Abstract
Background:
Pancreatic neuroendocrine tumors (pNETs) are rare; however, they are increasingly being detected. Although surgical resection remains the standard treatment, its invasiveness has prompted interest in less invasive alternatives, particularly for small non-functional pNETs (NF-pNETs) and insulinomas.
Objectives:
To evaluate the clinical efficacy and safety of endoscopic ultrasound-guided ethanol injection (EUS-EI) and radiofrequency ablation (EUS-RFA) for pNETs.
Design:
A systematic review and meta-analysis.
Data sources and methods:
A literature search of PubMed, MEDLINE, and Google Scholar was conducted (April 2005–April 2025). Studies were eligible if they reported clinical outcomes of EUS-EI or EUS-RFA in adult patients with insulinomas or NF-pNETs. The primary endpoints were clinical success (short-term symptom resolution or radiological response) and adverse event (AE) rates. Data were pooled using a random-effects model.
Results:
Twenty-six studies were included in the meta-analysis. For insulinomas, the pooled clinical success rate was 77% (95% confidence interval (CI), 59–88) for EUS-EI and 95% (95% CI, 89–97) for EUS-RFA. The pooled incidence of total AEs was 32% (95% CI, 17–51) for EUS-EI and 25% (95% CI, 15–39) for EUS-RFA. For NF-pNETs, the pooled clinical success rates were 76% (95% CI, 54–90) for EUS-EI and 85% (95% CI, 74–92) for EUS-RFA, and the pooled incidence of total AEs was 27% (95% CI, 20–35) and 26% (95% CI, 17–38), respectively. The most common moderate or severe AEs were pancreatitis in 12 patients (7.6%) after EUS-EI, and pancreatic fluid collection in 4 patients (1.9%) and pancreatic duct stricture in 3 patients (1.4%) after EUS-RFA. One fatal case occurred in a 97-year-old patient following EUS-RFA.
Conclusion:
Both EUS-EI and EUS-RFA are effective, relatively safe, and minimally invasive treatment options for pNETs. However, severe AE can occur, and careful patient selection and treatment indication are essential.
Trial registration:
Not registered.
Keywords
Introduction
Pancreatic neuroendocrine tumors (pNETs) are rare tumors. They account for approximately 1%–2% of all primary pancreatic malignancies. 1 Recently, their reported incidence has increased significantly, largely because of the widespread use of advanced endoscopic and radiological imaging techniques. 2 Functioning pNETs (F-pNETs), such as insulinomas, are often detected at an early stage because hormone secretion causes characteristic clinical syndromes that prompt diagnostic evaluation. Insulinomas are the most common type of F-pNETs and are typically small, benign lesions. 3 In contrast, non-functional pNETs (NF-pNETs) are usually asymptomatic, and when small, they are often detected incidentally during follow-up for other conditions or routine health checkups.
Treatment strategies for pNETs are determined based on the tumor size and the presence or absence of hormone-related symptoms.3–5 Surgical resection remains the standard of care, particularly for F-pNETs or large tumors. However, pancreatic surgery is associated with a relatively high rate of postoperative complications compared with other gastrointestinal procedures.3–7 Moreover, postoperative reductions in pancreatic endocrine and exocrine function may cause long-term issues such as diabetes or malabsorption.
The optimal management of small (<2 cm) and low-grade pNETs is an ongoing debate. Although surgery is recommended in current guidelines, its risks must be weighed against potential long-term complications.6,7 Minimally invasive local treatment options are attracting increasing interest. Among these, endoscopic ultrasound (EUS)-guided ablative therapies, including ethanol injection (EUS-EI) and radiofrequency ablation (EUS-RFA), have emerged as promising alternatives to surgery. 8 EUS-EI induces coagulative necrosis through the direct injection of ethanol into the tumor, whereas EUS-RFA involves using thermal energy to ablate the tumor tissue. Both treatments are primarily applied to small (<2 cm) low-grade pNETs confined to the pancreas. These approaches offer potential advantages, such as fewer complications and better preservation of pancreatic function than surgical resection.
Although several small case series have reported favorable outcomes with both techniques, high-quality evidence regarding their efficacy and safety remains limited. Therefore, we aim to conduct a systematic review and meta-analysis to evaluate the clinical success and adverse event (AE) profiles of EUS-EI and EUS-RFA for the treatment of insulinomas and NF-pNETs.
