Abstract
Background:
Inflammatory bowel disease (IBD) is not associated with worse coronavirus disease 2019 (COVID-19) outcomes. However, data are lacking regarding the long-term impact of severe acute respiratory syndrome coronavirus 2 infection on the disease course of IBD.
Objectives:
We aimed to investigate the effect of COVID-19 on long-term outcomes of IBD.
Design:
We performed a multicenter case–control study of patients with IBD and COVID-19 between February 2020 and December 2020.
Methods:
Cases and controls were individuals with IBD with presence or absence, respectively, of COVID-19-related symptoms and confirmatory testing. The primary composite outcome was IBD-related hospitalization or surgery.
Results:
We identified 251 cases [ulcerative colitis (n = 111, 45%), Crohn’s disease (n = 139, 55%)] and 251 controls, with a median follow-up of 394 days. The primary composite outcome of IBD-related hospitalization or surgery occurred in 29 (12%) cases versus 38 (15%) controls (p = 0.24) and on multivariate Cox regression, COVID-19 was not associated with increased risk of adverse IBD outcomes [adjusted hazard ratio (aHR): 0.84, 95% confidence interval [CI]: 0.44–1.42]. When stratified by infection severity, severe COVID-19 was associated with a numerically increased risk of adverse IBD outcomes (aHR: 2.43, 95% CI: 1.00–5.86), whereas mild-to-moderate COVID-19 was not (aHR: 0.68, 95% CI: 0.38–1.23).
Conclusion:
In this case–control study, COVID-19 did not have a long-term impact on the disease course of IBD. However, severe COVID-19 was numerically associated with worse IBD outcomes, underscoring the continued importance of risk mitigation and prevention strategies for patients with IBD during the ongoing COVID-19 pandemic.
Background
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative virus of coronavirus disease 2019 (COVID-19) pandemic, has infected more than 390 million people worldwide as of 1 February 2022 and continues to remain a global health crisis. 1 Inflammatory bowel diseases (IBD), comprising Crohn’s disease (CD) and ulcerative colitis (UC), are chronic immune-mediated inflammatory diseases (IMIDs) of the gastrointestinal tract that often require long-term immunosuppressive therapy for management.
Early in the onset of the pandemic there was concern that IBD, chronic immune dysregulation, and associated immunosuppression may represent a potential risk factor for SARS-CoV-2 infection or severe outcomes. 2 Subsequent studies have largely shown that IBD does not increase the risk of SARS-CoV-2 infection or worse COVID-19 outcomes.3–8 Data have also confirmed that biologic and novel small molecule therapies are not associated with worse COVID-19 outcomes.9,10 Furthermore, vaccines against SARS-CoV-2 are effective in reducing symptomatic or severe COVID-19, and early data have shown that in patients with IBD, vaccine efficacy is comparable with that in the general population.11,12
While these data are reassuring, the long-term impact of COVID-19 on the course of IBD remains largely unknown. Studies from the previous SARS epidemic of 2003 have shown a durable impact of infection with the SARS-CoV-1 virus on long-term pulmonary function, exercise capacity, psychiatric comorbidities, and chronic fatigue.13,14 Similarly, data suggest that COVID-19 is associated with long-term respiratory, neurologic, and psychiatric sequelae.15,16 Long-haul COVID-19 results in immune dysfunction that can persist up to 8 months after infection, and in the gastrointestinal tract, where angiotensin-converting enzyme 2 expression is increased and SARS-CoV-2 antigen can persist, it is associated with chronic abdominal pain, altered bowel motility, and irritable bowel syndrome.17–19 Furthermore, the cytokine storm associated with COVID-19 results in elevation of several pro-inflammatory cytokines, particularly interleukin (IL)-6, IL-10, IL-2, and IL-17, that are implicated in the pathogenesis of IBD. 20 Given these reported associations, the possible consequence of COVID-19 is an alteration of the inflammatory milieu with a downstream deleterious impact on IBD outcomes.
In this multicenter, case–control study, we aimed to determine the impact of COVID-19 on long-term outcomes of IBD.
