Abstract
Background:
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder worldwide, characterized by recurrent abdominal pain and altered bowel habits, which significantly impair quality of life and impose a socioeconomic burden. Traditional pharmacological treatments have limited efficacy and are often associated with side effects, highlighting the need for effective non-pharmacological interventions. Non-invasive transcutaneous nerve stimulation (NITNS) has shown potential in improving IBS symptoms and autonomic function, but systematic evidence is lacking.
Objectives:
To systematically evaluate the efficacy and safety of NITNS in IBS and explore its potential mechanisms.
Design:
Systematic review and meta-analysis.
Data sources and methods:
Randomized controlled trials (RCTs) were identified through PubMed, Embase, Cochrane Library, and Web of Science from database inception to September 28, 2025. Studies included patients aged 18–75 years who met IBS diagnostic criteria. Interventions involved NITNS, with sham-NITNS as the control. Primary outcomes were IBS symptom severity scale (IBS-SSS) and IBS quality of life (IBS-QOL). Secondary outcomes included visual analogue scale (VAS), Bristol stool form scale (BSFS), heart rate variability (HRV), and adverse events (AEs). Data extraction and risk-of-bias assessment were independently performed by two reviewers.
Results:
Four RCTs, including 170 patients, were analyzed. NITNS significantly reduced IBS-SSS scores (p = 0.0001) and increased IBS-QOL scores (p < 0.0001). VAS (p = 0.0003) and BSFS (p < 0.00001) scores also improved significantly. HRV results suggested NITNS might influence the autonomic nervous system function. The incidence of AEs did not differ significantly between NITNS and sham-NITNS groups (p = 0.64). Exploratory subgroup analysis indicated that differences in NITNS stimulation modalities and IBS subtypes might influence the therapeutic efficacy of NITNS. Overall evidence quality ranged from low to very low.
Conclusion:
NITNS is a safe and effective non-pharmacological intervention with potential value in the management of IBS. However, further large-scale, high-quality RCTs are needed to confirm their long-term efficacy and safety.
Trial registration:
PROSPERO CRD420251157617.
Keywords
Introduction
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder worldwide, characterized by recurrent abdominal pain associated with changes in bowel habits or stool form in the absence of identifiable organic lesions. 1 Owing to variations in diagnostic criteria, study design, and population characteristics, the reported prevalence of IBS varies across regions, with an overall global prevalence of approximately 4%–10%.2–4 Although IBS is not a life-threatening condition, its chronic, relapsing, and multifactorial nature imposes a substantial physical and psychological burden on patients. IBS greatly reduces quality of life and work productivity, increases healthcare utilization, and contributes to higher socioeconomic costs, posing a significant challenge to public health systems.5,6 Therefore, further research into its pathophysiology and effective therapeutic strategies remains urgently needed.
The treatment of IBS remains highly challenging, mainly due to its complex pathophysiology and marked symptom heterogeneity. 7 Conventional pharmacological therapies primarily aim to relieve symptoms, including antispasmodics, serotonin receptor modulators, antidiarrheal and laxative agents, prokinetics, and antidepressants, which are widely used in clinical practice. Although these medications can provide short-term relief for some patients, their overall efficacy remains unsatisfactory. Moreover, gastrointestinal discomfort, dizziness, and somnolence are common side effects, which not only impair quality of life but also reduce adherence to long-term treatment.8–11 With the growing understanding of the brain-gut axis in IBS, it has become increasingly clear that pharmacological therapy alone can not fully address its multifaceted pathophysiology. Thus, non-pharmacological approaches, such as dietary modification, psychological and behavioral therapies (e.g., cognitive behavioral therapy), and neuromodulation-based interventions, have emerged as research hotspots and potential therapeutic alternatives.12–14 These novel approaches aim to improve emotional regulation and gut function, ultimately promote symptom relief and enhance quality of life.
In recent years, with the rapid development of neuromodulation techniques in the field of functional gastrointestinal disorders, non-invasive transcutaneous nerve stimulation (NITNS) has become a promising therapeutic approach for IBS. NITNS is defined as a group of interventions with shared features that regulate autonomic nervous system function through transcutaneous and non-invasive ways, including transcutaneous auricular vagus nerve stimulation (taVNS) and transcutaneous electrical acustimulation (TEA). Compared with invasive transcutaneous nerve stimulation (ITNS), NITNS offers several advantages, including higher safety, better reproducibility, ease of use, and good patient tolerance. These features have led to its broad application in gastrointestinal motility treatment, postoperative functional recovery, and chronic pain management.15–17 NITNS delivers low-frequency (LF) electrical stimulation to specific skin regions innervated by target nerves, thereby directly or indirectly enhancing vagal activity and restoring autonomic nervous system balance.18,19 Previous studies have shown that NITNS not only improves gastrointestinal motility but also alleviates visceral hypersensitivity, reduces anxiety and depressive symptoms, and modulates gut microbiota composition by influencing the brain-gut axis.20–22 Through these mechanisms, NITNS may promote overall gastrointestinal functional recovery from multiple dimensions.
