Abstract
Background:
Proton pump inhibitors (PPIs) use has been linked to adverse outcomes in patients with inflammatory bowel disease (IBD). However, it remains unknown whether this is due to protopathic bias (i.e., when the outcome precedes exposure).
Objectives:
We aimed to conduct a propensity-weighted study of the association between PPI use and IBD-related hospitalizations and surgery in patients with IBD.
Design:
Historical propensity score (PS)-weighted cohort study.
Methods:
We identified all Danish residents diagnosed with IBD in 2000–2022 in the Danish National Patient Registry. We analyzed separate PPI treatment episodes allowing an individual to contribute with more than one PPI episode. We used PS-weighted Cox regression to estimate the hazard ratio (HR) for IBD-related hospitalization and surgery for current PPI-users compared with current nonusers.
Results:
We identified 50,460 patients with IBD (67% with ulcerative colitis, 33% with Crohn’s disease). Five years after their diagnosis, two-thirds of patients with IBD had used PPI at some point and 10% were in current treatment. The weighted HR for IBD-related hospitalizations was 1.45 (95% confidence interval (CI): 1.38–1.52) during the first year after PPI prescription, and 1.16 (95% CI: 1.05–1.28) thereafter. The weighted HR for IBD-related surgery was 1.21 (95% CI: 1.11–1.32) the first year and 1.35 (95% CI: 1.18–1.54) thereafter.
Conclusion:
We observed a 20%–40% higher rate of IBD-related hospitalization and surgery, the first year after PPI prescription in patients with IBD which most likely represents a protopathic bias, yet the rate of IBD-related surgery remained elevated more than 1 year after PPI prescription.
Plain language summary
We studied how the commonly used drug class proton pump inhibitors, that suppresses stomach acid production, affects the clinical course of inflammatory bowel diseases (Crohn’s disease and ulcerative colitis). We found that it is unlikely that proton pump inhibitors affects patients with Crohn’s disease, whereas they most likely increase the risk of having surgery performed in patients with ulcerative colitis.

Background
Inflammatory bowel diseases (IBD) comprise Crohn’s disease that may affect segments of the entire gastrointestinal tract, and ulcerative colitis that affects the colon.1,2 About 1% of the general population have IBD, and their clinical course is characterized by periods of remission interchanging with periods of inflammatory activity.1,3,4
Proton pump inhibitors (PPIs) are among the most prescribed drug classes in Western countries.5,6 Their clinical effect is mediated by inhibition of acid secretion from stomach parietal cells. 7 They relieve gastro-esophageal reflux, peptic ulcers, dyspepsia, and Crohn’s disease in the upper gastrointestinal tract. 8 Despite the perceived safety of PPIs, increasing evidence suggests that PPI treatment triggers bacterial enteritis, aggravation of chronic liver insufficiency, and Clostridioides difficile enterocolitis.7,9–12 Moreover, PPI treatment in the general population has been linked with incident IBD. 13 Although Abrahami et al. 14 suggest that this association is best explained by a protopathic bias. 15 This bias arises if PPI prescription marks the onset of symptoms from IBD flare-ups.16,17 On the other hand, it seems evident that PPI treatment alters the gut microbiota and injures intestinal tight junctions,18,19 which may trigger inflammation in IBD. 20 Five observational studies have raised suspicion that PPI treatment affects adverse outcomes in IBD (medication drug switch, IBD-related hospitalization, and surgery) across diverse populations.21–25 Yet, these studies did not address the possibility of protopathic bias.
Thus, it remains unresolved whether PPI use has an adverse impact on the clinical course of IBD, or whether an association between PPI use and deterioration of IBD is the result of bias. In line with Abrahami et al.’s 14 work, the insertion of a lag time between the exposure (PPI prescription) and the adverse outcomes would clarify the presence of protopathic bias. 26 The most meaningful outcome would have been clinical evidence of IBD-flare (biochemistry, radiology, endoscopy), but such detailed data are unavailable in Danish healthcare registers. Therefore, hospital IBD-related hospital admission and surgery served as proxies for IBD flare-up, despite that the need for surgery may have different interpretation in ulcerative colitis and Crohn’s disease.
We aimed to conduct a nationwide population-based patient cohort study designed to clarify whether the association between PPI prescription and IBD-related hospitalization and surgery in patients with IBD is explained by a protopathic bias.
Methods
The study aim was examined using Danish registry data from 2000 to 2022 in a matched cohort design using propensity score (PS)-weighting to balance PPI users and nonusers with respect to the propensity to use PPIs. We designed the study to adhere to the STROBE (the Strengthening the Reporting of Observational Studies in Epidemiology) guideline. The data were analyzed as separate PPI treatment episodes allowing an individual to contribute observation time from more than one PPI episode. Follow-up started at the index date. Each time a person contributed with a new PPI treatment episode or nonuser episode follow-up resumed to time zero.
Data sources
The 5.9 million residents of Denmark benefit from universal tax-funded access to work-up and treatment at general practitioners and hospitals. The Danish National Patient Registry (NPR) is a nationwide registry that covers nonpsychiatric hospital admissions since 1977 and emergency room and outpatient visits since 1995. The data include admission and discharge dates along with up to 20 diagnoses per discharge. These diagnoses are coded in accordance with the International Classification of Disease edition 10 (ICD-10) from 1994, and the ICD-8 before that.27–30 The NPR also holds data on surgical procedures coded in accordance with the Nordic Classification of surgical procedures (NCSP).
