Abstract
Background:
Prior studies suggest that longer drive time (DT) to specialists and higher area deprivation index (ADI) are linked to worse outcomes in inflammatory bowel disease (IBD), but their impact on postileocolic resection (ICR) care in patients with Crohn’s disease (CD) is not well defined.
Objectives:
We aimed to evaluate disparities in post-ICR care based on DT and ADI.
Methods:
Spatial analysis was performed with ArcGIS Pro to geocode patient and medical facility addresses with StreetMap Premium locators. High DT was defined as >60 min from the center. Data were analyzed using basic statistics and multivariate logistic regression.
Design:
A retrospective cohort study was performed of CD patients’ post-ICR care at two tertiary-care IBD centers (January 2018–March 2023).
Results:
Our study included 293 patients; 44% had high DT. High DT had a higher median number of preoperative advanced therapy (2 vs 1, p = 0.007). Despite this, there was no difference between cohorts in median days to postop ileocolonoscopy (IOE; 257 vs 332, p = 0.59) and surgical recurrence rate (21% vs 27%, p = 0.278). Tobacco use and perianal disease were associated with increased odds of postoperative IOE (adjusted odds ratio (aOR) 2.09) and advanced therapy initiation (aOR 2.00).
Conclusion:
We identified no differences in postoperative colonoscopy timing or surgical recurrence in patients with CD at two tertiary IBD centers based on DT or ADI. Given the lack of disparities in care delivery among patients treated in tertiary IBD centers, further comparative studies to care outside of specialized networks are needed to evaluate whether centralization to IBD centers is superior.
Plain language summary
This study assessed post-ICR care disparities in Crohn’s disease based on drive time and area deprivation. Among 293 patients, no differences in post-op care or recurrence were found, suggesting equitable care at tertiary IBD centers, regardless of geographic or socioeconomic factors.
Introduction
Although we have seen significant advances in our ability to achieve steroid-free remission among patients with Crohn’s disease (CD) using an increasing armamentarium of therapies, 1 patients with CD may still require ileocolic resection (ICR) in the event of disease complications, such as bowel obstruction, fistula, and abscess formation. 2 An estimated 80% of patients will have an ICR after 20 years of disease, with 30% needing an ICR within the first 5 years of disease.2,3 In those patients who undergo ICR, postoperative endoscopic monitoring with an ileocolonoscopy (IOE) is recommended 6–12 months after surgery. 4
Prior studies have demonstrated that patients from rural communities have lower rates of inflammatory bowel disease (IBD) office visits.5,6 In previous population-based cohort studies, rural patients with IBD demonstrated an increased risk for IBD-related hospitalizations, emergency department visits, and gastrointestinal/gastroenterology/gastroenterologist (GI) office visits when compared to urban area patients.7,8 Despite growing recognition of challenges in rural IBD management, few studies have evaluated geographic disparities in postoperative care—particularly following ICR, where specialized perioperative coordination is often required. This is an area ripe for disparities given the need for travel to multidisciplinary care centers for surgical and IBD expertise perioperatively.
Therefore, the aim of this study was to compare the postoperative management of patients with CD after ICR, with a focus on the impact of geospatial analysis as a potential method of identification of disparities in guideline-based postoperative management. 4 For these analyses, we focused on the association between drive time (DT) to an IBD center where surgery occurred and the Area Deprivation Index (ADI) as factors potentially influencing postoperative management. The primary outcome evaluated was time to initial postoperative IOE; however, we also evaluated the time to follow-up with a gastroenterologist, time to initiation of biologic therapy, and proportion of patients experiencing surgical recurrence after ICR for CD.
Methods
Data source
The Strengthening of the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were followed. 9 This retrospective cohort study was approved by the institutional review boards at the University of North Carolina (UNC) and Washington University (WashU). Informed consent was not required because patient data were de-identified.
