Abstract
Background:
The impact of dynamic changes in alpha-fetoprotein (AFP) levels on the prognosis of hepatocellular carcinoma (HCC) remains controversial.
Objectives:
This review aims to clarify the prognostic value of dynamic changes in AFP levels in patients with HCC treated with immune checkpoint inhibitors (ICIs).
Data sources and methods:
PubMed, EMBASE, Cochrane Library, and Web of Science were searched to collect eligible studies published up to July 29, 2024. The Newcastle–Ottawa Scale score was used to assess the quality of included studies, and Stata 15.1 statistical software was used for statistical analysis.
Results:
This review included 23 studies involving 2860 patients with HCC. Following treatment with ICIs, a dynamic decrease in AFP levels was significantly associated with improved overall survival (OS; hazard ratio (HR) 0.40, 95% confidence interval (CI), 0.34–0.48) and progression-free survival (PFS; HR 0.39, 95% CI, 0.34–0.45). Conversely, dynamically increased AFP levels were linked to poorer OS (HR 2.34, 95% CI, 1.69–3.23) and PFS (HR 2.62, 95% CI, 1.92–3.56). Subgroup analyses revealed that while the association with OS was influenced by factors such as treatment regimen and sample size, the prognostic value of AFP changes for PFS appeared more consistent across various subgroups.
Conclusion:
This review demonstrates that dynamic changes in AFP levels are significantly associated with OS and PFS in HCC patients treated with ICIs, particularly in those receiving combination ICI regimens.
Trial registration:
PROSPERO registration number CRD42024599795.
Plain language summary
The dynamic changes in alpha-fetoprotein (AFP) levels are an important tool for predicting the prognosis of hepatocellular carcinoma patients. Our meta-analysis showed that a dynamic decrease in AFP levels following treatment with immune checkpoint inhibitors (ICIs) was significantly associated with improved overall survival (OS) and progression-free survival (PFS). Conversely, dynamically increased AFP levels were linked to poorer OS and PFS. Due to the heterogeneity of the included studies and potential publication bias, these results should be interpreted with caution. High-quality prospective studies are needed in the future to validate these findings.
Introduction
Liver cancer is the sixth most common malignancy globally and represents the third leading cause of cancer-related mortality. Global cancer statistics from 2022 documented approximately 865,000 new cases and 757,948 deaths worldwide. 1 Hepatocellular carcinoma (HCC), comprising over 80% of liver cancer cases, carries a dismal prognosis with a 5-year survival rate of approximately 10%,2,3 primarily attributed to high recurrence rates and limited therapeutic efficacy.
Systemic therapies have substantially transformed HCC management, with approximately 50%–60% of patients now receiving such treatments. 4 The introduction of immune checkpoint inhibitor (ICI)-based combination regimens in 2020 marked a paradigm shift in HCC therapy, demonstrating promising survival benefits in selected patient populations. However, variable response rates to ICIs highlight the critical need for reliable predictive biomarkers. Identifying such markers is essential for advancing precision medicine and optimizing patient outcomes.
Alpha-fetoprotein (AFP), a glycoprotein synthesized by the fetal liver and yolk sac, remains at low concentrations in healthy adults but frequently becomes elevated in HCC patients. This characteristic has established AFP as a cornerstone biomarker for HCC diagnosis and monitoring. 5 Substantial evidence supports AFP’s utility in early detection6,7 and prognostication, with elevated levels correlating with adverse outcomes. 8 High preoperative serum AFP concentrations predict poorer postoperative survival, 9 while baseline levels exceeding 400 ng/ml are associated with significantly reduced survival compared to lower concentrations. 10 Additionally, dynamic AFP changes during treatment demonstrate strong prognostic associations. 11
Despite these findings, consensus regarding AFP dynamics as a predictive marker in ICI-treated HCC patients remains elusive. Initial meta-analyses suggested correlations between early AFP response and clinical outcomes, though these studies were limited by small sample sizes.12,13 Subsequent investigations yielded conflicting results,14,15 potentially reflecting heterogeneity in AFP response definitions and treatment protocols.
To resolve these inconsistencies, we conducted this comprehensive meta-analysis to evaluate the prognostic significance of dynamic serum AFP changes in HCC patients receiving ICI therapy. Through systematic synthesis of available evidence, we aimed to clarify AFP’s predictive utility and support the development of biomarker-guided treatment strategies to improve overall survival (OS) and quality of life in this patient population.
Materials and methods
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for systematic reviews and meta-analyses 16 (File S1). The study protocol was registered with the International Prospective Register of Systematic Reviews (CRD42024599795).
Search strategy
A comprehensive search was performed across PubMed, Embase, the Cochrane Library, and Web of Science databases, encompassing all publications up to July 29, 2024, and was limited to English-language articles. The search strategy combined Medical Subject Headings (MeSH) terms and free-text keywords, specifically “alpha-fetoprotein,” “hepatocellular carcinoma,” and “survival.” Detailed search strategies are provided in File S2. To ensure thoroughness, reference lists of relevant systematic reviews and pertinent articles were also examined.