The Danish National Prescription Database (NPD) holds data on all prescribed drugs redeemed in pharmacies across Denmark since 1995. The data include the date of the redemption, the civil registration number of the user, anatomical therapeutic chemical (ATC) code, dose of the drug, pack size, 24-h dose, and form of dose. 31
The Danish Central Office of Civil Registration tracks all Danish residents’ vital status, including the date of death or emigration. It also issues the unique 10 digit personal identifier that permits time-true individual-level linkage across the NPR, the NPD, and the civil registration system. 32 Use of this data required a project-specific permission from the Danish Data Protection Agency. The Danish Act on Processing of Personal Data is the legal basis for private and public institutions to obtain health data for research purposes. This Act protects against abuse of such data and balances the privacy rights of individuals and research purposes. 29
We identified all patients with two or more NPR diagnoses for IBD (Crohn’s disease (ICD-8: 56.300, 56.301, 56.302, ICD-10: K50.x) or ulcerative colitis (ICD-8: 56.319, 56.399, ICD-10: K51.x)) on inpatient contacts or outpatient visits, not including emergency room visits, within 2 years from 1977 and onwards. The date of the second diagnosis defined the diagnosis date. IBD was classified as Crohn’s disease or ulcerative colitis; in cases where a patient appeared with both diagnoses, the patient was classified as the most numerous diagnoses at time of PPI use. We used this data to create a cohort consisting of all Danes with a diagnosis of IBD from 2000 to 2022 and aged at least 15 years at first diagnosis. Individuals in the study population were further required to have been living in Denmark in the past 5 years before first PPI exposure to be able to calculate disease history.
PPI exposure
We used the cohort defined in the previous section to identify PPI treatment episodes. The PPI treatment episode started when patients with IBD redeemed a prescription for PPI (ATC: MA02BCxx). We defined the duration of each redeemed prescription based on the defined daily dose times the number of dispensed packages plus 28 days to separate exposed time from unexposed. A PPI treatment episode was defined as one or more redeemed PPI prescriptions, where each new prescription was redeemed before the end of the previous redeemed prescription, and the duration of this exposure time was truncated to the duration of the second redeemed prescription.
Each PPI treatment episode was matched 1:5 on age in 5-year intervals, sex and time since IBD diagnosis in 3-year intervals to a nonuser episode in a patients with IBD with no PPI use in the last 6 months and who had been living in Denmark for the past 5 years at the date of the PPI prescription. This initial matching was performed to assign an index date to the nonuser episodes: For PPI treatment episodes the index date was the date of the first PPI prescription and for the matched nonuser episodes the index date was the date of the first PPI prescription for the corresponding PPI user (see Supplemental Table 1). At each new PPI treatment episode/nonuser episode the index date was set as time zero the follow-up. The matched PPI nonusers were censored if they redeemed a PPI prescription during follow-up.
Patients in active PPI treatment were compared to “non-user” episodes in patients with IBD not in current PPI treatment who had not used PPI in at least 6 months before start of follow-up (the index date). A patient might enter with more than one PPI treatment episode if there was a washout period of at least 6 months between the PPI treatment episodes.
Outcomes
We defined two outcomes: (1) An in-hospital admission (IBD-related hospitalization) with a primary diagnosis for IBD or secondary diagnosis for IBD along with an IBD-related primary diagnosis. (2) An IBD-related surgical procedure (IBD-related surgery) defined as a procedure code for an operation on the colon or small intestine (NCSP: KJxx) at admission with a primary diagnosis for Crohn’s disease (ICD-10: K50x) or ulcerative colitis (ICD-10: K51x; NPR).
Statistical methods
We followed patients with IBD from the index date until an outcome, censoring at end of PPI treatment, death, emigration or until December 31, 2022. The effect of a PPI prescription on the hazard rates of the outcomes was analyzed with a Cox regression model using PS-weighting to account for potential confounders. 33 The PS-weights were calculated using logistic regression to estimate a PS for PPI treatment for each treatment episode. We included age, sex, time since IBD diagnosis, calendar year of diagnosis, IBD subtype, Charlson Comorbidity Index (CCI) score, previous IBD-related hospitalizations, previous IBD-related surgery, use of aminosalicylic acids, corticosteroids, thiopurines, antitumor necrosis factor-α agents, or NSAIDs on prescriptions in the year before the index date as predictors for PPI treatment, see details in Supplemental Material. The PS was used to construct standardized mortality ratio weights, PS/(1−PS) which were standardized to have mean 1. 34 These weights were used to re-weight the nonuser population to form a pseudo-population with same covariate distribution as the PPI-users. The matched PPI nonusers were censored if they redeemed a PPI prescription during follow-up.
