Abstract
Colorectal cancer (CRC) remains the second leading cause of global cancer-related mortality, with serrated colon neoplasia (SCN) accounting for 20%–30% of cases. SCN exhibits distinct clinicopathological and molecular features, whose superficial and pale appearance poses challenges for endoscopic detection. Linked Color Imaging (LCI) significantly improves lesion detection by enhancing color contrast through narrowband preprocessing and post-processing augmentation, demonstrating superior performance in large-lumen examinations. This review synthesizes current knowledge on the clinicopathological and molecular profiles of SCN, elaborates on the technical advantages of LCI over conventional imaging modalities, and highlights its diagnostic utility in SCN screening. Furthermore, we explore the innovative integration of LCI with artificial intelligence for real-time lesion recognition and with biomedical nanomaterials for targeted therapy, proposing a promising “detection–treatment” paradigm that may transform early CRC management.
Plain language summary
Colorectal cancer (CRC) is the second most common cause of cancer-related deaths globally, and serrated colon neoplasia is linked to 20% to 30% of cases. As a significant precancerous condition for CRC, serrated Colorectal cancer (CRC) remains the second leading cause of global cancer-related mortality, with serrated colon neoplasia (SCN) accounting for 20%–30% of cases. SCN exhibits distinct clinicopathological and molecular features, whose superficial and pale appearance poses challenges for endoscopic detection. Linked Color Imaging (LCI) significantly improves lesion detection by enhancing color contrast through narrow-band preprocessing and post-processing augmentation, demonstrating superior performance in large-lumen examinations. This review synthesizes current knowledge on the clinicopathological and molecular profiles of SCN, elaborates the technical advantages of LCI over conventional imaging modalities, and highlights its diagnostic utility in SCN screening. Furthermore, we explore the innovative integration of LCI with artificial intelligence for real-time lesion recognition and with biomedical nanomaterials for targeted therapy, proposing a promising “detection–treatment” paradigm that may transform early CRC management.
Keywords
Introduction
According to epidemiological data, colorectal cancer (CRC) is the third most common type of cancer worldwide and the second leading cause of cancer-related deaths. 1 CRC poses a significant burden on human health and presents a major challenge to healthcare systems in many countries. Numerous risk factors, including increased population longevity and unhealthy dietary habits, can significantly increase the incidence of CRC. 2 Previous studies suggest that approximately 20%–30% of CRC cases are associated with serrated colon neoplasia (SCN). 3 SCN, which serves as a precursor pathway to CRC, mainly consists of hyperplastic polyps (HP), traditional serrated adenoma (TSA), and sessile serrated lesions (SSL). 4 Furthermore, the serrated pathway plays a crucial role in the molecular onset and progression of CRC. 5 With unique histological and noticeable morphological traits, TSA is the least prevalent subtype of CRC precursors. Therefore, extending human longevity greatly depends on early detection and treatment of SCN.
Linked Color Imaging (LCI), developed by Fujifilm Corporation (Tokyo, Japan), is a revolutionary image-enhancing technology. It provides sufficient brightness to illuminate an extensive lumen. 6 The detection rate and diagnostic accuracy of gastrointestinal lesions are greatly enhanced by LCI, which was launched in 2014 and employs specialized pre and post-processing procedures to provide better and more accurate endoscopic images.7,8 LCI’s narrowband light and color enhancement technology significantly increases the contrast between lesions and normal mucosa, improving adenoma detection rates (ADR) and reducing polyp miss rates in the colon.9,10 LCI facilitates the diagnosis of superficial and mild lesions throughout the gastrointestinal system. It enables detailed visualization of microstructures and microvasculature on the mucosal surface.11,12 LCI has proven to be quite effective at detecting and diagnosing gastrointestinal lesions, particularly gastric cancer, and other malignant gastrointestinal tumors in their early stages. 13 Continued developments and wider applications will further enhance and promote the diagnostic utility of LCI in gastrointestinal disorders.
