Abstract
Background:
The usefulness of thiopurines has been poorly explored in pouchitis and other pouch disorders.
Objective:
To evaluate the effectiveness and safety of azathioprine as maintenance therapy in inflammatory pouch disorders.
Design:
This was a retrospective and multicentre study.
Methods:
We included patients diagnosed with inflammatory pouch disorders treated with azathioprine in monotherapy. Effectiveness was evaluated at 1 year and in the long term based on normalization of stool frequency, absence of pain, faecal urgency or fistula discharge (clinical remission), or any improvement in these symptoms (clinical response). Endoscopic response was evaluated using the Pouchitis Disease Activity Index (PDAI).
Results:
In all, 63 patients were included [54% males; median age, 49 (28–77) years]. The therapy was used to treat pouchitis (n = 37) or Crohn’s disease of the pouch (n = 26). The rate of clinical response, remission and non-response at 12 months were 52%, 30% and 18%, respectively. After a median follow-up of 23 months (interquartile range 11–55), 19 patients (30%) were in clinical remission, and 45 (66%) stopped therapy. Endoscopic changes were evaluated in 19 cases. PDAI score decreased from 3 (range 2–4) to 1 (range 0–3). In all, 21 patients (33%) presented adverse events and 16 (25%) needed to stop therapy.
Conclusion:
Azathioprine may be effective in the long term for the treatment of inflammatory pouch disorders and could be included as a therapeutic option.
Introduction
Restorative proctocolectomy with ileal pouch–anal anastomosis remains the gold-standard procedure for the surgical treatment of ulcerative colitis (UC).1,2 Pouchitis is a non-specific inflammatory disorder that affects some pouch patients. While its aetiology remains unknown, it has been associated with risk factors such as the previous presence of inflammatory bowel disease (IBD).3–5 In fact, pouchitis is the most frequent non-mechanical complication in pouch patients with UC [prevalence, 29% (8–41%)].4–8
Clinical manifestations include increased stool frequency, faecal urgency, bloody diarrhoea and abdominal pain. Pouchitis can manifest as an acute, recurrent or chronic disease that interferes with quality of life. 7 Inflammatory pouch disorders other than pouchitis, such as cuffitis and Crohn’s disease (CD) of the pouch, may also appear.8–10 Cuffitis involves inflammation of the rectal cuff and resembles ulcerative proctitis. 11 CD of the pouch is a heterogeneous entity that includes inflammation of the pouch and/or the afferent ileal limb and may give rise to complications such as the stricturing and fistulizing phenotypes beyond 6–12 months after pouch surgery.12,13 CD of the pouch could be present in up to 25% of pouch patients.13–16
The therapeutic approach to inflammatory pouch disorders has been poorly explored, and many recommendations are based on non-controlled studies.17–20 The recommended treatment of pouchitis, cuffitis and CD of the pouch includes antibiotics,21–23 mesalamine, oral budesonide 24 and biological therapy.25–29 However, the absence of studies on pouchitis and other inflammatory pouch disorders treated with immunosuppressants such as azathioprine means that these drugs are not included in the therapeutic arsenal despite their known effectiveness and role as steroid-sparing agents in IBD. Given the unmet needs in the management of inflammatory pouch disorders, it would be interesting to know the role of azathioprine in this scenario.
Therefore, the main objective of this study was to assess the long-term effectiveness of azathioprine prescribed for pouch inflammatory disorder treatment. We also assessed endoscopic findings, duration of treatment, safety and long-term outcomes.
Methods
Study design and endpoints
This was an observational, retrospective and multicentre nationwide study undertaken by the Young Members Group of the Spanish Working Group on Crohn’s Disease and Ulcerative Colitis (GETECCU).
We selectively identified pouch patients aged 18 years or older who had undergone surgery between 1995 and 2020. All patients had been diagnosed with UC (resection specimen with compatible histology) and subsequently with inflammatory pouch disorders (pouchitis, CD of the pouch or cuffitis) following the diagnostic criteria of the European Crohn’s and Colitis Organization, GETECCU and the International Ileal Pouch Consortium (IIPC).9,10,15 The conditions were being treated with azathioprine in monotherapy based on clinical decisions. Pouchitis was classified according to the criteria of the IIPC (acute, chronic, recurrent or antibiotic-responsive). 15 We excluded patients who received these therapies for other clinical indications (e.g. extraintestinal manifestations or as part of a solid organ transplant immunosuppressant regimen), patients who were taking azathioprine in combination with biological therapy and patients who were lost to follow-up after initiation of therapy.