We used data from institutional electronic health records from the included study sites. The study cites included in the study were tertiary IBD multidisciplinary centers at WashU in Saint Louis, Missouri, and the UNC in Chapel Hill, North Carolina. The IBD center was defined as both the main and satellite medical facility sites under one tertiary medical site that offered comprehensive multidisciplinary IBD care. WashU has three sites included in the study data, Kingshighway, West County, and South County, which encompass a less than 2-mile drive (<30-min DT) from each other. UNC had one main site.
Study cohort
We included encounter data from all adult patients aged 18 and above who had ICR at any of the tertiary sites between January 1, 2018, and March 31, 2023. Inclusion criteria for the study were any patient aged 18 or older who had undergone ICR for clinically established CD during the study period. CD diagnosis was assessed utilizing International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10) codes and confirmed with review of clinical documentation. Exclusion criteria included patients who did not have a diagnosis of CD, patients with a diagnosis of ulcerative colitis or IBD-undifferentiated, or those who were evaluated for CD but did not undergo resection between study dates and/or at the study centers.
Outcomes of interest
The primary outcome evaluated was time to initial postoperative IOE. This outcome was selected given the American Gastrointestinal Association guidelines recommending that all patients undergo IOE within 6–12 months after ICR for CD. 4 We also evaluated secondary outcomes of the time to follow-up with a gastroenterologist, time to initiation of biologic therapy, and proportion of patients experiencing surgical recurrence after ICR for CD. Any missing data for these outcomes is noted in the primary analyses and is not included in multivariable analyses.
Covariates
Each patient’s electronic medical record was reviewed to extract clinical and demographic data that may have influenced the postoperative management of CD, and ultimately the recurrence of CD after ICR, using a standardized case extraction form. Data collected included Montreal classification of CD, history of tobacco use, body mass index at the time of surgery, and the use of postoperative therapy. Additionally, data were collected regarding emergency department visits and hospital admissions in the postoperative period.
Statistical analysis and spatial analysis
Continuous variables were summarized using medians and interquartile range (IQR) and compared using the Wilcoxon–Rank-sum test. Categorical variables were expressed as proportions and compared using Fisher’s exact and Chi-square testing as appropriate.
Spatial analysis was performed with ArcGIS Pro to geocode patient and medical facility addresses with StreetMap Premium (SMP) locators. The distance comparison between the closest medical facility and the patient’s address was tabulated with the Network Analysis Extension and the SMP routing dataset. Then, characteristics of the surrounding addresses were determined utilizing the US Census Bureau and University of Wisconsin ADI data. The ADI was created utilizing Health Resources & Services Administration and census block groups to rank neighborhoods based on socioeconomic factors. ADI is a satellite for disadvantage and is calculated for each U.S. census block group utilizing weighted indicators of socioeconomic status as defined by Singh.10,11 These include but are not limited to the median family income, monthly mortgage, percentage below poverty level, and percentage of single-family households.
In our study, DT was defined as the total transport duration time from the listed patient zip code to the closest medical center site by motor vehicle under normal traffic patterns. High DT was defined as a total DT of greater than 60 min from the IBD center based on findings that this is a threshold for delay in care. 12 High ADI is defined as >75, given that higher ADI is associated with greater disadvantage. These parameters were used to divide the study population into separate cohorts: high DT, low DT, high ADI, and low ADI.
All factors were analyzed in multivariable logistic regression models to adjust for potential confounders. Variables were selected for potential inclusion in the multivariable models using backward selection (where the threshold to remain in the model was 0.10), along with the forced variables of DT or ADI. Collinearity was assessed as part of model selection. Other than the spatial analyses described above, all other analyses were performed using SAS (version 9.4) statistical software (SAS Institute, Cary, NC, USA). There was no formal sample size calculation, as this was a retrospective study of events that had already occurred, and the largest available sample size to address the topic of geospatial analysis in postoperative CD management to date. The study protocol was approved by the respective Institutional Review Boards at the UNC at Chapel Hill and WashU in St Louis.