Early detection and treatment of CRC, particularly SCN, during the screening procedure are essential for maintaining a favorable prognosis. They also contribute to a better quality of life. LCI has shown significant clinical value in the early identification, precise differentiation, and therapeutic monitoring of SCN due to its exceptional image-enhancement features.14,15 In this review, we provide a thorough overview of the molecular and clinicopathological characteristics of SCN, the current state and developments of LCI, its applications in screening and diagnosis, and its potential in treating SCN when combined with artificial intelligence (AI) and biomedical nanomaterials.
Clinicopathological and molecular correlations of SCN
SCN has recently attracted considerable attention and is a fundamental pathway in the development of CRC. SCN, as a crucial precursor lesion of CRC, is characterized histologically by serrated structures in the colonic crypts. These structures are closely associated with decreased apoptosis and increased cell senescence in the crypt epithelial cells. 16 According to the World Health Organization (WHO) classification, serrated lesions are categorized into HP, SSL, TSA, and serrated adenomas-unclassified (Figure 1). 17 HP is the most common type of serrated lesion, accounting for approximately 80% of SCN. 18 SL plays a primary role as a precursor lesion in the serrated pathway. This pathway is driven by molecular mechanisms such as MLH1 promoter methylation and microsatellite instability (MSI) during progression to CRC. 18 In contrast, TSA is the rarest type of serrated polyp in the colorectum and exhibits substantial morphological and molecular heterogeneity. Its histological features include abundant eosinophilic cytoplasm, elongated and distorted nuclei, ectopic crypt formation, and cleft-like serrated structures. 19 A Japanese clinical study of 128 cases found that TSA often harbors various genetic alterations in the WNT and MAPK pathways. 20 Additionally, the observed correlation between morphological and genetic features indicates that TSA’s histological diversity reflects its underlying molecular heterogeneity. Molecular profiles of serrated lesions may vary significantly among different populations. Furthermore, a case study demonstrated that enormous pedunculated polyps may represent SSL in the rectum. This finding highlights the need for endoscopists to be aware of uncommon morphological variations of SSL during clinical practice. 21

Pathogenesis and progression of serrated colorectal neoplasia. The transition from normal colonic epithelium to serrated neoplasia is influenced by a combination of innate and external factors. This process can progress through various precursor lesions, including HP, SSL, and TSA, driven by specific molecular alterations.
Recent fecal metabolomic studies have revealed notable distinctions between the metabolic profiles of patients with HP and those with typical adenomas. The metabolomic signature of HP is characterized by an enrichment of glycerophospholipids and alterations in xanthine, purine, and pyrimidine metabolic pathways. In contrast, the metabolomic profile of typical adenomas shows enrichment in exogenous disaccharides and phenolic compounds. 22 An immunological analysis of 74 SCN cases revealed that the serrated neoplasia pathway development correlates with intraepithelial lymphocyte density and PD-1/PD-L1 expression. 23 A cohort study at Massachusetts General Hospital showed that patients undergoing TSA removal had an increased risk of developing CRC, particularly within the first 3 years post-resection. 24 Clarifying the distinctions between SCN and the typical adenoma-carcinoma sequence is especially important. This is due to the distinct clinical, pathological, and molecular features of this condition. The traditional adenoma-carcinoma sequence is primarily driven by mutations in the APC gene and activation of the Wnt signaling pathway. On the other hand, the serrated pathway is characterized by BRAF mutations, the CpG island methylator phenotype (CIMP), and MLH1 methylation. 25 Serrated lesions are identified by their serrated architecture, while lesions in the traditional adenoma-carcinoma sequence typically exhibit tubular or villous structures. 25 Additionally, lesions in the traditional adenoma-carcinoma sequence are more commonly found in the distal colon and rectum, whereas serrated lesions are more frequently located in the proximal colon anatomically. 26 These unique characteristics imply that the traditional adenoma-carcinoma sequence and the serrated pathway may cause CRC through distinct mechanisms.