We anticipated that the rate for both outcomes interacted with time since PPI prescription, because we suspected that a PPI prescription may reflect the onset of symptoms of IBD, and that these symptoms later resulted in IBD-related hospitalization/surgery (protopathic bias). Such a bias would result in a short-term rather than a long-term association between PPI prescription and IBD-related hospitalization or surgery. 26 For this reason, we computed and reported the rate of both outcomes by time since the index date in intervals of 20 days (IBD-related hospitalizations) and 100 days (IBD-related surgery). This diagnostic analysis showed a clear interaction between time since the index date and PPI treatment. Therefore, we decided not to report a single weighted hazard ratio (HR) for the entire follow-up, but to divide follow-up into two time-periods: The first year of follow-up after the index date, and the remaining follow-up, and reported the PS-weighted HR for each time-period. Presumably, previous IBD-related surgery in IBD patients signify a high risk of repeated IBD-related surgery. To clarify how PPI treatment affects the large pool of patients without previous IBD-related surgery, we repeated our main analysis but excluded patients with IBD and disease-related surgery.
Results
Among all IBD contacts in the NPR, we identified 50,460 patients with IBD, 15 years or older, who had two or more IBD hospital registrations within 2 years and with first ever IBD contact after January 1, 2000 and before December 31, 2022. In this population, 67% had a diagnosis of ulcerative colitis, 33% with Crohn’s disease, 53.2% were women, and the mean age at diagnosis was 41.8 years (interquartile range 31.3 years; Table 1).
Patient characteristics for PPI use and PPI nonuse episodes before and after SMRW at the index date.

Note that patients were matched on age, sex, time-period, and time since IBD diagnosis before the weighting.