Methods
Study population
A multicenter, case–control, observational cohort was performed by five academic medical centers within the New York Crohn’s and Colitis Organization. After Institutional Review Board approval at each participating site, electronic medical records were reviewed to identify patients with an established diagnosis of IBD (CD, UC, or IBD-undefined) for at least 1 year who were confirmed to have COVID-19 (cases) or not (controls) between February 2020 to December 2020.
COVID-19 definitions and variables
COVID-19 cases were defined as individuals with the presence of COVID-19 symptoms (fever, cough, dyspnea, anosmia, dysgeusia, pharyngitis, diarrhea, arthralgia–myalgia/ asthenia, rhinitis, dysphonia) with a confirmatory positive nasopharyngeal SARS-CoV-2 PCR test at the time of symptoms and/or subsequent positive SARS-CoV-2-IgG antibody test in 2020. Controls were individuals with the absence of COVID-19 symptoms (excluding diarrhea) and both one negative SARS-CoV-2 PCR and at least one negative SARS-CoV-2 IgG in 2020 in testing performed at the academic medical centers involved in this study. Severe COVID-19 was defined as need for hospitalization, mechanical ventilation, or intensive care unit admission.21,22 Cases and controls were matched 1:1 based on age, sex, and type of IBD. Sample size was determined by the number of all eligible cases within the study period.
We collected the following initial data on demographics and disease characteristics: age, sex, IBD disease extent and phenotype (Montreal classification), history of IBD-related surgery and/or hospitalizations, and IBD treatment history; and for cases, COVID-19 disease course and management. These data were entered into a REDCap electronic database accessible to participating centers. Long-term outcomes for each patient were then prospectively collected from April to December 2021 and updated in the REDCap database.
Outcomes
The primary outcome was a composite of IBD-related hospitalization (non-elective) or surgery at any time during follow-up. Secondary outcomes included biologic dose escalation, switch to a new biologic or small molecule therapy, corticosteroid initiation, change in UC extent, change in CD location or behavior, development of non-IBD IMID, initiation of new psychiatric care or medication, and interval COVID-19, time to first follow-up, and delay of next biologic dose. For each outcome, patients were censored at date of first outcome event, date of interval COVID-19 (if a patient in the control group was subsequently infected), or date of last follow-up, whichever was earliest. For the primary outcome, the study groups were further stratified by COVID-19 severity (non-severe versus severe).
Statistical analysis
Continuous variables were summarized using means with standard deviation or medians with interquartile ranges (IQRs) and were compared using Student’s t test or Wilcoxon rank-sum test. Chi-squared tests of independence and Fischer’s exact tests were used for categorical variables between groups and Kruskal–Wallis tests for continuous variables. Missing variables were excluded from statistical analysis. Time to the primary outcome was compared between the study groups with Kaplan–Meier survival curves using log-rank analysis. Cox proportional hazards models were used to identify the risk of the primary outcome adjusted for COVID-19 severity, indicators of IBD disease severity (prior IBD hospitalization or surgery, duration of disease), disease activity (current steroids or biologic use), and delays in care (time to first follow-up or postponement of biologics). Statistical analysis was performed using IBM SPSS 25 (IBM, Armonk, NY, USA). The reporting of this study conforms to the STROBE statement. 23
Results
Cohort baseline characteristics
We identified 251 patients with IBD [UC (n = 112, 45%), CD (n = 139, 55%)] and confirmed COVID-19 matched to 251 controls [UC (n = 112), CD (n = 139)]. The median age in the overall cohort was 39 years (IQR: 30–52), and 45% of patients were males (Table 1). There were no differences between study groups in age, race, ethnicity, duration of IBD, or smoking status. A total of 5642 person-months of follow-up were assessed, with a median follow-up of 417 days (IQR: 304–495; range: 43–581) for controls and 358 days (IQR: 238–459; range: 32–576) for COVID-19 cases. The majority of cases (n = 208, 81%) occurred between March and May 2020.
Demographics, baseline IBD characteristics.