Although several randomized controlled trials (RCTs) have recently explored the use of NITNS in patients with IBS, the current evidence remains limited.15,16,22,23 The reported findings on its efficacy and safety are inconsistent, likely due to variations in stimulation modalities, parameters, target sites, and IBS subtypes. Moreover, most studies have small sample sizes, short follow-up durations, and variable methodological quality, which restrict the reliability and generalizability of their conclusions. Previous reviews have mainly focused on invasive or semi-invasive neuromodulation techniques, while systematic evidence on NITNS remains lacking so far.19,24
Based on the current state of research, we conducted a systematic review and meta-analysis on the efficacy and safety of NITNS in patients with IBS. This study aimed to: (1) evaluate the efficacy of NITNS in IBS patients after a 4-week treatment period; (2) explore potential mechanisms of action, particularly its influence on autonomic nervous function; and (3) systematically assess the safety during NITNS treatment. Through this study, we wish to supplement the evidence and limitations for NITNS in patients with IBS and further provide a theoretical basis for the future optimization and mechanism exploration of non-invasive neuroregulation therapies in functional gastrointestinal disorders.
Methods
Search strategy
This study was registered in the International Prospective Register of Systematic Reviews (PROSPERO; registration number: CRD420251157617). The systematic review and meta-analysis were conducted in strict accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and the Cochrane Handbook for Systematic Reviews of Interventions.25,26 We systematically searched PubMed, Embase, Cochrane Library, and Web of Science from database inception to September 28, 2025, for all relevant studies. The search strategy combined MeSH terms, keywords, and their occurrences in titles and abstracts, using search phrases such as “IBS” and “transcutaneous nerve stimulation.” To ensure comprehensive coverage, the reference lists of included studies were also manually screened to identify additional potentially eligible studies. No language restrictions were applied during the search. The full search strategy is provided in Supplemental Material.
Inclusion and exclusion criteria
Inclusion criteria were as follows:
(1) Study population: Adults aged 18–75 years who met the diagnostic criteria for IBS.
All included patients were diagnosed according to the Rome IV criteria, which require recurrent abdominal pain on average at least 1 day/week over the past 3 months, associated with two or more of the following: 1) pain related to defecation; 2) changes in stool frequency; and 3) changes in stool form.
IBS was defined as meeting these criteria for the past 3 months, with symptom onset at least 6 months prior to diagnosis. 27
(2) Intervention and control: The intervention group received NITNS, while the control group received sham-NITNS. Sham-NITNS was implemented in one of two ways: 1) electrodes were placed at the same anatomical sites as the intervention group but without electrical stimulation, or 2) electrodes were placed in non-target areas (e.g., outside the vagal nerve distribution), with the same stimulation parameters and procedures as the intervention group. Apart from the intervention, participants in both groups did not receive any other pharmacological or non-pharmacological treatments that could affect the outcomes during the treatment period. The stimulation sites of NITNS are shown in Figure 1.
(3) Outcomes: The primary outcomes were the IBS symptom severity scale (IBS-SSS) and the IBS quality of life (IBS-QOL), which together assessed symptom improvement and the impact on patients’ quality of life. Secondary outcomes included: 1) visual analogue scale (VAS), for assessing the intensity of abdominal pain or bloating; 2) Bristol stool form scale (BSFS), for evaluating stool form and bowel function; 3) heart rate variability (HRV), as an indirect measure of autonomic nervous system activity and regulation of the brain-gut axis; and 4) adverse events (AEs), to evaluate the safety profile of NITNS. Detailed definitions of all outcomes are provided in Supplemental Material.
(4) Study: RCTs only.
Exclusion criteria were as follows:
(1) Non-original research, such as reviews, commentaries, conference abstracts, letters, case reports, or studies with unavailable full-text data.
(2) Studies in which the intervention group did not receive NITNS alone (e.g., combined with pharmacological, acupuncture, or psychological/behavioral therapies), or the control group received interventions beyond sham stimulation.
(3) Duplicate publications, overlapping results, or studies in which independent samples could not be clearly distinguished.

Stimulation sites of NITNS. Red dots indicate the stimulation sites.
Data extraction and assessment of risk of bias
Study selection and data extraction were independently performed by two reviewers (Y.B.W. and Z.H.S.) according to predefined inclusion and exclusion criteria. Extracted data included publication year, first author, country of study, sample size, age range and sex, details of the intervention (including stimulation modality, target site, and stimulation parameters), treatment duration and frequency, as well as outcomes. All extracted data were cross-checked, and any discrepancies were resolved through discussion with a third reviewer (X.Y.) to reach consensus, ensuring the accuracy and consistency of the data.
Concurrently, the two reviewers independently assessed the risk of bias for the included studies using the Rob bias assessment tool, following the guidance of the Cochrane Handbook for Systematic Reviews of Interventions. The assessment covered seven domains: (1) random sequence generation (selection bias); (2) allocation concealment (selection bias); (3) blinding of participants and personnel (performance bias); (4) blinding of outcome assessment (detection bias); (5) incomplete outcome data (attrition bias); (6) selective reporting (reporting bias); and (7) other bias. Any disagreements were resolved through discussion among the three reviewers to ensure a consistent and rigorous evaluation of bias.
