Abstract
Background:
Endoscopic ultrasonography is crucial for diagnosing solid pancreatic lesions. Tissue acquisition using rapid on-site evaluation (ROSE) significantly improves diagnostic efficiency.
Objectives:
This study evaluates the efficacy of tissue acquisition from pancreatic tumors with and without ROSE.
Design:
Systematic review and meta-analysis.
Data sources and methods:
A search was conducted in PubMed and other databases covering the period up to February 2024. Eligible randomized control trials (RCTs) reporting data on comparing the efficacy of ROSE and no ROSE were included in this study. We compare the two groups using odd ratios (OR) and mean difference approach.
Results:
This meta-analysis included seven RCTs, including 1723 patients (909 in the ROSE and 814 in the no-ROSE group) with pancreatic masses. The fundamental characteristics of the studies were almost identical in both groups. There was no significant difference (
Conclusion:
The ROSE and non-ROSE approaches showed similar outcomes regarding mean needle passes, sample adequacy, diagnostic accuracy, and adverse event rates.
Trial registration:
This systematic review and meta-analysis was registered at PROSPERO (https://www.crd.york.ac.uk/PROSPERO/) with PROSPERO Number CRD42024520977.
Plain language summary
Endoscopic ultrasonography (EUS) with tissue acquisition is widely used for diagnosing solid pancreatic lesions. ROSE is thought to improve diagnostic efficiency by providing immediate results. This meta-analysis shows no significant difference between ROSE and non-ROSE approaches in diagnostic outcomes or procedure time. Both methods yielded similar results for sample adequacy, accuracy, and adverse events. ROSE may not be essential for improving pancreatic lesion diagnosis, leading to more cost-effective practices. Future research may explore other methods to enhance diagnostic efficiency.
Keywords
Introduction
Endoscopic ultrasonography (EUS) is essential for diagnosing solid pancreatic lesions. Morphological examination alone is insufficient to accurately characterize pancreatic masses. To achieve cytopathological and histological diagnoses, EUS-guided fine-needle aspiration (FNA) and fine-needle biopsy (FNB) are necessary.1–3 When EUS-FNA is used to get cell samples, the surrounding tissues essential for diagnosing are often lost due to the fibrotic character of such diseases and the significant necrosis that frequently follows them. EUS-FNA specimens might not include sufficient cells. EUS-FNB, especially with end-cutting styles, is another more modern technique. It keeps the structure of the tissue and allows for a more accurate diagnosis. This aids in detecting cancer and other diseases that can manifest in a wide range of solid tissues.4–7
By providing immediate feedback on the appropriateness of the sample retrieved and decreasing the number of needle passes necessary, rapid on-site cytological evaluation (ROSE) is a valuable adjunct EUS-FNA. Although the results have been contradictory, ROSE is an effective method of enhancing EUS-precision FNAs and their efficiency. Interestingly, EUS-FNB with ROSE has shown equal efficacy to EUS-guided FNB without ROSE, even when using current end-cutting needles.8–10 However, because of the high cost and time commitment involved in training cytopathologists in pancreatic cancer and the limited accessibility of on-site cytologic testing, ROSE has been used only in a limited capacity.11,12 As a result, it is essential to understand and evaluate the usefulness of ROSE for detecting pancreatic lesions. Recent retrospective studies suggest that EUS-FNB + ROSE with cytological and histological specimens may increase diagnostic yield over EUS-FNB alone.13–15 ROSE real-time sample adequacy evaluation can guide sampling and improve diagnostic accuracy. ROSE can also deliver touch-imprint cytology specimens for fast cellular morphology assessment. 16 ROSE’s advantages over EUS-FNB or FNA alone need further exploration. Because ROSE may increase procedural time and cost, patient variables and professional judgment should be considered while choosing a sample strategy. 17
This meta-analysis aims to evaluate the efficacy of ROSE compared to no-ROSE for tissue acquisition of pancreatic masses in randomized control trial (RCT) studies.