ASA, 5-aminosalicyclic acid; CD, Crohn’s disease; IBD, inflammatory bowel disease; NSAID, nonsteroid anti-inflammatory drugs; PPI, proton pump inhibitors; SMRW, standardized mortality ratio weighting; TNF, tumor necrosis factor; UC, ulcerative colitis.
At the IBD diagnosis, 36.0% of patients with IBD had at least one prescription of PPI and 12.6% were in current treatment with PPI (Table 2). The proportions of users were 1.5 times higher for patients with CD than patients with UC (Table 2). Five years after the IBD diagnosis, 48.0% of patients with IBD had used PPI at some point and 11.5% were in current treatment.
Prevalence of past and current PPI use at diagnosis of IBD and 5 years after diagnosis for all IBD and by IBD subtype.

CD, Crohn’s disease; IBD, inflammatory bowel disease; PPI, proton pump inhibitors; UC, ulcerative colitis.
In total, there were 21,171 patients with IBD who used PPI at some point after IBD diagnosis and who contributed with 82,266 separate PPI treatment episodes, which were matched to nonuser episodes. Table 2 and Figure 1 show an overview of PPI treatment episodes and nonuser’s episodes, before and after the standardized mortality ratio weighting. Age, sex, calendar time, and time since IBD diagnosis did not affect the weighting because of the preceding matching. Yet, the PS showed that patients with comorbidity (CCI >0), previous NSAID treatment, or corticosteroid treatment were more likely to receive PPI treatment that resulted in higher weighting of nonuser episodes with these characteristics, and lower weighting of patients treated with 5-ASAs. There were only small differences in the prevalence of predictors for PPI treatment between PPI treatment and nontreatment episode after weighting (gray dots Figure 1).
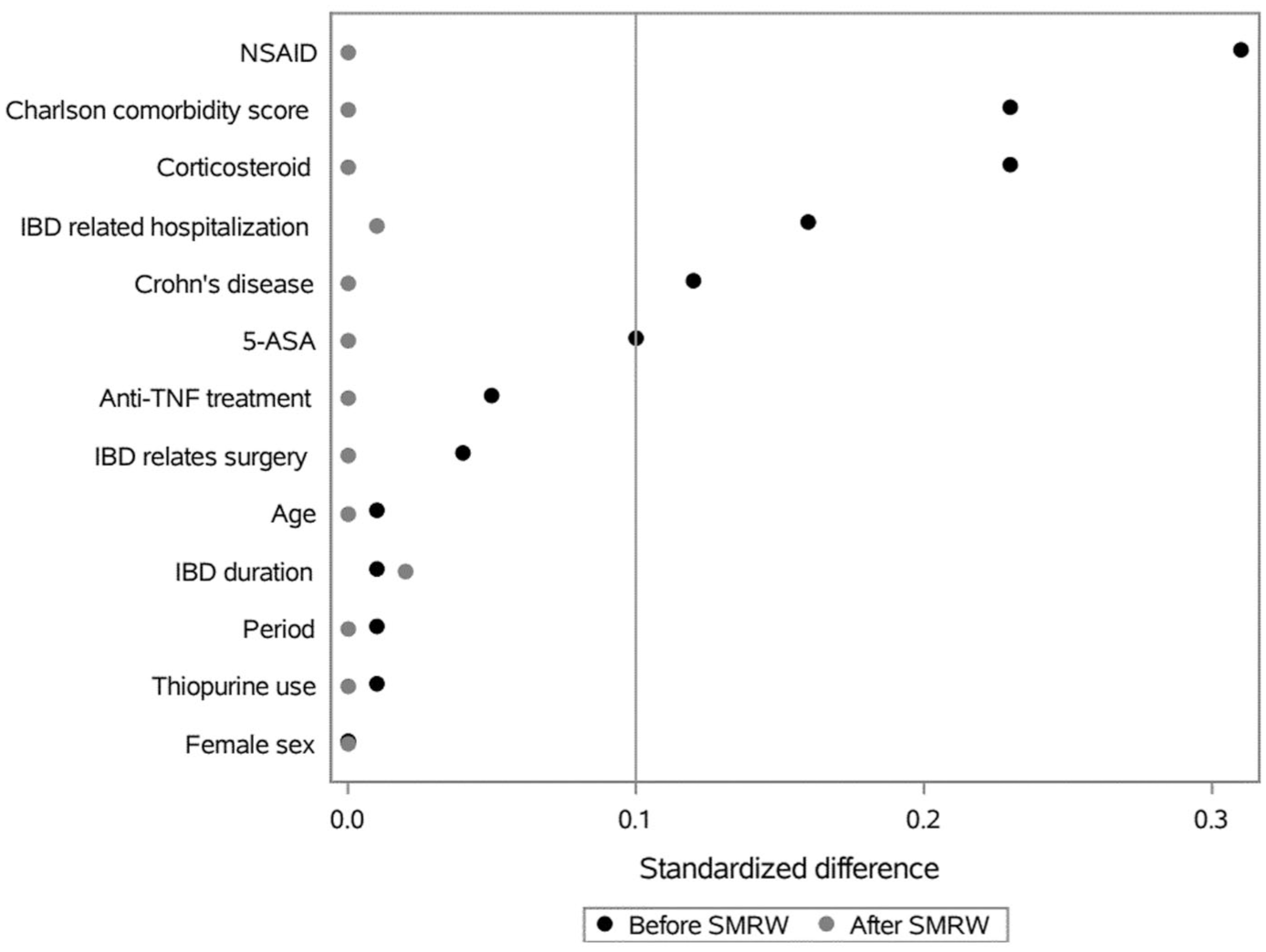
Standardized difference between PPI users and nonusers before and after propensity score-weighting using SMRW.