Methods
Search strategy
A bibliographic search of PubMed, MEDLINE, and Google Scholar databases was conducted on October 11, 2025, covering studies published between April 2005 and April 2025 using the following search string: “(“EUS” OR “EUS-guided” OR “Endosonography” OR “Endoscopic ultrasound”) AND (ablation OR ablative OR Radiofrequency OR ethanol) AND (neuroendocrine OR insulinoma OR endocrine).” Detailed search strategies for each database are provided in the Supplemental Table 1. Two independent reviewers (K.M. and Y.F.) scrutinized the titles and abstracts of the articles and eliminated duplicates. Prospective or retrospective studies, conference proceedings, and abstracts were assessed. In the case of overlapping publications on the same population, the main reference was included after excluding case reports and non-English manuscripts. A single reviewer (K.M.) finally selected the relevant articles after reading them.
Selection criteria
The studies included in this systematic review and meta-analysis were original studies that met the following inclusion criteria: (i) patients: adult patients with NF-pNETs or insulinoma; (ii) intervention: EUS-EI or EUS-RFA; (iii) comparator: none; and (iv) outcomes: for NF-pNETs, studies that reported radiological treatment response were included, whereas for insulinomas, studies that described the resolution of hypoglycemia were included. Only studies that reported the presence or absence of AEs were included. We excluded (i) case reports, (ii) studies not reporting any of the outcomes, and (iii) studies evaluating other locoregional ablative techniques (i.e., percutaneous ultrasound-guided ablation therapies).
Data extraction
For each study, the following data were collected: first author’s name, year of publication, study type, number of pNETs, type of pNETs (insulinoma or NF-pNET), patient age, tumor size, needle gauge, ethanol concentration, EUS-RFA power, type of EUS-RFA probe, number of ethanol ablation sessions, volume of ethanol injected during the first session, complete radiological response rate for NF-pNETs, clinical response rate for insulinomas, incidence and severity of AEs, mortality, and follow-up duration.
Outcome definitions
Clinical success was defined as the study-specified short-term resolution of hormone-related symptoms (i.e., hypoglycemia) in patients with insulinoma after EUS-EI or EUS-RFA. 8 In patients with NF-pNETs, clinical success was defined as the absence of enhancement within the tumor on contrast-enhanced computed tomography (CE-CT) image. 8 The AE rate was calculated as the number of AEs divided by the number of patients who underwent either EUS-EI or EUS-RFA. AEs were graded according to the American Society for Gastrointestinal Endoscopy guidelines. 9
Statistical analysis
The methodological quality and risk of bias of the included studies were assessed independently by two reviewers using the Newcastle–Ottawa Scale (NOS) for nonrandomized studies. The NOS evaluates three domains—Selection (0–4 points), Comparability (0–2 points), and Outcome (0–3 points)—with a total score ranging from 0 to 9. Discrepancies were resolved by consensus. The results of the quality assessment are summarized in Supplemental Table 2. Study outcomes were pooled using a random-effects model based on the DerSimonian–Laird test, and the results were expressed as proportions (%) and 95% confidence intervals (CIs). Heterogeneity was assessed using I2 tests: I2 <30%, 30%–60%, and >60% were considered low, moderate, and high levels of heterogeneity, respectively. Potential publication bias was verified through visual assessment using funnel plots. To address 0 or 100% event rates, a continuity correction of 0.5 was applied to the number of events and the total sample size before logit transformation. This allowed for the inclusion of studies with extreme proportions in the meta-analysis and assessment of publication bias. Pooled estimates were presented to two decimal places. For studies originating from the same institution with overlapping enrollment periods, the most recent and comprehensive report was included in the quantitative synthesis to avoid double-counting. Earlier reports from the same center were retained for descriptive purposes only. All analyses were performed using the R version 4.4.2 (R Foundation for Statistical Computing, Vienna, Austria). This systematic review and meta-analysis was not prospectively registered in PROSPERO or any other database.