Inclusion and exclusion criteria
Eligible studies for this meta-analysis were required to: (1) include patients diagnosed with HCC who had received treatment with ICIs, whether as monotherapy or in combination with other treatments; (2) report on dynamic changes in AFP levels (defined as per the criteria of each primary study, typically an AFP increase or AFP decrease relative to baseline, or stable AFP levels serving as the reference); (3) be observational in design; and (4) provide survival outcomes, specifically progression-free survival (PFS) and OS.
Studies were excluded if they involved animal or in vitro experiments, were laboratory-based studies, case reports, reviews, meta-analyses, conference abstracts, letters, or similar types of publications. Additionally, studies were excluded if they contained incomplete or erroneous data, were duplicate publications, or if the full text was inaccessible.
Data extraction and quality assessment
The selected articles were imported into EndNote, after which two researchers (Y.Z. and J.-L.G.) independently screened the titles and abstracts based on the predefined inclusion and exclusion criteria. Subsequently, the full texts of potentially relevant studies were reviewed in a second screening round. Any discrepancies between the researchers were resolved through discussion or by consulting a third researcher (J.-J.L.) for further evaluation. Data extraction from the final included studies was performed independently by two researchers using Excel 2019 (Microsoft Corporation, Redmond, WA, USA). The extracted data encompassed the first author, publication year, country, study design, sample size, gender distribution, age, HCC classification, tumor staging, treatment regimen, baseline AFP levels, definitions of AFP response (including criteria for AFP increase, AFP decrease, and stable AFP levels), timing of AFP response assessment, data sources, outcome measures, and hazard ratios (HRs) with their corresponding 95% confidence intervals (CIs). The reference group for assessing AFP changes, generally termed “stable AFP levels,” was adopted as defined by each primary study. These definitions for what constituted “stable” or “no significant change” in AFP levels (i.e., changes not meeting the predefined thresholds for an increase or decrease) typically varied across the included literature, and this variability was noted during data extraction. For studies reporting both univariate and multivariate HR analyses, multivariate results were prioritized. Attempts were made to contact the authors of primary studies via email for critical missing data or clarification if required; however, no responses providing additional data were received during the study period.
Study quality was assessed using the Newcastle–Ottawa Scale (NOS), 17 which evaluates three domains: selection of exposed and non-exposed cohorts (4 points), cohort comparability (2 points), and outcome assessment (3 points). Each cohort study could achieve a maximum of 9 points, with studies scoring above 6 points deemed to be of high quality.
Statistical analysis
Statistical analyses were performed using Stata 15.1 (StataCorp, College Station, TX, USA). The results of the meta-analysis were illustrated through forest plots to estimate the overall HR. Heterogeneity among the included studies was quantitatively evaluated using the I2 statistic. Significant heterogeneity was prespecified as an I2 value >50% or a p-value <0.10 for the Chi-squared test. In such cases, a random-effects model (DerSimonian and Laird method) was employed to synthesize the results; otherwise, a fixed-effects model (Mantel-Haenszel method) was used if heterogeneity was not substantial I2 ⩽ 50% and p ⩾ 0.10). To explore potential sources of any identified heterogeneity and to assess the consistency of effects across different study characteristics, subgroup analyses were conducted based on prespecified factors. Sensitivity analyses were also performed by systematically excluding individual studies one at a time to assess the robustness of the overall findings. Publication bias was evaluated using Egger’s test and visual inspection of funnel plots. A two-sided p-value of less than 0.05 was considered statistically significant for all analyses unless otherwise specified.
Results
Literature search and screening process
A comprehensive search retrieved 27,114 articles. After the removal of 18,839 duplicates, an initial screening of titles and abstracts led to the exclusion of 18,756 articles. The remaining studies were assessed in full, and based on the inclusion and exclusion criteria, 23 studies were ultimately included in the final analysis.15,18–39 The detailed screening process is illustrated in Figure 1.