The demographic and clinical characteristics of the patients were recorded based on the Montreal and the IIPC classification. Perianal fistulas, strictures and penetrating complications were recorded if they appeared 6–12 months after stoma closure. We also recorded the presence of extraintestinal manifestations (articular, ocular, cutaneous, hepatic and other).15,30 Prior treatments and the indication for azathioprine therapy were also collected.
Participants were followed until withdrawal of therapy or their last clinical visit.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 31
Definitions
Effectiveness was evaluated using clinical definitions. Clinical remission was defined as the normalization of stool frequency (this was the recovery of basal stool frequency) with the absence of abdominal pain, rectal bleeding, faecal urgency or cessation of fistula drainage. Clinical response was defined as any improvement in these parameters (over clinical worsening) without remission. Non-response was defined as no change in symptoms.25–29 This evaluation was used for pouchitis and CD of the pouch. We only considered that patients had experienced a clinical response and remission provided they were not receiving concomitant steroid therapy. Changes in endoscopic activity were evaluated (only for patients with baseline endoscopy assessment) using the endoscopic sub-score of the modified Pouchitis Disease Activity Index (PDAI). 32
Effectiveness was evaluated at 12 months and to the maximum follow-up. Treatment was discontinued in the case of non-response, loss of remission/response, adverse events, maintained remission and patient decision.
Finally, we also analysed the persistence of therapy defined as the time under active treatment.
Statistical analysis
In the descriptive analysis, categorical variables were expressed as absolute and relative frequencies. Quantitative variables were expressed as the mean and standard deviation (SD) or as the median and interquartile range (IQR) when they were not normally distributed. In the univariate analysis, categorical variables were compared using the chi-square test, and quantitative variables were compared using the appropriate test depending on the normality of the distribution. Factors that were found to be significantly associated with clinical response and remission were further explored in a multivariate logistic regression analysis with the odds ratio and 95% confidence interval. Persistence of therapy was assessed by the Kaplan–Meier method whereby patients in whom therapy was discontinued for any reason were right-censored at the time of discontinuation. Variables were included in the analysis if their p value was <0.1. Statistical significance was set at p < 0.05 for the rest of the statistical analysis. The analysis was performed using IBM SPSS Statistics for Windows, Version 24.
Results
Patient baseline characteristics
The cohort of the RESERVO study comprised 338 patients with inflammatory pouch disorder from 46 centres in Spain. A total of 93 patients from this cohort (27%) were treated with immunosuppressants. We excluded 25 patients who started immunosuppressants as combination therapy with biologics and 5 treated with other immunosuppressants (cyclosporine, methotrexate and tacrolimus). After applying the inclusion/exclusion criteria, the final study cohort consisted of 63 patients (Figure 1).

Flowchart of population included.
The main demographic and disease-related characteristics of the study patients are provided in Table 1. Before therapy, 59 (94%) of participants had received antibiotics; of these, 65% were refractory or dependent. In all, 28 (46%) patients received probiotics (failure in 43%, recurrence in 13%), 37 (64%) rectal and/or oral mesalamine (38% without response) and 30 (50%) oral and/or topical steroids (29% were refractory, 42% developed dependence and 2 stopped due to adverse events). No patients received biological therapy before azathioprine for the treatment of their pouch disorders.
Demographic and disease-related characteristics of patients treated with azathioprine.

Acute (<4 weeks of symptoms), chronic (⩾4 weeks of symptoms), recurrent and antibiotic dependent (⩾3–4 episodes per year). 15
B1, inflammatory; B2, stricturing; B3, penetrating.
Treatment characteristics
The indications for azathioprine therapy were a CD of the pouch (25, 40%), recurrent pouchitis (20, 32%) and antibiotic-refractory pouchitis (17, 27%). None of the patients who received this treatment presented cuffitis.
