Abstract
Background:
Endoscopy units throughout the world have seen a substantial increase in colonoscopy activity. One way to keep patient waiting time acceptable could be to introduce alternative diagnostic modalities. Colon capsule endoscopy has been proven to have a high diagnostic accuracy, yet the re-investigation rates reported are too high. Faecal haemoglobin concentration may offer a valid measure for triaging patients between diagnostic modalities, due to its association with colonic pathology.
Objective:
To investigate the re-investigation rate and risk classification in complete colon capsule endoscopies in a screening population stratified by faecal haemoglobin concentration.
Design:
Cross-sectional analyses of a data set derived from the intervention arm of a large randomised controlled trial, CareForColon2015, conducted in the Region of Southern Denmark between August 2020 and December 2022.
Methods:
Complete colon capsule endoscopy investigations were identified, and the proportions of investigations leading to re-investigation by colonoscopy were calculated, stratified by faecal haemoglobin concentration. Further, the odds of re-investigation were estimated by logistic regression models and the odds of increased risk classification by ordinal regression models.
Results:
The re-investigation rate was 58.6% in 1413 complete colon capsule endoscopies out of 2030 procedures. There were no significant differences (p = 0.312) in re-investigation rates between faecal haemoglobin concentration subgroups with 58.2%, 61.1% and 62.7% in the groups of 100–249, 250–499 and >499 ng hb/mL buffer, respectively. The odds of referral for re-investigation did not differ significantly either. The odds of increased risk classification (i.e. higher than level one) was 1.17 (CI 95% 0.92; 1.49, p = 0.211) for concentrations between 250 and 499 ng hb/mL buffer, and 1.40 (CI 95% 1.12; 1.77, p = 0.003) for concentrations above 499 ng hb/mL buffer, compared to 100–249 ng hb/mL buffer.
Conclusion:
Faecal haemoglobin concentration did not prove to be a stand-alone selection parameter for diagnostic modality in a faecal immunochemical test-positive colorectal cancer screening population, although it was significantly associated with an increased risk classification.
Plain language summary
Colon capsule endoscopy is a less invasive investigation modality used for diagnostic purposes in the large bowel, as an alternative to the conventional colonoscopy. In 2030, colon capsule endoscopy investigations revealed that 72.2% needed a follow-up investigation. This is too many to accept on the basis of patient experience and cost. In our analyses, we have investigated whether the concentration of blood in the stool is a good indicator of whether an individual would need a follow-up investigation. If that were to be the case, we could direct patients with a high blood concentration in the stool directly to colonoscopy to avoid too many re-investigations. However, this turned out not to be the case, as the proportion needing an additional investigation only differed between 58.2% and 62.7% between the groups of lowest and highest blood concentration.
Introduction
The demand for alternative endoscopic diagnostic modalities is needed to relieve endoscopic units, limiting the patient waiting time. The increased activity of the European endoscopy units have not only, but especially, been caused by the introduction of structured population-based colorectal cancer screening. The use of colon capsule endoscopy has increased in the last decade, although not yet to numbers that cause a significant reduction in colonoscopy activity. In order to facilitate clinical use, one should focus on quality, complication rate, patient experience, costs, the use of specialised labour and accessibility. Three of these important issues depend directly upon the re-investigation rate—the costs, the use of specialised labour and the patient experience. The re-investigation rate depends upon technical issues that are being addressed (bowel cleanliness and transit time), but it also very much depends upon the rate of significant positive findings needing therapy or biopsy.
Larger clinical trials and feasibility clinical services are promising and may potentially relieve endoscopy units. Colon capsule endoscopy has been introduced in Scotland and England as a result of increasing demands, where faecal haemoglobin concentration is used to triage patients between investigation modalities. 1 If the rate of colon capsule endoscopies resulting in a referral for further diagnostic or therapeutic endoscopic procedures is too high, the cost-effectiveness and the patient experience will be affected. The European Society of Gastrointestinal and Abdominal Radiology (ESGAR) guidelines do not suggest colon capsule endoscopy as a first-line screening modality, but do so in cases with positive faecal immunochemical test and incomplete or unfeasible colonoscopy (weak recommendation, moderate quality evidence). 2 Colon capsule endoscopy has been tested in faecal immunochemical test-positive screening participants, with completion rates of 54%–81% and polyp detection rates of 51%–74%,3,4 indicating a high re-investigation rate.
