Abstract
Background:
After multiple uncovered self-expandable metal stent (UCSEMS) deployments, endoscopic revision becomes complex and potentially challenging. A novel stent delivery system with a dilation function has recently been developed in Japan. This study evaluated the technical feasibility of this device for patients with unresectable malignant hilar biliary obstruction (MHO) after multiple UCSEMS deployments.
Method:
Participants comprised consecutive patients with unresectable MHO who had undergone multiple UCSEMS deployments and required endoscopic revision for the right or left hepatic bile ducts, as decided by cholangiography under endoscopic retrograde cholangiopancreatography (ERCP) guidance.
Results:
A total of 14 patients requiring endoscopic revision of right and left hepatic bile ducts were enrolled in this study. Among these 14 patients, guidewire insertion through the mesh of previously placed stents failed in 2 patients. As a result, these two patients underwent only unilateral UCSEMS deployment. Because clinical success was not obtained, transmural placement of the UCSEMS under Endoscopic Ultrasound (EUS) guidance to the segment of the liver not being drained was performed in those patients. The molting technique was attempted in 12 patients, proving successful in all patients. The technical success rate was therefore 92.8% (12/14). Mean procedure time was 23.2 min.
Conclusion:
This novel stent delivery system with a dilation function may be useful in endoscopic revision techniques for bilateral UCSEMS deployment.
Introduction
Unresectable malignant hilar biliary obstruction (MHO) is usually treated by stent deployment under endoscopic retrograde cholangiopancreatography (ERCP) guidance.1,2 Compared with plastic stents, an uncovered self-expandable metal stent (UCSEMS) may offer longer stent patency.3,4 Recent randomized controlled trials have shown favorable results such as longer stent patency or prolonged overall survival with unilateral UCSEMS deployment compared with bilateral UCSEMS. 5 In addition, bilateral drainage may be needed based on drainage volume. 6 Bilateral UCSEMS deployment is performed using stent-in-stent (SIS) or side-by-side (SBS) techniques. Although no clinical differences in technical and clinical success rates or survival rates have been identified, a recent randomized controlled trial found stent patency rates at 3 or 6 months were more favorable with the SIS technique. 7
On the contrary, endoscopic revision after multiple stent deployment should be considered because of recent improvements in chemotherapy. 8 After multiple UCSEMS deployments, endoscopic revision becomes complex and sometimes challenging. During endoscopic revision, even if the guidewire can be successfully inserted through the mesh to the target area or the mesh of the UCSEMS is successfully dilated, insertion of the stent delivery system into the target area through the mesh is sometimes difficult because the delivery system can become stuck within the mesh of the previously deployed stent. To overcome this issue, a novel stent delivery system with a dilation function has been developed in Japan. The present study evaluated the technical feasibility of this device for patients with unresectable MHO after multiple UCSEMS deployments.
Patients and method
Consecutive patients with unresectable MHO who underwent multiple UCSEMS and required endoscopic revision were enrolled between October 2020 and May 2021. All patients were complicated with obstructive jaundice due to UCSEMS obstruction according to computed tomography. The drainage area was determined by cholangiography under ERCP guidance. All study protocols were approved by the institutional review board of our hospital. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in the a priori approval given by the human research committee at Osaka Medical College (IRB no. 2021-028). The requirement for informed consent was waived due to the retrospective nature of this study.
Technical tips for endoscopic revision using the novel stent delivery system with a dilation function (molting technique)
All procedures were performed by two experienced endoscopists who were trained in therapeutic ERCP (T.O. and A.O.). Figure 1 shows the novel stent delivery system with a dilation function (EndoSheather; Piolax Medical Devices, Kanagawa, Japan). The diameter of the outer sheath for this device is 7.2 Fr, and the inner sheath is 5.9 Fr. In addition, the tip of this device is extremely tapered (3.9 Fr), conforming to a 0.035-inch guidewire. These characteristics allow penetration of stricture sites, and after removal of the inner sheath, various devices under 5.9 Fr can be inserted. First, the inner sheath is removed (Figure 1(a)) and then the proximal site of the outer sheath is cut to allow device insertion (Figure 1(b)). Next, the stent delivery system is inserted into the outer sheath (Figure 1(c)) and deployed to the appropriate site (Figure 1(d)) (Supplemental Video 1).