Figures 2 and 3, along with Tables 3–6, show the crude incidence rates (IRs) of IBD-related hospitalizations and surgery after the index date and their rate of IBD-related hospitalizations and surgery was much higher the first year after initiating PPI use. Importantly, it also shows that these rates flattened about 1 year after the index. Therefore, we divided follow-up into the first year and the remaining follow-up. Tables 3–6 also show that the absolute risk—in terms of the IR of hospitalizations and surgical procedures—were slightly higher in patients aged less than 50 years and that patients with Crohn’s disease were twice as likely to be hospitalized and to undergo surgery compared with patients with ulcerative colitis. Consequently, the IR of surgical procedures was lowest among patients with ulcerative more than 1 year after the index date.

Propensity score-weighted rate of hospitalization, and time from the index date (the first PPI prescription after the IBD diagnosis). The dots represent hospitalization rates per 20 days intervals. The curve represents a smoothing of this rate.
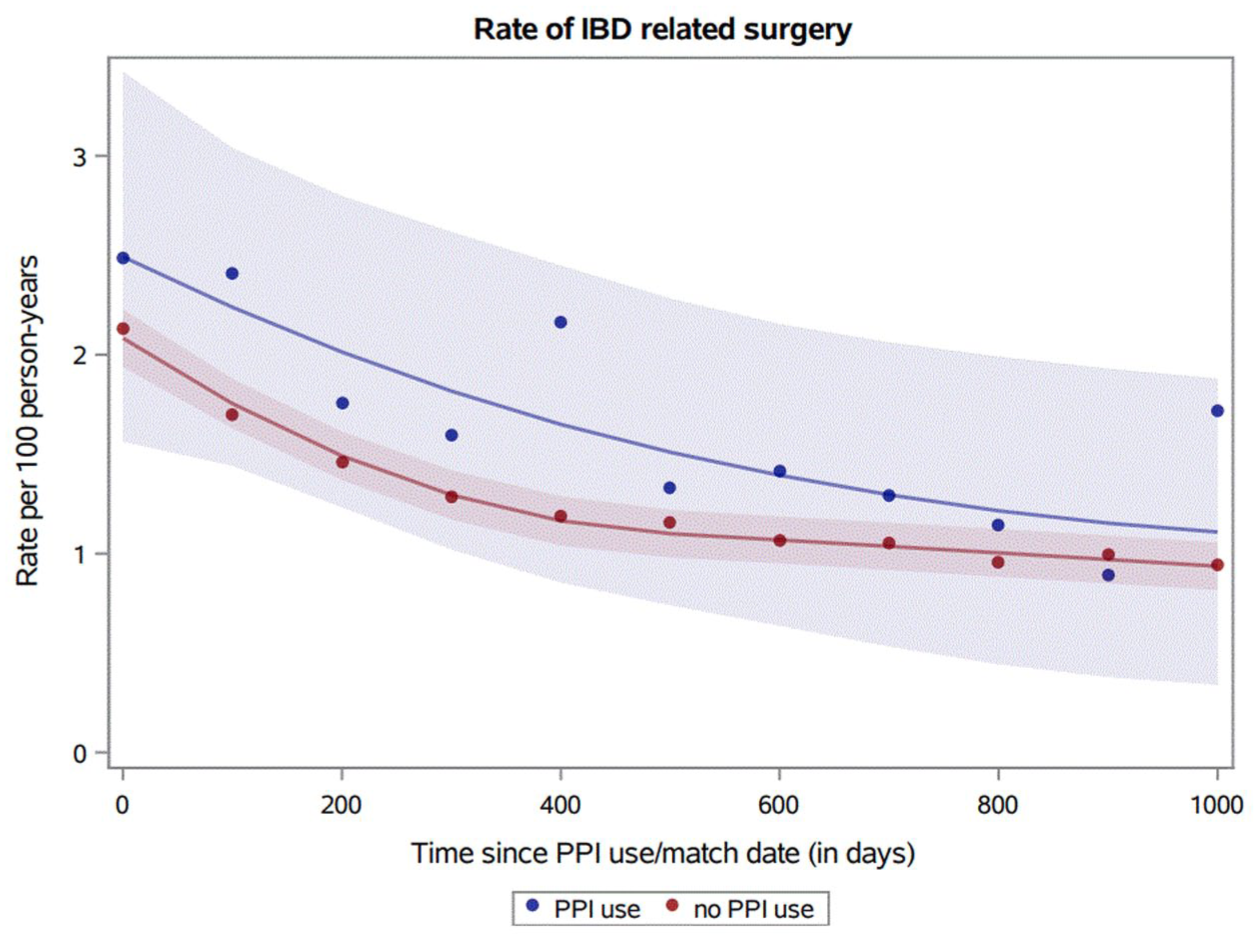
Propensity score-weighted rate of surgery, and time from the index date (the first PPI prescription after the IBD diagnosis). The dots represent surgery rates per 100 days intervals. The curve represents a smoothing of this rate.
Number of IBD-related hospitalization (N), PY, IR, and propensity score-weighted HR (aHR) for the first year of follow-up by disease category, sex, age group, and calendar period with 95% CIs.

The number of weight of events, PY, and IRs are propensity score-weighted for the non-user group.
CD, Crohn’s disease; CI, confidence intervals; HR, hazard ratio; IBD, inflammatory bowel disease; IR, incidence rate; PY, person-years; UC, ulcerative colitis.
Number of IBD-related hospitalization (N), PY, IR, and propensity score-weighted HR (aHR) remaining follow-up by disease category, sex, age group, and calendar period with 95% CI.
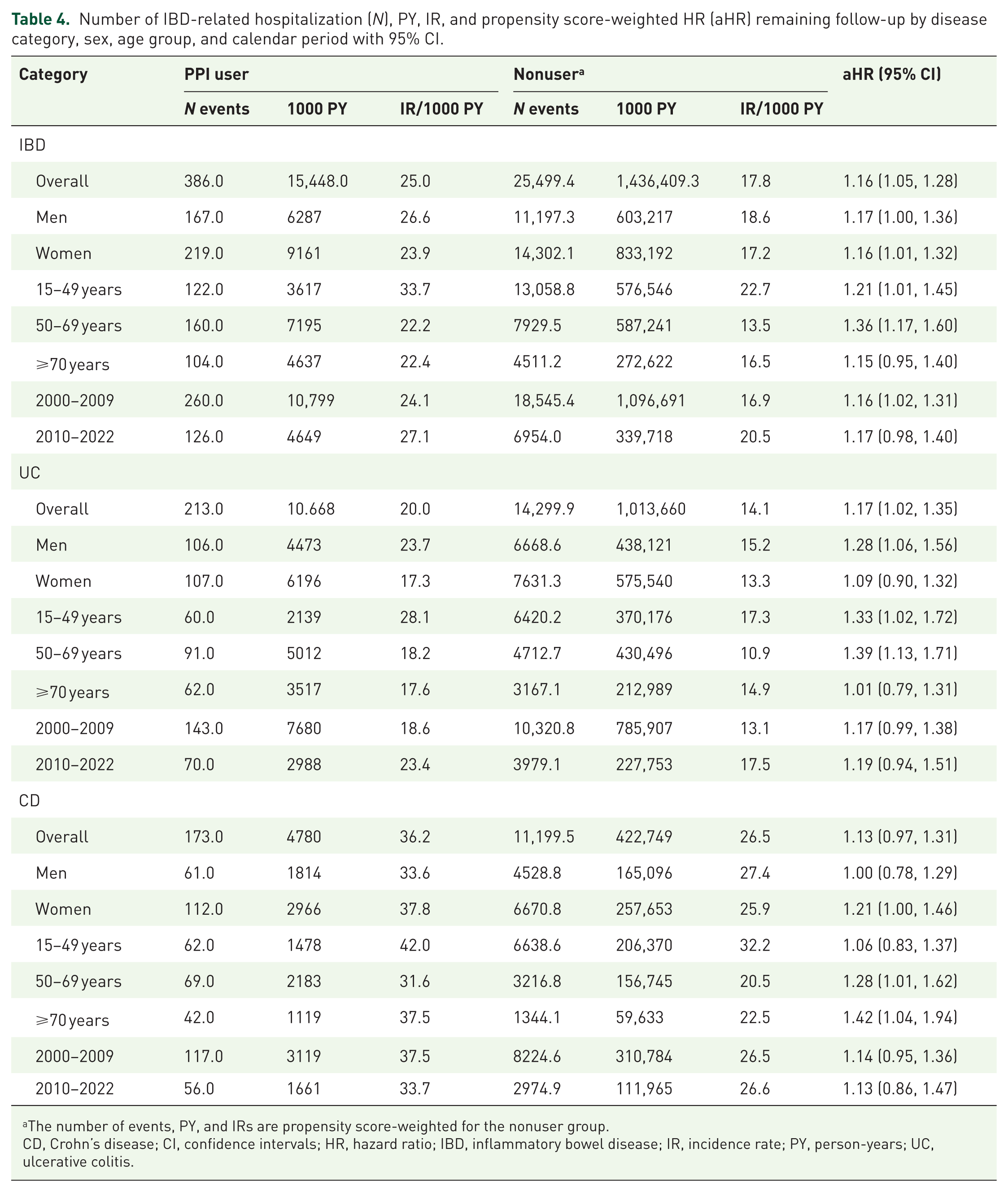
The number of events, PY, and IRs are propensity score-weighted for the nonuser group.