Quality assessment
Two independent reviewers assessed the quality of evidence for the included studies using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system and summarized the results in a summary of findings table. Evidence grading was performed by GRADEpro GDT (version 3.6.1; Evidence Prime, Hamilton, ON, Canada) software. Following GRADE guidelines, the quality of evidence for each outcome was systematically evaluated, considering factors for downgrading, including risk of bias, inconsistency, indirectness, imprecision, and publication bias, as well as factors for upgrading, including large effect, plausible confounding, and dose-response gradient. Any disagreements during the review process were resolved through discussion with a third reviewer to reach a consensus.
Statistical analysis
Meta-analysis of the included studies was performed primarily using Review Manager 5.4 to estimate pooled effect sizes. Mean difference (MD) was used for continuous outcomes, while risk ratio (RR) was used for dichotomous outcomes, with corresponding 95% confidence interval (CI) calculated. Statistical heterogeneity among studies was assessed using the I2 statistic, with p < 0.05 or I2 > 50% indicating significant heterogeneity. A fixed-effects model was applied when heterogeneity was not significant, whereas a random-effects model was used in the presence of significant heterogeneity. To further explore the potential impact of NITNS stimulation modalities and IBS subtypes on heterogeneity, exploratory subgroup analysis was conducted. Sensitivity analyses were performed to assess the robustness of the meta-analysis results. Potential publication bias was evaluated using funnel plots and Egger’s test. All statistical tests were two-sided, with a significance threshold of p < 0.05.
Results
Literature retrieval and study characteristics
A total of 359 records were initially identified from 4 databases. After removing 91 duplicates, 268 potentially relevant studies remained. Screening of titles and abstracts excluded 182 studies related to animal experiments or non-original research, including systematic reviews and meta-analyses, narrative reviews, trial registry records, study protocols, conference abstracts, letters, and case reports. An additional 60 studies were excluded due to non-RCT study designs. Full-text assessment of the remaining studies identified 15 RCTs using ITNS, such as electroacupuncture or percutaneous electrical nerve field stimulation, which did not meet the focus of this study on NITNS and were excluded. Five studies were further excluded because their participants had other functional gastrointestinal disorders rather than IBS. Two studies were excluded for not reporting primary outcomes or for including only a single-session intervention. Ultimately, 4 RCTs involving a total of 170 patients with IBS met the inclusion criteria and were included in this systematic review and meta-analysis (Figure 2). All studies were published between 2021 and 2024, with individual sample sizes ranging from 40 to 52. The mean age of IBS patients ranged from 41.5 to 51.4 years. The intervention period was 4 weeks in all studies, with the intervention group receiving NITNS and the control group receiving sham-NITNS. Baseline comparisons of outcome measures between intervention and control groups showed no statistically significant differences (p > 0.05). Detailed characteristics of the included studies are presented in Table 1.

PRISMA flow diagram of study selection process.
Characteristics of the included studies.
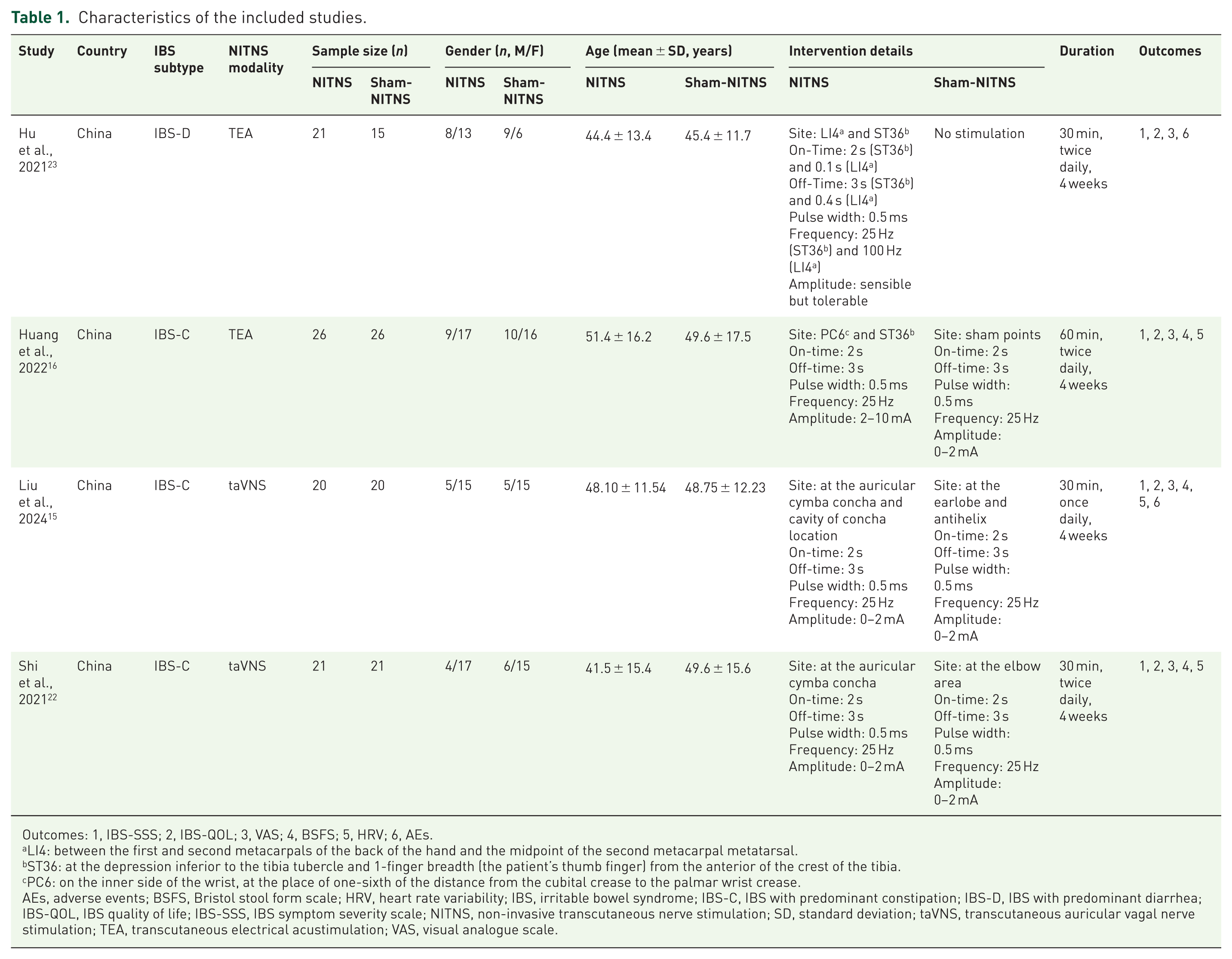
Outcomes: 1, IBS-SSS; 2, IBS-QOL; 3, VAS; 4, BSFS; 5, HRV; 6, AEs.