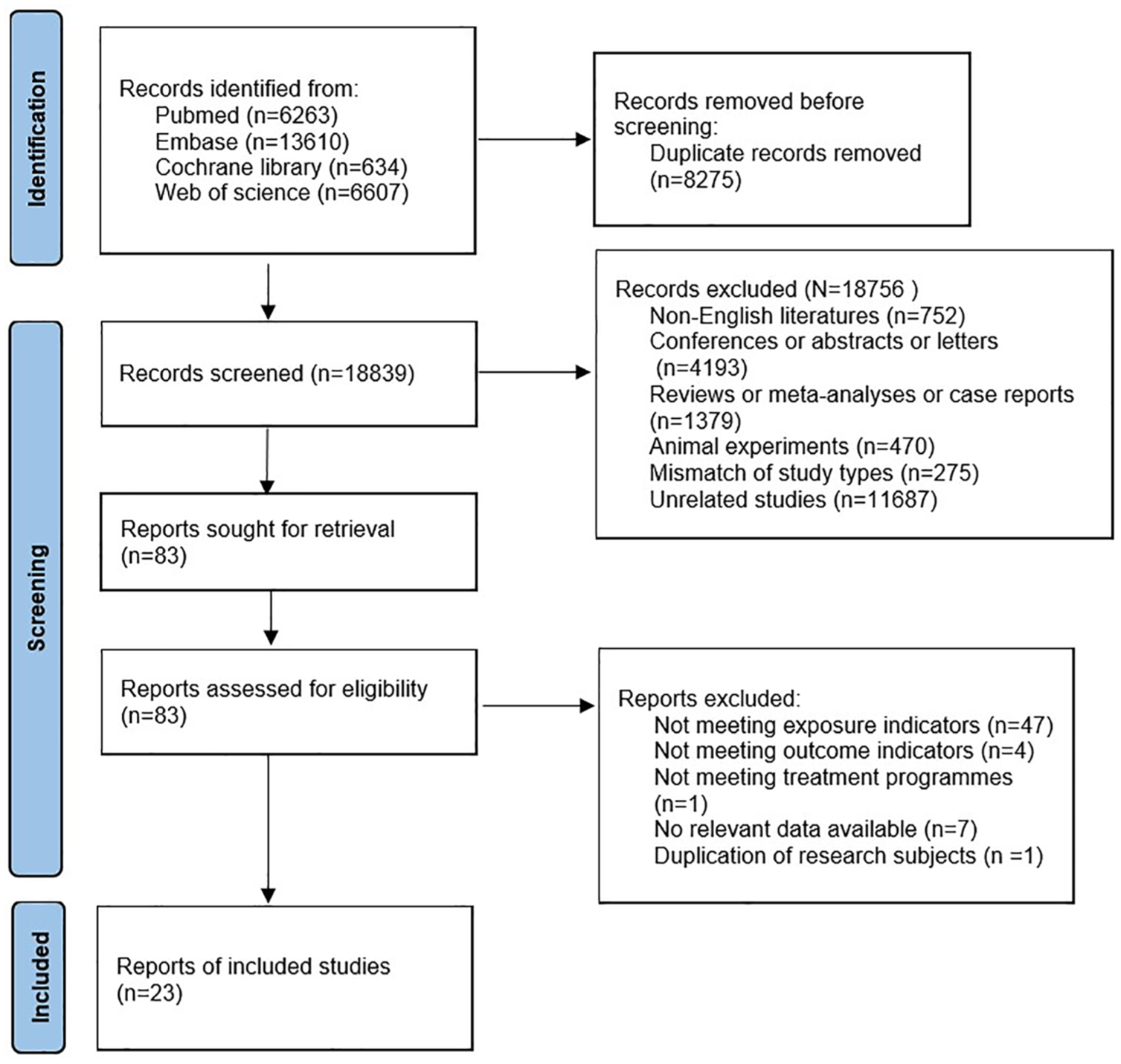
Flow diagram of the literature screening process.
Basic characteristics and quality assessment of included studies
The 23 included studies were published between 2019 and 2024. Seventeen studies originated from China,15,18–24,26,30,31,33,34,36–39 two27,32 from Japan, one each from Korea 25 and the United States, 35 and the remaining two28,29 were multinational, multicenter studies. Collectively, these studies encompassed 2860 patients, comprising 2409 males and 451 females. Twenty-one studies15,18–29,31–38 reported the mean or median age of participants, ranging from 51 to 74 years. Among the included studies, two18,23 utilized ICIs alone, while the others employed combination ICI therapy. The basic characteristics of the included studies are summarized in Table 1.15,18–39 Quality assessment using the NOS yielded scores between 7 and 9 points, indicating that all studies were of high quality (File S3).
Basic characteristics of the included studies.

AFP, alpha-fetoprotein; BCLC, Barcelona Clinic Liver Cancer; F, female; HCC, hepatocellular carcinoma; ICIs, immune checkpoint inhibitors; IQR, interquartile range; M, male; NA, not available; OS, overall survival; PCS, prospective cohort study; PFS, progression-free survival; RCS, retrospective cohort study.
Meta-analysis results
Overall survival
Eighteen studies, involving 1984 patients, were included in the analysis of the association between an AFP decrease and improved OS. Heterogeneity analysis revealed low heterogeneity I2 = 20.5%, p = 0.195); thus, a fixed-effects model was applied. The pooled results demonstrated that patients with an AFP decrease had significantly better OS compared to those with stable AFP levels (HR 0.40, 95% CI, 0.34–0.48, p < 0.001; Figure 2 and Table 2).

Forest plot showing the relationship between the AFP dynamic decrease and OS.
Analysis of the impact of AFP dynamic changes on OS.

AFP, alpha-fetoprotein; CI, confidence interval; HR, hazard ratio; ICIs, immune checkpoint inhibitors; NA, not available; OS, overall survival.
Five studies encompassing 993 patients were included in the analysis of AFP increase associated with prognosis. Heterogeneity analysis showed no significant heterogeneity (I2 = 0.0%, p = 0.520), supporting the use of a fixed-effects model. The pooled results revealed that patients with an AFP increase had significantly poorer OS compared to those with stable AFP levels (HR 2.34, 95% CI, 1.69–3.23, p < 0.001; Figure 3 and Table 2).