The analysis of molecular markers during SCN progression provides important insights for clinical diagnosis and treatment. Daiki Hirano and colleagues classified early-stage SCN into distinct molecular subtypes. They also elucidated the clinicopathological features and genetic alterations associated with each subtype. 27 According to a previous Korean study, HPs in the proximal colon of the Korean population exhibit distinct molecular characteristics compared to TSA and SSL, especially regarding BRAF mutations and aberrant DNA methylation. 28 Detecting BRAF mutations and high-level CIMP facilitates early diagnosis of high-risk serrated lesions, enabling prompt and effective intervention. 29 Previous research confirmed that Annexin A10 expression is associated with serrated pathway features in microsatellite unstable CRC. 30 A separate study from Korea revealed that neurotrophic receptor tyrosine kinase oncogenic fusions are exclusively linked to the serrated neoplasia pathway in the colorectum. These fusions typically originate in SSLs. 31 Furthermore, detecting MLH1 methylation and MSI can help assess lesion invasiveness and prognosis. 32 Moreover, a single-cell transcriptomic analysis from China uncovered early molecular and immune alterations linked to the serrated tumor pathway leading to CRC. 33 Additionally, a study in the United States found that SSL and HP can be differentiated by platform-independent gene expression profiles. 34 A recent retrospective study from Japan found that TSA typically carry BRAF mutations but not KRAS mutations and exhibit several distinct endoscopic findings, including sessile morphology, absence of a pine-cone or coral-like appearance, pale red color, and a mucus cap. 35 As molecular diagnostic technologies advance, molecular marker-based diagnostic tools are expected to become more common in clinical practice, improving early detection and management of SCN.
Basics and characteristics of LCI
Due to the unique clinical, pathological, and molecular phenotypes of SCN, its endoscopic manifestations are often subtle and prone to being missed. Therefore, the imaging principle of LCI makes it particularly suitable for identifying SCN through enhanced color contrast. 17 Endoscopy has long been recognized as a vital diagnostic tool for gastrointestinal disorders. Chromoendoscopy, known for improving diagnostic accuracy, has historically played a pivotal role in this field. 36 An excellent illustration is indigo carmine staining, a popular and successful chromoendoscopic method for defining lesion borders and mucosal surface patterns. 37 As medical technology rapidly advances, image-enhanced endoscopy (IEE) has emerged as a groundbreaking technology, steadily leading gastrointestinal diagnostics. With major improvements in image contrast, resolution, and color distinction, IEE technology has transformed diagnostic methods, enabling more accurate lesion detection and significantly improving diagnostic efficiency and accuracy. 38 Among the various IEE technologies, narrowband imaging (NBI), blue laser imaging (BLI), and LCI are the most prominent and clinically significant. 39 While each possesses unique characteristics, they collectively aim to optimize endoscopic visualization. Notably, LCI has become Fujifilm’s signature IEE technology, distinguishing it from competitors. Developed by Fujifilm in 2014, LCI was hailed as a groundbreaking diagnostic technique that could deliver remarkable brightness and color contrast in wide-lumen settings. 40 This unique feature ensures exceptional performance across various endoscopic settings, consistently delivering sharp images under all lighting conditions.
LCI is based on the characteristics of lasers at short wavelengths (Figure 2). At short wavelengths (such as 410 and 450 nm), the laser travels a short distance to reach the mucosal surface layer and is specifically absorbed by hemoglobin. This results in high-contrast imaging. 41 This high contrast allows LCI to clearly detect mucosal surface morphology, including microstructures, and emphasize subtle color differences. The subtle color differences may arise from structural disparities, histological variations, mucosal blood flow, and mucus content within the gastrointestinal mucosa. 42 Different tissue structures exhibit distinct light reflection and absorption characteristics. Moreover, changes in mucosal blood flow affect mucosal color, and mucus content also influences the image. LCI can sensitively capture these subtle differences and translate them into clear, enhanced image information. This capability provides strong support for physicians’ diagnosis. At a wavelength of 410 nm, LCI can simultaneously highlight the microstructures and microvessels in the superficial layer of the mucosa, making it easier for endoscopists to identify suspicious lesions during screening. 43 In addition, the digital image processing in the LCI system focuses on subtle color differences and enhances color contrast, especially in the red range. 44 By amplifying the intensity of red and white, the color contrast is increased while preserving the natural colors, resulting in areas that appear redder and whiter. This processing method produces more vivid and distinct images, helping physicians more accurately assess the nature and extent of lesions. The increased color contrast is like wearing enhancement glasses, making lesions more prominent in the images and facilitating their detection, inflammation identification, and more accurate delineation.