LI4: between the first and second metacarpals of the back of the hand and the midpoint of the second metacarpal metatarsal.
ST36: at the depression inferior to the tibia tubercle and 1-finger breadth (the patient’s thumb finger) from the anterior of the crest of the tibia.
PC6: on the inner side of the wrist, at the place of one-sixth of the distance from the cubital crease to the palmar wrist crease.
AEs, adverse events; BSFS, Bristol stool form scale; HRV, heart rate variability; IBS, irritable bowel syndrome; IBS-C, IBS with predominant constipation; IBS-D, IBS with predominant diarrhea; IBS-QOL, IBS quality of life; IBS-SSS, IBS symptom severity scale; NITNS, non-invasive transcutaneous nerve stimulation; SD, standard deviation; taVNS, transcutaneous auricular vagal nerve stimulation; TEA, transcutaneous electrical acustimulation; VAS, visual analogue scale.
Risk of bias
We further assessed the risk of bias in the four included RCTs, with the results shown in Figure 3. Overall, all studies were judged to have a low risk of bias for allocation concealment, incomplete outcome data, and selective reporting, indicating good internal consistency of the findings. Regarding random sequence generation, three of the four studies clearly described their randomization methods, suggesting that the allocation process was generally appropriate. For blinding, three RCTs employed a single-blind design, resulting in a high risk of bias for blinding of participants and personnel. Two studies explicitly reported that outcome assessments were conducted independently and in a blinded manner, resulting in a low risk of bias for blinding of outcome assessment. Overall, the included studies were of moderate-to-high quality, with the main source of bias arising from limitations in blinding, while randomization, data completeness, and reporting transparency were generally satisfactory.
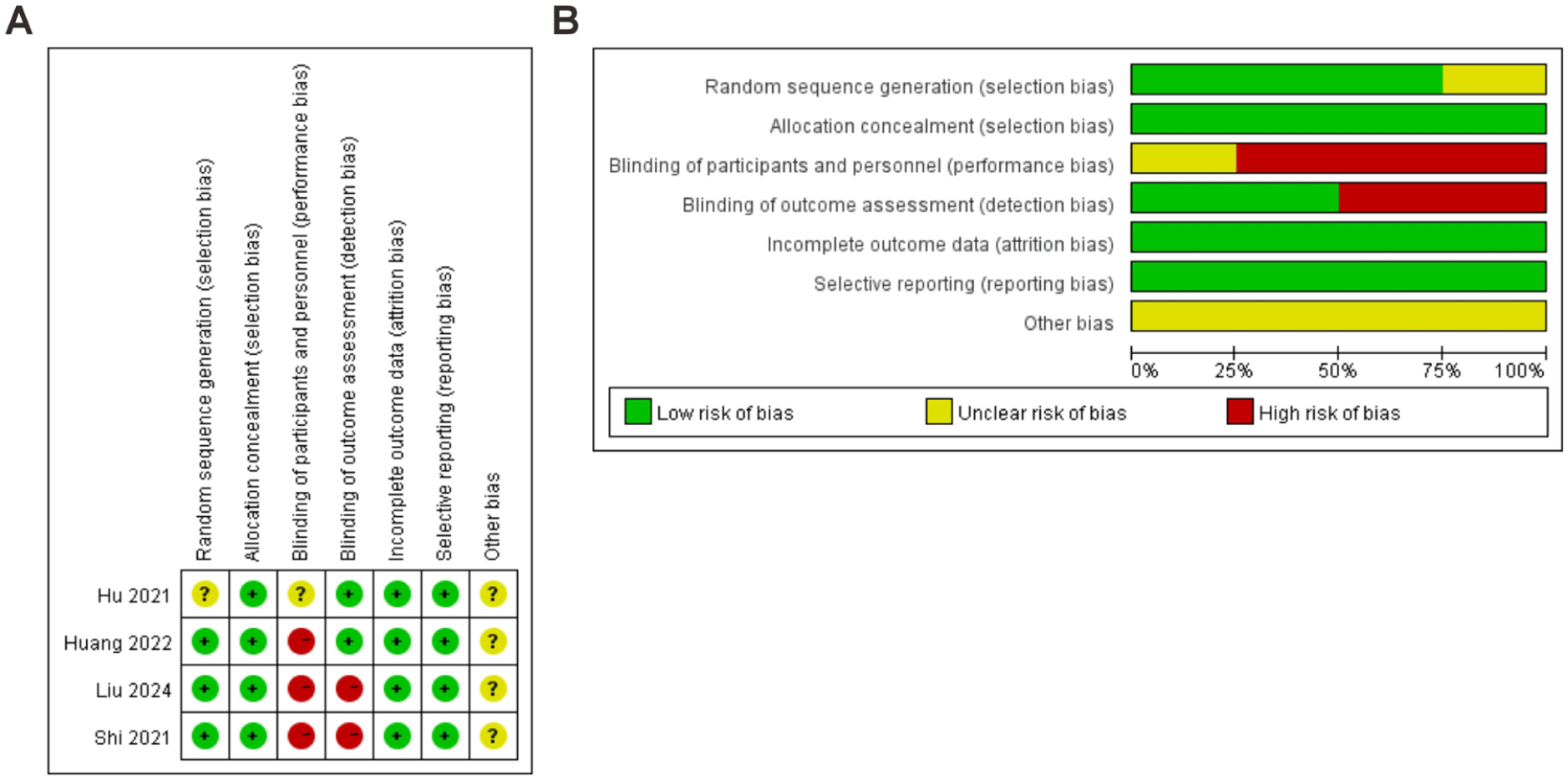
Risk of bias summary (A) and graph (B).
Primary outcomes
All four studies reported the effects of a 4-week NITNS intervention on IBS-SSS (Figure 4(A)). Compared with sham-NITNS, the NITNS group showed a significant reduction in IBS-SSS scores after 4 weeks of treatment, with a pooled MD of −67.69 (95% CI: −102.46 to −32.93). Heterogeneity analysis indicated moderate heterogeneity (I2 = 67%, p = 0.03), and the overall effect was statistically significant (Z = 3.82, p = 0.0001). Similarly, four studies reported IBS-QOL outcomes. However, the study by Liu et al. did not calculate IBS-QOL scores using a standardized method, and the scoring approach differed