Forest plot showing the relationship between AFP dynamic increases and OS.
Progression-free survival
Eighteen studies involving 1870 patients were included in the analysis of AFP decrease associated with PFS. The heterogeneity analysis showed no significant heterogeneity I2 = 0.0%, p = 0.905), allowing for the application of a fixed-effects model. The pooled results demonstrated that patients with an AFP decrease had significantly better PFS compared to those with stable AFP levels (HR 0.39, 95% CI, 0.34–0.45, p < 0.001; Figure 4 and Table 3).
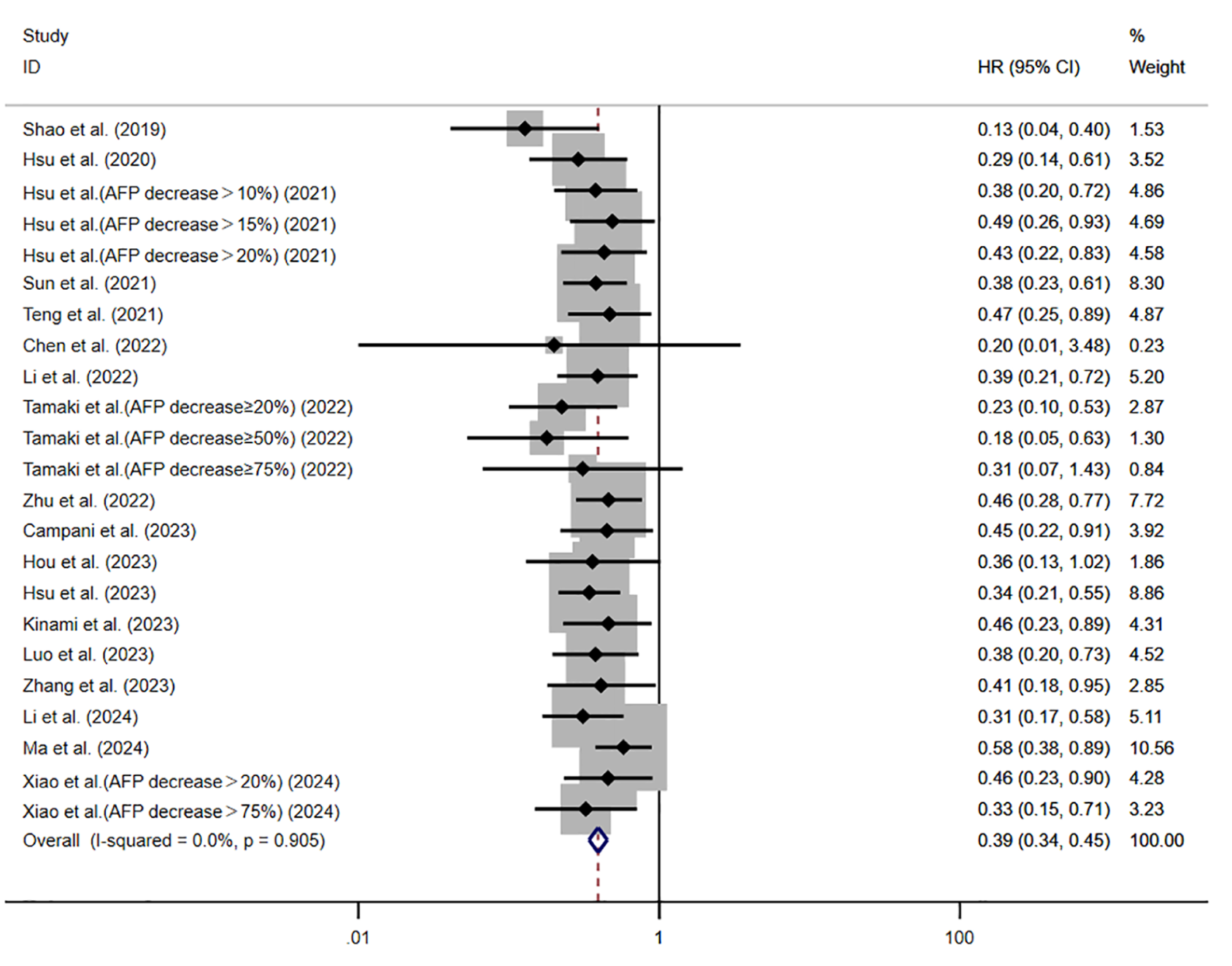
Forest plot showing the relationship between AFP dynamic decrease and PFS.
Analysis of the impact of AFP dynamic changes on PFS.

AFP, alpha-fetoprotein; CI, confidence interval; HR, hazard ratio; ICIs, immune checkpoint inhibitors; NA, not available; PFS, progression-free survival.