(a) The International Commission on Illumination 1976 L*a*b* color space. (b) Efficient screening of patients was conducted using Linked color imaging with the special preprocess and post-process.
Previous studies have shown that LCI overcomes image blurring caused by the large distance within the gastric cavity. Even during long-distance observation in large lumens such as the stomach, LCI can effectively screen gastrointestinal lesions. 45 A systematic review and meta-analysis from Korea revealed that LCI exhibited higher sensitivity in the endoscopic diagnosis of Helicobacter pylori (H. pylori) infection compared to standard white light imaging (WLI). 46 This implies that LCI can detect H. pylori infection more accurately, enabling earlier patient treatment. Kitagawa et al. 47 confirmed in the previous research that LCI significantly improved the visibility of early gastric cancer after H. pylori eradication and reduced the missed diagnosis rate of these lesions compared to WLI. Helicobacter pylori infection is a major risk factor for gastric cancer, and its eradication reduces cancer risk. However, detecting early gastric cancer after eradication remains challenging. LCI technology provides a new method to overcome these detection difficulties. A multicenter prospective study further supported that LCI greatly enhanced the visibility and detection rate of gastric cancer. This improvement was observed when considering lesion size and endoscopic intestinal metaplasia. 48 LCI images share the same wavelength spectrum as blue laser imaging-bright mode (BLI-BRT), but include additional post-processing. This additional post-processing enhances LCI images by improving color contrast and lesion visibility. A study from Japan indicated that in the absence of background staining, LCI may make superficial esophageal squamous cell carcinoma more apparent than BLI-BRT. 49 With an expanded color range, LCI better distinguishes subtle mucosal color differences, aiding the diagnosis of cancerous versus noncancerous tissue. A key feature of LCI is its enhanced color contrast, which helps distinguish suspicious lesions from surrounding mucosa. A randomized clinical trial from Japan revealed that LCI mode was more effective and clearer than WLI in delineating tumor lesions in the pharynx, esophagus, and stomach. 50 LCI is also useful for detecting gastritis, assessing gastric mucosal intestinal metaplasia as a precancerous lesion, and identifying early gastric cancer. A systematic review and meta-analysis demonstrated that LCI improves the diagnostic efficacy for H. pylori infection, 51 highlighting its broad application and significant value in diagnosing digestive system diseases. LCI enables clear visualization of structures with an ultrathin scope, without the need for magnification. By utilizing LCI, it has become possible to diagnose inflammatory changes that are difficult to distinguish from cancer. Previous case reports and studies have reported that LCI is easier to identify early gastric cancer compared to traditional WLI technology. 6 Lu et al. 52 found that LCI could narrow the gap in the detection rate of high-grade gastric intraepithelial neoplasia between junior and senior endoscopists. LCI combines a unique imaging principle, advanced digital signal and image processing technology, and an expanded color range to provide physicians with clearer and more accurate diagnostic images. This advancement improves diagnostic accuracy and efficiency in digestive system diseases, strongly supporting early diagnosis and treatment.
The application of LCI in the diagnosis and screening of SCN
SCN, an important precancerous lesion of CRC, plays a crucial role in the early detection and prevention of CRC. Early identification of SCN allows timely interventions to prevent malignant transformation, significantly reducing CRC incidence and mortality. LCI, an emerging endoscopic technique, has shown clear advantages in detecting SCN and is gaining increasing attention from the medical community (Figure 3). Table 1 summarizes the randomized controlled trials (RCTs) comparing LCI with other methods for colon examination53,54 (Table 1). A past study revealed that LCI was the most sensitive modality for detecting SSL, outperforming BLI and BLI-bright (BLI-b) in both static image analyses and prospective RCTs. 55 This advantage likely arises from LCI’s unique imaging principle. A systematic review and meta-analysis confirmed the superiority of LCI in detecting polyps and adenomas. Compared to WLI, LCI has a higher detection rate for polyps, including previously missed adenomas. 56 By enhancing color contrast, LCI facilitates the detection of minute lesions that are hard to discern under WLI. These subtle lesions have important clinical implications in early stages; timely detection and management can effectively prevent CRC development.