As previously mentioned, 50% of patients had previously been exposed to steroids, and they experienced refractoriness or dependence in 29% and 42%, respectively. Therapy was started at a median of 23 months (IQR 7–190 months) after the diagnosis of pouch disorder and this was longer for patients diagnosed with CD of the pouch (median, 89 months, IQR 25–205, difference 66 months, IQR 15–112 (p = 0.002).
Effectiveness of azathioprine
A total of 63 patients received azathioprine as monotherapy. The standard dose used was 2.5 mg/kg/day. At 12 months, 19 patients (30%) were in clinical remission and 33 (52%) had achieved a clinical response. In all, 11 patients (18%) achieved no response.
No differences in clinical response, clinical remission or non-response at 1 year were detected for various indications (recurrent pouchitis versus antibiotic-refractory pouchitis versus CD of the pouch). The only factor associated with clinical response and/or remission in the univariate analysis was steroid-dependent disease as the indication for azathioprine (85% versus 15%, p = 0.01) (Table 2). Therefore, the multivariable analysis was not performed.
Factors associated with clinical response and remission at 1 year: results of univariate analysis.
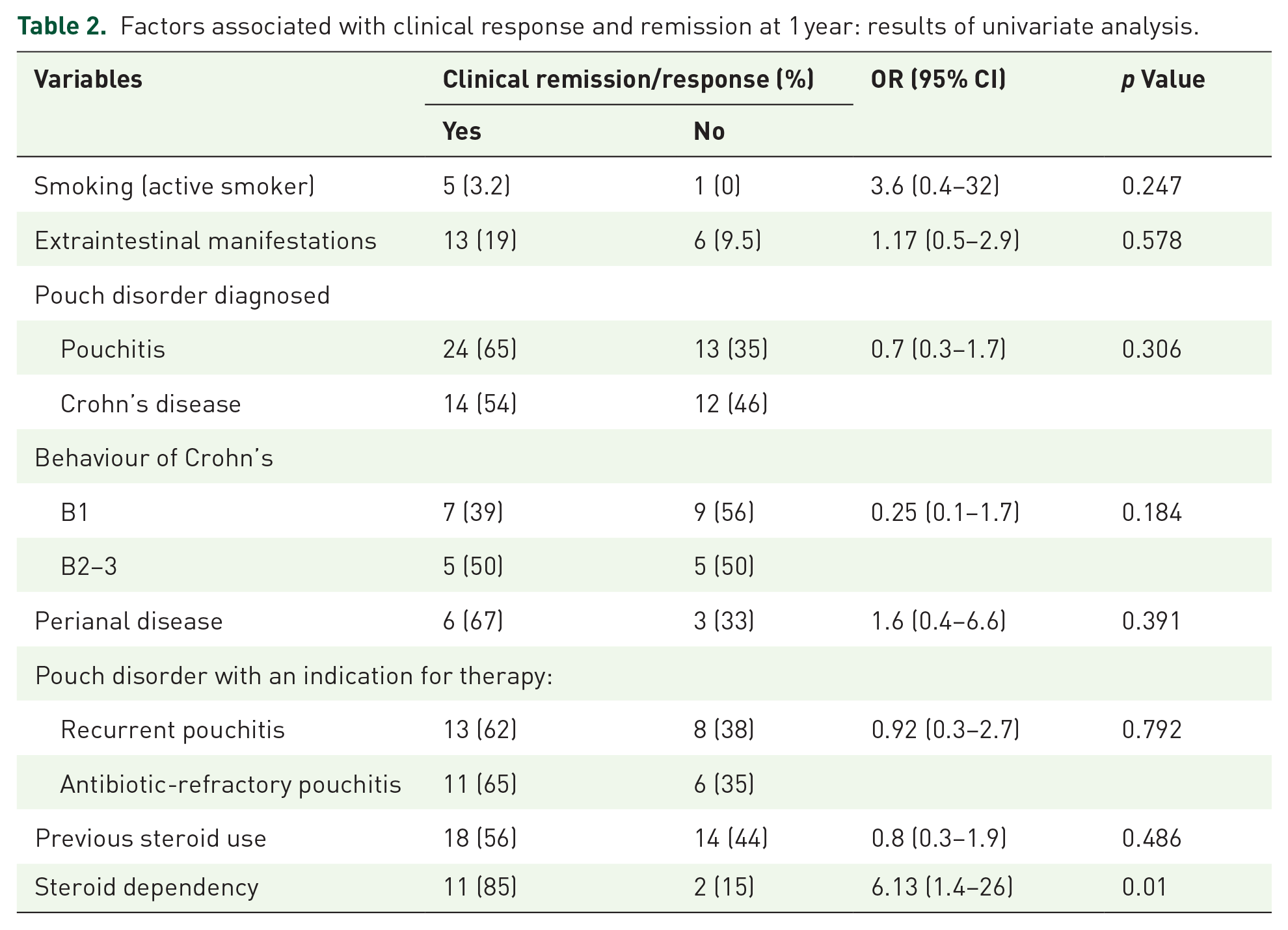
In long term, after a median follow-up of 23 months (IQR 11–55) from the start of azathioprine, 19 patients (30%) were in clinical remission, 21 (33%) had achieved a clinical response and 12 (19%) who had initially experienced a clinical response/remission lost their response.