from that of the other studies. To ensure comparability and robustness of the analysis, this study was excluded from the meta-analysis of IBS-QOL. Consequently, data from three RCTs were included in the quantitative analysis (Figure 5(A)). The NITNS group showed a significant improvement in IBS-QOL scores compared with sham-NITNS (MD = 9.41; 95% CI: 4.86–13.95; Z = 4.06, p < 0.0001), with low heterogeneity (I2 = 0%, p = 0.48). Sensitivity analyses for both IBS-SSS and IBS-QOL indicated that sequential removal of individual studies did not substantially alter the direction or magnitude of the pooled effect (Figures 4(B) and 5(B)). Publication bias was assessed using funnel plots and Egger’s test (Figures 4(C) and 5(C)). The Egger’s test p-values were 0.361 (Figure 4(D)) and 0.924 (Figure 5(D)). However, due to the small number of included studies, the statistical power for detecting publication bias was limited, and these results should be interpreted with caution.

Meta-analysis of IBS-SSS in IBS patients after treatment with NITNS. (A) The forest plot showed IBS-SSS. (B) Sensitivity analysis demonstrated the reliability of NITNS to affect IBS-SSS. (C) Filled funnel plot for the publication bias. (D) Egger’s funnel plot analysis revealed potential publication bias.

Meta-analysis of IBS-QOL in IBS patients after treatment with NITNS. (A) The forest plot showed IBS-QOL. (B) Sensitivity analysis demonstrated the reliability of NITNS to affect IBS-QOL. (C) Filled funnel plot for the publication bias. (D) Egger’s funnel plot analysis revealed potential publication bias.
Secondary outcomes
Visual analogue scale
Four RCTs reported VAS outcomes. In Hu et al., the change from baseline in VAS was reported, but the raw data were unavailable. The remaining three studies reported VAS scores at the end of the 4-week NITNS treatment. To ensure consistency and comparability, we used post-treatment VAS scores as the secondary outcome for analysis. Meta-analysis indicated that NITNS significantly reduced VAS scores compared with the control group (MD = −1.75; 95% CI: −2.70 to −0.80; p = 0.0003; Figure 6(A)). However, substantial heterogeneity was observed (I2 = 80%, p = 0.006), and a random-effects model was applied. Sensitivity analysis was shown in Supplemental Figure 1(A). Publication bias was further evaluated using funnel plots and Egger’s test (p = 0.713; Supplemental Figure 1(B) and (C)). Nevertheless, due to the limited number of included studies, the statistical power for detecting publication bias was low, and the conclusion remains somewhat uncertain.