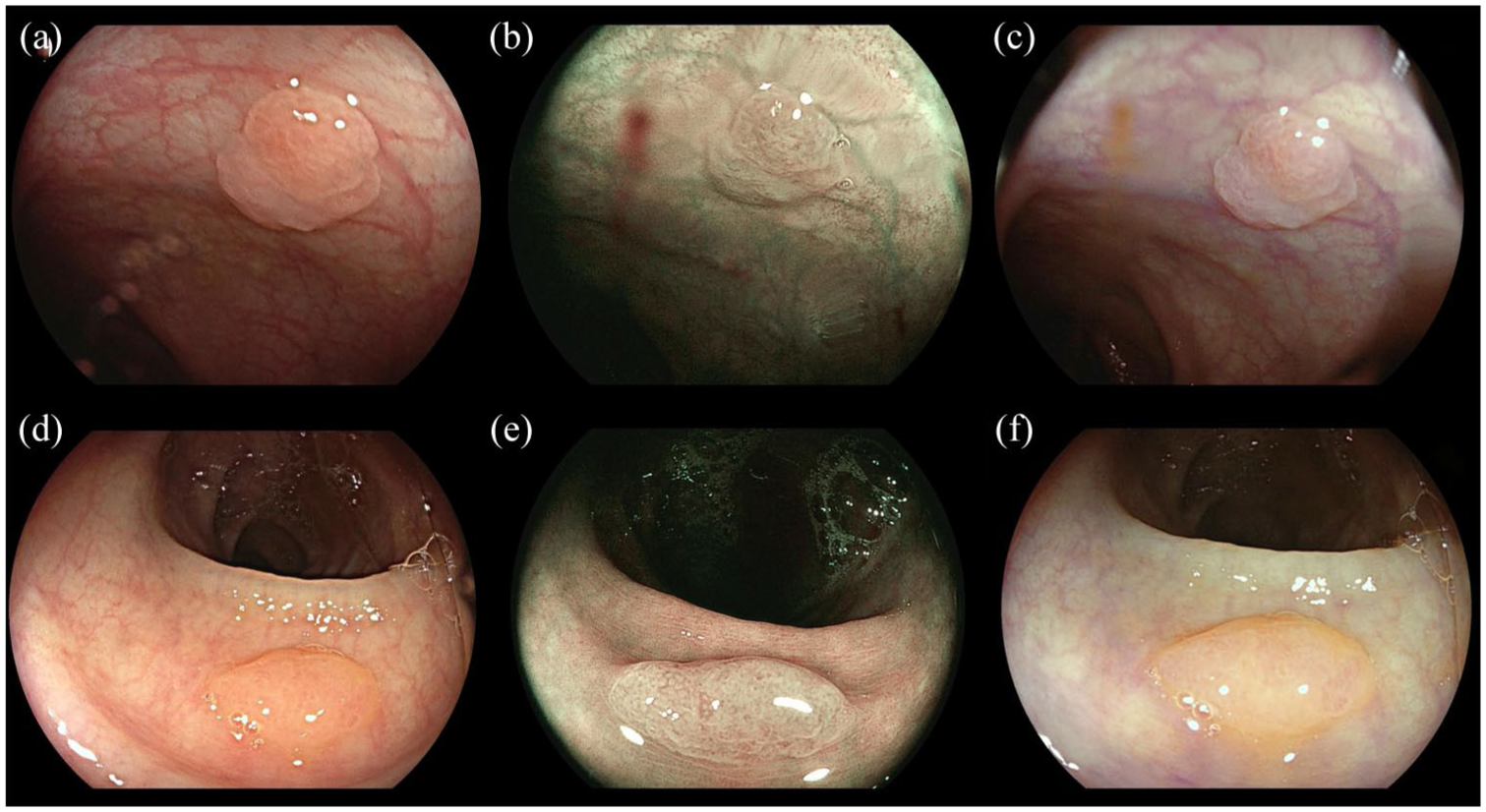
Comparison of WLI, BLI, and LCI in the visualization of serrated colon neoplasia. (a, d) WLI images of serrated lesions in two patients. Under conventional white light, the lesions exhibit subtle color differences and marginally indistinct borders against the surrounding mucosa. (b, e) BLI images of the same lesions. BLI enhances mucosal surface patterns and microvascular details through narrowband laser illumination, offering improved contrast compared to WLI. (c, f) LCI images demonstrating significant improvement in lesion detectability. The linked color processing algorithm markedly enhances color contrast and border sharpness (reddish lesions against surrounding mucosa), facilitating clearer delineation and easier recognition of serrated neoplasia compared to WLI and BLI.