Results
Among the 2 centers, 293 patients with CD who underwent ICR between January 1, 2018, and March 31, 2023, were included. Eleven out of 293 patients were lost to follow-up with no postoperative GI office visits during the entire study period. The median age of the cohort was 39 years (IQR 29–54; Table 1). Among the entire population, 128 patients (44%) had a high DT (Figure 1) and 94 patients (32%) had a high ADI (Figure 2) at the time of ICR for CD. Fifty-six patients (19%) had both high ADI and high DT.
Clinical and demographic characteristics of patients undergoing ileocolonic resection for CD between 2018 and 2023.

BMI, body mass index; CD, Crohn’s disease; IBD, inflammatory bowel disease; IQR, interquartile range.

Drive time (in minutes) to WUSM (a) and UNC facilities (b).

Area Deprivation Index as Rank and Block groups in area surrounding WUSM (a) and UNC facilities (b).
When comparing those patients with high and low DT at ICR for CD, the median number of preoperative biologic therapies was significantly higher among patients with high DT (2 vs 1, p = 0.007, Table 2). There were no other significant differences when comparing the two groups, including age, sex, and CD behavior. In evaluating postoperative outcomes, there was no statistically significant difference in time to postoperative ileo-colonoscopy (median 332 vs 257 days, p = 0.593). Of note, there were four patients (1%) with missing data regarding postoperative ileo-colonoscopy. There was also no difference when comparing the high DT cohort to the low DT cohort in median days to postoperative follow-up with a gastroenterologist (77 vs 63, p = 0.345) or median days to start of postoperative biologic therapy (66 vs 65, p = 0.902) over the entire follow-up period. Among 125 patients with endoscopic assessments by Rutgeerts’ scoring, there was no difference in endoscopic recurrence when evaluated by high and low DT (35% vs 38%, p = 0.669). The proportion of patients experiencing surgical recurrence between high and low DT was also similar (27% vs 21%, p = 0.278).