It has been established that faecal haemoglobin concentration is associated with colorectal cancer incidence and mortality, as well as adenoma detection rates.5–11 If the re-investigation rate differs substantially between subgroups of faecal haemoglobin concentrations, these concentrations may provide a beneficial selection parameter for which screening participants should be offered colon capsule endoscopy as a filter test, and who should be referred straight to colonoscopy. This strategy has been suggested previously, 12 but has not been tested in a screening population. As discussions of the risk of leaving small polyps untreated are ongoing, future guidelines may suggest lowering the risk estimations of individuals with several small or diminutive polyps. It is also of interest to see how this would affect the re-investigation rate in colon capsule endoscopy.
We aimed to investigate the re-investigation rate and risk classification in complete colon capsule endoscopies in a screening population stratified by faecal haemoglobin concentration to evaluate its value in triaging patients for diagnostic strategy.
Methods
This manuscript was prepared in concordance with the STROBE checklist (Supplemental material). 13 In a randomised controlled trial, conducted in the Region of Southern Denmark between August 2020 and December 2020 (described previously14–17), 2031 participants underwent colon capsule endoscopy following positive faecal immunochemical test (100 ng hb/mL buffer or higher) as a filter test before colonoscopy in the Danish colorectal cancer screening programme. Patient were eligible for inclusion if they were invited for colorectal cancer screening in the inclusion period, but were excluded if they fulfilled one of the following criteria; history of abdominal surgery (excluding appendectomy), symptomatic Crohn’s disease, cardiac pacemaker, known renal insufficiency, pregnancy/breastfeeding, allergy towards bowel preparation or booster medication, known severe constipation or disability to comply either cognitively or physically. The result of each colon capsule endoscopy led to a risk classification determining whether or not to refer the individual for colonoscopy (re-investigation). All incomplete colon capsule endoscopies (i.e. inadequate bowel preparation or incomplete transit) would elicit a colonoscopy. All complete colon capsule endoscopies were included in this study.
Faecal immunochemical test
The Danish colorectal cancer screening programme provides citizens aged 50–74 with the OC-Auto Sampling bottle 3 (Eiken Chemical Co, Japan) biennially. Participants then place approximately 10 mg of faeces in a vial containing 2.0 mL of buffer and send it to the laboratory, where the faecal immunochemical test is performed using the OC-Sensor Diana instruments (Eiken Chemical Co, Japan), which has a reported lower limit of detection at 50 ng hb/mL buffer. 6 The concentration reported from the laboratory is a numerical value ranging from 35 (incl. those below) to 1000 (incl. those above) ng hb/mL buffer. The threshold for positivity is set at 100 ng hb/mL buffer in the Danish screening programme. In this trial, we divided the participants into groups of faecal haemoglobin concentrations ranging from 100–249, 250–499 and above 499 ng hb/mL buffer.