Additionally, three studies involving 776 patients were analyzed to assess the impact of AFP increase on PFS. The heterogeneity analysis again revealed no significant heterogeneity I2 = 0.0%, p = 0.395), supporting the use of a fixed-effects model. The pooled results indicated that patients with an AFP increase had significantly poorer PFS compared to those with stable AFP levels (HR 2.62, 95% CI, 1.92–3.57, p < 0.001; Figure 5 and Table 3).
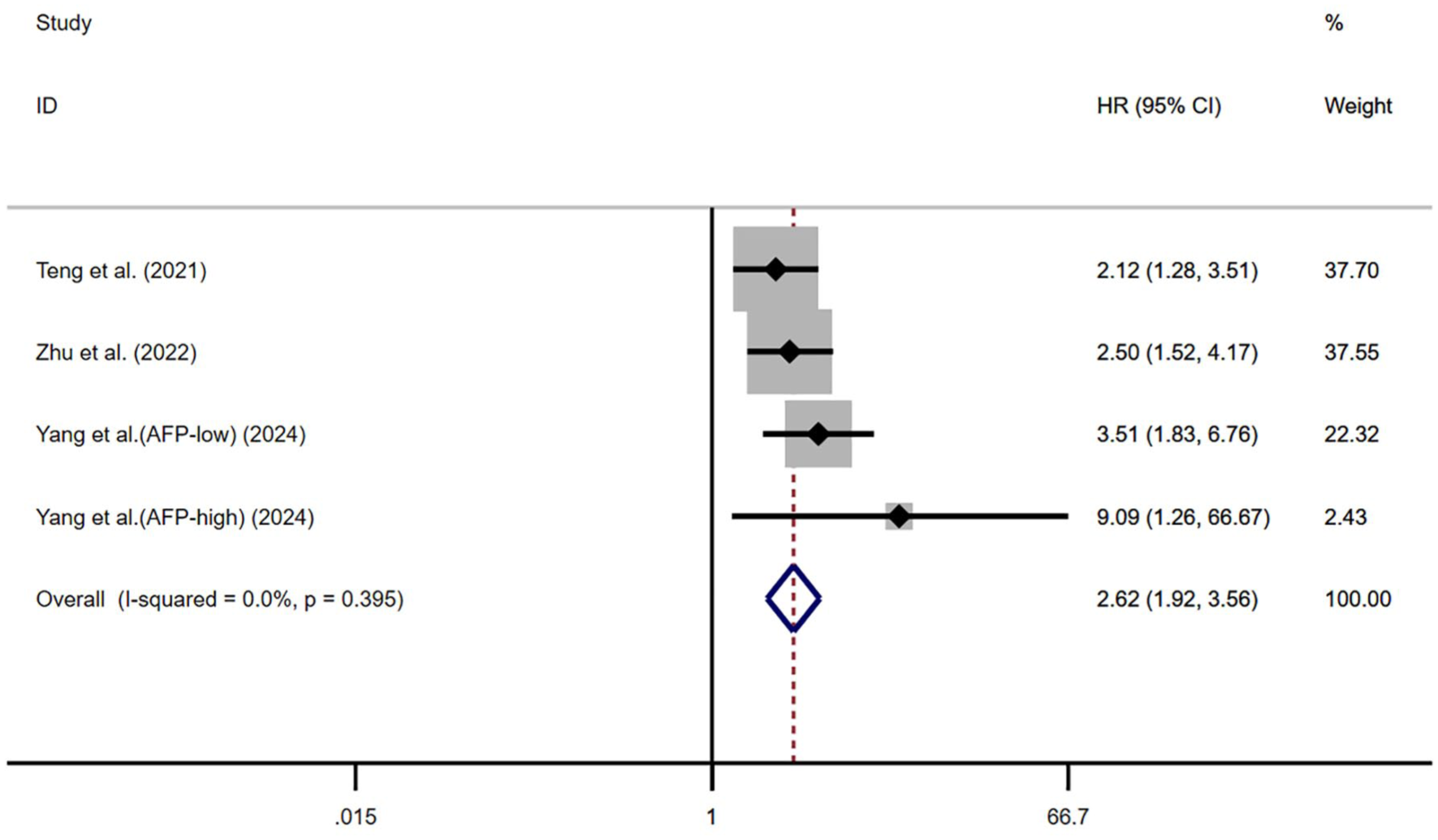
Forest plot showing the relationship between AFP dynamic increases and PFS.
Subgroup analysis
Although overall heterogeneity for OS and PFS in the meta-analysis of dynamic AFP changes was low, we conducted subgroup analyses to explore potential influences from clinical characteristics such as region, sample size, treatment regimen, data source, timing of AFP response assessment, and AFP response cutoff values. Subgroup analyses were pre-specified based on factors with known or suspected influence on HCC prognosis or ICI treatment effect.
Overall survival
Subgroup analyses stratified by geographic region, sample size, data source, timing of AFP response assessment, AFP response cutoff values, and treatment modality showed that an AFP decrease was consistently correlated with improved OS compared to no changes in AFP levels. However, this correlation was not observed in patients treated with ICI monotherapy (File S4 and Supplemental Figure 1(A)–(F); Table 2).
