controls; LTG, lamotrigine; OL, open-label; OXC, oxcarbazepine; PC, placebo-controlled; PHB, phenobarbital; PHT, phenytoin; rand, randomized; retro, retrospective; SB, single-blind; TPM, topiramate; VGB, vigabatrin; VPA, sodium valproate.
In relation to the effects of other AEDs on cognitive function, valproate has been suggested to be preferable to carbamazepine [Forsythe et al. 1991], phenobarbital [Vining et al. 1987] and topiramate [Sun et al. 2008; De Araujo Filhou 2006; Meador et al. 2003]. However, a recent study by Glauser and colleagues reported that attentional dysfunction was more common with valproic acid than ethosuximide (in 49% of the children compared with 33%) [Glauser et al. 2010]. In another study [Capovilla et al. 1999] ethosuximide led to no side effects in nine patients with partial epilepsy, and improvements in cognitive performance with the use of ethosuximide have been reported for individuals both with and without epilepsy [Browne et al. 1975]. Ethosuximide may therefore prove a more favourable option than valproate.
Tiagabine
A few CAEs have been reported with the use of tiagabine, such as deterioration in verbal memory from 3 months after baseline tests [Fritz et al. 2005]. However, some studies, including double-blind, placebo-controlled trials, have reported no negative impact on cognition [Aikiä et al. 2006; Dodrill et al. 1997; Kälviäinen et al. 1996; Sveinbjornsdottir et al. 1994]. It appears that most reports indicate potential for positive influences, such as improved motor speed, concentration, and verbal fluency after use [Dodrill et al. 1998]. An add-on study in adults with partial seizures documented similar effects on verbal fluency in addition to faster perceptual/motor speed for tiagabine in comparison with carbamazepine [Dodrill et al. 2000]. Patient-perceived cognition has also been shown to improve soon after treatment commencement [Cramer et al. 2001].
Vigabatrin
Vigabatrin has demonstrated few CAEs when compared with placebo in double-blind studies [Dean et al. 1999; Dodrill et al. 1993; Gillham et al. 1993] and fewer adverse effects than carbamazepine in a small, open-label, randomized, parallel-group study [Kälviäinen et al. 1995]. Vigabatrin had no detrimental effects on cognitive functions at 12 months, and memory retrieval and mental flexibility improved significantly. One double-blind, placebo-controlled, randomized study reported impaired performance relating to motor speed and design learning in adolescents and adults with partial epilepsy [Grunewald et al. 1994]. However, such negative effects were only reported in nonresponders who poorly tolerated the drug. There was also evidence of the development of tolerance in relation to CAEs. Overall, vigabatrin monotherapy seems well tolerated in relation to cognition [Monaco et al. 1997; Provinciali et al. 1996; Ylinen et al. 1995], including in patients with intellectual disability [Ylinen, 1998].
In a double-blind study involving 100 adult epilepsy patients, 52 taking vigabatrin and the rest placebo [Guberman and Bruni, 2000], there was a tendency for improvement on most tests of cognitive function: 15 showed improvement, one showed no change, and scores worsened for five tests. Other studies report improved cognition in children [Camposano et al. 2008] and in 66% of patients with epilepsy and psychosis [Veggiotti et al. 1999]. Improvements include better episodic memory, semantic memory and flexibility of mental processing in adolescents and adults [Kälviäinen et al. 1995] and decreased arithmetic response time [McGuire et al. 1992].
The main limitation with the use of vigabatrin is visual field constriction, which may affect 30–50% of patients [Gonzalez et al. 2006]. The prevalence of such difficulties may be lower in children than in adults, and the cumulative dose of vigabatrin or length of treatment may add to the personal predisposition for developing visual field constriction [Vanhatalo et al. 2002]. Caution in prescribing is merited, given reports of slowed response time linked to visual processing during use and possible difficulties detecting peripheral moving objects, which have implications for driving [Naili et al. 2009].
Clobazam
One study reported no difference between the performance of patients taking clobazam or carbamazepine on tests assessing intelligence, memory, attention, psychomotor speed, and impulsivity [Bawden et al. 1999]. There was little evidence of CAEs in an open study involving children [Jan and Shaabat, 2000]. In another study involving children, attention and alertness improved with clobazam [Munn and Farrell, 1993]. Although 72% reported improvements in cognition, 26% reported deterioration in at least one aspect of cognition. An open study of adolescents and adults [Montenegro et al. 2001] identified CAEs in just two of 97 patients.