Finally, the persistence of azathioprine is represented in Figure 2. The percentage of patients who maintained treatment was 66%, 59% and 48% at 1, 2 and 3 years.

Persistence of azathioprine, survival analysis.
A total of 42 (66%) patients stopped therapy due to failure [non-response/remission or loss of response; 20 (47.6%)], adverse events (16, 38%) and sustained remission (6, 14%).
Re-evaluation of endoscopic activity
Endoscopic activity was re-evaluated in 19 patients (30%). After a median 9 months of endoscopic follow-up, the median PDAI endoscopic sub-score dropped from 3 (range 2–5) to 1 (range 0–5) (p = 0.020) (Figure 3). Histological data were not systematically obtained and could not be analysed.

Endoscopic activity changes.
Safety
A total of 21 patients (33%) experienced adverse events secondary to azathioprine (Table 3). Finally, 16 (25%) patients were forced to stop therapy due to adverse effects.
Adverse events associated with azathioprine (N = 63).

Ovarian neoplasm, leukaemia.CMV, citomegalovirus.
Duration and outcomes
The median follow-up was 23 months (IQR 11–55). A total of 42 patients (66%) suspended therapy due to failure (20, 32%), adverse events (16, 25%) and sustained remission (6, 9%). After treatment withdrawal, 6 patients received another immunosuppressant, and 36 patients started biological therapy. Nevertheless, 22 patients needed pouch surgery owing to treatment refractoriness and, finally, 16 required a permanent ileostomy.
Discussion
To our knowledge, this nationwide study comprises the largest series to date of pouch patients with IBD and subsequent inflammatory pouch disorders (mainly chronic pouchitis) treated with azathioprine. One in four patients from the RESERVO study used this drug as a therapeutic option, even before biological therapy, resulting in an effective long-term strategy in around two-thirds of patients. However, half of them interrupted therapy for several reasons.
Various pharmacologic options are available for the management of inflammatory pouch disorders. However, published data are often scarce, and the strongest recommendations are based on antibiotic and/or biological therapy.9,18,19 Although immunosuppressants such as thiopurines and methotrexate have proven effective in the management of CD and UC, these drugs have been poorly explored in the case of pouch disorders, although they may be more widely used than previously thought. 33 To date, only two studies have evaluated the effectiveness of thiopurines in CD of the pouch (two case reports and a short clinical series of eight patients).34,35 Based on this evidence, GETECCU 18 recommended against the use of immunosuppressants in the case of chronic refractory pouchitis. However, clinical experience with thiopurines has been reported, since many studies that have evaluated the effectiveness of biological therapy in pouch disorders included patients previously exposed to thiopurines.25,26,28,29,36 Consensus guidelines from the IIPC are also based on limited evidence in favour of immunosuppressants for chronic pouchitis and CD of the pouch; nevertheless, the authors suggest the potential use of immunosuppressants (thiopurines) as monotherapy with a low grade of evidence, supported mainly by expert opinion. 19 This is supported as thiopurines are used in IBD, an entity that shares many characteristics with pouch inflammatory disorders. 37
Our results show that azathioprine seems an interesting additional option in patients with inflammatory pouch disorders even in the biological era (in this series, more than 60% of patients received this treatment after 2010, data not shown). We found this therapy was associated with clinical response and remission during the first year in more than 80% of cases and with clinical remission in the long term in 30% of cases. The profile of patients in this study includes those whose previous treatment (antibiotics, mesalamine, probiotics and steroids) had failed. It is important to highlight that azathioprine was chosen as a therapeutic option first than biological therapy. This was a clinical decision and it could be due to several reasons such as clinical experience with thiopurines or the scarce and limited evidence of biological therapy in previous decades among others.