Evaluation of clinical and demographic characteristics among patients undergoing ileocolonic resection for CD, stratified by DT.
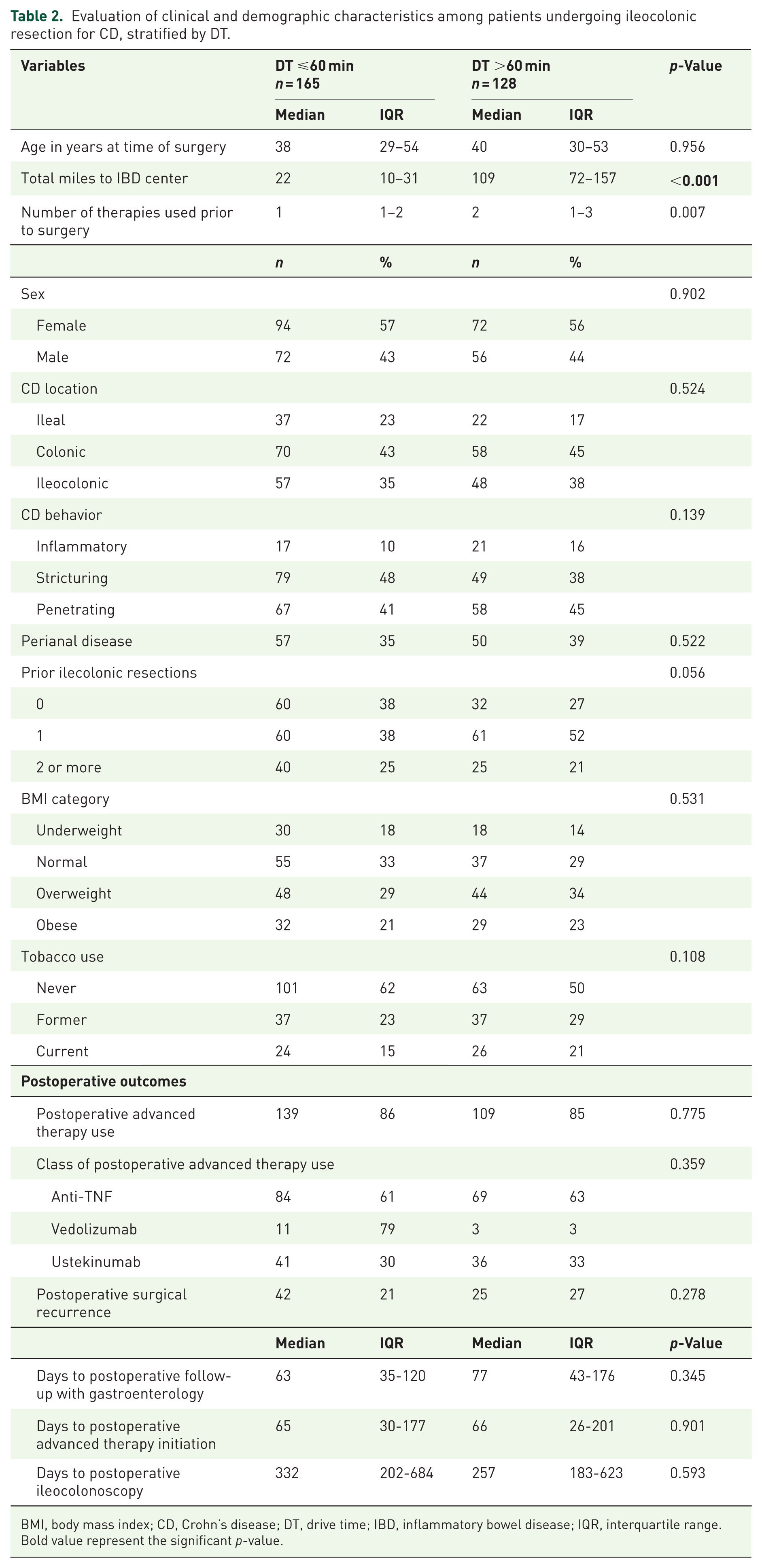
BMI, body mass index; CD, Crohn’s disease; DT, drive time; IBD, inflammatory bowel disease; IQR, interquartile range.
Bold value represent the significant p-value.
In evaluating the impact of ADI, patients with high ADI used a greater median number of biologics prior to ICR (2 vs 1, p = 0.002) and had a lower proportion of patients with no history of tobacco use (43% vs 64%, p = 0.002) and a lower proportion of patients with no prior ICR (24% vs 37%, p = 0.015, Table 3). Patients with high ADI also drove a greater number of total miles to reach the IBD center (median 69 vs 33, p = 0.002). When comparing patients with high ADI compared to the remainder of the population, there was no difference in median days to a postoperative colonoscopy (327 vs 302, p = 0.593), to a follow-up visit with a gastroenterologist (77 vs 64 days, p = 0.469) or to advanced therapy initiation after ICR (70 vs 63 days, p = 0.541). There was also no difference in median days to surgical recurrence (1159 vs 1048, p = 0.830). Among patients undergoing endoscopy with Rutgeerts’ scoring, there was no difference in endoscopic recurrence when evaluated by high and low ADI (41% vs 35%, p = 0.603).
Evaluation of clinical and demographic characteristics among patients undergoing ileocolonic resection for CD, stratified by ADI.

ADI, Area Deprivation Index; BMI, body mass index; CD, Crohn’s disease; IBD, inflammatory bowel disease; IQR, interquartile range.
Bold value represent the significant p-value.
When evaluating the odds of undergoing interval colonoscopy within 12 months postoperatively in a multivariable model, only current tobacco use remained significantly associated with an increased odds of undergoing IOE within 12 months after adjusting for travel time (adjusted odds ratio (aOR) 2.09, 95% CI 1.08–4.03, Supplemental Table 1). Similarly, current tobacco use was associated with an increased odds of undergoing IOE within 12 months of ICR after adjusting for ADI (aOR 1.98, 95% CI 1.02–3.84).