Results
Study characteristics
A total of 176 full-text articles were identified through the literature search. After excluding 73 review articles and meta-analyses, 103 articles were selected for a full-text review. Subsequently, 24 studies were included in this review after excluding case reports (n = 43), letters (n = 9), books (n = 2), and studies that did not target pNETs or lacked sufficient detail (n = 27). The literature search process according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses reporting guidelines is summarized in Figure 1.

PRISMA flow diagram of the study.
Endoscopic ultrasound-guided ethanol injection
Insulinoma
Six studies involving 31 patients were included in the meta-analysis (Table 1). The median/mean age ranged from 47 to 75 years, and tumor sizes ranged from 9 to 17 mm. Needle gauges varied (22- or 25-gauge), and the ethanol concentrations ranged from 95% to 99%. Four studies reported multiple injection sessions with initial ethanol volumes ranging from 0.4 to 2.2 mL. The median follow-up periods after treatment ranged from 5 to 62 months.
Endoscopic ultrasound-guided ethanol ablation therapy for insulinomas.

The age, diameter, percentage of clinical success rate, and median follow-up duration were rounded to the nearest whole number.
n/a, not available.
The pooled clinical success rate was estimated at 77% (95% CI, 59–88), with no heterogeneity among studies (I2 = 0%). The pooled total AE rate was estimated at 32% (95% CI, 17–51), with no heterogeneity among studies (I2 = 0%). Moderate or severe AEs were rare; only one case of moderate pancreatitis treated conservatively was reported, and the pooled incidence of moderate/severe AEs was 10% (95% CI, 4%–26%; Figure 2).

Forest plot showing pooled rates of clinical success (a), total AEs (b), and moderate or severe AEs (c) after EUS-guided ethanol injection for insulinomas.
Non-functional neuroendocrine tumor
Six studies involving 171 patients were extracted (Table 2). The median/mean age ranged from 54 to 75 years, and tumor sizes ranged from 10 to 13 mm. The ethanol concentrations ranged from 99% to 100%, and five studies reported multiple sessions using ethanol volumes of 0.9–3.1 mL. The median follow-up periods after treatment ranged from 9 to 26 months. Because three studies were conducted at the same institution with overlapping enrollment periods, a total of 127 patients from three studies were included in the meta-analysis.
Endoscopic ultrasound-guided ethanol ablation therapy for pancreatic non-functional neuroendocrine tumors.

The age, diameter, percentage of clinical success rate, and median follow-up duration were rounded to the nearest whole number. Four studies originated from the same institution with overlapping enrollment periods (Refs. 12, 13, 16, 18). To avoid double-counting, the most recent and comprehensive report (Ref 16) was used in the pooled analysis.
1:1 volume ratio injection of ethanol and lipiodol.
Thirty-three patients with 40 lesions were evaluated.
IQR, interquartile range; n/a, not available; PD, pancreatic duct; WON, wall off necrosis.
The pooled clinical success rate was 76% (95% CI, 54–90) with moderate heterogeneity (I2 = 58%). The pooled total AE rate was estimated at 27% (95% CI, 20–35), with no heterogeneity among studies (I2 = 0%). Regarding moderate or severe AEs, there were 10 cases of pancreatitis, one case of pancreatic duct (PD) stricture, and one case of walled-off necrosis (WON). PD plastic stenting was performed for PD strictures, and EUS-guided drainage was performed for WON. All the patients were successfully managed with conservative and endoscopic treatments. The pooled incidence rate for moderate or severe AEs was estimated to be 11% (95% CI, 7–18), with no heterogeneity (I2 = 0%; Figure 3).

Forest plot showing pooled rates of clinical success (a), total AEs (b), and moderate or severe AEs (c) after EUS-guided ethanol injection for non-functional pancreatic neuroendocrine tumors.
Publication bias was assessed using funnel plots (Supplemental Figure 1). Funnel plot asymmetry suggesting potential publication bias was observed for total AE rates in both the insulinoma (panel b) and NF-pNET (panel e) groups. Clinical success rates (panels a and d) and moderate or severe AE rates (panels c and f) appeared largely symmetrical.
Endoscopic ultrasound-radiofrequency ablation
Insulinoma
Eight studies involving 136 patients were included in the meta-analysis (Table 3). The median/mean age ranged from 45 to 81 years, and the tumor sizes ranged from 11 to 17 mm. Most studies used 19-gauge RFA needles, and the power settings ranged from 10 to 50 W. The median follow-up period after treatment ranged from 6 to 42 months.