CD, Crohn’s disease; CI, confidence intervals; HR, hazard ratio; IBD, inflammatory bowel disease; IR, incidence rate; PY, person-years; UC, ulcerative colitis.
Number of IBD-related surgical procedures (N), PY, IR, and propensity score-weighted HR (aHR) the first year of follow-up by disease category, sex, age group, and calendar period with 95% CI.

The number of events, PY, and IRs are propensity score-weighted for the nonuser group.
CD, Crohn’s disease; CI, confidence intervals; HR, hazard ratio; IBD, inflammatory bowel disease; IR, incidence rate; PY, person-years; UC, ulcerative colitis.
Number of IBD-related surgical procedures (N), IR, and propensity score-weighted HR (aHR) remaining follow-up by disease category, sex, age group, and calendar period with 95% CI.

The number of events, person-years, and IRs are propensity score-weighted for the nonuser group.
CD, Crohn’s disease; CI, confidence intervals; HR, hazard ratio; IBD, inflammatory bowel disease; IR, incidence rate; UC, ulcerative colitis.
The main results of PS-weight regression analysis are also shown in Tables 3–7. The weighted HR for hospitalizations for PPI-users versus nonusers was 1.45 (95% confidence interval (CI): 1.38–1.52) during the first year, and 1.16 (95% CI: 1.05–1.28) for the remaining follow-up. In contrast, the weighted HR for IBD-related surgery for PPI-users compared to nonusers increased rather than decreased after 1 year of follow-up, being 1.21 (95% CI: 1.11–1.32) the first year after the index data and 1.35 (95% CI: 1.18–1.54) for the remaining follow-up. The long-term association between PPI use (>1 year after index) and IBD-related surgery was most pronounced in women 1.52 (1.29, 1.80), those aged more than 49 years on the index date, and in patients with ulcerative colitis (HR = 1.50, 95% CI: 1.25–1.79). Patients with ulcerative colitis also demonstrated a increased long-term risk for hospitalizations among PPI users (HR = 1.17, 95% CI: 1.02–1.35). In patients with ulcerative colitis, the pattern of a higher rate of hospitalizations and IBD-related surgery in PPI treated patients recurred in women and those aged more than 49 years (Table 7). In patients with Crohn’s disease the long-term association between PPI use and hospitalization (HR = 1.13; 95% CI: 0.97–1.31) or IBD-related surgery (HR = 1.17; 95% CI: 0.95–1.43) was less striking.
Propensity score-weighted HR (aHR) overall, and by disease category period with 95% CIs for patients with no previous IBD-related surgical procedures.
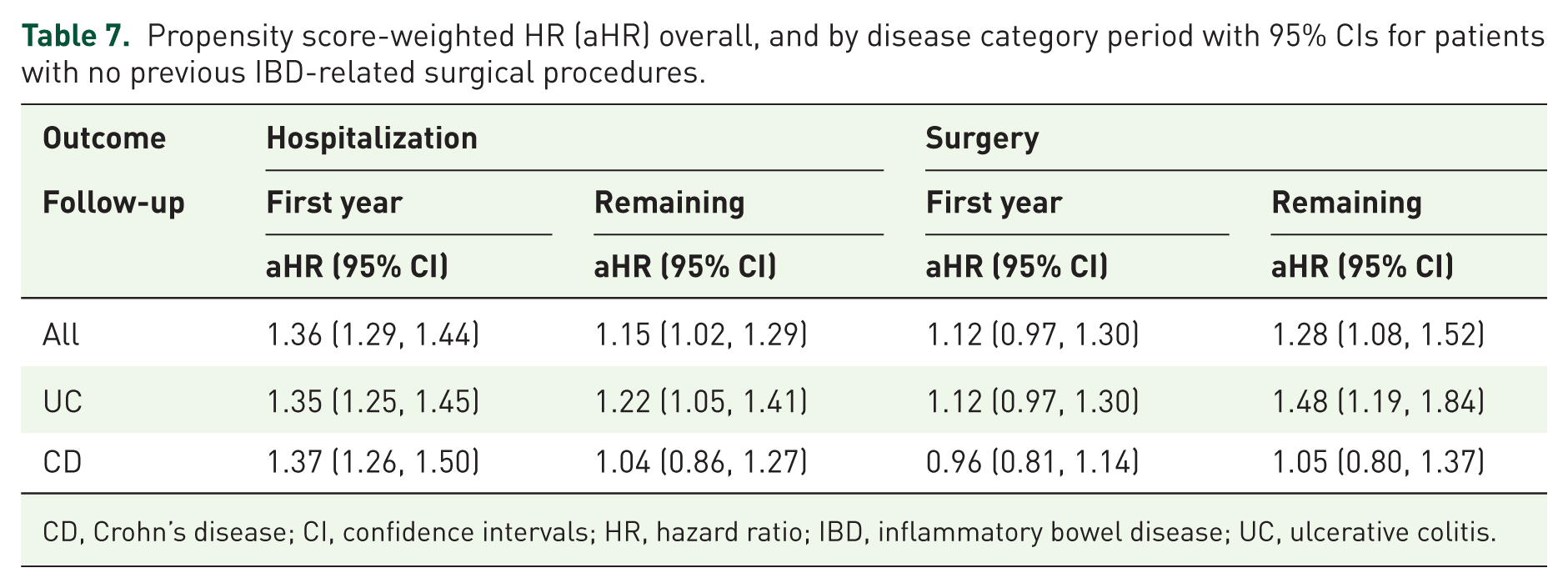
CD, Crohn’s disease; CI, confidence intervals; HR, hazard ratio; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Importantly, the association between PPI use and hospitalizations/surgical procedures in patients with UC more than 1 year after the index was essentially unchanged at 1.22 (1.05, 1.41) and 1.48 (1.19, 1.84), respectively (Table 7).