Zonisamide
Although CAEs are often the most common reason for discontinuation of zonisamide, perhaps only 5.8% will withdraw from treatment for this reason [White et al. 2010]. There are conflicting findings as to the prevalence of CAEs. One retrospective analysis showed memory loss in 35% and attentional problems in 27% of 60 patients [Park et al. 2007], while other reports indicate cognitive problems in 4–12% of children [Kothare et al. 2006; Kim et al. 2005]. Berent and colleagues reported difficulties with learning in association with zonisamide in a small, add-on study [Berent et al. 1987]. Cognitive difficulties were also reported by an open-label investigation [Park et al. 1999]. After 1 year of treatment, 16 patients (47%) complained of cognitive deficits. Worse performance was apparent in tasks involving attention, memory and verbal fluency, and was related to dose. These authors concluded that zonisamide has adverse effects on cognition, even after 1 year of treatment.
Gabapentin
Most studies tend to report little or no cognitive impairment in association with gabapentin, including double-blind studies [Dodrill et al. 1999; Meador et al. 1999; Leach et al. 1997], and there may be potential for improved performance in cognitive measures [Mortimore et al. 1998].
Pregabalin
In one study, only 4% of patients taking pregabalin reported cognitive problems [Valentin et al. 2009]. When present, possible CAEs include deterioration in verbal and visual episodic memory [Ciesielski et al. 2006]. One double-blind, randomized, placebo-controlled study involving a large sample of adolescents and adults with partial epilepsy [French et al. 2003] reported abnormal thinking in 2% of the placebo group, compared with 3.4% of patients taking 50 mg pregabalin, 7% taking 150 mg, 7.8% taking 300 mg and 4.5% taking 600 mg. An open study of add-on pregabalin noted few reported CAEs [Jan et al. 2009]. Other reports indicate that abnormal thinking may be rare [Arroyo et al. 2004], although one report indicated this CAE could be 9–12 times more likely with pregabalin than with placebo [Beydoun et al. 2005].
Topiramate
Studies are listed in Table 2. Reports indicate that CAEs are not uncommon with topiramate [Bootsma et al. 2006], and may constitute a large proportion of the AEs in children [Mohamed et al. 2000]. One study found 44% of patients reported experiencing CAEs after 1 year of treatment [Lee et al. 2006]. However, a double-blind, randomized trial in older adults reported CAEs including memory worsening and language difficulties in only 1–5% of patients using topiramate [Ramsay et al. 2008]. Whatever the frequency of CAEs, they are the most likely reason for treatment withdrawal [Tatum et al. 2001].
Studies on the cognitive effects of topiramate in patients with epilepsy.

AEDs, antiepileptic drugs; CAEs, cognitive adverse effects; CBZ, carbamazepine; cont, controlled; DB, double-blind; ERP, event Related Potential; ETX, ethosuximide; GBP, gabapentin; HCs, healthy controls; LTG, lamotrigine; OL, open-label; OXC, oxcarbazepine; PAE, psychiatric adverse effects; PC, placebo-controlled; PHB, phenobarbital; PHT, phenytoin; rand, randomized; retro, retrospective; SB, single-blind; TPM, topiramate; VGB, vigabatrin; VPA, sodium valproate.
Cognitive side effects can include impaired concentration [Froscher et al. 2005], cognitive dulling [Coppola et al. 2002], psychomotor slowing [Tatum et al. 2001], language and comprehension problems [Fritz et al. 2005], detrimental effects on short-term memory [Gomer et al. 2007; Aldenkamp et al. 2000] and working memory [Jung et al. 2010; Lee et al. 2006; Fritz et al. 2005], poor verbal fluency [Jung et al. 2010; Gomer et al. 2007; Lee et al. 2006, 2003; Fritz et al. 2005; Thompson et al. 2000] and word-finding deficits [Mula et al. 2003b], reduced IQ score [Sun et al. 2008; Lee et al. 2003; Thompson et al. 2000] and cognitive speed [Bootsma et al. 2008a; Gomer et al. 2007], and abnormal thinking [Froscher et al. 2005]. Other studies report improvements in cognitive measures after topiramate withdrawal [Kockelmann et al. 2003]. For example, in one study, verbal fluency, verbal and spatial span and attention improved after treatment withdrawal, and patients then performed similarly to untreated patient controls [Kockelmann et al. 2003]. EEG changes may be seen in frontal regions, along with deterioration in cognitive performance [Jung et al. 2010].