Conversely, among patients with baseline AFP elevation, subgroup analyses stratified by region, timing of AFP response assessment, sample size greater than 100, treatment regimen (combination therapies), data source (univariate vs multivariate analysis), and cutoff values of 20%, 10%, or unspecified thresholds indicated that an AFP increase was significantly associated with poorer OS. However, this association was not significant in subgroups with sample sizes of 100 or fewer, those receiving ICI monotherapy, studies based on survival curve analysis, or when cutoff values were set at 50% or 25% (see File S4 and Supplemental Figure 2(A)–(F); Table 2).
Progression-free survival
Subgroup analyses for PFS, considering region, sample size, treatment regimen, data source, timing of AFP response assessment, and AFP response cutoff values, demonstrated that an AFP decrease was significantly associated with better PFS, whereas an AFP increase correlated with poorer PFS (see File S4 and Supplemental Figures 3(A)–(F) and 4(A)–(F); Table 3).
Sensitivity analysis and publication bias
Sensitivity analyses, conducted by omitting one study at a time, demonstrated that the pooled HRs for OS and PFS associated with AFP changes remained robust, and no single study unduly influenced the overall significant results (Figures 6–9). Egger’s test did not detect significant publication bias for the association between AFP increase and OS (p = 0.298) or PFS (p = 0.131). In contrast, while Egger’s test showed no significant publication bias for AFP decrease and OS (p = 0.123), a significant publication bias was identified for AFP decrease and PFS (p = 0.005). The trim-and-fill method was applied to assess the potential impact of this asymmetry; while this method did not impute any studies, the presence of publication bias suggested by Egger’s test cannot be dismissed. Therefore, the pooled estimate for AFP decrease and PFS should be interpreted with caution. (Figures 10–13).

Sensitivity analysis of the effect of AFP dynamic decrease on OS.

Sensitivity analysis of the effect of AFP dynamic increases on OS.
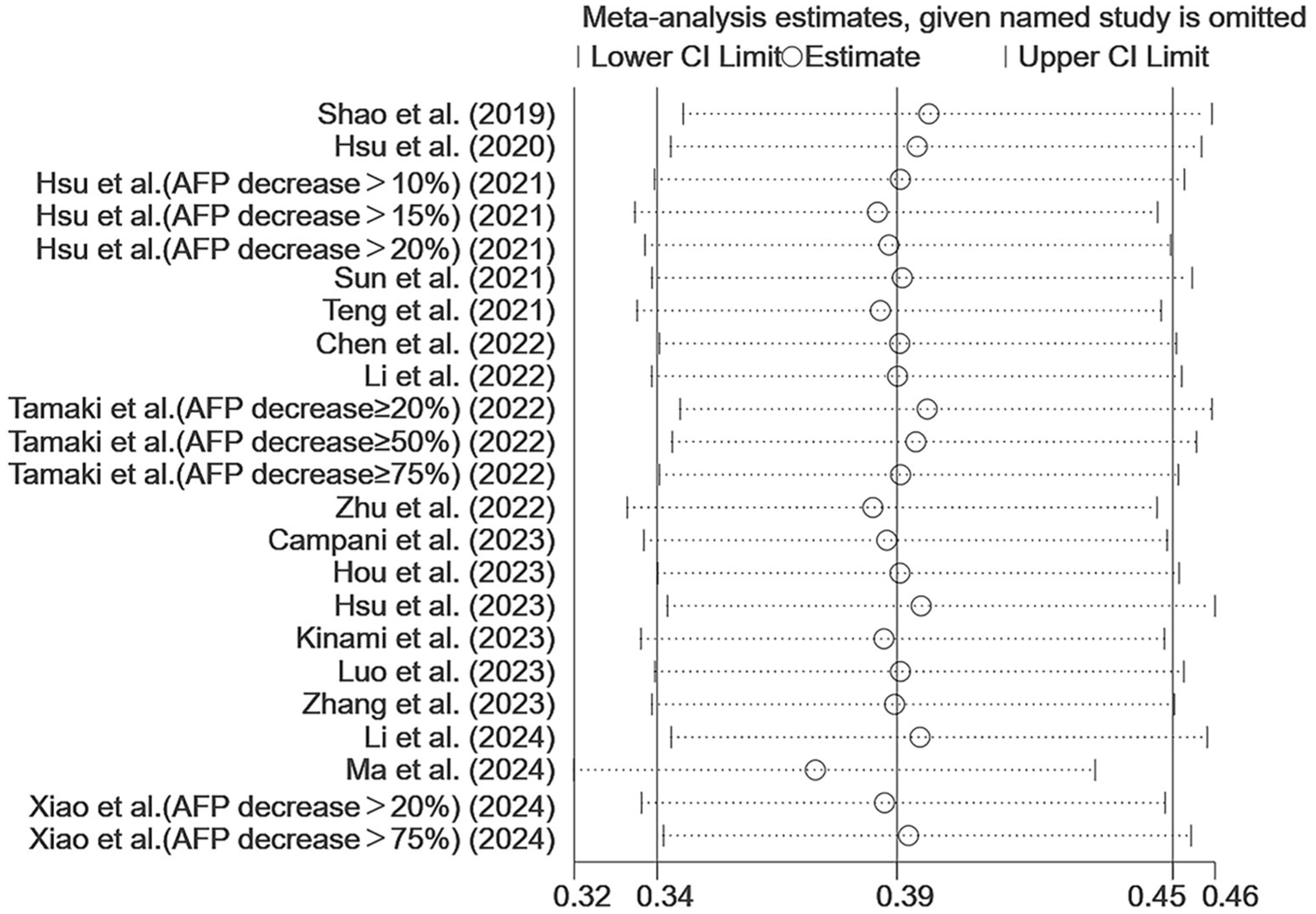
Sensitivity analysis of the effect of AFP dynamic decrease on PFS.