The summary of randomized controlled trials comparing LCI with other methods for colon examination.

ADR, adenoma detection rate; APC, adenoma per colonoscopy; ASDR, adenoma and SSA/P detection rate; BLI, blue-laser imaging; BLI-b, BLI-bright; CI, confidence interval; LCI-A, linked color imaging-add 30 s; NBI-A, narrowband imaging-add 30 s; SSA/P, sessile serrated adenoma/polyp; SSL, sessile serrated lesions; WLI, white light imaging; WLI-A, WLI-add 30 s.
Due to its special anatomical location and physiological environment, the detection of lesions in the right-sided colon is relatively challenging. However, a study from Italy involving randomized tandem colonoscopy demonstrated that LCI reduces, to a certain extent, the miss rate of tumorous lesions in the right-sided colon. 57 This improvement is likely due to the color-enhancing function of LCI, which facilitates the discovery of lesions easily overlooked under conventional imaging, particularly those with pale colors or atypical morphologies. This significantly improves early diagnosis of right-sided colon lesions, allowing patients to receive timely treatment. A meta-analysis of an RCT revealed that LCI had more advantages compared to WLI in terms of the detection efficacy for adenomas, SSL, serrated lesions, and advanced adenomas. 58 Nevertheless, a recent multicenter tandem RCT reported no significant difference in the miss rates of SSL, advanced adenomas, and polyps in the proximal colon between WLI and LCI. 59 However, Yoshida et al. 60 found that an additional 30-s observation with LCI improved the detection of adenomas and SSL in the right-sided colon. In addition, Hashimoto et al. showed that lesion detection in the right-sided colon did not differ between Add-30-s LCI and NBI overall. Add-30-s observations with both LCI and NBI significantly improved the adenoma and SSL detection rate and ADR. 61 This suggests that spending more observation time on LCI may help detect lesions often missed during routine checks. The extra observation time allows endoscopists to observe the subtle changes in the mucosa more carefully. When combined with LCI’s color-enhancing function, the extra observation time can greatly improve lesion identification.
The inconsistency of the conclusions from different studies further highlights the complexity and multiple influencing factors involved in evaluating this enhanced imaging technique, LCI. Firstly, the operational experience and proficiency of endoscopists are key variables. The interpretation of LCI image features requires a specific learning curve, and experienced physicians can more effectively utilize their color contrast advantages, while novices may not be able to fully exert their efficacy. Secondly, the withdrawal time is a core confounding factor. Studies have shown that a sufficient withdrawal time (such as ⩾6 min) alone can improve ADR. 62 If the withdrawal time is not strictly defined and standardized in the research, the benefits brought by the longer observation time may be wrongly attributed to the LCI technology, and vice versa. Thirdly, the hardware and software differences of the equipment itself may also have an impact. The algorithms and color-rendering effects of the LCI mode can vary between manufacturers and even between different generations of endoscopy systems, which may influence color representation and lesion recognition. Finally, the characteristics of the lesions themselves, such as the SSL in the right colon, often appear flat, with blurred edges and low color contrast compared to the surrounding mucosa. This inherent high rate of missed diagnosis makes the incremental benefits of any technology fluctuate among different populations. Therefore, future research should ensure that all participating physicians are proficient in LCI technique through standardized training and follow uniform operating procedures (such as clearly defining and supervising the minimum withdrawal time) to minimize bias from human factors. Ultimately, it is necessary to design rigorous multicenter, large-sample, RCTs to control these variables and verify the true benefits and clinical value of LCI in the detection of right colon lesions more objectively and accurately.