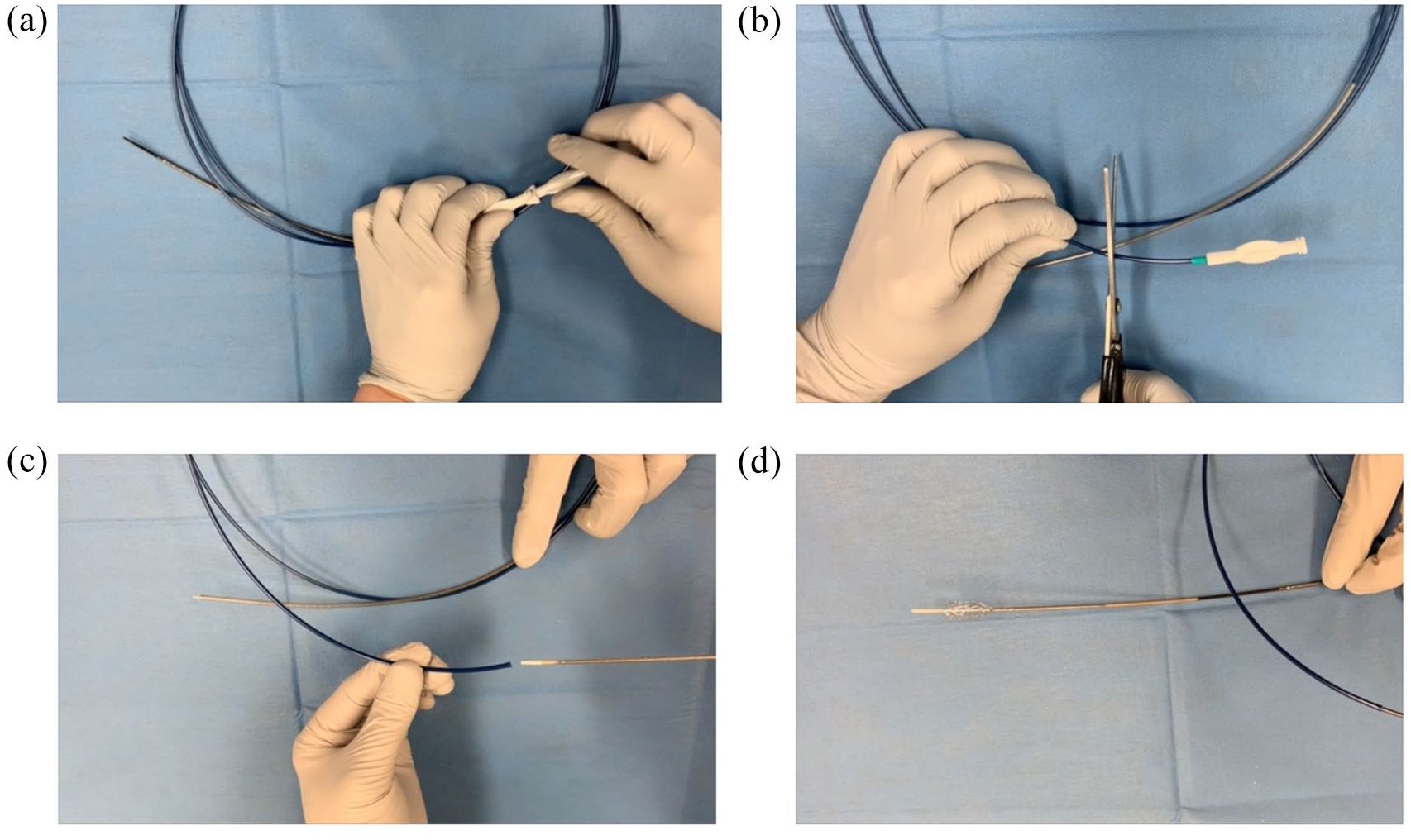
The novel stent delivery system with a dilation function (EndoSheather; Piolax Medical Devices, Kanagawa, Japan). The diameter of the outer sheath for this device is 7.2 Fr. After removing the inner sheath (a), the proximal outer sheath is cut (b). A device with a diameter of up to 5.9 Fr can be inserted into this outer sheath (c, d).
Figures 2 and 3 show technical tips for revision procedures after multiple UCSEMS deployments using SIS deployment, which we call the ‘molting technique’. In this case, cholangiography reveals left and right hepatic bile duct obstruction (Figure 2(a)). After guidewire deployment (0.025-inch, VisiGlide 2; Olympus Medical Systems, Tokyo, Japan or 0.035-inch, RevoWave SeekMaster; Piolax Medical Devices) into both bile ducts, the EndoSheather is inserted into the left hepatic bile duct (Figure 2(b)), then the inner sheath is removed. Next, the stent delivery system for the UCSEMS (BILERUSH Selective; Piolax Medical Devices) is inserted into the outer sheath (Figure 2(c)), and only the outer sheath is removed. In so doing, the UCSEMS is placed at the left hepatic bile duct across the previously deployed UCSEMS. The new UCSEMS is then deployed (Figure 2(d)). Next, the guidewire is advanced into the right hepatic bile duct across the mesh of the previously placed UCSEMS. Although the ERCP catheter cannot be inserted into the right hepatic bile duct, insertion of the EndoSheather into the right hepatic bile duct is successfully performed (Figure 3(a)). The inner sheath is removed (Figure 3(b)), and the stent delivery system is then inserted within the outer sheath. Subsequently, the outer sheath is removed (Figure 3(c)). Finally, UCSEMS deployment is successfully performed (Supplemental Video 2).