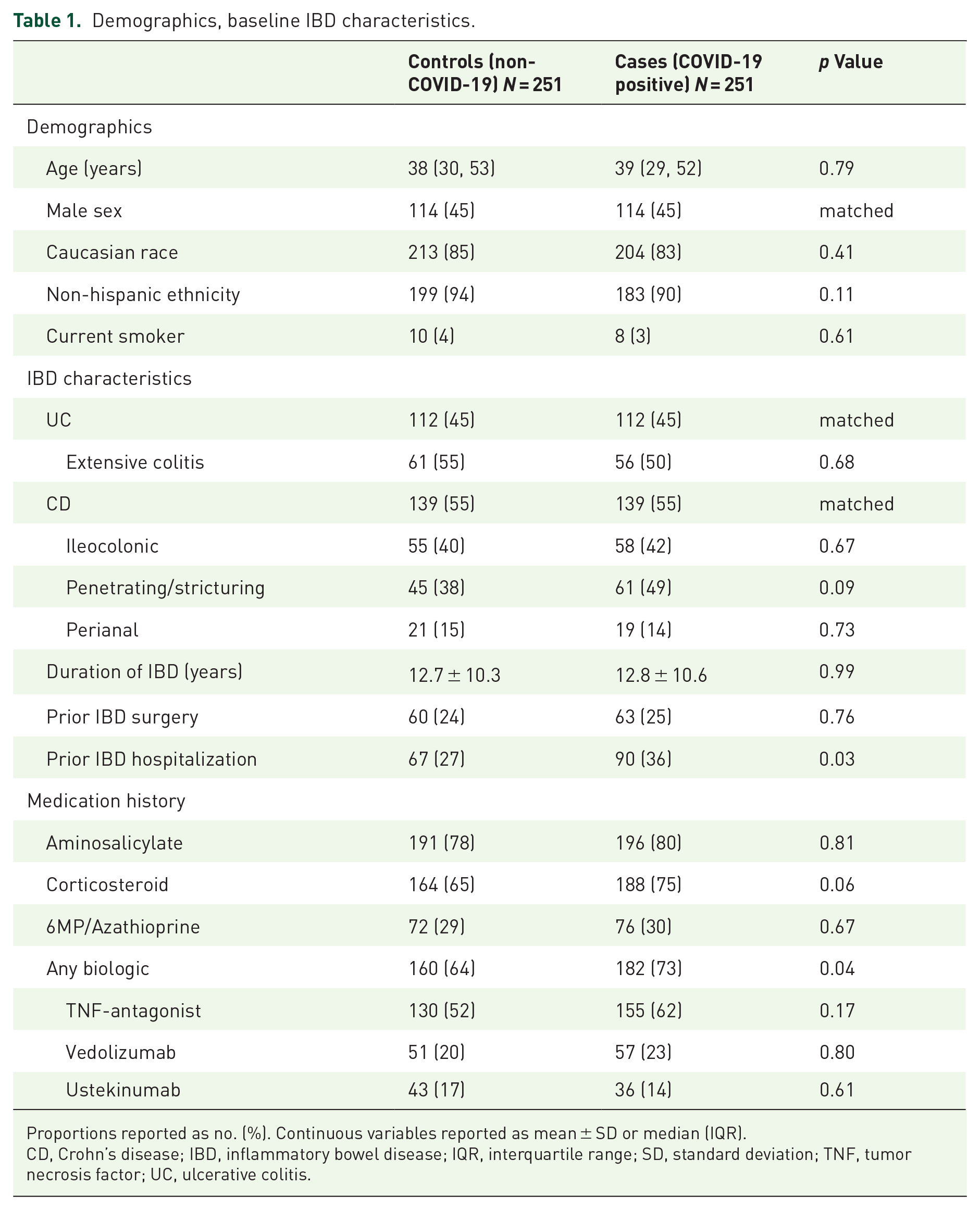
Proportions reported as no. (%). Continuous variables reported as mean ± SD or median (IQR).
CD, Crohn’s disease; IBD, inflammatory bowel disease; IQR, interquartile range; SD, standard deviation; TNF, tumor necrosis factor; UC, ulcerative colitis.