In an evaluation of the odds of initiating an advanced therapy within the first 6 months of ICR, patients with a history of perianal disease (PD) demonstrated an increased odds for initiation of advanced therapy (aOR 2.00, 95% CI 1.07–3.72), while current tobacco users were less likely to initiate advanced therapy (aOR 0.33, 95% CI 0.17–0.68) after adjusting for travel time (Supplemental Table 1). In a model accounting for ADI, patients with a history of PD were also at increased odds for initiating advanced therapy within 6 months after ICR (aOR 1.97, 95% CI 1.05–3.66), while current tobacco users were less likely (aOR 0.31, 95% CI 0.15–0.63).
Discussion
In our study, we found that patients with high DT and high ADI had a greater median number of preoperative biologics compared to their low DT and low ADI counterparts. Additionally, tobacco use and PD were associated with increased rates of post-ICR IOE and therapy initiation, respectively. However, we did not note any disparities in postoperative care after ICR in patients with CD at tertiary IBD centers, regardless of DT or ADI.
Patient distance or DT to specialist care is a known variable in the likelihood of patient follow-up. 12 In our study, we did not find a difference in surgical recurrence between high and low DT (27% vs 21%, p = 0.278). 13 Borren et al. 14 found that patients in the higher distance quartile had an increased risk for biologic use and surgery than their counterparts. However, distance was concluded to have no effect on surgical needs in a separate study by Grob et al. 15 It should be noted that Grob and Borren had differing upper limits of distance, 48.7 versus 81.8 km, respectively, which may have affected their ability to detect outcome differences. Overall, Borren’s conclusion is consistent with the literature finding that longer distance or DT is associated with worse health outcomes. 16 In IBD, this is reflected as longer distance patients having lower rates of office visits, lower rates of endoscopy, and increased hospitalizations.5,7,8
Though we did not find a difference in need for surgery, our study found higher median numbers of presurgical biologics in patients with longer DT. This finding may be partially explained by the longer distance having an inverse effect on adherence by hindering the patient–provider relationship.17,18 Additionally, patients of longer distance may have difficulty with drug monitoring or switch agents to delay surgical interventions. 19 Patients at a longer distance may be referred to IBD centers for advanced management given medical complexity.
Current guidelines recommend smoking cessation to avoid worsening disease activity. Our study found that current tobacco use was associated with an increased likelihood of undergoing an IOE within 12 months post-ICR. Active smoking has been deemed an independent risk factor for postoperative recurrence in CD. Several studies have shown that tobacco users have worse disease phenotypes, increased odds for flares, increased need for maintenance treatment, and surgery.20,21 For example, Reese et al. 22 ’s meta-analysis found that smokers had a 2.5-fold increased risk of surgical recurrence and a 2-fold risk of clinical recurrence. Patient smoking status and symptoms likely motivated the preferential timing of post-ICR endoscopy among tobacco users. However, smokers in our study also had biologic treatment initiation later than their nonsmoking counterparts postoperatively. It is unclear what factors, such as patient preference, provider awareness, or lack of follow-up, led to biologic delay in the current smoker population.
Likewise, patients with PD were associated with initiating advanced therapy within the first 6 months of ICR. This is most likely due to impaired quality of life and the recognition that PD represents an increased risk for recurrence after surgery. 4 Patients with symptoms related to perianal CD, such as pruritus, abscess, and pain with defecation, have higher healthcare resource utilization and costs. 23 Current guidelines on PD management recommend early combined surgical and medical management. 24 Given that high-risk patients in our population (those with PD and tobacco users) were more likely to receive guideline-recommended care, our tertiary care IBD centers demonstrated an ability to provide timely follow-up to those patients at the highest risk of recurrence, regardless of DT or potential sociodemographic disadvantages.