Colon capsule endoscopy
The bowel preparation regimen before colon capsule endoscopy started 3 days prior to capsule ingestion. Participants drank a polyethylene glycol solution (13.8 g sachet, Movicol®) 3 and 2 days before, followed by a split-dose polyethylene glycol solution (1 + 1 litre, MoviPrep®) the night before and morning of the capsule ingestion. Forty-five to sixty minutes before capsule ingestion, participants received one tablet of prucalopride (2 mg, Resolor®). Prucalopride was added to the regimen following the interim analysis of the randomised trial.15,16 After capsule ingestion, the participants drank a sulphate-based solution (330 mL, Eziclen®) at each of three capsule signals. A bisacodyl suppository (10 mg) was administered if the capsule had not been excreted by signal four. The capsule ingestion was supervised by experienced colon capsule endoscopy nurses and the PillCam™ COLON 2 capsules (Medtronic, Minneapolis, MN, US) were used. The videos from colon capsule endoscopy were reviewed by experienced readers using the PillCam™ Web Software (Given Imaging Inc., USA). The readers were blinded to faecal haemoglobin concentration. The readers delivered a report describing bowel cleanliness as well as images and size estimation of each detected polyp. A doctor from the clinical team then reviewed the report and determined whether to refer the patient for a colonoscopy based on the risk stratification (described below). Inter-observer agreement was not continuously monitored throughout the trial. However, we did test inter-observer agreement for landmark identification between the team of readers and internal readers of different experience levels afterwards on an image level, 18 and for bowel cleansing assessment using an artificial intelligence algorithm for whole investigations. 19 In those comparisons, substantial inter-observer variations were found, which is common in capsule reading.20,21
Risk classification and re-investigation
The Danish colorectal cancer screening applies the risk classification for surveillance after polypectomy as suggested by the European guidelines for quality assurance in colorectal cancer screening and diagnosis.22,23 For this current trial, the risk classification was simplified since histology is not available from colon capsule endoscopy, but only size and number of polyps. Therefore, the sizes of polyps were estimated by the readers using PillCam™ Web Software (Given Imaging Inc., USA), and no indications of neoplasia were available. The simplified risk classifications applied in this study were:
- Level one: no findings
- Level two: less than three polyps, all smaller than 10 mm in size
- Level three: three to four polyps, any size or at least one polyp 10–19 mm in size
- Level four: More than four polyps, or at least one polyp larger than 19 mm in size
Participants were referred for re-investigation after colon capsule endoscopy in case of level three or four risk.
Statistical analyses
The differences in re-investigation rate between faecal haemoglobin concentration subgroups were tested using the chi-square test. The odds ratios for re-investigation were estimated using univariate and multivariate logistic regression models. The odds ratios for increased risk classification were estimated using univariate and multivariate ordinal logistic regression models. The multivariate models were adjusted for sex (male and female) and age (50–59, 60–69 and 70 years and up). Further, theoretical re-investigation rates were calculated relying exclusively on the size of the largest polyp for risk stratification, while ignoring the number of polyps, as proportions of colorectal polyps carry no potential for malignant transformation.5,24 Data management was performed using SAS 9.4 (SAS Institute Inc., SAS 9.4. Cary, NC, USA) and R statistical software package, version 1.4.1717 (R Core Team, Vienna, Austria). 25
Results
Within the sample of 2031 CCEs performed, 1 participant withdrew consent, and 617 investigations were incomplete, leaving 1413 complete investigations for inclusion (Figure 1). Male percentage was 54.4% (n = 768), and the median age was 62.9 years (IQR 55.2–68.3). Faecal samples with haemoglobin concentrations between 100 and 200 ng hb/mL buffer were the most common, and the frequency decreased with increasing concentration level until reaching a cluster of faecal immunochemical test results of 1000 ng hb/mL buffer or higher (Figure 2). The overall polyp detection rate was 78.8%. The polyp detection rates for any size polyps were 77.3%, 80.6% and 80.3% respectively, for faecal haemoglobin concentrations of 100–249, 250–499 and above 499 ng hb/mL buffer. The mean polyp count was similar between faecal haemoglobin concentration subgroups (Table 1). The polyp detection rate as well as the mean polyp count was higher in males compared to females and increased with age groups. Detailed polyp detection rates across polyp size thresholds and mean polyp counts are provided in Table 1.

Flow chart of colon capsule endoscopies including their risk classification and re-investigation rate.