Material and methods
Search strategy
We conducted a comprehensive literature search in various medical databases, including PubMed, Embase, Scopus, and others, to identify studies relevant to our topic. The search was completed in February 2024. We used a combination of keywords such as “endoscopic ultrasound,” “fine-needle biopsy,” “fine-needle aspiration,” “rapid on-site evaluation,” “ROSE,” and “pancreatic lesions” to ensure a thorough and inclusive search strategy (Supplemental Data 1). In addition to the database searches, we also reviewed relevant reviews and meta-analyses focusing on the application of EUS in solid pancreatic lesions to identify additional suitable studies for inclusion in our analysis. This meta-analysis was registered (PROSPERO Number CRD42024520977).
Studies selection
We adopted the following criteria for determining the inclusion and exclusion of studies. Studies were eligible for inclusion if they were RCTs directly comparing the use of EUS-FNB/FNA with ROSE versus EUS-FNB/FNA alone or providing subgroup analysis based on the application of ROSE, conducted on patients with solid pancreatic lesions, and reporting sample adequacy or mean number of passes (or data that could be used for this calculation). We excluded retrospective studies, noncomparative single-arm studies, case studies, studies lacking subgroup analysis on pancreatic masses or failing to compare with ROSE, studies not reporting any of the required results, and studies published in languages other than English.
Data extraction
Two authors independently extracted data from the selected studies, including country, publication year, mean age, gender proportion, number of patients in both groups, lesion size, needle type, number of passes, procedure time, sample adequacy, sensitivity, accuracy, specificity, and adverse events. The extracted data were divided into the ROSE and no ROSE groups. As most studies presented data in various formats, it was necessary to convert them into consistent units for analysis. Sensitivity, accuracy, and specificity data were primarily presented as percentages. To compute the risk difference between the two groups, we obtained the number of cases by multiplying the total number of patients by the percentage and dividing the result by 100. In the absence of actual case numbers, we assumed the cases were based on the total number of patients for comparison purposes. This study adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) requirements. 18
Outcome and definitions
The primary outcome of our study was the mean difference in the number of needle passes required to obtain adequate samples. Secondary outcomes included mean difference in procedure time (total time taken to perform the procedure), sample adequacy (percentage of specimens satisfying the criteria for a proper diagnosis), diagnostic accuracy (combined number of true positives (TPs) and true negatives divided by the total number of patients), diagnostic sensitivity (calculated as the number of accurately determined positives (TPs) divided by the incidence of the condition in the research group (TPs + false negatives)), and adverse events (any adverse outcomes resulting from the procedure). Additionally, we performed subgroup analyses comparing FNA + ROSE versus FNA, FNA + ROSE versus FNB, and FNB + ROSE versus FNB to further evaluate the impact of ROSE on different EUS-guided sampling techniques.
Quality assessment of studies
This study utilized the risk-of-bias assessment method developed by the Cochrane Collaboration. 19 The assessments were classified as “low” if there was a limited risk of bias, “high” if there was a significant risk of bias, and “some concerns” if the available evidence could not evaluate the possibility of bias.
Statistical analysis
The study findings from both groups were combined and compared using a random-effects model using the continuous inverse variance method. The results were reported as odd ratios (OR) and pooled mean differences, along with their corresponding 95% confidence intervals (95% CI).
Results
Characteristics of included studies and search results
From the articles that were retrieved from the search of medical databases, we included seven RCTs in this meta-analysis (Figure 1). These studies comprised 1723 patients, 909 of whom were assigned to the ROSE group and 814 of whom were enrolled in the no ROSE group. Among the seven studies, three investigated the comparison between FNA + ROSE and FNA alone21–23; three evaluated the comparison between FNA + ROSE and FNB alone,8,24,25 and only one study compared FNB + ROSE and FNB alone. 26 Two studies were carried out in the United States,22,25 with one study conducted in each of the following countries: Brazil, 21 Philippines, 24 China, 23 Italy, 26 and Canada. 8 Three studies were performed as single-center RCTs, and four were conducted as multicenter RCTs.