Topiramate has been shown to exert more negative effects on cognition than a range of other AEDs. Poorer verbal fluency, attention and inhibitory performance have been reported than with lamotrigine [Blum et al. 2006; Kockelmann et al. 2004], and verbal fluency and working memory are also worse than with oxcarbazepine [Kim et al. 2006]. Memory and attention effects may be worse than with valproate [De Araujo Filhou et al. 2006], and verbal fluency, cognitive speed and short-term memory were worse than with levetiracetam in one study [Gomer et al. 2007].
Although some studies argue that topiramate may be more commonly linked to CAEs, there are some reports indicating these effects are rather infrequent [Brandl et al. 2010; Majkowski et al. 2005; Reith et al. 2003; Baker et al. 2002]. The rate of discontinuation due to CAEs may also be low, especially with low doses [Arroyo et al. 2005]. In one study, more than half the children taking topiramate experienced adverse effects that could interfere with learning at school, but 20% demonstrated increased alertness or improved behaviour [Moreland et al. 1999]. Reassuringly, CAEs tend to reverse with discontinuation [Lee et al. 2006; Rosenfeld et al. 1997]. Studies have also indicated that certain individuals may be more vulnerable to CAEs with topiramate, including those with temporal lobe epilepsy [Mula et al. 2003b], existing cognitive difficulties [Coppola et al. 2008] or a past psychiatric history [Kanner et al. 2003].
Lamotrigine
Studies are listed in Table 3. One double-blind, placebo-controlled crossover study of 81 patients with refractory partial seizures demonstrated no adverse effect on cognition in association with lamotrigine [Smith et al. 1993], and other studies have reported similar findings [Bootsma et al. 2008b; Gillham et al. 2000; Aldenkamp et al. 1997]. For example, in a double-blind, placebo-controlled study involving children, there were no differences in the results of cognitive tasks assessing reaction time, recognition memory, attention, visual search and working memory [Pressler et al. 2006]. Positive effects on cognitive function in epilepsy have also been reported [Placidi et al. 2000; Banks and Beran, 1991]. Attention processes, short-term memory, and motor and mental speed were investigated in 25 epilepsy patients taking carbamazepine plus lamotrigine as add-on therapy [Aldenkamp et al. 1997]. There were no significant score decreases after adding lamotrigine, and most changes were in a positive direction. It has been suggested that positive effects on cognition are related to EEG changes [Marciani et al. 1998]. Lamotrigine has also been shown to have a favourable cognitive profile in comparison with other AEDs. Verbal fluency and attentional/inhibitory performance is better than with carbamazepine [Lee et al. 2011], list learning may be better than with oxcarbazepine [Seo et al. 2007], and a double-blind, randomized study of adults with partial seizures showed performance was better with lamotrigine than topiramate for verbal fluency, the Stroop test and the DSST [Blum et al. 2006].
Studies on the cognitive effects of lamotrigine in patients with epilepsy.

AEDs, antiepileptic drugs; CAEs, cognitive adverse effects; CBZ, carbamazepine; cont, controlled; DB, double-blind; ETX, ethosuximide; GBP, gabapentin; HCs, healthy controls; LTG, lamotrigine; OL, open-label; OXC, oxcarbazepine; PC, placebo-controlled; PHB, phenobarbital; PHT, phenytoin; rand, randomized; retro, retrospective; SB, single-blind; TPM, topiramate; VGB, vigabatrin; VPA, sodium valproate.
Oxcarbazepine
Studies have indicated no deterioration in learning, memory or attention in patients treated with oxcarbazepine [Donati et al. 2006; McKee et al. 1994], and little evidence of cognitive problems after 4–12 months of treatment [Aikiä et al. 1992]. A multicentre, randomized, open-label study in children and adolescents reported little evidence of CAEs [Donati et al. 2007], while another study involving children with benign epilepsy with centrotemporal spikes showed no deterioration in cognitive tasks, and evidence of mild improvement in some cases [Tzitiridou et al. 2005]. One study documented improvement in an information processing speed task in patients with partial epilepsy [Donati et al. 2006].