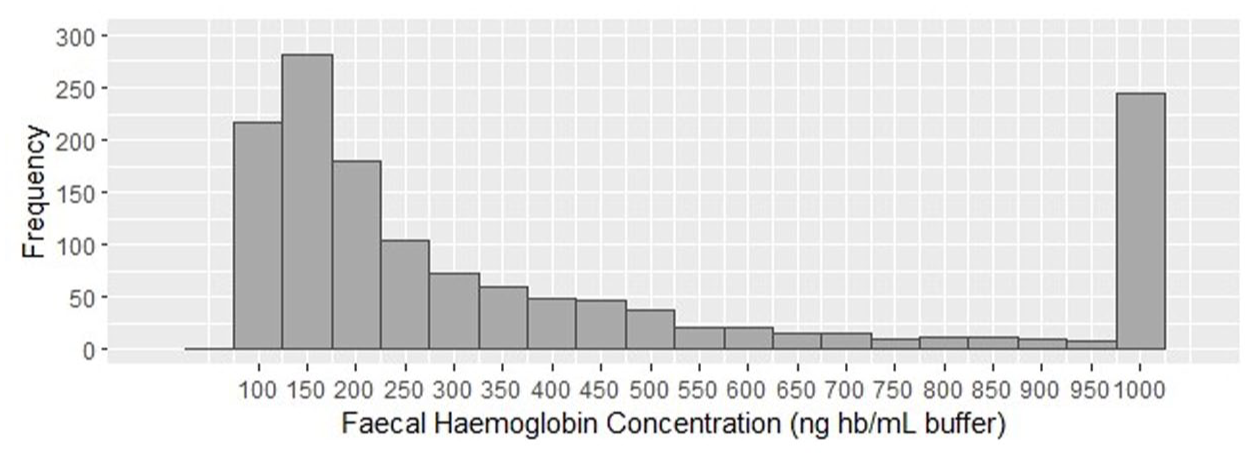
Distribution of faecal haemoglobin concentrations in participants undergoing complete colon capsule endoscopy, n = 1413.
Polyp detection rates for each polyp size threshold and mean polyp count per individual across faecal haemoglobin concentration, sex and age categories.

Re-investigation rate following complete colon capsule endoscopy
Level 1 or 2 findings were identified in 41.4% (n = 585) of the complete CCE investigations, returning the participant to the colorectal cancer screening. Level 3 or 4 findings were identified in 58.6% (n = 828) of the complete CCE investigations, resulting in referrals for follow-up colonoscopy (Figure 1). The re-investigation proportion increased with increasing faecal haemoglobin concentration from 58.2% to 61.1%, and 62.7% in the groups of 100–249, 250–499 and >499 ng hb/mL buffer (Figure 3). These differences in re-investigation proportions were not significantly different (p = 0.312).

Re-investigation rates by faecal haemoglobin concentration in complete colon capsule endoscopies, n = 1413.
The odds ratio for referral for re-investigation compared to 100–249 ng hb/mL buffer was 1.13 (CI 95% 0.86; 1.49, p = 0.391) for concentrations between 250 and 499 ng hb/mL buffer, and 1.21 (CI 95% 0.94; 1.56, p = 0.143) for concentrations above 499 ng hb/mL buffer. Adjustments for sex and age had little effect on these odds ratios as they changed to 1.12 (CI 95% 0.85; 1.49, p = 0.416) and 1.23 (CI 95% 0.95; 1.60, p = 0.109), respectively (Figure 4).

Odds of re-investigation based on faecal haemoglobin concentration estimated from univariate and multivariatea logistic regression models, n = 1413.
Limiting the risk classification from colon capsule endoscopy to rely exclusively on the size of the largest polyp would have reduced the re-investigation rate in the sample to 46.2%. As all incomplete colon capsule endoscopy investigations also resulted in referral for colonoscopy, the total re-investigation rate in the entire sample (n = 2030) was 72.2%.
Risk classification from colon capsule endoscopy
The proportions of colon capsule endoscopy investigations resulting in a level four classification were 28%, 26% and 30% across faecal haemoglobin concentration subgroups. Level three was evident in 29%, 33% and 31% across faecal haemoglobin concentration subgroups, whereas level two risk was seen in 21%, 21% and 19%, respectively. These differences in risk classification proportions were not significantly different (p = 0.653). When limiting the risk classifications to rely exclusively on size, the risks were downgraded overall. Especially, proportions of level four risk classification decreased to 9%, 11% and 17%, respectively, compared to the original 28%, 26% and 30% across faecal haemoglobin concentration subgroups (Figure 5).