Forest plots showed (A) VAS, (B) BSFS, (C) HF, (D) LF/HF, and (E) AEs after NITNS treatment.
Bristol Stool Form Scale
Additionally, 3 RCTs including 134 IBS patients reported the BSFS as an outcome. Pooled analysis showed that NITNS significantly improved BSFS scores in IBS patients (MD = 1.28; 95% CI: 0.85–1.72; I2 = 54%; p < 0.00001; Figure 6(B)). Sensitivity analysis confirmed that the exclusion of individual studies did not materially alter the pooled effect (Supplemental Figure 1(D)). Funnel plots were relatively symmetrical, and Egger’s test (p = 0.191) did not indicate significant publication bias (Supplemental Figure 1(E) and (F)). Similarly, given the limited number of studies, the statistical power for detecting publication bias was low.
Heart rate variability
Furthermore, three RCTs evaluated the effects of NITNS on HRV. Specifically, one study reported both high-frequency (HF) and the ratio of LF to HF (LF/HF), while the other two studies reported either HF or LF/HF. Pooled analysis of two studies including 94 IBS patients showed that NITNS significantly increased HF values (MD = 0.13; 95% CI: 0.06–0.19; I2 = 0%; p = 0.0001; Figure 6(C)). Additionally, based on two studies involving 92 patients, NITNS significantly reduced LF/HF ratios (MD = −0.26; 95% CI: −0.50 to −0.02; I2 = 56%; p = 0.04; Figure 6(D)). Sensitivity analyses and funnel plots for HF and LF/HF are shown in Supplemental Figure 1(G)–(J). Egger’s test could not be performed for the limited number of studies, and these findings require confirmation in further high-quality RCTs.
Adverse events
Regarding safety, 2 RCTs reported data on AEs, including a total of 76 IBS patients. Pooled analysis showed no significant difference in the incidence of AEs between the NITNS and sham-NITNS groups (RR = 1.50; 95% CI: 0.28–8.04; p = 0.64; Figure 6(E)). Reported AEs were generally mild and included auricular skin rash, dizziness, palpitations, and transient diarrhea, with no serious AEs observed. Due to the limited number of studies, sensitivity analysis and publication bias assessment were not performed.
Subgroup analysis
Due to the limited number of included studies, exploratory subgroup analysis was conducted for primary outcomes (IBS-SSS and IBS-QOL). Compared with sham-NITNS, IBS-SSS scores were significantly reduced in the taVNS (MD = −93.89; 95% CI: −117.55 to −70.22; p < 0.00001) and TEA (MD = −37.26; 95% CI: −72.70 to −1.82; p = 0.04) subgroups, while IBS-QOL scores were significantly increased (taVNS: MD = 13.70; 95% CI: 3.17–24.23; p = 0.01; TEA: MD = 8.42; 95% CI: 3.39–13.46; p = 0.001) after 4 weeks of NITNS (Figure 7(A) and (C)). Notably, a significant difference between taVNS and TEA subgroups was observed for IBS-SSS scores (p = 0.009). In contrast, no significant difference was found between the two stimulation modalities for IBS-QOL scores (p = 0.38).

Forest plots of subgroup analysis for the effects of NITNS on IBS-SSS and IBS-QOL. (A, B) Effects of NITNS on IBS-SSS by stimulation modalities (A) and IBS subtypes (B). (C, D) Effects of NITNS on IBS-QOL by stimulation modalities (C) and IBS subtypes (D).
In the exploratory subgroup analysis based on IBS subtypes, patients with IBS with predominant constipation (IBS-C) showed a significant reduction in IBS-SSS scores (MD = −85.89; 95% CI: −107.25 to −64.54; p < 0.00001) and a significant increase in IBS-QOL scores (MD = 10.86; 95% CI: 5.57–16.16; p < 0.0001). In contrast, compared with sham-NITNS, patients with IBS with predominant diarrhea (IBS-D) did not show significant changes (IBS-SSS: MD = −20.00, 95% CI: −63.43 to 23.43, p = 0.37; IBS-QOL: MD = 5.34, 95% CI: −3.51 to 14.19, p = 0.24; Figure 7(B) and (D)). Similarly, the reduction in IBS-SSS scores after 4 weeks of NITNS was significantly greater in IBS-C (p = 0.008), whereas no significant difference between IBS-C and IBS-D was observed for IBS-QOL scores (p = 0.29). Funnel plots for the subgroup analysis are presented in Supplemental Figure 2. Given the limited number of included studies, these conclusions remain tentative.
Quality assessment of evidence
The certainty of evidence and strength of recommendations for the primary and secondary outcomes are summarized in Table 2. Overall, the quality of evidence for IBS-SSS, IBS-QOL, VAS, BSFS, and HF was rated as low. The quality of evidence for LF/HF and AEs was rated as very low.
Summary of findings for included studies.

GRADE Working Group grades of evidence: High quality, further research is very unlikely to change our confidence in the estimate of effect; Moderate quality, further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate; Low quality, further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate; Very low quality, we are very uncertain about the estimate.
The quality of the evidence for all outcomes is shown in bold.