(a) Obstruction of bilateral uncovered self-expandable metal stents is observed. (b) The novel device is inserted into the left hepatic bile duct across the occluded stent. (c) A stent delivery system is inserted within the novel device. (d) Endoscopic revision of the left hepatic bile duct using the uncovered self-expandable metal stent (8 mm × 8 cm) is successful.

(a) The novel device is inserted into the right hepatic bile duct. (b) The inner sheath is removed. (c) A stent delivery system is inserted within the novel device, and the outer sheath is removed. (d) Endoscopic revision of the left hepatic bile duct using the uncovered self-expandable metal stent (8 mm × 8 cm) is successful.
Definitions and statistical analysis
Technical success was defined as successful endoscopic revision using the molting technique. If stricture dilation was achieved using the previous crawling technique, the result was considered technical failure. Clinical success was defined as a decrease in serum bilirubin level to <50% of the pre-procedure level or to a normal level (<1.3 mg/dL) within 14 days. Procedure resolution was measured from scope insertion to removal. Adverse events were graded according to the severity grading system of the American Society for Gastrointestinal Endoscopy lexicon. 9
Descriptive statistics are presented as median (interquartile range), mean (± standard deviation), and frequency for continuous and categorical variables, respectively. The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement. 10
Results
A total of 14 patients (median age, 65.0 years; 9 men) requiring endoscopic revision of the right and left hepatic bile ducts were enrolled in this study (Table 1). Number of previous metal stents was mainly two. As kinds of previous deployment UCSEMS, dedicated UCSEMS for hepatic hilar obstruction (Moving cell stent, M-Hilar stent) had been previously deployed.11,12
Baseline characteristics of patients.

IQR, interquartile range; SD, standard deviation.
Table 2 shows outcomes and adverse events for the bilateral SIS molting technique. Among the 14 patients, guidewire insertion through the mesh of previously placed stents failed in 2 patients. As a result, these two patients underwent only unilateral UCSEMS deployment. Because clinical success was not obtained, Endoscopic Ultrasound (EUS)-guided biliary drainage was performed in those patients. The molting technique was attempted in 12 patients, proving successful in all cases. The technical success rate was therefore 92.8% (12/14). Mean procedure time was 23.2 min, and clinical success was obtained in all patients who underwent bilateral UCSEMS deployments. Finally, adverse events were observed in two patients (acute pancreatitis, n = 1; cholangitis, n = 1) and were successfully resolved with conservative treatment in both cases.
Outcomes and adverse events.

SD, standard deviation.
Discussion
Technical failure for endoscopic revision after bilateral UCSEMS deployment using the SIS technique can occur for various reasons. Okuno et al. 13 evaluated endoscopic revision for bilateral UCSEMS obstruction. Among their 31 patients who required endoscopic revision, technical failure was observed in 6 patients. Guidewire insertion across the UCSEMS failed in three of those six patients. Indeed, guidewire insertion failed in two patients in our study. Son et al. 14 also evaluated endoscopic revision after bilateral UCSEMS obstruction. In that study, the technical success rate was 76.3% (29/38). Of the nine patients for whom endoscopic revision failed, failure in six patients was due to tight biliary stricture. Improvement of the technical success rate requires a better dilation device. However, after dilation of the mesh of the previously placed UCSEMS, insertion of stent delivery systems across the mesh is sometimes challenging if the tip of the stent delivery system becomes stuck in the mesh of the previously placed UCSEMS. The EndoSheather may prove clinically useful in overcoming this issue. This device offers several advantages. First, the mesh of a previously deployed UCSEMS can be easily dilated because the EndoSheather shows the good dilation function. Second, after successful insertion of this device across the mesh of a previously placed UCSEMS, stent deployment can be ensured using the molting technique. This may have contributed to the improved technical success rate and reduced procedure time.