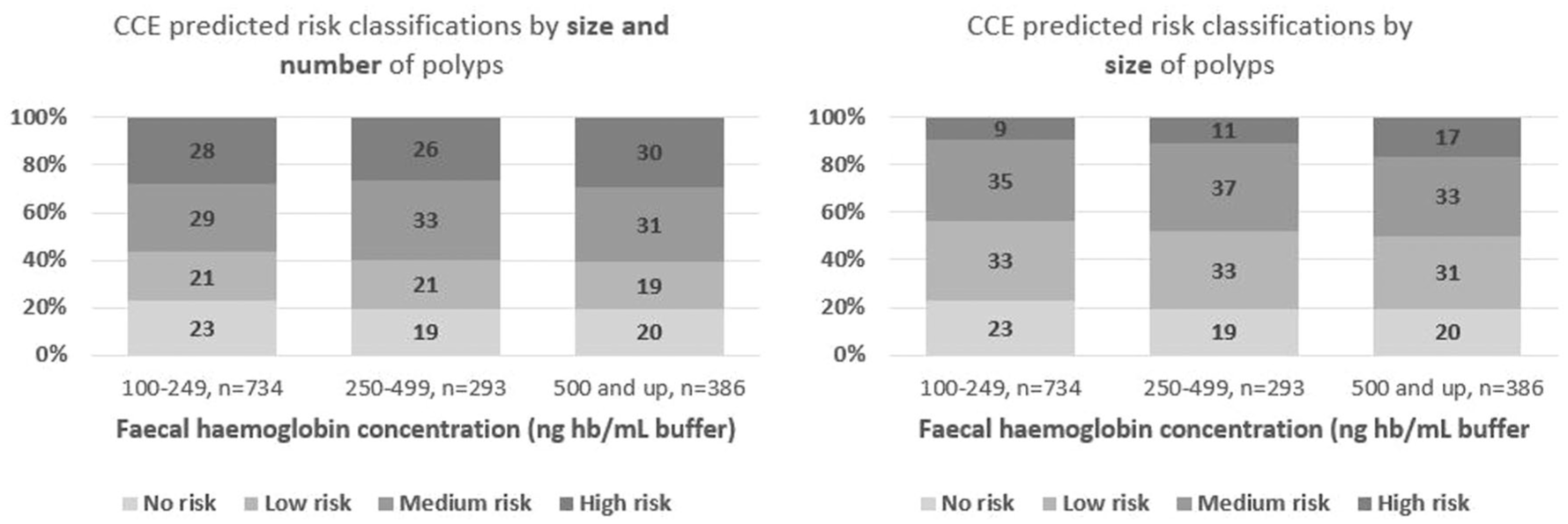
Distribution of colon capsule endoscopy predicted risk classifications within each faecal haemoglobin concentration stratum, n = 1413.
The odds ratio for increased risk classification (i.e. higher than level one) was 1.17 (CI 95% 0.92; 1.50, p = 0.199) for concentrations between 250 and 499 ng hb/mL buffer, and 1.36 (CI 95% 1.09; 1.71, p = 0.008) for concentrations above 499 ng hb/mL buffer, compared to 100–249 ng hb/mL buffer. Adjustments for sex and age had little effect on these odds ratios as they changed to 1.17 (CI 95% 0.92; 1.49, p = 0.211) and 1.40 (CI 95% 1.12; 1.77, p = 0.003), respectively (Figure 6).

Odds of increased risk classification based on faecal haemoglobin concentration estimated from univariate and multivariatea ordinal logistic regression models, n = 1413.