The sigmoid colon is a common site for colorectal polyps. Observation during the insertion phase of colonoscopy is crucial for early polyp detection. A retrospective study confirmed the effectiveness and feasibility of LCI in enhancing the detection of colorectal polyps in the sigmoid colon, particularly during the insertion phase. 63 LCI provides clearer images during insertion, helping endoscopists promptly identify polyps in the sigmoid colon. An international clinical trial from Asia has demonstrated that LCI is beneficial for both experts and nonexperts, and is particularly advantageous for endoscopists with relatively lower detection performance when using WLI. 64 In clinical practice, less-experienced endoscopists vary in lesion detection ability; LCI can help them in improving detection rates and reducing missed diagnoses. Previous studies have suggested that cold snare polypectomy under LCI may be an effective treatment in reducing tumor residue. 65 LCI enhances color contrast, making the borders of polyps clearer. This helps endoscopists determine the scope of polyps more accurately, thereby reducing the risk of tumor residue during resection. Past research has indicated that the LCI virtual chromoendoscopy technique is superior to traditional high-definition white light colonoscopy in detecting colorectal polyps and adenomas. 66 Yoshida et al. 67 confirmed through a multicenter pilot study that LCI outperforms BLI-b in adenoma visibility and rapid observation. The clear images provided by LCI help endoscopists accurately assess the size, morphology, and characteristics of SCN, enabling more personalized and precise treatment plans that improve efficacy.
The improvement prospects of LCI in the treatment of SCN
Combined with AI
CRC screening technologies continue to evolve. LCI has shown significant advantages in the early detection and diagnosis of serrated colon neoplasms, mainly because of its unique color-enhancement capabilities. However, endoscopists’ experience largely determines the interpretation of LCI images, which leads to interobserver variability. Recently, integrating AI has significantly advanced the clinical application of LCI. AI’s potential in LCI image analysis focuses on lesion identification and risk stratification. A clinical randomized trial conducted in Japan revealed that LCI combined with a computer-aided detection system based on deep learning, can process real-time endoscopic video streams, automatically identify and mark the areas of suspected colorectal adenomas, thereby increasing ADR by 18.7% compared to LCI alone (p < 0.01). 68 Similarly, Zhao et al. employed a deep learning model based on the YOLOv5 object detection architecture combined with an active learning strategy. Through a convolutional neural network, they automatically extracted the mucosal color and texture features enhanced by color enhancement technology in LCI images, significantly improving the detection rate of early gastric cancer. The sensitivity and specificity reached 92.3% and 89.5%, respectively. 69 A diagnostic system for Barrett’s Esophagus was established using LCI and AI. This system achieved diagnostic accuracy comparable to that of senior clinical endoscopists, further validating AI’s reliability in analyzing LCI images. 70 A prior single-center prospective study indicated that the developed AI shows excellent capabilities in diagnosing H. pylori infection using both BLI-b and LCI. 71 Through deep learning algorithms, AI can analyze images acquired by LCI in real time, automatically marking suspicious lesions to assist endoscopists in quickly localizing and characterizing lesions. Yasuda et al. utilized an interpretable Support Vector Machine model and employed color space analysis combined with feature engineering to achieve automatic diagnosis of H. pylori infection status in LCI images. The accuracy, sensitivity, specificity, positive predictive value, and negative predictive value reached 87.6%, 90.4%, 85.7%, 80.9%, and 93.1%, respectively. 72 Furthermore, AI can integrate multimodal data, including patient history and genetic testing results. This integration allows the construction of a risk prediction model for malignant transformation of serrated lesions, which guides personalized follow-up strategies. Recent studies have demonstrated that AI, fine-tuned with specific datasets, can detect non-red and non-polypoid polyps under LCI. 73 In the treatment decision-making process for serrated lesions, the synergistic effect of LCI and AI can optimize the selection of surgical plans. For high-risk SSL, particularly those with dysplasia, AI can combine microscopic structural information from LCI to predict the depth of lesion invasion and the risk of lymph node metastasis. This assists endoscopists in determining whether endoscopic mucosal resection or surgical intervention is required.