Azathioprine was especially useful for previously steroid-dependent patients, with 84% long-term steroid-free clinical remission and response. However, two out of three patients needed to stop therapy in the long term owing to non-response (32%) or adverse events (25%), and more than half switched to biological therapy.
Our findings lead us to ask how to include and position thiopurines in the treatment algorithm of inflammatory pouch disorders. However, thiopurines are not an appropriate choice for inducing remission. Biological therapy is a good therapeutic option in patients with chronic refractory pouchitis and CD of the pouch.25–29,33,36,38 One meta-analysis reported that clinical remission after induction of anti-TNF agents was more common in CD than in chronic pouchitis (64% versus 10%). 27 Nonetheless, our data show that they would be a useful alternative long-term option, especially for patients with previous exposure to steroids who develop dependence (85% versus 15%, p = 0.01). Considering effectiveness and safety data, the different maintenance therapeutic options should be discussed and balanced with the patient.
The patients in our cohort achieved similar clinical benefits with azathioprine, irrespective of the type of inflammatory pouch disorder [chronic recurrent (62%), refractory (64%) and CD of the pouch (60%)]. In their single-centre study of 21 patients with CD of the pouch, Haveran et al. reported infliximab and/or azathioprine to be effective. 34 Eight patients received thiopurines in monotherapy, and the success of treatment was defined as complete resolution of or significant improvement in symptoms without requiring ileostomy. Treatment proved successful for all patients after a median of 38 months. This excellent result may be because physicians selected patients who received thiopurines in monotherapy instead of biological therapy or, even, combination therapy.
Finally, safety is one of the most relevant factors in therapy with azathioprine. In our study, 33% of patients presented adverse events, mainly affecting the haematological and digestive systems. More than three out of four were forced to stop therapy because of the event. These results resembled those reported in the literature and could limit the use of this therapy in many cases. 39 However, there are several strategies to optimize the management of azathioprine (some of them to improve tolerance) that have not been analysed in this study and could related to the high rate of discontinuation. We did not evaluate whether patients were exposed to azathioprine before colectomy or whether they had tolerated treatment previously.
This study is subject to a series of limitations. First, the design was retrospective and the number of patients included was limited to obtain firm conclusions. Nevertheless, pouchitis and other pouch inflammatory disorders are uncommon diseases within IBD. Second, baseline clinical activity and effectiveness were not evaluated using scores such as the PDAI or modified PDAI: the retrospective nature of this study meant that we used a less standardised definition, as analysed in many previously published studies.25–29 Moreover, none of these activity indices have previously been validated. A recent expert consensus group used a RAND/UCLA process to help clinicians and investigators assess the activity of pouchitis based on clinical factors such as stool frequency, faecal urgency and endoscopic and histological activity. 40 In our cohort, 19 patients (30%) were also evaluated using endoscopy, which revealed a decrease in the PDAI sub-score. On the other hand, ours is the first publication to date to report the effectiveness of azathioprine in inflammatory pouch disorders and to include a relevant number of patients belonging to a large multicentre and nationwide cohort affected by a rare condition within IBD. Furthermore, it enabled us to explore future options for this complex scenario.
In conclusion, the therapeutic arsenal for inflammatory pouch disorders remains open to various pharmacologic options. Azathioprine would also be a feasible maintenance therapeutic option for these disorders, especially in the case of steroid dependence. Further studies may confirm our findings and position these old but effective drugs appropriately.