Our study is not without limitations. ADI has been critiqued for its overemphasis on home values, which can mischaracterize high-cost-of-living urban areas with high deprivation. 25 At the time of our study, we were not aware of more accurate indices or the balanced ADI with updated metrics to reduce dependence on housing.26–28 Thus, the use of traditional ADI could have led to misinterpretation of the social determinants of health. Additionally, ADI does not look at individual-level factors that affect follow-up, such as family or community support. Additionally, driving time as a marker for access to health care facilities was employed instead of distance. We elected to use travel time to account for the variable traffic patterns in the metropolitan areas of our study sites: Saint Louis, MO, and Chapel Hill, NC. Sixty minutes was the marker to separate data into binary groups (high and low DT) due to association with diagnostic delay. 12 A limitation of travel time is that it carries the assumption of motor vehicle transport, does not account for alternative modes of travel, or factor in wait times for public transportation. Despite this, we believe that DT is the best way to account for infrastructure differences that affect patient follow-up between our two sites. Lastly, there is potential for selection bias in patients lost to follow-up. Although not explored in our study, patients may have elected to follow-up locally or been deterred by travel time. All these limitations are noted given the possibility of Type II error due to lack of statistical power as an explanation of the lack of difference in key clinical outcomes. However, we would note that the patients with high DT had shorter times to postoperative colonoscopy and lower rates of surgical recurrence (favoring those patients with potential geographic disparities).
Further interventions are needed to extend a uniform level of monitoring to all CD patients. Of our studied patients, 7.5% engaged in telemedicine follow-up visits after surgery. Telemedicine is promising as a method to overcome distance barriers by improving access to specialist care.29–31 Huang et al. 29 showed that distance management through patient use of technology to interact with providers and self-managed care was effective in decreasing clinic visits without increasing relapses or hospitalizations. Additionally, nudge interventions can improve guideline adherence through electronic medical system reminders aimed at providers and patients to schedule follow-up visits, postoperative endoscopies, or start biologics.32–34 Our study had multiple strengths, including the inclusion of patients from two hospital catchment areas. The diversity of geography and patient population adds validity to our results and makes it readily applicable to other tertiary centers in the USA. It is not readily applicable to nontertiary or community-based practices. However, we did not account for the ways geography, infrastructure, and climate influenced postoperative care by study sites (Illinois vs North Carolina).
Despite the high DT cohort having a higher median ADI and preoperative use of biologic therapies, our tertiary IBD centers provided equivalent post-ICR care. Previous studies have shown that both surgeons’ volume of ICRs and hospital surgical volume impact outcomes in patients with CD undergoing surgery.35–37 Our study’s findings may suggest that once patients can access tertiary IBD centers, subsequent synchronization of surgical and medical care is able to diminish disparities due to distance from the IBD center or due to ADI. However, the superiority of tertiary IBD centers cannot be validated given the lack of comparison to nontertiary and community-based practices.
Conclusion
In conclusion, we demonstrated that despite a greater preoperative use of biologic therapies in those with a high DT, no disparities were seen in postoperative care after ICR at two tertiary IBD centers. Our findings suggest centralization of CD care at tertiary centers may benefit patients. Further studies are needed to establish the superiority of such models comparing tertiary to nontertiary IBD centers and identify scalable interventions to ensure equitable postoperative care.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261436999 – Supplemental material for Lack of disparities in postoperative care after ileocecal resection in patients with Crohn’s disease at tertiary inflammatory bowel diseases centers
Supplemental material, sj-docx-1-tag-10.1177_17562848261436999 for Lack of disparities in postoperative care after ileocecal resection in patients with Crohn’s disease at tertiary inflammatory bowel diseases centers by Shannon Coombs, Molly Powell, Kemmian Johnson, Jessica Hu, Mollie Webb, Philip McDaniel, Parakkal Deepak and Edward L. Barnes in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848261436999 – Supplemental material for Lack of disparities in postoperative care after ileocecal resection in patients with Crohn’s disease at tertiary inflammatory bowel diseases centers
Supplemental material, sj-docx-2-tag-10.1177_17562848261436999 for Lack of disparities in postoperative care after ileocecal resection in patients with Crohn’s disease at tertiary inflammatory bowel diseases centers by Shannon Coombs, Molly Powell, Kemmian Johnson, Jessica Hu, Mollie Webb, Philip McDaniel, Parakkal Deepak and Edward L. Barnes in Therapeutic Advances in Gastroenterology