At baseline, COVID-19 patients had higher rates of previous IBD-related hospitalizations (36% versus 27%; p = 0.03), corticosteroid use (75% versus 65%; p = 0.06), and biologic exposure (73% versus 64%; p = 0.04) than controls (Table 1). There were no differences in extent of UC or CD location or behavior between groups, or in prevalence of coexisting comorbidities (Supplemental Table 1). Among cases of COVID-19, 39 (16%) were severe and 212 (84%) were mild–moderate. Among patients with COVID-19, 63 (25%) had their next biologic dose delayed, with a median delay of 14 days (Supplemental Table 2). Overall, 49 (20%) patients received COVID-19-related therapy, with a higher rates of use in severe COVID-19 cases than in mild–moderate cases (72% versus 10%, p<0.001)
Long-term primary and secondary outcomes
The primary composite outcome of IBD-related hospitalization or surgery occurred at similar rates between COVID-19 cases (12%, n = 29) and controls (15%, n = 38; p = 0.24; Table 2) and there was no difference in times to these outcomes (log-rank p = 0.70, Figure 1). Between COVID-19 cases and controls, there were no differences in unadjusted rates of hospitalization or surgery, biologic dose escalation, switch to new biologic or corticosteroid initiation, and progression of UC or CD extent or phenotype. Control patients were more likely to subsequently receive a COVID-19 vaccine in than were cases (74% versus 51%, p<0.001). On multivariable analysis, after adjusting for prior IBD hospitalization or surgery, penetrating or stricturing CD, extensive UC, steroid use, biologic or small