Endoscopic ultrasound-guided radiofrequency ablation for insulinomas.
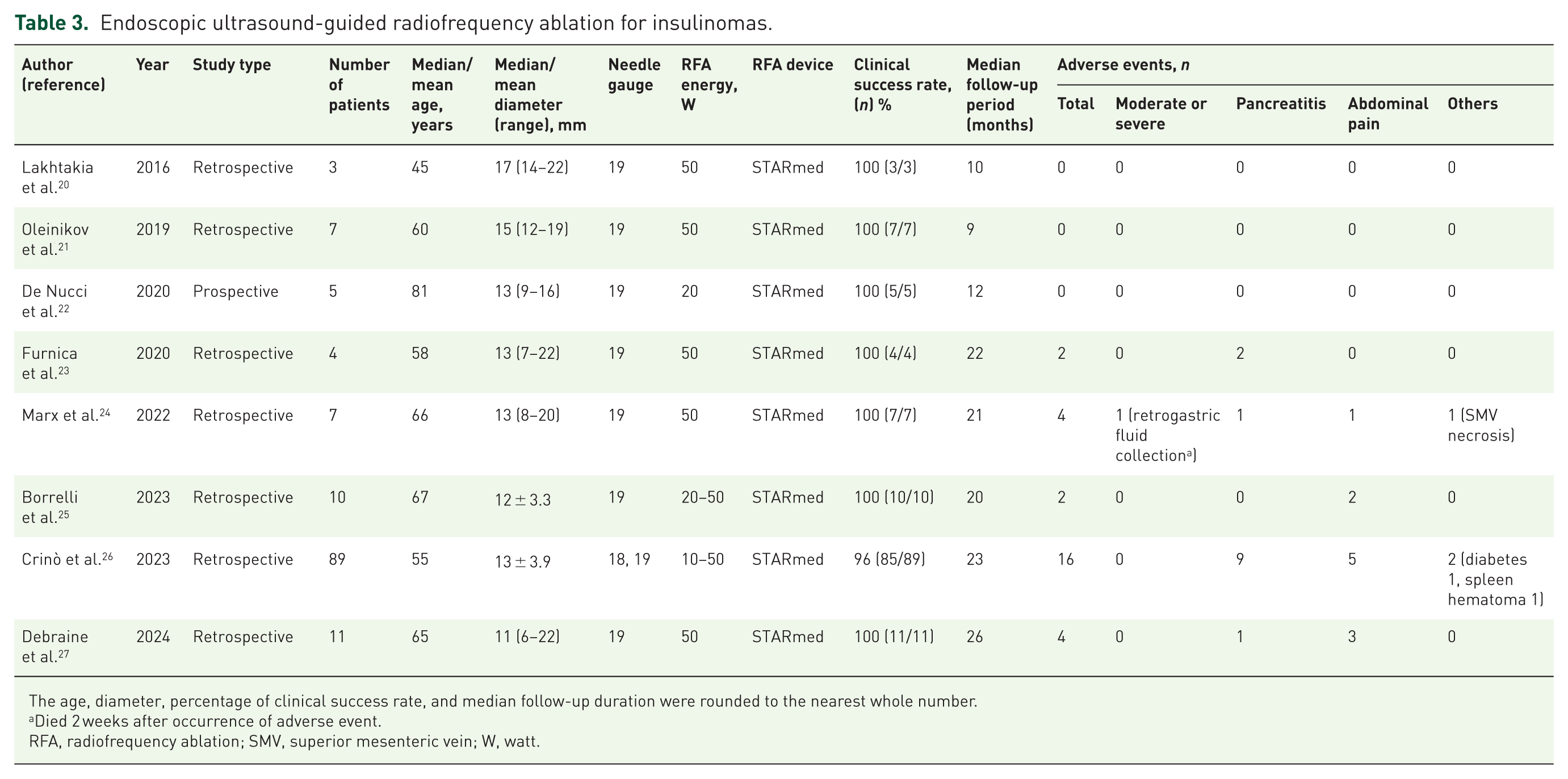
The age, diameter, percentage of clinical success rate, and median follow-up duration were rounded to the nearest whole number.