The corresponding risk (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
AEs, adverse events; BSFS, Bristol Stool Form Scale; CI, confidence interval; GRADE, Grading of Recommendations Assessment Development and Evaluation; HF, high-frequency; IBS, irritable bowel syndrome; IBS-QOL, IBS quality of life; IBS-SSS, IBS symptom severity scale; LF, low-frequency; RR, risk ratio; VAS, visual analogue scale.
Discussion
This systematic review and meta-analysis comprehensively evaluated the efficacy and safety of NITNS in patients with IBS. Pooled results from four RCTs indicated that NITNS might significantly reduce IBS-SSS scores (low quality of evidence) and improve IBS-QOL scores (low quality of evidence). It also showed a significant improvement in VAS scores (low quality of evidence) and BSFS scores (low quality of evidence), without significantly increasing the risk of AEs (very low quality of evidence). Furthermore, the therapeutic effects of NITNS might be mediated through enhanced vagal activity (low quality of evidence) and modulation of autonomic nervous system balance (very low quality of evidence). Exploratory subgroup analysis further indicated that taVNS might be more effective than TEA in improving IBS symptoms and that different IBS subtypes might exhibit variable responsiveness to neuromodulation.
Although only 4 RCTs comprising a total of 170 patients with IBS were included, all eligible studies adopted randomized controlled designs and demonstrated relatively high overall methodological quality. During the statistical analysis, we strictly adhered to established methodological principles for meta-analyses. Therefore, we believe that, based on the current evidence, this meta-analysis provides preliminary and exploratory quantitative evidence regarding the potential efficacy of NITNS in IBS. Nevertheless, the findings should be interpreted with caution. For the primary outcomes, IBS-SSS and IBS-QOL, although the observed improvements were statistically significant, it is important to distinguish statistical significance from clinical significance. Previous studies have shown that the minimal clinically important difference (MCID) for the IBS-SSS is 50 points, with a change of this magnitude generally considered to reflect a meaningful improvement in symptoms and commonly used as a response threshold in clinical trials. 28 With respect to IBS-QOL, an RCT in patients with diarrhea-predominant IBS showed that, compared with placebo, a significantly higher proportion of patients treated with rifaximin achieved an improvement of at least 14 points in the total IBS-QOL score, supporting the practical use of 14 points as the MCID. 29 Although the improvement in IBS-QOL observed in our study did not reach this threshold, symptom relief in IBS is often accompanied by meaningful gains in quality of life. Therefore, we consider that the improvements observed with NITNS in both IBS-SSS and IBS-QOL may be of clinical relevance. The low to very low quality of evidence does not imply that NITNS is ineffective. Rather, it reflects limitations in the current evidence base in terms of study number, methodological quality, and duration of follow-up.
As a non-invasive neuromodulation intervention, NITNS demonstrated potential in this study to improve IBS symptoms, enhance quality of life, and modulate autonomic nervous system function. Its high portability, ease of use, and favorable safety profile make it more acceptable for IBS patients. In contrast, although traditional ITNS has been shown in multiple studies to effectively relieve abdominal pain and improve bowel function, its clinical application is limited by procedural complexity, invasiveness, and the risk of serious AEs such as pain, infection, or electrode displacement, which can reduce patient adherence.19,24,30–32 Therefore, with a clearer understanding of its mechanism and confirmed efficacy, NITNS has strong potential to surpass the long-studied ITNS as a primary neuromodulation approach for IBS treatment.
To further elucidate the mechanisms underlying NITNS, this study analyzed autonomic function indicators, including HF and LF/HF. The results suggest that NITNS significantly increased HF values and reduced LF/HF ratios. This seems to indicate that it enhances vagal activity while suppressing sympathetic responses, thereby maintaining autonomic balance. However, although the included studies commonly used HRV as a representative index of autonomic nervous system function and drew conclusions regarding autonomic function based on it, we consider that this approach remains methodologically limited. HRV is only an indirect index of autonomic nervous system function and can not directly measure vagal nerve activity or neurotransmitter release. An increase in HF is generally considered to reflect enhanced vagal activity, but this relationship can be influenced by sympathetic tone, breathing, physical activity, and other factors. 33 At the same time, the reliability of the LF/HF ratio as a measure of sympathovagal balance has also been questioned. Therefore, the findings of this study only suggest a potential for autonomic regulation, and direct neurophysiological evidence is still needed, such as vagal nerve recordings, neurotransmitter measurements, or brain imaging.
It is worth noting that this neuromodulatory effect may underlie its benefits in alleviating abdominal pain, improving bowel function, and enhancing quality of life and is closely associated with activation of the cholinergic anti-inflammatory pathway. 34 Bowles et al. 35 demonstrated in a mouse model that optogenetic inhibition of cholinergic neurons blocked the motor learning enhancement induced by vagal stimulation, highlighting the central role of the cholinergic pathway in vagal regulation. Another study showed that the improvement of gastric motility disorders induced by colonic distension via vagal stimulation could be abolished by the cholinergic antagonist atropine, further confirming the dependence on cholinergic activity. 36 Several preclinical studies have shown that vagal stimulation in rodents activates the cholinergic anti-inflammatory pathway, with α7 nicotinic acetylcholine receptors subsequently engaged. The binding of acetylcholine inhibits the NF-κB signaling pathway and reduces the release of pro-inflammatory cytokines such as TNF-α and IL-1β, thereby attenuating low-grade inflammation.37,38 This mechanism has been systematically reviewed elsewhere. 39 Additionally, Atalar et al. 40 found that vagal activation promotes the release of the anti-inflammatory cytokine IL-10 while suppressing excessive pro-inflammatory cytokine production, a finding supported by clinical observations from Hu et al. 23 Taken together, these studies appear to support that NITNS may improve IBS symptoms by enhancing vagal activity and activating cholinergic anti-inflammatory pathways, which is broadly consistent with our findings. However, such multilevel inferences from indirect indices to mechanistic conclusions should be interpreted with caution and viewed as hypothesis-generating rather than definitive evidence of underlying mechanisms.