PRISMA flow chart.
There were no significant differences in the fundamental characteristics of the included patients between the two groups. In the studies, the lesion site and size were nearly identical. The included studies employed various types of 19G, 20G, 22G, and 25G needles for tissue acquisition (Tables 1 and 2).
Characteristics of included studies.
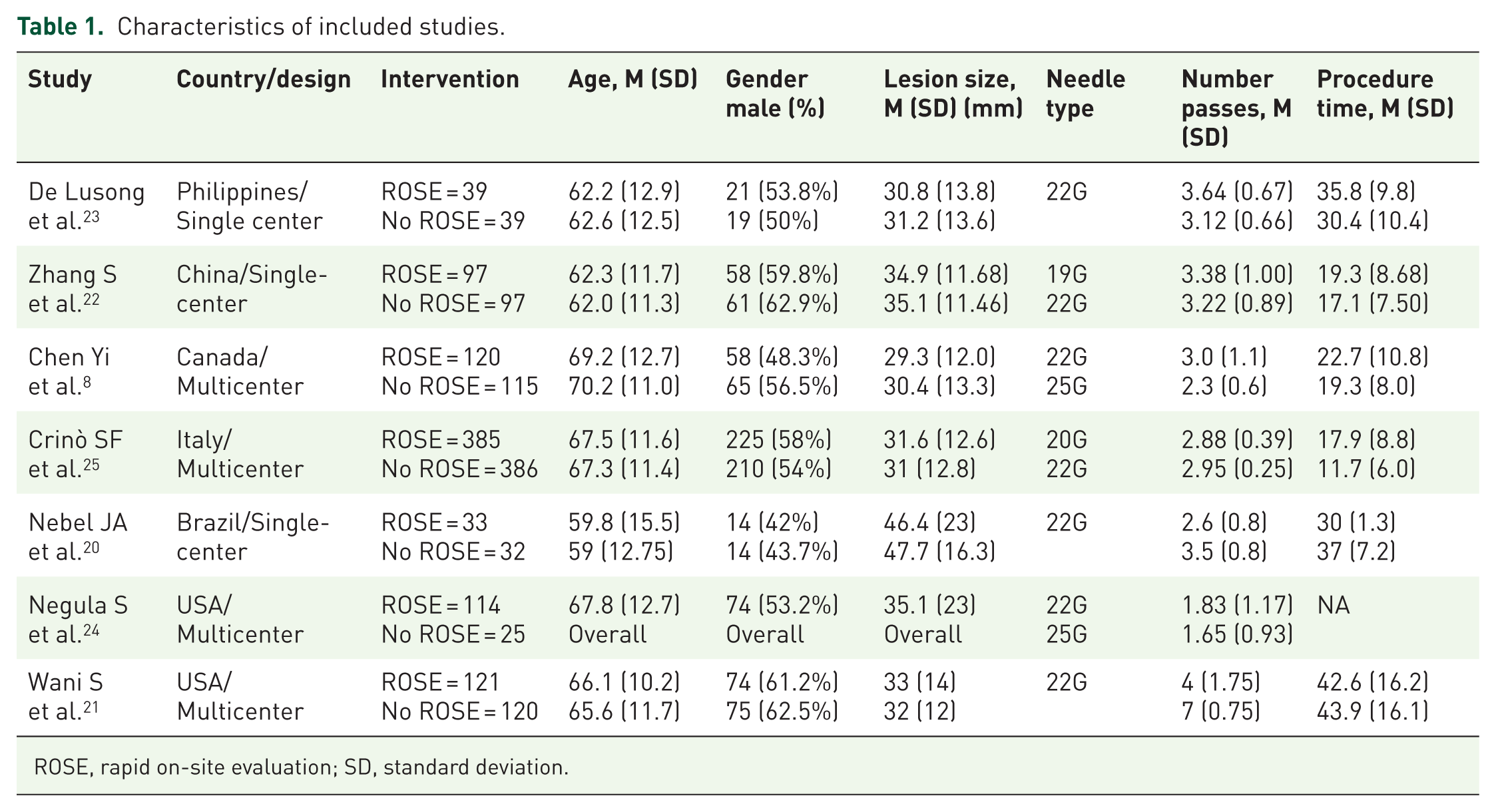
ROSE, rapid on-site evaluation; SD, standard deviation.