molecule, biologic delay, and time to first follow-up, compared to controls, COVID-19 was not associated with an increased risk of the primary composite outcome [adjusted hazard ratio (aHR): 0.84, 95% confidence interval (CI): 0.44–1.42], whereas prior hospitalization or surgery (aHR: 2.71, 95% CI: 1.53–4.80) (Table 3).
Long-term outcomes in COVID-19 and IBD.
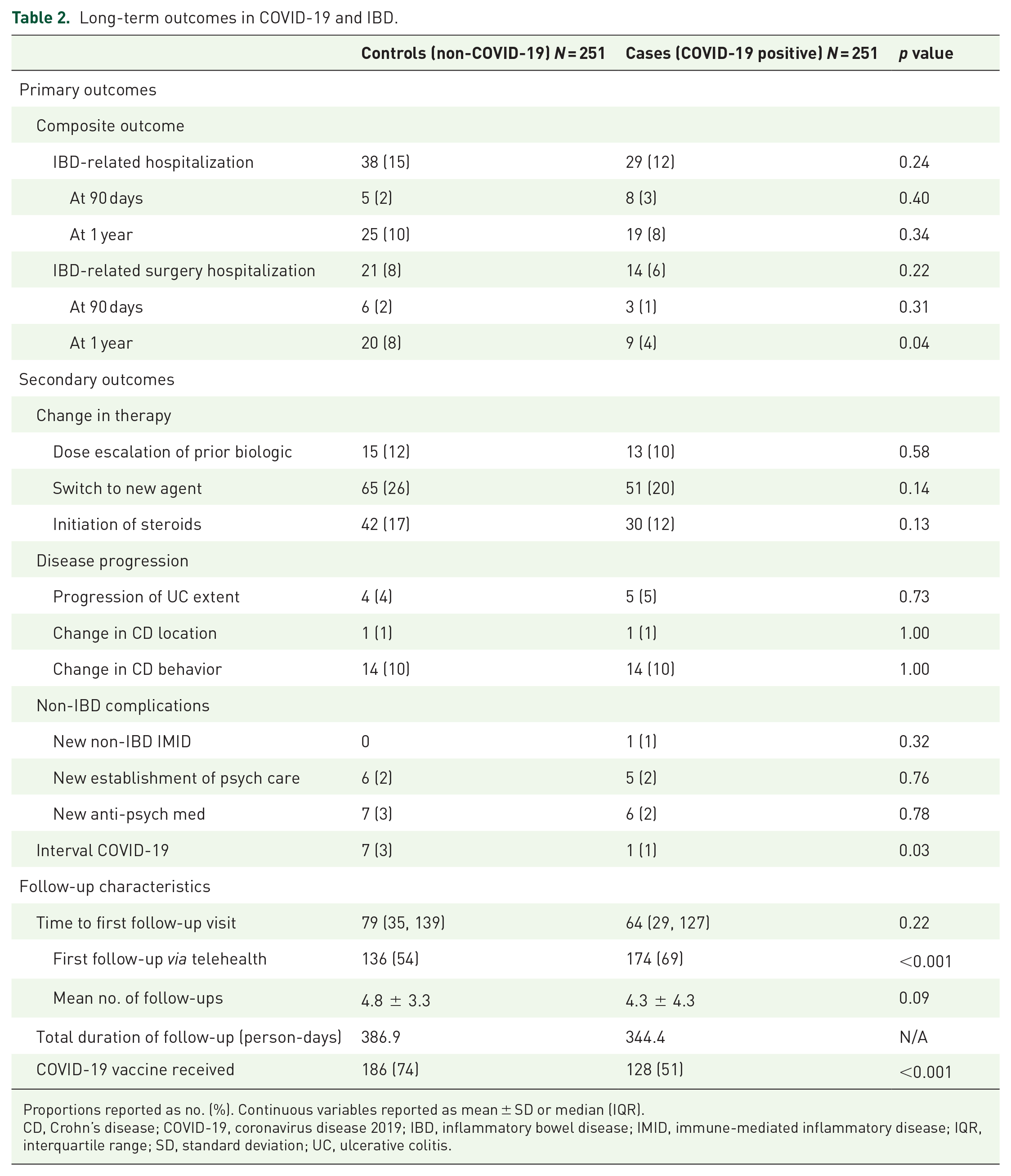
Proportions reported as no. (%). Continuous variables reported as mean ± SD or median (IQR).
CD, Crohn’s disease; COVID-19, coronavirus disease 2019; IBD, inflammatory bowel disease; IMID, immune-mediated inflammatory disease; IQR, interquartile range; SD, standard deviation; UC, ulcerative colitis.
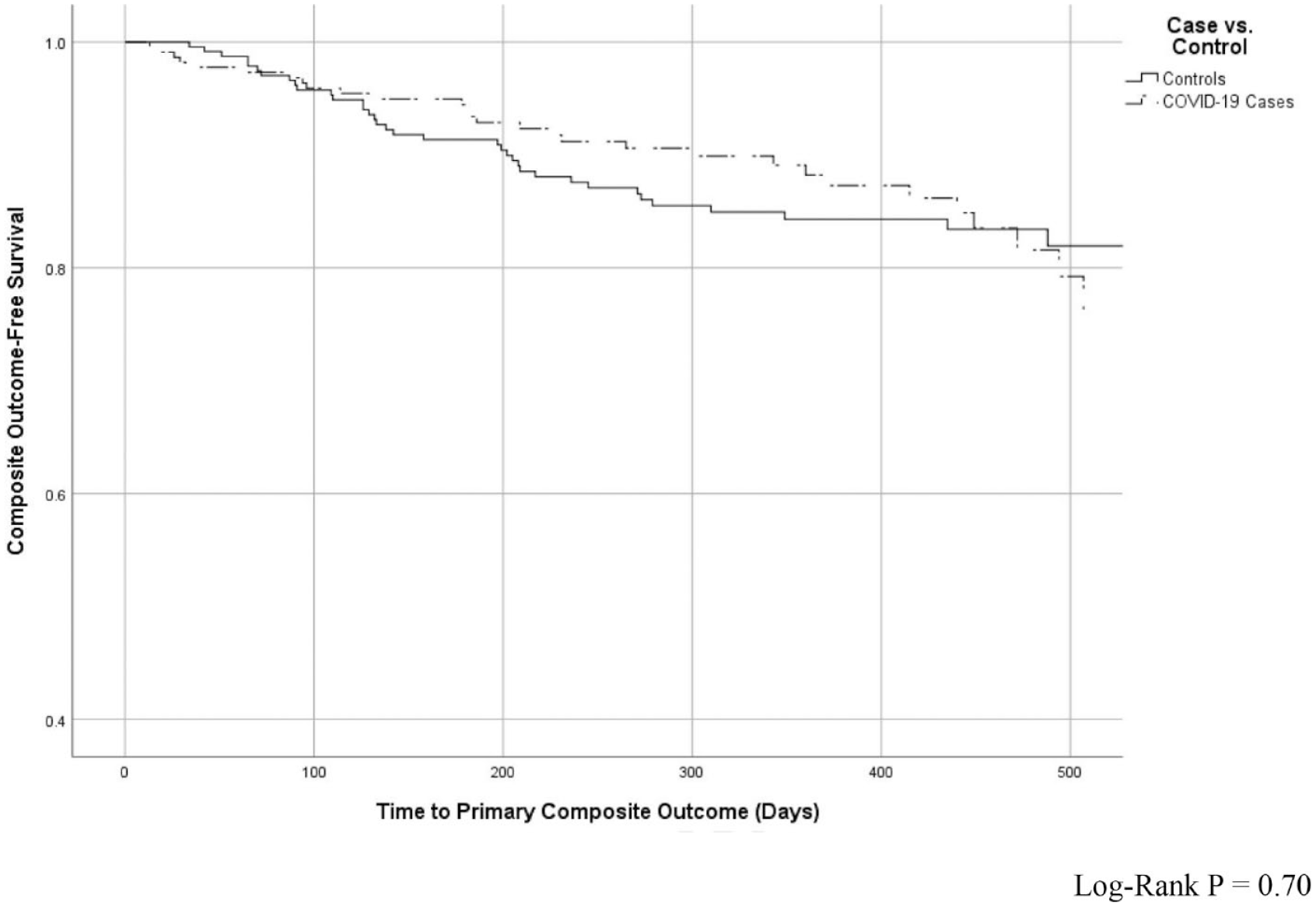
Kaplan–Meier curve of IBD hospitalizations or surgeries over time.
Multivariate cox proportional analysis of risk factors for composite outcome of IBD hospitalization or surgery.*

All variables included in the Cox proportional hazards regression model are reported in the table.
CD, Crohn’s disease; COVID-19, coronavirus disease 2019; IBD, inflammatory bowel disease; ICU, intensive care unit; UC, ulcerative colitis.
Sub-group analysis: stratified by COVID-19 severity
When further stratified by COVID-19 severity, the primary composite outcome occurred in 21% (8/39) of patients with severe COVID-19 compared with 11% (21/212) of those with mild-to-moderate COVID-19 and 15% (38/251) of controls (log-rank p = 0.02; Supplemental Figure 1). When COVID-19 severity was included in the multivariable model described above, severe COVID-19 was associated with a