Levetiracetam
Studies are listed in Table 4. In one study of patients with intractable epilepsy, levetiracetam was compared with topiramate [Huang et al. 2008]. There were no significant differences in cognition for these AEDs, and no significant negative effects were reported with the use of levetiracetam in this study. Many other studies have reported no detrimental effects on cognition [Levisohn et al. 2009; Huang et al. 2008; Gomer et al. 2007; Piazzini et al. 2006]. No significant changes in psychomotor function, memory or information processing were found in a study of 10 patients when levetiracetam was added to carbamazepine or phenytoin [Neyens et al. 1995]. In fact, levetiracetam seemed to improve reaction time, tapping rate and memory.
Studies on the cognitive effects of levetiracetam in patients with epilepsy.

AEDs, antiepileptic drugs; CAEs, cognitive adverse effects; CBZ, carbamazepine; cont, controlled; DB, double-blind; ETX, ethosuximide; GBP, gabapentin; HCs, healthy controls; LTG, lamotrigine; OL, open-label; OXC, oxcarbazepine; PC, placebo-controlled; PHB, phenobarbital; PHT, phenytoin; rand, randomized; retro, retrospective; SB, single-blind; TPM, topiramate; VGB, vigabatrin; VPA, sodium valproate.
Other studies have reported improved cognitive functioning with levetiracetam. An open study reported improvements in cognition/behaviour in 10 of 39 children [Wheless and Ng, 2002], while another study involving children with atypical benign childhood epilepsy found that 10 of 32 patients reported improvements in cognition, while the remainder stated no differences [Von Stülpnagel et al. 2010]. One controlled study provided more conservative estimates of improvement [Helmstaedter and Witt, 2010], reporting that executive functions improved in 15% but deteriorated in 5% of patients. Some studies report improved cognition according to quality-of-life scores [Wu et al. 2009; Cramer et al. 2000].
Levetiracetam may have the potential to improve a range of cognitive abilities. These include visual short-term memory [Ciesielski et al. 2006], working memory [López-Góngora et al. 2008; Rosche et al. 2004], motor functions [López-Góngora et al. 2008], psychomotor speed and concentration [Helmstaedter et al. 2008], and fluid intelligence [Rosche et al. 2004]. A randomized, double-blind, placebo-controlled study reported improved performance in set-shifting, attention and delayed logic memory tasks in patients with partial seizures in comparison with controls [Zhou et al. 2008]. One study reported that improvements in prospective memory, working memory and motor functions were related to seizure reduction [López-Góngora et al. 2008]. However, improvements in psychomotor speed, concentration and remote memory were not related to type of epilepsy, medication parameters or psychiatric history in another study [Helmstaedter et al. 2008].
Individuals with existing cognitive weaknesses may benefit most from levetiracetam. A blinded study showed recent memory improved most in patients with poor baseline scores [Huang et al. 2008]. In addition, at least two studies have reported improvements on the Mini-Mental State Examination with use [Lippa et al. 2010; Wu et al. 2009], one of these in elderly patients with cognitive impairment [Lippa et al. 2010].
Discussion
Summary of study findings
The older agents likely to have the greatest cognitive toxic potential are phenobarbital and perhaps primidone. Carbamazepine has the potential to lead to mild but sometimes significant difficulties relating to motor speed and performance on more attention-demanding tasks. The cognitive effects associated with phenytoin may be more obvious but are generally restricted to visually guided motor functions. Minimal difficulties are also likely with sodium valproate taken at low doses. Further investigation is needed, but ethosuximide may be the older drug that shows the best cognitive profile.
With regards to newer drugs, topiramate is associated with more consistent evidence of detrimental influences on cognition. Little data are available at present for zonisamide, but findings so far indicate that adverse long-term effects are possible. Study findings for tiagabine are relatively promising, with the potential for positive effects on verbal fluency and visuomotor performance, although it is unclear whether these may be the result of seizure control. There is currently limited evidence of CAEs with the use of clobazam, and reported ‘abnormal thinking’ in association with pregabalin requires further specification. There is only limited evidence that vigabatrin interferes with cognition and this agent may exert a range of cognition-enhancing effects, including improvements in memory and mental flexibility. However, its use is restricted owing to reported visual field defects. More favourable options may include gabapentin and oxcarbazepine, which have been associated with only minor cognitive difficulties. Based on the evidence reviewed, the agents least likely to interfere with cognitive processes are levetiracetam and lamotrigine. The current review indicates the most consistent evidence of widespread positive effects on cognition is for levetiracetam, which may be particularly beneficial in cases with existing cognitive limitations.