Died 2 weeks after occurrence of adverse event.
RFA, radiofrequency ablation; SMV, superior mesenteric vein; W, watt.
The pooled clinical success rate was estimated at 95% (95% CI, 89–97), with no heterogeneity among studies (I2 = 0%). The pooled total AE rate was estimated at 25% (95% CI, 15–39), with low heterogeneity among the studies (I2 = 27%). One severe AE was reported. A 97-year-old patient developed retrogastric fluid collection 2 weeks after EUS-RFA and declined further intervention. Despite receiving the best supportive care, the patient died 2 weeks later. The pooled incidence rate for moderate or severe AEs was estimated to be 6% (95% CI, 2–15), with no heterogeneity (I2 = 0%; Figure 4).

Forest plot showing pooled rates of clinical success (a), total AEs (b), and moderate or severe AEs (c) after EUS-guided radiofrequency ablation for insulinomas.
Non-functional-pancreatic neuroendocrine tumors
Eight studies involving 75 patients were included in the meta-analysis (Table 4). The median/mean age ranged from 62 to 77 years, and the tumor diameter ranged from 9 to 28 mm. Most studies used 19-gauge needles and power settings between 10 and 50 W. The median follow-up period after treatment ranged from 6 to 16 months.
Endoscopic ultrasound-guided RFA for non-functional pancreatic neuroendocrine tumors.

The age, diameter, percentage of clinical success rate, and median follow-up duration were rounded to the nearest whole number.
Twelve patients with 14 lesions were evaluated.
Two patients were not evaluated.
PD, pancreatic duct; RFA, radiofrequency ablation; W, watt.
The pooled clinical success rate was estimated at 85% (95% CI, 74–92), with no heterogeneity among studies (I2 = 0%). The pooled total AE rate was estimated at 26% (95% CI, 17–38), with no heterogeneity (I2 = 0%). Two cases of PD stricture, two cases of retrogastric fluid collection, and one case of pancreatic necrosis were observed. PD plastic stenting was performed for PD strictures, and EUS-guided drainage was performed for retrogastric fluid collection. All the patients were successfully managed with conservative and endoscopic treatments. The pooled incidence rate for moderate or severe AEs was estimated to be 10% (95% CI, 5–19), with no heterogeneity (I2 = 0%; Figure 5).
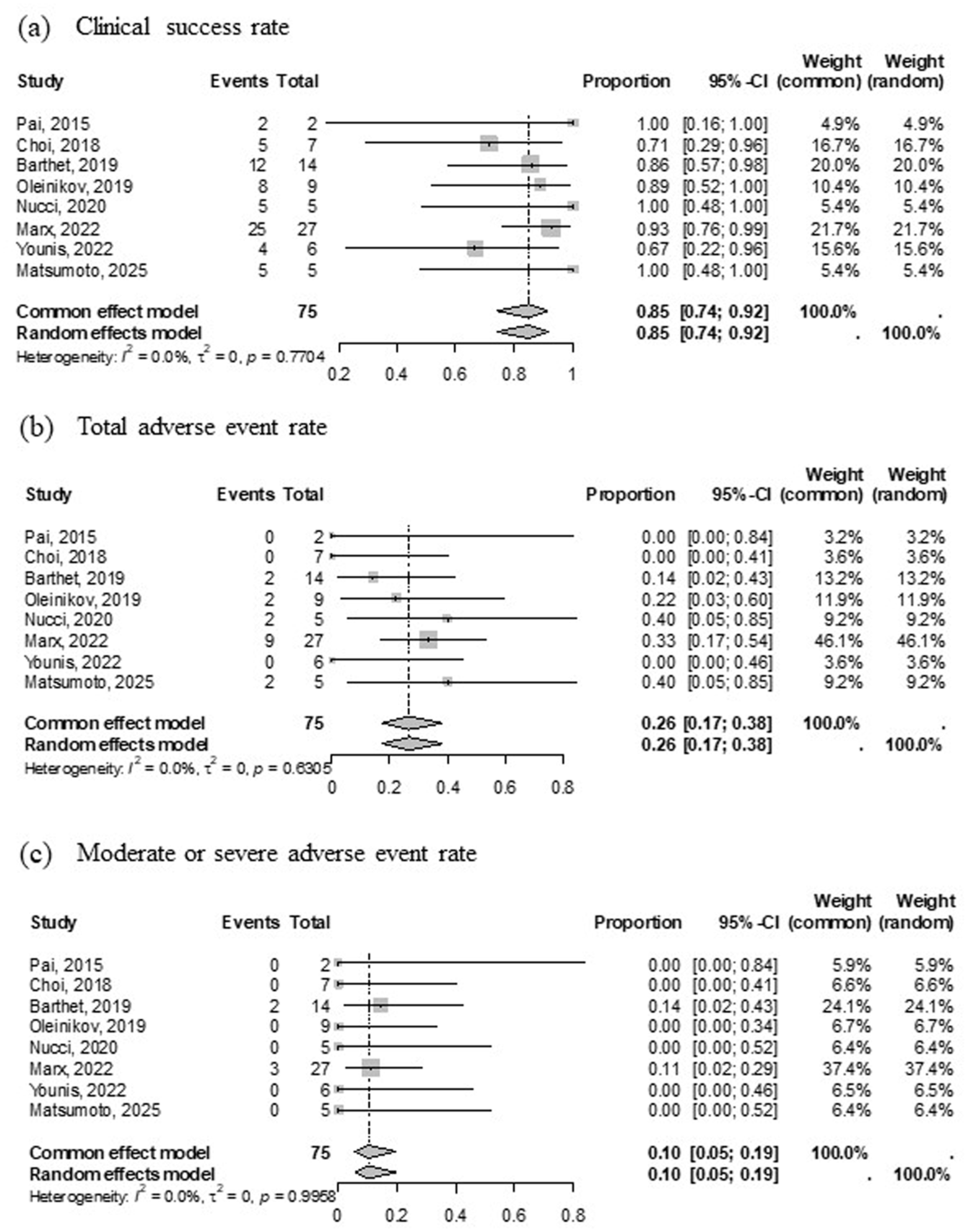
Forest plot showing pooled rates of clinical success (a), total AEs (b), and moderate or severe AEs (c) after EUS-guided radiofrequency ablation for non-functional pancreatic neuroendocrine tumors.