Characteristics of included studies.

NPV, negative predictive values; NR, Not reported; ROSE, rapid on-site evaluation; PPV, positive predictive values.
Two studies were rated as having “some concern” by the risk of bias assessment, whereas others had a low risk of bias (Supplemental Figure 5). Except for Nabel et al. in the mean procedure time analysis, no study had significant effects on the final results, according to the study’s impact analysis. As number of studies included were less than 10, so no other publication bias was found.
Two studies were rated as having “some concern” by the risk of bias assessment, whereas others had a low risk of bias (Supplemental Figure 5). Except for Nabel et al. in the mean procedure time analysis, no study had significant effects on the final results, according to the study’s impact analysis. As number of studies included were less than 10, so no other publication bias was found.
Primary outcomes
The number of needle passes
Data from seven studies were utilized to calculate the mean difference in the number of needle passes between the two groups. The analysis revealed no statistically significant difference of –0.34 passes (95% CI –1.00, 0.32;
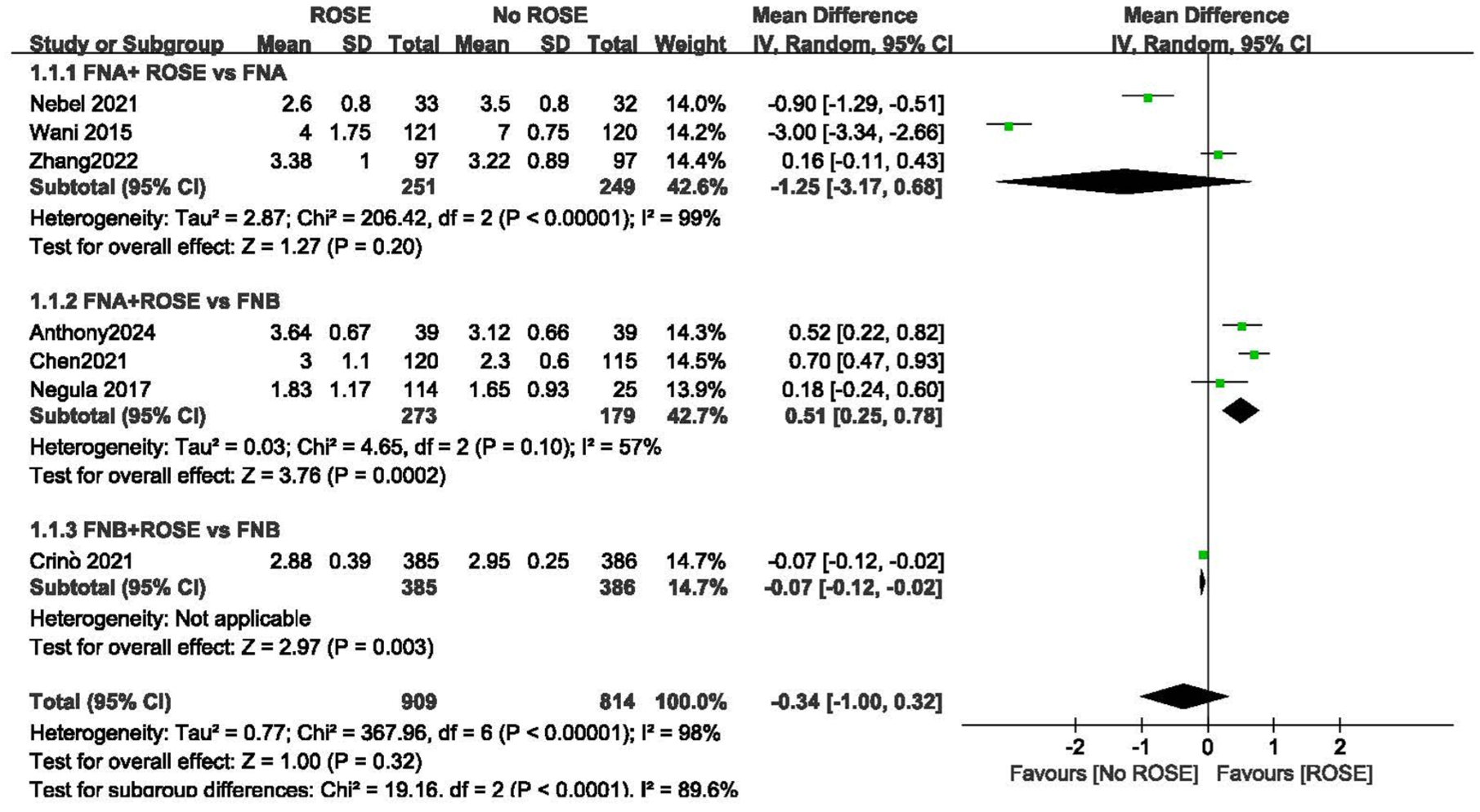
Forest plot of the number of needle passes.
Secondary outcomes
Procedure time
Six studies included in the analysis included data on the length of time of the procedure for both the ROSE and no ROSE groups. The procedure time did not show a statistically significant mean difference of 1.49 min (95% CI –2.76, 5.75;