Some outcome measures in this study exhibited moderate heterogeneity. Due to the limited number of included studies, we were unable to explore the specific sources of heterogeneity via meta-regression. Based on previous literature and clinical experience, we hypothesized that heterogeneity might primarily arise from differences in NITNS stimulation modalities and IBS subtypes, both of which could partially influence the stability of the pooled effect estimates. This hypothesis is partially supported by our exploratory subgroup analysis. Regarding stimulation modalities, subgroup analysis indicated that taVNS may be more effective than TEA. taVNS directly stimulates the vagus nerve via auricular branches, whereas TEA primarily exerts indirect modulation through somatic acupoints. However, given the limited number of included studies, this conclusion should be interpreted with caution. Concerning IBS subtypes, NITNS appeared to improve symptoms in IBS-C more effectively than in IBS-D. However, this finding should also be interpreted cautiously. Evidence for IBS-D is limited and based on a single study, which restricts the ability to draw general conclusions about the effectiveness of NITNS in this subtype. This represents a clear gap in the evidence and highlights the need for well-designed, adequately powered prospective controlled trials specifically targeting IBS-D populations. Differences in stimulation parameters are also considered a key factor affecting the comparability of NITNS studies. 41 Although we did not perform subgroup analysis based on stimulation parameters, the included studies used relatively similar settings in terms of pulse width, frequency, and amplitude, suggesting that parameter variation likely had a limited impact on overall results. The selection of stimulation sites in TEA may play an important role in efficacy differences, too. Commonly used acupoints reported in previous studies include ST36, LI4, PC6, ST37, SP6, CV17, and LI11, with ST36, LI4, and PC6 being the most frequently combined.16,23,17,42–44 Different acupoints may influence the brain-gut axis via distinct neural reflex pathways, affecting vagal activity and inflammatory regulation.
This study has several limitations. Firstly, the number of included studies and the overall sample size were relatively small, which may have reduced statistical power and the stability of the findings, thereby limiting a full estimation of the overall effect, despite strict adherence to established meta-analytic methods. Secondly, differences across studies in NITNS stimulation modalities, stimulation sites, and the IBS subtypes enrolled may have introduced heterogeneity. Although exploratory subgroup analysis was performed, the results should be interpreted with caution, given the limited number of studies and participants. In addition, HRV cannot be equated with autonomic nervous system function and serves only as an indirect index. Mechanistic inferences based on HRV remain uncertain. IBS is a chronic condition, yet all included studies applied a 4-week intervention period, which is insufficient to adequately assess the durability of treatment effects. Consequently, the conclusions of this meta-analysis are largely confined to the short-term efficacy and safety of NITNS in IBS and cannot be extended to long-term clinical benefit or symptom maintenance. Overall, future RCTs should focus on larger sample sizes, standardized NITNS intervention protocols, stratified or subtype-specific designs for different IBS subtypes, longer intervention and follow-up periods, and the incorporation of direct neurophysiological measures, in order to provide higher-quality evidence and clarify the long-term efficacy and underlying mechanisms of NITNS in IBS.
Conclusion
In conclusion, the current evidence supports NITNS as a safe and effective non-pharmacological intervention with potential value in the management of IBS, providing new insights and an evidence-based rationale for its use. However, given the limited number of included studies and the low overall quality of evidence, further large-scale, high-quality RCTs are needed to confirm their long-term efficacy and elucidate underlying mechanisms.
Supplemental Material
sj-doc-1-tag-10.1177_17562848261436121 – Supplemental material for Efficacy and safety of non-invasive transcutaneous nerve stimulation in patients with irritable bowel syndrome: a systematic review and meta-analysis
Supplemental material, sj-doc-1-tag-10.1177_17562848261436121 for Efficacy and safety of non-invasive transcutaneous nerve stimulation in patients with irritable bowel syndrome: a systematic review and meta-analysis by Yanbin Wei, Zihe Shi, Shanshan Wu and Xin Yao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-doc-2-tag-10.1177_17562848261436121 – Supplemental material for Efficacy and safety of non-invasive transcutaneous nerve stimulation in patients with irritable bowel syndrome: a systematic review and meta-analysis
Supplemental material, sj-doc-2-tag-10.1177_17562848261436121 for Efficacy and safety of non-invasive transcutaneous nerve stimulation in patients with irritable bowel syndrome: a systematic review and meta-analysis by Yanbin Wei, Zihe Shi, Shanshan Wu and Xin Yao in Therapeutic Advances in Gastroenterology