Publication bias was assessed using funnel plots (Supplemental Figure 2). Funnel plot asymmetry suggesting potential publication bias was observed for total AE rates in both insulinoma (panel b) and NF-pNET (panel e) groups. No obvious asymmetry was detected for clinical success rates (panels a and d) or moderate/severe AE rates (panels c and f).
Discussion
This systematic review and meta-analysis evaluated the efficacy and safety of EUS-EI and EUS-RFA for treating pNETs, including insulinomas and NF-pNETs. Pooled data suggest that both EUS-EI and EUS-RFA are promising minimally invasive therapies. Although both modalities demonstrated favorable clinical outcomes, their efficacy and safety profiles may vary depending on tumor functionality. Endoscopic approaches may be particularly beneficial for elderly patients, those with significant comorbidities, and those who are unsuitable or unwilling to undergo surgery.
Although the pooled clinical success for EUS-RFA in insulinomas reached 95% (95% CI, 89–97), this estimate primarily reflects short-term outcomes. Crinò et al. 26 compared 89 patients with insulinoma treated using EUS-RFA and 89 treated surgically, using propensity score matching to adjust for baseline characteristics. Clinical efficacy, defined as the resolution of hypoglycemic symptoms, was 100% in the surgery group and 95.5% in the EUS-RFA group (p = 0.160), indicating a comparable effectiveness between the two modalities. However, symptom recurrence was reported in 15 patients (16.9%) after a mean follow-up of 9.5 months. Among them, 11 underwent a second EUS-RFA session, and 4 ultimately required surgical resection. While EUS-RFA achieves a high initial symptom resolution rate, close follow-up is warranted because of the potential for symptom recurrence within 1 year, likely caused by microscopic residual tumors. The recent review, which summarized current evidence for EUS-RFA in insulinomas, emphasizing its high efficacy and acceptable safety profile while underscoring the need for further prospective studies with standardized protocols and longer follow-up. 34 EUS-RFA also showed high efficacy for NF-pNETs, with a pooled clinical success rate of 85% (95% CI, 74–92), however, the complete ablation rate varied widely, ranging from 67% to 100%. This suggests the possible influence of tumor size, location, or patient selection criteria across studies. In addition, the ablation protocol for EUS-RFA has not yet been standardized, with reported power settings ranging from 10 to 50 W across studies.