During the molting technique, not only a fine-gauge stent delivery system but also flexibility may be important. We therefore consider that laser cut-type UCSEMS with a flexible, fine-gauge stent delivery system, such as BILERUSH selective (Piolax Medical Devices), YABUSAME (Kaneka, Tokyo, Japan), and ZEO stent (Zeon Medical Co., Tokyo, Japan). However, these stent delivery systems are fine. In this context, whether these small-caliber stents could also potentially still be inserted without using the molting technique. However, the tip of EndoSheather is extremely tapered with a fine gauge (3.9 Fr) and offers favorable push-ability. We therefore believe that this system may better suit than stent delivery systems for insertion across a previously deployed SEMS. This strategy should be evaluated in a comparison study between a stent delivery system insertion group and a molting technique group.
Lee et al. 7 recently conducted a randomized trial comparing SIS (n = 34) and SBS (n = 35) for unresectable MHO. The technical success rates for stent deployment were similar between groups (SIS group, 100% versus SBS group, 91.4%; p = 0.633), as were clinical success rates (SIS group, 94.1% versus SBS group, 90.6%; p = 0.668). In addition, rates of adverse events did not differ significantly between groups (SIS group, 23.5% versus SBS group, 28.6%; p = 0.633). However, although no significant differences were confirmed, stent patency rates at 3 and 6 months tended to be higher for SIS than for SBS (3 months: 85.3% versus 65.7%, p = 0.059; 6 months: 47.1% versus 31.4%, p = 0.184). In addition, SBS shows several limitations such as compression of adjacent vascular structures. Further high-quality randomized trials including a larger patient cohort might confirm SIS as more suitable for bilateral UCSEMS deployment. Therefore, according to these results, UCSEMS using an SIS technique appears useful. However, we should also consider endoscopic revision because of recent developments in chemotherapy, which may have clinical benefits for prolonging patient survival. Several studies have shown the clinical efficacy of endoscopic revision after bilateral UCSEMS using the SIS technique.14,15 Given the above reasons, the present technique may be helpful as an alternative revision technique.
For revision, a plastic stent may be easier to use than a UCSEMS. According to a meta-analysis regarding UCSEMS and plastic stents, 16 UCSEMS showed a lower 30-day occlusion rate than plastic stents in MHO [odds ratio (OR), 0.36; 95% confidence interval (CI), 0.19–0.39], and a lower long-term occlusion rate compared with plastic stents in MHO (OR, 0.28; 95% CI, 0.19–0.39). Clinical success rate was lower when using plastic stents (13%) compared with UCSEMS (7%; OR, 0.43; 95% CI, 0.27–0.67). Revision rate was also lower with UCSEMS than with plastic stent (mean difference, −0.49; 95% CI, −0.8 to −0.19), and the incidence of cholangitis was lower with UCSEMS (8% versus 21%; OR, 0.41; 95% CI, 0.22–0.76). UCSEMS may therefore have clinical benefits compared with plastic stents. However, in clinical practice, plastic stent exchange may be more commonly performed. This factor is worth considering when selecting stents. Moreover, one of the advantages of plastic stents is the ability to perform intra-ductal radiofrequency ablation at every stent exchange. Gao et al. 17 recently conducted significant multicenter randomized controlled trial regarding the benefit of intra-ductal radiofrequency ablation (I-RFA). In that study, groups were divided into patients receiving either a plastic stent alone or a plastic stent with I-RFA. Median overall survival was significantly longer in plastic stent with I-RFA group (14.3 months) than in the plastic stent group (9.2 months; hazard ratio, 0.488; 95% CI, 0.351–0.678; p < 0.001). The role of plastic stents may therefore change in the near future. Under the current situation, as per the American Society for Gastrointestinal Endoscopy recommendation, 2 if an optimal drainage strategy has not been established such as having the possibility of curative surgery after neo-adjuvant chemotherapy, a plastic stent may be suitable.
Additional limitations should be considered in our study. First, all procedures were performed by experienced endoscopists because the molting technique may not be performed with same technical success rate. Second, in this study, patients had generally two previous deployments of UCSEMS. In addition, these stents were dedicated for hepatic hilar obstruction, so endoscopic revision of occluded UCSEMS using the molting technique may have been easier than with some other types of UCSEMS. Finally, our study was retrospective in nature.
In conclusion, the molting technique using the novel stent delivery system with a dilation function may be useful in endoscopic revision techniques for bilateral UCSEMS deployment.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211062803 – Supplemental material for Endoscopic bilateral revision after metal stent deployment for hepatic hilar obstruction using molting technique (with videos)
Supplemental material, sj-docx-1-tag-10.1177_17562848211062803 for Endoscopic bilateral revision after metal stent deployment for hepatic hilar obstruction using molting technique (with videos) by Masahiro Yamamura, Takeshi Ogura, Saori Ueno, Atsushi Okuda, Nobu Nishioka, Masanori Yamada, Kazuya Ueshima, Jun Matsuno, Yoshitaro Yamamoto and Kazuhide Higuchi in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material (videos) for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