numerically increased risk of the primary composite outcome (aHR 2.43: 95% CI: 1.00–5.86), whereas mild-to-moderate COVID-19 was not (aHR: 0.68, 95% CI: 0.38–1.23; Table 4). Inclusion of administration of COVID-19 therapies in the multivariable model did not impact the primary outcome (aHR 0.62: 95% CI: 0.19–2.01), or decrease the impact of COVID-19 severity (aHR: 3.39, 95% CI: 0.40–1.31).
Subgroup analysis: multivariate cox proportional analysis of risk factors for composite outcome of IBD hospitalization or surgery, stratified by COVID-19 severity.*

All variables included in the Cox proportional hazards regression model are reported in the table.
COVID-19 course requiring hospitalization, oxygen supplementation, mechanical ventilation, or ICU admission.
CD, Crohn’s disease; COVID-19, coronavirus disease 2019; IBD, inflammatory bowel disease; ICU, intensive care unit; UC, ulcerative colitis.
Discussion
In this multicenter, case–control, prospective observational cohort of IBD patients from five academic centers in the New York City area, COVID-19 was not associated with long-term worsening of disease course of IBD, defined as subsequent need for hospitalization, surgery, or other non-IBD adverse outcomes such as the development of other IMIDs or psychiatric comorbidities. However, severe COVID-19 did appear to increase the risk of future adverse IBD outcomes. This is the largest study to date to assess the long-term impact of COVID-19 in IBD and the first to use a case–control, matched study design, to specifically determine the independent effect of COVID-19 on the course of IBD.