Implications for clinical practice
Importantly, the data reviewed suggest that the effects exerted by an AED could vary depending on factors linked to patient characteristics and individual susceptibility. Although reported findings are mixed, many of these potentially influential treatment and patient variables clearly compel further investigation in more focused and rigorous experimental designs.
The amount of an AED administered, or AED combinations, may be linked to the incidence of cognitive effects. CAEs may be dose related for phenytoin [Gillham et al. 1990], zonisamide [Park et al. 1999] and carbamazepine [Gillham et al. 1988], with the strongest evidence available for topiramate [Kim et al. 2006; Lee et al. 2006; Froscher et al. 2005; Thompson et al. 2000]. However, other studies report no relationship between CAEs and dose for valproate [Ristic et al. 2006; Gillham et al. 1991], topiramate [Kockelmann et al. 2004; Kockelmann et al. 2003; Gerber et al. 2000] and levetiracetam [Helmstaedter et al. 2008; Gomer et al. 2007]. Drug concentration has been linked to CAEs for valproate [Forsythe et al. 1991; Galassi et al. 1990], carbamazepine [Gillham et al. 1988; O’Dougherty et al. 1987] and topiramate [Froscher et al. 2005]. Other studies have reported no such association for valproate [Brouwer et al. 1992], topiramate [Kockelmann et al. 2003] and phenobarbital [Manni et al. 1993]. Titration rate is also potentially influential, and there is mixed evidence regarding whether this factor may lead to increased risk of CAEs with topiramate [Mula et al. 2003b; Tatum et al. 2001; Aldenkamp et al. 2000].
The use of concurrent medications appears to increase the likelihood of CAEs with many AEDs such as valproate [Gillham et al. 1991], carbamazepine [Gillham et al. 1988] and topiramate [Brandl et al. 2010; Mula et al. 2003b], but is perhaps less relevant in relation to levetiracetam [Helmstaedter et al. 2008]. Combining topiramate with lamotrigine led to an increased likelihood of cognitive problems in one study [Gerber et al. 2000].
Certain individuals may be more vulnerable to the CAEs associated with particular AEDs. These include refractory cases, patients with different kinds of epilepsy and younger or older patients. One study of vigabatrin reported CAEs in a relatively limited set of individuals classified as nonresponders [Grunewald et al. 1994], although the characteristics of these individuals were poorly specified. Improvements in seizure control are particularly important in relation to attributing the cause of positive effects on cognition. In relation to levetiracetam, some studies report associations between enhanced cognition and seizure reduction [Von Stülpnagel et al. 2010], while others argue improvements may be independent of seizure reduction [Wheless and Ng, 2002]. Although AEDs such as topiramate and carbamazepine could have the potential to affect cognitive function in a wide variety of cases, some studies have reported that temporal lobe epilepsy may be a risk factor for the development of CAEs [Mula et al. 2003a, 2003b; Gigli et al. 1996]. Word-finding difficulties in particular have been linked to the presence of simple partial seizures and left temporal EEG focus [Mula et al. 2003b]. There is little evidence that patients with a longer history of epilepsy are more likely to be susceptible to CAEs with sodium valproate [Ristic et al. 2006; Galassi et al. 1990], although duration of drug intake has been shown to be a related factor for CAEs with carbamazepine [Shehata et al. 2009]. Age has sometimes been shown to be unrelated to the presence of CAEs [Ristic et al. 2006], but is likely to play a role in the expression of cognitive difficulties, and one complicating factor in the examination of age effects is the use of differing instruments with individuals of different age groups.
Current or previous cognitive or psychiatric problems may also be linked to the cognitive effects of an AED. With topiramate, CAEs may be most likely in individuals with intellectual disability [Coppola et al. 2008] or previous cognitive difficulties [Gerber et al. 2000]. Other risk factors associated with the use of topiramate in adults include the occurrence of psychiatric adverse events and having a past psychiatric history [Kanner et al. 2003], and depression and hippocampal sclerosis in temporal lobe epilepsy [Mula et al. 2003a]. In relation to levetiracetam, one study reported that psychiatric history was not associated with cognitive effects [Helmstaedter et al. 2008], although another report indicated this agent could be more likely to lead to cognitive improvements in cases with existing cognitive difficulties [Huang et al. 2008].