Discussion
In this nationwide population-based cohort study of more than 50,000 individuals with IBD followed longitudinally, PPI-use was associated with a 40% increased rate of hospitalizations and 20% increased rate of IBD-related surgery in patients with IBD the first year after PPI prescription. In patients with ulcerative colitis, the rate of IBD-related surgery remained 50% elevated following the first year of follow-up, which was not the case in patients with Crohn’s disease. Yet, the IR of surgical procedures was low in patients with ulcerative colitis.
Ulcerative colitis is a disease of colonic mucosa, whereas inflammation in Crohn’s disease is transmural and hence not in direct contact with the colon microbiota. PPI treatment disrupts the natural antimicrobial barrier generated by gastric acids 35 and increases intestinal permeability, 19 thereby it facilitates translocation of oral flora to the colon, 18 and reduce diversity of bacterial taxa in the colon.18,36 PPIs also increase the risk for enteric infection, 37 in particular C. difficile infection,9,38 which constitutes a fulminant disturbance of the gut microbiome in the colon and is known to be a risk factor for colectomy in patients with IBD. 39 Thus, both gut dysbiosis in general and susceptibility to C. difficile in particular are candidates for a likely link between PPI-treatment and an increased risk of IBD-related hospitalization and surgery in patients with ulcerative colitis.
When studying the association between PPI use and outcome of IBD, it essential to apply a study design aimed at identifying and quantifying the possibility of a “protopathic bias.” This bias occurs when a drug is prescribed for early symptoms of the condition under study and would arise in this study if symptoms from an IBD flare-up result in PPI prescription, and later, these symptoms result in IBD-related surgery or hospital admission. This scenario will inflate the results in favor of a harmful effect of PPIs. The investigation of an interaction between time since PPI prescription and the rate of the outcome may qualify the extent of such bias. 26 Abrahami et al. 14 used this method to examine the association between PPI use in the general population and incident IBD, but this has not been the case in previous studies on PPI use and outcome of IBD.21–25 Similarly, to our findings during the first year of follow-up, Fossmark et al. 25 found a HR for colectomy of 1.52 (95% CI: 1.17–1.98) in patients with UC, and Juillerat et al. 23 found a HR for IBD-related surgery of 1.31 (0.76–2.27) in PPI exposed patients with IBD. These studies were both derived from an unselected patient material, but they did not specifically aim to identify and quantify a protopathic bias, and they only incorporated a 1-month lag-time period between PPI exposure and their outcome.23,25 As we demonstrated in Figure 1, a 1 month of lag-time between exposure and outcome may be insufficient to address a protopathic bias. Our study confirms the association between PPI treatment and IBD flare-up.21–25 Yet, it also qualifies our understanding since it was strongest in patients with CD during the first year of follow-up but only present and intensified in UC patients for the remaining follow-up. Consequently, our study is the first to demonstrate that the concern on PPI treatment in CD may be exaggerated by protopathic bias, whereas the concern in UC patients persists.
The strength of our study is its foundation in the nationwide Danish healthcare registers. This ensures population-based coverage and complete follow-up. Repeated studies have confirmed a positive predictive value of the IBD diagnoses of more than 95% in the NPR, when only included patients with two or more admissions with a diagnosis for IBD.40,41 Finally, we see no reason to believe PPI treatment affects the validity of the IBD diagnosis. The uncharged access and financial support to prescribed drugs in the Danish healthcare system limit diagnostic delay and increase compliance. The interpretation of our results in resource-limited settings may therefore be different. On the other hand, we have every reason to believe that our clinical hypothesis of the effect of PPIs on the clinical course of IBD applies to all populations regardless of ethnicity.