Reassuring early data from New York City, the epicenter of the COVID-19 pandemic in the United States, showed that the majority of COVID-19 cases in patients with IBD were mild-to-moderate and not associated with poor outcomes. 5 The international SECURE-IBD (Surveillance Epidemiology of Coronavirus Under Research Exclusion) registry confirmed this at a global level, although a more recent population-based study by Attauabi et al. 8 did show that patients with IBD have worse COVID-19 outcomes.9,10,24 When assessing the impact of COVID-19 on IBD, Lukin et al. 25 found that patients with IBD and COVID-19 did not have worsening of their IBD clinical disease activity, endoscopic scores or biomarkers of inflammation up to 210 days after infection.
Although our study similarly showed overall non-severe outcomes in patients with COVID-19 and IBD, when stratified by infection severity, the subgroup of patients with severe COVID-19 had numerically increased rates of future IBD-related hospitalization or surgery, hospitalization at 1 year, surgery at 1 year, and initiation of new biologic agent (Supplemental Table 3). These findings are notable but should be interpreted with caution in this smaller sample. It is possible that severe COVID-19 is a reflection of, rather than risk factor for, uncontrolled IBD as studies have shown that baseline steroid use and disease activity are associated with worse COVID-19 outcomes.5,26 Delays in care or postponement of biologics due to COVID-19 may also lead to worse IBD outcomes, as has been reported in one study by Li et al. 27 We demonstrate, however, that transient postponement of biologics in the setting of COVID-19 did not have an adverse impact on IBD outcomes. On the other hand, administration of COVID-19-related therapies did not appear to mitigate the risk of future adverse IBD outcomes.
After multivariable analysis adjusting for various indicators of disease activity and delays in care, COVID-19 was not an independent risk factor for subsequent IBD-related hospitalization or surgery. This especially notable given that the COVID-19 cases in our study had more severe IBD at baseline, with higher rates of penetrating or stricturing CD, prior hospitalizations, steroid use, and biologic use. This suggests that COVID-19 alone does not appear to alter the future course of IBD. However, patients who experience a severe course of COVID-19 and its resultant hyper-inflammatory cytokine storm may potentially have worse future IBD outcomes, although this requires further study. 28
Our study has limitations, including the risk of misclassification, as patients may have had undocumented or undiagnosed COVID-19 despite our use of fairly stringent criteria for identification of controls. We defined outcomes as discrete events of hospitalization and surgery to ensure consistency of outcome assessment across all study sites, but it is possible that other outcomes such as clinical disease scores, biomarkers, and endoscopic disease activity which were not evaluated may have differed between study groups. Lastly, the entry period of this study spanning 2020 captured COVID-19 cases likely from the original SARS-CoV-2 B.1.1.7 variant (Alpha), but these findings may not reflect the transmission and virulence profiles of later emergent strains such as the B.1.617.2 (Delta) and B.1.1.529 (Omicron) variants.
Conclusion
In this multicenter study, COVID-19 did not alter the long-term course of IBD; however, severe COVID-19 may portend a worse IBD disease course and prognosis. Risk mitigation and vaccination remain important strategies in the care of IBD patients during the ongoing COVID-19 pandemic.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221132363 – Supplemental material for COVID-19 is not associated with worse long-term inflammatory bowel disease outcomes: a multicenter case–control study
Supplemental material, sj-docx-1-tag-10.1177_17562848221132363 for COVID-19 is not associated with worse long-term inflammatory bowel disease outcomes: a multicenter case–control study by Simon J. Hong, Sumona Bhattacharya, Aiya Aboubakr, Devika Nadkarni, Diana Lech, Ryan C. Ungaro, Manasi Agrawal, Robert P. Hirten, Ruby Greywoode, Anjali Mone, Shannon Chang, David P. Hudesman, Thomas Ullman, Keith Sultan, Dana J. Lukin, Jean-Frederic Colombel and Jordan E. Axelrad in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