Khoury et al. 35 reported a meta-analysis of EUS-RFA for pNETs that involved 11 studies with 292 patients. The pooled complete radiological response was 87.1% (95% CI, 80.1%–92.8%), and the pooled clinical response rate for functional pNETs was 94.9% (95% CI, 90.7%–97.9%). The pooled incidence of AEs was 20.0% (95% CI, 14.0%–26.7%); most AEs were mild to moderate in grade, while the pooled incidence of severe AEs was 0.9% (95% CI, 0.2%–2.3%). Although our findings are generally consistent with previous study, 32 our analysis differs in that we evaluated EUS-EI and EUS-RFA separately according to tumor type. Regarding the pooled incidence of moderate or severe AEs, our analysis included only studies that provided detailed data, resulting in a relatively small sample size. In addition, some studies reported no AEs. After applying a logit transformation to account for zero-event studies, the pooled incidence was calculated to be higher than that reported in previous meta-analyses. Interestingly, the main types of moderate or severe AEs differed between the two treatment modalities: pancreatitis was predominant in EUS-EI, whereas peripancreatic fluid collection and PD stricture were the main events in EUS-RFA. These findings highlight the distinct safety profiles of the two techniques and may help clinicians anticipate procedure-specific risks and optimize perioperative management.
EUS-EI yielded a pooled clinical success rate of 77% (95% CI, 59–88) for insulinomas and 76% (95% CI, 54–90) for NF-pNETs. Although effective, these rates were lower than those observed for EUS-RFA. So et al. 18 reported a complete ablation rate of 65% (63/97) in 97 patients with NF-pNET who underwent EUS-EI. During the follow-up, local recurrence after complete ablation was observed in 29 patients (46.0%). The median time from the initial treatment to recurrence was 34.5 months (interquartile range: 28.9–48.9). Among these, one patient in the complete ablation group required surgical resection 6 years after EUS-EI due to recurrence, while another patient in the partial ablation group underwent surgery 4 years after EUS-EI because of tumor growth noted during follow-up. Thus, although salvage interventions are possible after EUS-EI, the risk of local recurrence remains high. Therefore, it is recommended that patients undergo follow-up for at least 5 years after treatment. Matsumoto et al. 17 reported a complete ablation rate of approximately 60% after a single ethanol injection. To improve this outcome, the authors implemented a protocol involving two planned treatment sessions. A CE-CT scan was performed 3 days after the initial procedure, and an additional ablation session was conducted if any residual tumor was detected. A final complete ablation rate of 88% (22/25 patients) was achieved. The operator-dependent nature of ethanol distribution and variability in injection techniques may also contribute to the heterogeneity of outcomes.
Although both EUS-EI and EUS-RFA are minimally invasive compared with surgical resection and are associated with lower morbidity and shorter hospital stays,19,26 the pooled overall AE rates ranging from 25% to 32% are not negligible. In surgical series, postoperative complication rates often exceed 40%–60%19,26 and include major events such as pancreatic fistula. Most AEs after EUS-guided ablation can be managed conservatively or through endoscopic interventions such as PD stenting and EUS-guided drainage.12,14,16,18,19,24,30,31 Nevertheless, pancreatitis and ductal injuries can occasionally be severe, underscoring the need for careful patient selection, procedural expertise, and close follow-up to minimize the risk of procedure-related complications. Park et al. 12 reported the use of EUS-EI in nine patients with NF-pNET. The mean diameter of the lesions treated using EUS-EI was 11 mm, and the mean volume of ethanol injected per session was 2.1 mL (range: 0.5–7.0 mL). The complete ablation rate was 67% (6/9), and mild pancreatitis occurred in three of the nine patients (one patient had a PD stricture as a late AE). They reported that the risk of pancreatitis was associated with the total amount of ethanol injected during the treatment session, because all procedure-associated cases of pancreatitis occurred when >2 mL of ethanol was administered in a session. Interestingly, in insulinoma cases, only one moderate or severe AE event occurred in each treatment group, which may be attributed to the fact that the primary endpoint was improvement of hypoglycemic symptoms rather than complete ablation. 24 Consequently, a wide ablation margin was not considered necessary.