A potential limitation is that nonusers have purchased PPIs as over-the-counter drugs. Therefore, PPI exposure may be subject to nondifferential misclassification that leads to an unpredictable bias. Yet, 96% of PPIs sold in Denmark are bought on prescription. 42 Consequently, we see no reason to believe misclassification of PPI use is a thread to our conclusion.
Our study also has potential limitations. Ideally, we could have circumvented confounding by means of a randomized controlled trial. Yet, our study followed several studies that question the safety of PPIs in IBD.21–23 Thus, despite their limitations, observational studies remain a relevant and unique source for information on side effects of drugs. We strove to minimize confounding in our study design and used a PS-weighted Cox model to address differences in age, sex, IBD-treatment intensity, and comorbidity between PPI users and nonusers. Smoking is a key candidate to confounding of the association between PPI-use and IBD-related hospitalization or surgery. Unfortunately, the NPR does not hold data on smoking status, but we adjusted for the CCI which included several comorbidities linked to smoking (chronic obstructive pulmonary disease, peripheral vascular disease). Besides, although smoking might be associated with PPI use and is a risk factor for flare-up of Crohn’s disease, smoking does not increase the risk for flare-up in ulcerative colitis.1,2 In fact, we found the strongest association between PPI use and IBD-related hospitalization or surgery in patients with ulcerative colitis. Thus, there are strong arguments why smoking is an unlikely confounder in this setting.
We stress cautious interpretation of our results concerning chronic disease activity. Evidently, patients with IBD with chronic abdominal pain are more likely to be hospital-admitted and prescribed PPIs. Abdominal pain is a disease-defining feature of Crohn’s disease, and patients with Crohn’s disease are prescribed PPIs at a higher rate immediately before their diagnosis. 17 Conversely, abdominal pain is an uncommon symptom of ulcerative colitis. Therefore, we adjusted for previous hospitalization, previous surgery, and TNF-α therapy, and other IBD-related treatment before the index date as proxies for chronic disease activity, and we acknowledge that the absence of key clinical information (pain severity, stool frequency) and biochemical measures (C-reactive protein, albumin, hemoglobin) was a limitation of our data.
Finally, body composition, diet, and physical activity are other potential confounders which to a certain extent are addressed by adjustment for comorbidity. Thus, we have done our best to reduce and address sources of confounding. Nevertheless, we acknowledge that possibility of residual confounding from differences in chronic disease activity, smoking, alcohol intake, diet, and other lifestyle factors between PPI using and nonusing IBD patients may influence our results.
Crohn’s disease in the esophagus, stomach, and duodenum is rare and constitutes the only indications for PPI treatment in patients with IBD. 1 Nonetheless, we found that approximately 10% of patients with CD and UC were using PPI when they were diagnosed which was unchanged after 5 years of follow-up, and that half the patients with IBD were treated with PPI at least once during the first 5 years of follow-up. Since PPIs may be linked to an increased risk of flare-up combined with our clinical impression that patients often are treated with PPIs without work-up to ensure its indication, we suggest a more conservative use of PPIs in patients with IBD. Importantly, we do not recommend stopping PPI treatment for well-founded indications, but we suspect that many patients with IBD could be tapered off PPIs without putting them at risk. 43 Moreover, even if the interpretation of our results was that chronic disease activity is a contributing factor to flare-ups rather than PPIs, the clinical lesson remains to reconsider the indication for PPI treatment and to consider work-up for IBD flare-up.
Conclusion
We designed this study to address whether a protopathic bias explained the association between PPI prescription and IBD flare-up. It showed that more than two-thirds of patients with IBD were prescribed PPIs during the first 5 years follow-up, and that PPI-using patients with IBD had about 45% higher rate of IBD-related hospitalization and about 21% higher rate of IBD-related surgery, the first year after starting PPI treatment which most likely reflects a protopathic bias. Yet, the rate of IBD-related surgery remained elevated after 1 year of follow-up and was 50% higher in patients with ulcerative colitis that continue PPI treatment for more than 1 year, which is unlikely to be the result of protopathic bias. Residual confounding may have influenced our results, but even if chronic disease activity or other factors was the cause of flare-ups and PPI treatment in our data, the requirement for long-term PPI treatment in patients with IBDs calls for work-up rather than continuation of PPIs, particularly in those with ulcerative colitis.
Supplemental Material
sj-doc-2-tag-10.1177_17562848251393884 – Supplemental material for The effect of proton-pump inhibitor therapy on disease outcomes in inflammatory bowel disease: a population-based cohort study
Supplemental material, sj-doc-2-tag-10.1177_17562848251393884 for The effect of proton-pump inhibitor therapy on disease outcomes in inflammatory bowel disease: a population-based cohort study by Thomas Deleuran, Gry Juul Poulsen, Lone Larsen, Jan Fallingborg, Peter Jepsen and Tine Jess in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-1-tag-10.1177_17562848251393884 – Supplemental material for The effect of proton-pump inhibitor therapy on disease outcomes in inflammatory bowel disease: a population-based cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848251393884 for The effect of proton-pump inhibitor therapy on disease outcomes in inflammatory bowel disease: a population-based cohort study by Thomas Deleuran, Gry Juul Poulsen, Lone Larsen, Jan Fallingborg, Peter Jepsen and Tine Jess in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