Forest plot of the mean difference in procedure time.
Sample adequacy
The OR in the sample adequacy indicates no statistically significant difference between the two groups. The OR was 1.34 (95% CI: 0.29, 6.25;

Forest plot of sample adequacy.
Diagnostic sensitivity
The diagnostic sensitivity between the ROSE and no ROSE groups was reported in six trials as a percentage. The pooled mean diagnostic sensitivity percentage for the ROSE group was 93.3%, and 85.9% for the no ROSE group. The OR technique to compute the difference between the two groups. The analysis yielded an OR of 1.95 (95% CI: –0.79, 4.82;
Diagnostic specificity
We calculated the diagnostic specificity difference between the two groups using the Risk difference (RD) approach. The pooled mean diagnosis specificity for the ROSE group was 100%, whereas the no ROSE group was 95.83%. As most of studies reported 100% specificity, so we were unable to calculate OR. These results indicate no significant difference in the diagnostic specificity between the two groups.
Diagnostic accuracy
The ROSE group had 93.05% pooled mean diagnosis accuracy, while no ROSE had 89.67% showing no significant difference. There was OR of 1.28 (95% CI 0.54, 3.00;
Diagnostic positive predictive value
The diagnostic tests’ positive predictive value was about 100% in both groups. Therefore, calculating the OR between the two groups was unnecessary.