Limitations and recommendations for future research
Particular AEDs may have greater potential for negative or beneficial impacts on cognition in epilepsy, and these effects may be mediated by both treatment and patient characteristics. However, it is difficult to compare findings across studies owing to variation in study design, treatment group and assessment tools. Analysis is also limited by the availability of studies for particular agents. Many investigations have been conducted into agents including topiramate, whereas there are limited data available for AEDs such as ethosuximide and pregabalin.
There are significant difficulties relating to the consistency of available information pertaining to study methodology. Many studies fail to assess cognitive effects using a wide variety of tasks. Some reports fail to clearly indicate patient characteristics such as epilepsy type, seizure type or focus (left/right/frontal/temporal). Other useful information such as effect size is rarely available. Furthermore, many reports have failed to investigate predictive factors for the development of CAEs. Future studies should seek to determine whether reported changes in cognition are linked to drug efficacy in treating seizures rather that direct effects on cognition per se.
It can be particularly difficult to differentiate between AEDs on the basis of cognitive findings. In some cases there could be age-difference effects, which could partly reflect the assessment of children through behavioural observation and parent report, by comparison with older patients’ self report. The inclusion of different kinds of patient groups (e.g. treatment-naïve patients, refractory patients, patients with epilepsy after brain injury) and control measures (healthy individuals or nonmedicated patients; no treatment, placebo or alternative medication etc.) further complicate comparisons across studies. The CAEs reported by these studies will at least partly reflect the differing clinical characteristics and specific vulnerabilities of the groups compared.
Another problem relates to poor definitions of cognitive functions, and lack of consensus in the terms used to refer to cognitive abilities. Some studies refer to ‘mental processing speed’, others refer to ‘psychomotor speed’, and yet others use terms such as ‘information processing rate’. It is often unclear as to whether such descriptions refer to the same ability. Vague terms are also used, such as ‘cognitive dulling’ and ‘abnormal thinking’. These need to be better specified in order to be informative. In addition, particular tasks are sometimes reported as measuring slightly different cognitive abilities that are often closely linked, such as memory, learning and attention. For example, tasks such as the Stroop test may be considered to make demands in terms of attention, concentration, response inhibition and maybe even memory. Future studies need to use consistent, more precisely defined terms and consider the individual contributions of different cognitive abilities or processing stages involved in tasks and assessments.
A number of randomized, double-blind trials have been conducted. These studies appear favourable as they control for bias, although this approach could lack ecological validity. Add-on trials may be problematic because of a possible increase in the incidence of side effects due to the potentiation of toxicity. However, these studies do have value in relation to current prescribing practices, as many patients are only successfully treated with a combination of different AEDs. What may constitute a more rigorous investigation may provide data that are not so readily applicable in everyday clinical practice. Consistent evidence across studies using rigorous and more naturalistic methods will provide a good indication of the reliability of study findings.
More specific, systematic studies are needed to fill the gaps in our knowledge and follow up reported patterns in the literature about the cognitive effects linked to AEDs. Longer-term investigations would also offer critical insight, because individuals may gradually develop tolerance to an AED’s side effects. It is also informative for studies to investigate whether performance alters on discontinuation, in order to determine if effects are reversible. Systematic trials are needed that compare the effects of commonly prescribed agents across subgroups of patients with epilepsy (e.g. in partial versus generalized epilepsy). Comparing age-defined subgroups could also be useful, as drug effects are likely to differ between younger and older children.
A wide range of assessments clearly need to be included in future research. Studies investigating cognition should include tests of a range of executive functions including working memory; verbal fluency; response inhibition; set-shifting and measures assessing transient, sustained and divided attention; short and long-term aspects of verbal and visual memory; and motor functioning including dexterity and reaction time tests. One useful outcome of future research would be the identification or development of tailored instruments that are sensitive to the specific cognitive changes associated with particular AEDs.
Footnotes
Acknowledgements
The authors had no assistance with study design, data collection, data analysis or manuscript preparation.
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