Discussion
We found that the re-investigation rate following complete colon capsule endoscopies did not differ significantly between subgroups of increasing faecal haemoglobin concentration, even though the risk classification did. The rate of investigations resulting in a referral for re-investigation was 58.4%, comparable to 63% in the Scottish symptomatic population and 72% in the Scottish surveillance population. 1 The polyp detection rate in the current study was 78.8%. This was comparable to other screening populations with polyp detection rates of 86.0% 4 and 77.2% 3 , while it was 74.4% in Scottish symptomatic patients.1,26
It seems there is a misalignment between the risk prediction made from colon capsule endoscopy and the actual risk of the findings, as the correlation between faecal haemoglobin and colorectal cancer and mortality has been well established.5–10 However, faecal haemoglobin concentration does not translate into risk of re-investigation in our sample. There is a general agreement that very few diminutive and small polyps bleed, and that the majority of those found in screening, should be considered as incidental findings in persons who have a positive faecal immunochemical test for other, non-neoplastic reasons. There is an increasing awareness internationally of the limited risk associated with leaving small polyps in situ, even though there are more of them simultaneously, although large trial evidence on this is still missing, since most small polyps are resected and their possible progression is therefore unknown. A large proportion is even resected and discarded.
As of now, it is not possible to determine the malignant potential of polyps in situ when performing a colon capsule endoscopy. Therefore, a large proportion of the detected polyps, utilised to predict risk, will be hyperplastic polyps and diminutive adenomas with no risk to the patient. The proportion of polyps that can safely be left in situ has been reported as high as 86.5% of rectosigmoid polyps <6 mm, while as much as 69% of all polyps are non-adenomatous. 24 As long as these polyps are still being included in the risk classification, the risk predictions being made by adopting the simplified classification model, as in this study, may severely overestimate the actual risk, compared to the classification determined by histology. On top of this, we know the sizes of the polyps are being overestimated in colon capsule endoscopies using the rapid reader software,27,28 and that colon capsule endoscopy holds a risk of double reporting of polyps, which may also result in substantial overestimation of risk. As a result of risk overestimation, large proportions of individuals undergoing colon capsule endoscopy may be referred for re-investigations such as colonoscopy, CT colonography or sigmoidoscopy, although the patient’s benefit from this is highly questionable. This greatly affects the cost-effectiveness of implementing colon capsule endoscopy as a filter test in colorectal cancer screening and adds additional discomfort and risk of complications to the patients. Prior simulation studies have already indicated the inferior cost-effectiveness of colon capsule endoscopy screening, and the re-investigation rate is a major contributor to this.29,30 Faecal haemoglobin concentration, sex and age are all readily available information in screening participants who have provided a faecal sample. However, other characteristics would also be of relevance to try to predict the risk of re-investigation, such as those associated with pathology (e.g. tobacco use and family history of colorectal pathology). This should be investigated further, and modelling using artificial intelligence may additionally improve risk predictions, but in order to easily implement these predictions as decision tools for triaging, it would be simpler to rely on information already available rather than requiring patient-reported information.
The development of artificial intelligence algorithms that are able to classify the polyps in situ, and thereby their inherent risk of progression to colorectal cancer, may offer a solution.31–34 If such algorithms prove continuously safe, accurate and reliable, an adoption of them into clinical use may be realistic and could leave current risk predictions, relying on size and number of polyps, obsolete. As the current study describes a secondary outcome from a randomised controlled trial, no formal sample size calculation was performed. However, the sample size derived remains one of the largest colon capsule endoscopy cohorts recruited. Another limitation is the nature of the sample, which is from a screening population. Other patterns of re-investigation might have been revealed below the threshold for a positive faecal immunochemical test in the Danish screening programme.
Conclusion
Faecal haemoglobin concentration did not prove to be an optimal selection parameter for diagnostic modality in a colorectal cancer screening population, although it was significantly associated with an increased risk classification from colon capsule endoscopy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261434425 – Supplemental material for Re-investigation rate following colon capsule endoscopy stratified by faecal haemoglobin concentration in a screening population
Supplemental material, sj-docx-1-tag-10.1177_17562848261434425 for Re-investigation rate following colon capsule endoscopy stratified by faecal haemoglobin concentration in a screening population by Ulrik Deding, Benedicte Schelde-Olesen, Thomas Bjørsum-Meyer, Lasse Kaalby, Issam Al-Najami, Anastasios Koulaouzidis, Morten Kobaek-Larsen and Gunnar Baatrup in Therapeutic Advances in Gastroenterology