Sensitivity analysis of the effect of AFP dynamic increases on PFS.

Funnel plot for the effect of AFP dynamic increases on OS.

Funnel plot for the effect of AFP dynamic increases on PFS.

Funnel plot for the effect of AFP dynamic decrease on OS.

Funnel plot for the effect of AFP dynamic decrease on PFS.
Discussion
This meta-analysis, to our knowledge, is the first comprehensive and systematic examination of dynamic changes in AFP and their association with prognosis in HCC patients undergoing treatment with ICIs. Our analysis of 23 eligible studies demonstrated that an AFP decrease correlated with improved OS and PFS, with PFS remaining independent of region, sample size, treatment regimen, data source, timing of AFP response assessment, or AFP response cutoff values. In contrast, OS was affected by the treatment regimen. Conversely, an AFP increase was associated with poorer OS and PFS. While PFS remained unaffected by the aforementioned factors, OS was influenced by sample size, treatment regimen, data source, and AFP response cutoff values.
Our conclusions align with previous research, 12 which also reported that an AFP decrease is linked to better OS and PFS, whereas an AFP increase predicts poorer outcomes compared to baseline levels. Additionally, some studies have identified that higher baseline AFP levels in HCC patients receiving ICIs are indicative of a poor prognosis.40,41
AFP is a widely utilized serum biomarker in the clinical management of HCC. 42 While this meta-analysis did not directly investigate biological pathways, the observed prognostic associations of AFP changes can be considered in the context of AFP’s known biological roles in HCC. Research has demonstrated that AFP stimulates cell proliferation by activating the protein kinase B (AKT) signaling pathway and its downstream target, mammalian target of rapamycin (mTOR), thereby promoting metastasis through the activation of components essential for epithelial-mesenchymal transition. 43 Furthermore, AFP evades anti-tumor immunity by suppressing dendritic cells, natural killer cells, T lymphocytes, and macrophages.44,45 Ma et al. reported that AFP activates the vascular endothelial growth factor (VEGF) pathway, potentially inhibiting anti-tumor immune responses by modulating immune cell function and infiltration. 46 AFP also suppresses programmed cell death in tumor cells. Wang et al. demonstrated that in HCC, AFP inhibits phosphatase and tensin homolog (PTEN) activity, activating the phosphoinositide 3-kinase (PI3K)/AKT/mTOR pathway and upregulating mTOR expression, thereby suppressing autophagy and promoting malignant behavior. 47 Yang et al. showed that AFP inhibits apoptosis through the tumor protein 53 (p53)/Bax/cytochrome c/caspase-3 signaling pathway. 48 Elevated AFP indicates increased tumor invasiveness, while AFP declines may reflect treatment-induced hypoxia and tumor necrosis. 11 Therefore, post-treatment AFP reduction likely suggests a favorable therapeutic response, whereas AFP elevation indicates poor response or disease progression. Alternatively, dynamic changes in AFP may not directly modulate ICI response but could rather serve as a sensitive surrogate for overall changes in tumor burden and activity resulting from treatment. Further research is needed to elucidate the precise interplay between AFP dynamics, the tumor microenvironment, and ICI efficacy.
Subgroup analyses stratified by region, sample size, treatment regimen, data source, timing of AFP response assessment, and AFP response cutoff values revealed that dynamic changes in AFP levels are closely linked to PFS in patients with HCC. An AFP decrease correlates with improved PFS, whereas an AFP increase suggests poorer outcomes. However, in patients receiving ICI monotherapy, a decline in AFP did not significantly correlate with OS, contrasting with the findings of Tian 12 showing a significant association between the decline of AFP and improved OS in patients with HCC receiving ICI treatment. In the current study, the negative predictive result for OS in the ICI monotherapy subgroup could be related to a delayed AFP response. The tumor-promoting and immunosuppressive properties of AFP may impede the efficacy of ICIs. 34 Most prior investigations examined ICIs in combination regimens, which bolster anti-tumor activity and prompt a faster AFP response. In contrast, ICI monotherapy may delay this response. 23 Subgroup analyses of AFP elevation showed no significant correlation with poorer OS in cohorts with ⩽100 patients, ICI monotherapy, data derived from survival curves, and cutoff values of AFP at 50% or 25%. In some studies, survival curves were the only data source available, and the use of Engauge Digitizer software to extract numerical values may have introduced errors. Moreover, certain subgroup analyses included only a single study, and the limited number of studies may have contributed to the lack of a significant association between AFP elevation and OS. A smaller sample size and shorter follow-up duration could also have influenced these results.