Looking ahead, the integration of LCI and AI continues to face challenges in technical standardization and clinical validation. On the one hand, it is essential to establish unified LCI image databases and standardized AI training models to ensure diagnostic consistency across different devices and operators. On the other hand, large-scale prospective studies are needed to confirm how AI-assisted LCI diagnosis reduces CRC incidence and mortality. The cost-effectiveness of AI in serrated lesion screening should be further explored to balance healthcare resource allocation and patient benefits. The future development of AI models requires not only high accuracy but also the ability to provide a decision-making basis to ensure that endoscopists understand and trust the output, which is particularly important when dealing with ambiguous or unclear lesions.
Combined with biomedical nanomaterials
LCI has been widely applied for the early diagnosis of SCN. Combining LCI with biomedical nanomaterials has opened novel avenues for therapeutic interventions. Biomedical nanomaterials leverage their unique physicochemical properties, including targeted drug delivery and controlled drug release, to form an integrated “diagnosis-therapy” synergistic system with LCI. This system optimizes the entire management strategy for SCN.74,75
SCN exhibits heterogeneous characteristics, including lesion size, depth of invasion, and molecular subtypes. These factors limit the efficacy of traditional chemotherapy drugs and cause significant side effects. Through surface modification, nanomaterials precisely deliver chemotherapeutic agents or molecular-targeted drugs to the lesion site. 76 Li et al. 77 developed a novel multifunctional nanocarrier named ASCP (AuNRs@MS/CpG ODN@PEG-bPEI)/siPD-L1, which can enhance the gene silencing and immunotherapy effects through mild photothermal therapy, and significantly inhibit the growth of CRC. Yang et al. 78 developed a pH/glutathione dual-responsive nano-herb delivery system (PMDC NP) for targeted delivery of dihydroartemisinin, along with abundant carbon monoxide release, for enhanced immunotherapy of CRC. Sun’s research team combined nanotechnology with probiotic therapy to develop SeNVs@NE-IL32-EcN, which helps to enhance CD8 T-cell-mediated immunity and overcome the immune treatment resistance of CRC. 79 Previous studies have demonstrated that hyaluronic acid–manganese dioxide nanoparticles catalyze the decomposition of intratumoral hydrogen peroxide. This reaction generates oxygen, which alleviates hypoxia and promotes T-cell function. 80 In the future, combined with the real-time imaging capabilities of LCI, nanomaterials will be able to mark lesion boundaries preoperatively, facilitate local drug delivery via endoscopic injection or spraying during surgery, and monitor drug distribution and lesion regression postoperatively. 81 Guided by LCI imaging, nanomaterials can precisely accumulate in the lesion area, disrupt tumor microvasculature, induce apoptosis, and simultaneously minimize damage to surrounding normal tissues. 82 Combined with real-time LCI imaging, nanomaterials can dynamically monitor immunotherapy responses and enable timely treatment adjustments. Moreover, nanomaterials can precisely stratify lesion risk by integrating multimodal imaging with LCI. 83
Despite the immense potential of integrating LCI with nanomaterials, its clinical translation faces multiple challenges. First, the regulatory and safety science of biomedical nanomaterials must be advanced by conducting large-scale clinical trials and meticulously tracking both short- and long-term outcomes. Second, standardized operational protocols for the combined application of LCI and nanomaterials are essential to ensure consistent diagnostic and therapeutic outcomes across different devices and operators. For instance, developing a unified LCI-nanomaterial imaging database and training AI models to automatically recognize lesion features and optimize nanomaterial delivery pathways are necessary.
Conclusion
This review provides a comprehensive analysis of the clinicopathological and molecular features of SCN. It also discusses the current applications and future potential of LCI in diagnosing and managing this condition. As an emerging endoscopic imaging technology, LCI significantly improves the detection and diagnosis of SCN by enhancing color contrast. The integration of LCI with AI and biomedical nanomaterials is expected to further enhance its clinical utility in treating SCN. AI-assisted real-time image analysis can help endoscopists identify lesions more precisely, while nanomaterials enable targeted drug delivery and photothermal/photodynamic therapies, facilitating more precise and personalized treatment strategies. Despite challenges such as technical standardization and clinical validation, the combination of LCI, AI, and nanomaterials holds great promise for revolutionizing the early diagnosis and treatment of SCN. Future research should focus on developing and validating integrated “diagnosis-therapy” platforms in real clinical environments, as well as evaluating their usability, cost-effectiveness, and impact on the incidence and mortality of CRC.