Although no direct comparative studies are available between EUS-EI and EUS-RFA for pNETs, recent reports of EUS-RFA have generally demonstrated higher ablation rates and are increasingly represented in the literature. This trend may suggest potential advantages of RFA, similar to the established evidence in hepatocellular carcinoma. However, it is important to recognize technical limitations: current RFA devices require a 19G needle, whereas EUS-EI can be performed using thinner 22–25G needles. Thus, depending on the intrapancreatic location of the tumor, RFA puncture can be difficult or even infeasible. These considerations highlight that, despite the promising efficacy of RFA, and direct head-to-head comparisons will be essential to validate its role.
Given the lack of randomized controlled trials directly comparing EUS-guided ablation with surgery or active surveillance, careful patient selection remains essential. EUS-guided ablation appears most suitable for elderly patients or those with significant comorbidities who are at increased surgical risk. In addition, this approach may be considered for small, well-differentiated (G1) pNETs measuring ⩽2 cm in diameter, particularly when performed at institutions with established expertise in interventional EUS. 36 Further prospective studies are warranted to refine patient selection criteria and long-term outcomes. 37
This study has some limitations. First, all the included studies were non-randomized, which introduces a risk of selection bias. The methodological quality of included studies was assessed using the NOS, which indicated that most studies were of moderate or low quality. Our meta-analysis did not have a pre-registered protocol, including the absence of PROSPERO registration. As a result, the search could not comprehensively cover all potential sources or achieve full reproducibility. These issues reduce the methodological transparency of the review and should be considered when interpreting the findings. Therefore, potential selection and reporting biases cannot be excluded and may have influenced the pooled estimates. Second, the sample size was small. Consequently, although significant heterogeneity was observed in this subgroup, further sensitivity or meta-regression analyses were not feasible due to the limited number of studies and patients. These factors may indeed influence outcomes, but robust evaluation requires large-scale prospective studies with standardized reporting. Third, long-term outcomes have not been consistently reported across studies. Most included studies had short follow-up durations and reported no recurrence during the limited observation window. Therefore, the long-term durability of EUS-guided ablation remains uncertain and may be influenced by tumor biology and malignant potential. Moreover, data on progression-free survival and overall survival are lacking and warrant further investigation. Fourth, because of the limited number of included studies, the funnel plot analyses had low statistical power to detect potential publication bias, and the results should therefore be interpreted with caution. Prospective multicenter trials with standardized treatment protocols and long-term follow-up are needed to validate these findings.
Conclusion
Both EUS-EI and EUS-RFA are effective, relatively safe, and minimally invasive treatment options for managing pNETs. However, severe AE can occur, and careful patient selection and treatment indication are essential. Comparative studies on EUS-EI, EUS-RFA, and surgical resection are essential to determine the most effective and safest approach for each patient subgroup.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251410769 – Supplemental material for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848251410769 for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis by Kazuyuki Matsumoto, Yuki Fujii, Daisuke Uchida, Yasuto Takeuchi, Toshiharu Mitsuhashi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251410769 – Supplemental material for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tag-10.1177_17562848251410769 for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis by Kazuyuki Matsumoto, Yuki Fujii, Daisuke Uchida, Yasuto Takeuchi, Toshiharu Mitsuhashi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-3-tag-10.1177_17562848251410769 – Supplemental material for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis
Supplemental material, sj-jpg-3-tag-10.1177_17562848251410769 for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis by Kazuyuki Matsumoto, Yuki Fujii, Daisuke Uchida, Yasuto Takeuchi, Toshiharu Mitsuhashi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-4-tag-10.1177_17562848251410769 – Supplemental material for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis
Supplemental material, sj-jpg-4-tag-10.1177_17562848251410769 for Clinical efficacy and safety of endoscopic ultrasound-guided ablation therapies for pancreatic neuroendocrine tumors: a systematic review and meta-analysis by Kazuyuki Matsumoto, Yuki Fujii, Daisuke Uchida, Yasuto Takeuchi, Toshiharu Mitsuhashi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