Although ICIs represent considerable progress in cancer therapy, their efficacy varies substantially among patients. Currently, no unified prognostic biomarker has been established for HCC patients receiving ICI therapy. AFP is minimally invasive, cost-effective, and readily accessible. Unlike a single measurement, continuous AFP monitoring may better capture the actual tumor state. Our findings suggest that continuing the existing treatment regimen could benefit HCC patients who show a dynamic decrease in AFP. Conversely, a dynamic increase in AFP may indicate emerging resistance, highlighting the need for early adjustments to improve treatment outcomes.
Subgroup analyses were conducted to examine the influence of clinical characteristics on study outcomes. In contrast to previous research, 49 our findings reveal that the relationship between dynamic changes in AFP and both PFS and OS is not significantly impacted by the timing of the AFP response assessment or the specific cutoff values used. Specifically, dynamic changes in AFP effectively predicted PFS/OS regardless of whether measured at less than 4 weeks, 4–8 weeks, or beyond 8 weeks, suggesting that the predictive ability of AFP is not affected by the specific timing of assessment. Consequently, early and continuous monitoring of AFP levels in clinical practice may be a valuable tool for physicians to consider, enabling them to promptly assess whether HCC patients are potentially benefiting from their current treatment regimen. Similarly, regardless of whether the AFP response is defined by a reduction of 10%, 15%, 18%, 20%, 25%, or 50%, a dynamic decrease in AFP effectively predicts PFS and OS. This indicates that the predictive capability of AFP changes does not rely on specific cutoff thresholds. Such findings may help minimize diagnostic errors, prevent unnecessary adjustments to effective treatment protocols, and ensure that patients continue to benefit from their therapies.
This study has several limitations. Firstly, a major limitation is the substantial heterogeneity in AFP response definitions across the included studies. This variability fundamentally complicates the pooling of data and challenges the robustness of the summary estimates. While we performed subgroup analyses by reported cutoff values, these may not fully resolve this issue. Secondly, the timing of AFP assessment also varied considerably, which could introduce heterogeneity that subgrouping alone cannot fully address. AFP assay results may vary across testing platforms; however, since the included studies did not clearly mention the specific system used, it is not possible to assess the potential impact of this factor on the results. Thirdly, the predominance of retrospective studies with relatively small sample sizes may affect result reliability. Fourthly, the study population consists mainly of Asian participants (17 studies from China, 2 from Japan, 1 from Korea), potentially limiting result generalizability to other populations with different HCC etiologies or genetic backgrounds. Fifthly, significant publication bias was detected for the association between AFP decrease and PFS using Egger’s test, suggesting that studies with non-significant or unfavorable results might be underrepresented, potentially leading to an overestimation of the true effect size. Our analysis included only English-language studies, and some subgroup analyses were based on limited studies, which may also contribute to publication bias. Sixthly, another concern is the prevalence of combination ICI therapy studies (21 out of 23), with fewer investigations of ICI monotherapy (only 2 studies), which severely underpowers the monotherapy subgroup analysis and restricts the broader application of our findings primarily to combination regimens. The exclusion of some negative results due to missing HR data may introduce further publication bias. Furthermore, this meta-analysis was based on aggregate data extracted from published studies, not on individual patient data (IPD). The lack of IPD prevented more granular analyses, such as standardizing AFP response definitions across all patients, detailed exploration of interactions between AFP changes and various patient/tumor characteristics, and more robust adjustment for confounders (e.g., baseline liver function, tumor burden, prior treatments) at the patient level. This reliance on study-level data is an inherent limitation that may affect the precision and depth of our findings.
Conclusion
Our study demonstrates that among HCC patients receiving ICIs, dynamic changes in AFP show a significant association with OS and PFS, primarily driven by results from combination ICI therapy studies. This association was less clear or not statistically significant for ICI monotherapy in the case of OS and is subject to limitations stemming from inter-study heterogeneity in AFP definitions and assessment timing, as well as potential publication bias for one outcome. While these findings suggest AFP’s potential as a prognostic marker in HCC patients receiving ICI-based therapies, particularly combination regimens, the heterogeneity of included studies and potential publication bias necessitate cautious interpretation. Further validation in prospective, standardized studies adopting uniform AFP response criteria and assessment timings is crucial before AFP can be unequivocally established as a reliable biomarker for routine clinical decision-making in this context.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251387501 – Supplemental material for Dynamic changes in serum alpha-fetoprotein predict prognosis in hepatocellular carcinoma treated with immune checkpoint inhibitors: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848251387501 for Dynamic changes in serum alpha-fetoprotein predict prognosis in hepatocellular carcinoma treated with immune checkpoint inhibitors: a systematic review and meta-analysis by Yong Zeng, Junlin Gu, Wensheng He and Jianjun Luo in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