Diagnostic negative predictive value
Diagnostic negative predictive value (NPV) was also reported as a percentage. The mean diagnostic NPV for the ROSE group was 52.78%, while it was 51.08% for the no ROSE group. The OR of 1.05 (95% CI 0.54, 2.06;
Adverse events
There was no significant difference in the rate of adverse events between the two groups. The results showed an OR of 0.87 (95% CI 0.16, 4.87;
Subgroup analyses
FNA + ROSE versus FNA alone
Subgroup analyses of three studies comparing FNA + ROSE and FNA alone revealed no significant differences in the mean difference of number of needle passes (–1.25, 95% CI –3.17, 0.68;
FNA + ROSE versus FNB alone
Subgroup analyses of three studies comparing FNA + ROSE and FNB alone demonstrated significant differences in the mean difference of number of passes (0.51, 95% CI 0.25, 0.78;
FNB + ROSE versus FNB alone
A single study that compared FNB + ROSE and FNB alone found that the mean difference in the number of passes was –0.07 (95% CI –0.12, –0.02,
Discussion
This systematic review and meta-analysis includes seven RCTs to assess the efficacy of ROSE and no ROSE for acquiring pancreatic lesion tissues for diagnostic purposes. There were no significant differences (
EUS is used to obtain tissue samples from solid pancreatic lesions for diagnosis, either through FNA or FNB. FNB needles, designed to obtain larger and more accurate tissue samples, may enhance diagnostic accuracy compared to FNA needles.15,27 The necessity of ROSE for sample assessment during procedures remains debatable. Obtaining high-quality core biopsy tissues for histologic inspection is crucial for molecular characterization and identifying treatment classification indicators. 28 EUS-FNB may become the dominant method for identifying molecular alterations in the pancreas postchemotherapy, providing valuable information on molecular changes after treatment to guide future therapy decisions.29,30
The pooled analysis revealed no significant difference in the mean number of needle passes required to obtain adequate samples between the ROSE and no ROSE groups, with a mean difference of –0.34 passes (95% CI –1.00, 0.32;
The sample adequacy analysis indicated no statistically significant difference (
Adverse events have been reported in four investigations, and the analysis of the risk difference revealed no statistically significant difference in the incidence of these adverse events between the two groups. The adverse events observed in these studies included bleeding, acute pancreatitis, and pain. These occurrences were managed using conservative treatment methods, and the symptoms resolved following the therapies.
Based on these results, it is recommended that the centers that already have a ROSE service reconsider its necessity. In contrast, institutions without a ROSE service can achieve excellent diagnostic accuracy using EUS-FNB/FNA alone.
Our study has several limitations that should be considered when interpreting the results. First, the small number of studies included in the analysis and the significant heterogeneity identified in most of the analyses necessitate cautious interpretation of the findings. Unfortunately, we were unable to identify the sources of heterogeneity. Second, all studies reported diagnostic efficacy as a percentage and 95% CI. To compare the two groups, we converted these values into the number of cases by multiplying the total number of patients by the percentage and dividing the result by 100. As the actual number of cases was not available, we assumed the cases were per total patient for comparison purposes. This assumption may introduce some degree of uncertainty in the analysis. Third, our study did not compare the types of needles used for tissue acquisition, which may have an impact on the diagnostic yield and other outcomes. Furthermore, the size of the tumor is an important factor impacting the diagnostic yield, we were unable to perform subgroup analysis on this basis, due to lack of specific data and less studies with almost similar size. 32 Future studies should consider evaluating the influence of needle type on the effectiveness of ROSE in EUS-guided tissue sampling of solid pancreatic lesions, as well as the relationship between FNA/FNB with ROSE and FNA/FNB alone.
Conclusion
For the collection of tissue samples from pancreatic masses using two different procedures: ROSE and no ROSE. There is no statistically significant differences in the mean number of needle passes, procedure time, sample adequacy, diagnostic accuracy, or adverse events.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251381182 – Supplemental material for The role of rapid on-site evaluation in pancreatic lesion: a systematic review and meta-analysis of randomized control trials
Supplemental material, sj-docx-1-tag-10.1177_17562848251381182 for The role of rapid on-site evaluation in pancreatic lesion: a systematic review and meta-analysis of randomized control trials by Bing Chen, Hanbei Lv, Zhangpeng Feng and Guoping Jiang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251381182 – Supplemental material for The role of rapid on-site evaluation in pancreatic lesion: a systematic review and meta-analysis of randomized control trials
Supplemental material, sj-docx-2-tag-10.1177_17562848251381182 for The role of rapid on-site evaluation in pancreatic lesion: a systematic review and meta-analysis of randomized control trials by Bing Chen, Hanbei Lv, Zhangpeng Feng and Guoping Jiang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
