Abstract
Background:
Endoscopic ultrasound-guided gallbladder drainage (EUS-GBD) using lumen apposing metal stent (LAMS) has emerged as a treatment option in patients with acute cholecystitis (AC) deemed unfit for surgery. Prior to this technique, percutaneous transhepatic gallbladder drainage (PT-GBD) was the only drainage modality available for these patients.
Objectives:
This study compares clinical outcomes of EUS-GBD versus PT-GBD in high-risk surgical patients with AC.
Design:
From July 2019 to October 2023, all consecutive patients with AC undergoing EUS-GBD (using LAMS) and PT-GBD at a single academic medical center were retrospectively reviewed and analyzed.
Methods:
A propensity score-matched analysis using age, sex, and Charlson Comorbidity Index was performed. This was used to obtain a 1:1 ratio of PT-GBD:EUS-GBD patients. Technical success was defined as successful placement of LAMS or percutaneous cholecystostomy tube in the gallbladder. Clinical success was defined as resolution of patients’ symptoms and normalization of white cell count within 96 h post procedure without recurrence of AC. Outcomes were analyzed using Fisher’s exact test and Student’s t test.
Results:
Following propensity score matching, 57 EUS-GBD patients were matched with 57 PT-GBD patients. Technical success was seen in 96% (55/57) in the EUS-GBD group and 98% (56/57) in the PT-GBD group (p > 0.99). Clinical success was observed in 93% (52/56) in EUS-GBD group and 80% (45/56) in PT-GBD group (p = 0.093). PT-GBD patients underwent more procedures than the EUS-GBD group (median 3 vs 2, p < 0.0001) and had more complications (44% vs 16%, p = 0.0010). The median survival was 573 days for EUS-GBD and 452 days for PT-GBD (p = 0.77).
Conclusion:
EUS-GBD is superior to PT-GBD, requiring fewer gallbladder-related procedures and lower rates of adverse events. Given these benefits, it has emerged as the preferred non-surgical alternative in the management of patients with AC who are poor surgical candidates.
Introduction
Acute cholecystitis (AC) is estimated to occur in 6%–11% of patients with symptomatic gallstones. 1 Currently, laparoscopic cholecystectomy is the standard of care treatment. In the United States, laparoscopic cholecystectomy is the sixth most commonly performed surgery, with over 600,000 operations performed annually.2,3 Surgery, however, may not be feasible in certain patients due to advanced age, medical comorbidities, or severe acute illness.
Traditionally, percutaneous transhepatic gallbladder drainage (PT-GBD) has been the principal alternative to surgery and is still recommended as such in the most recent international guidelines on cholecystitis. 4 Notable advantages of PT-GBD include widespread availability, high clinical efficacy, and minimal anesthesia requirements.4,5 Despite its benefits, PT-GBD is associated with a moderate adverse event rate and frequent need for re-interventions which negatively impact patient’s quality of life.1,5 Additionally, cholecystitis recurs in up to 47% of patients. 1
Endoscopic ultrasound-guided transmural gallbladder drainage (EUS-GBD) has emerged as an alternative to PT-GBD for AC in non-surgical patients. First described in 2007, EUS-GBD initially involved placement of a plastic double pigtail stent (DPT) into the gallbladder via a transmural approach under EUS guidance. 6 With the advent of the lumen apposing metal stent (LAMS), stent insertion was simplified to a single step which has led to more widespread utilization of this technique. 7 EUS-GBD for AC has demonstrated high rates of technical and clinical success with acceptable adverse event rates.8–12 In comparative studies, EUS-GBD was associated with similar rates of technical and clinical success with fewer adverse events, re-interventions, and AC recurrences compared to PT-GBD.13,14
In this study, we aimed to compare clinical outcomes between EUS-GBD using LAMS and PT-GBD in patients with AC who were not surgical candidates using a propensity score-matched analysis.
Methods
Study design, subjects, and data collection
Consecutive non-surgical patients (based on the ASA class and ECOG performance status) undergoing EUS-GBD using a LAMS for AC from July 2019 through October 2023 at a single tertiary care center were eligible for inclusion. Additionally, patients who underwent PT-GBD for AC over the same time period at the same institution were also included. Exclusion criteria consisted of patients with perforated or gangrenous gallbladder on imaging, age <18 years, platelet count <50,000, and international normalized ratio >1.5. A preliminary review of the electronic health record was completed for each patient, and age, gender, and medical comorbidities were recorded. Charlson Comorbidity Index (CCI), a validated prognostic score used to predict mortality based on comorbidities, was then calculated for each patient. 15
Propensity score matching was then performed to reduce the impact of confounding variables and treatment allocation bias. Matching was completed in a 1:1 ratio of EUS-GBD:PT-GBD patients. Complete retrospective chart review of matched patients was performed with procedure notes, progress notes, discharge summaries, clinic notes, laboratory studies, and imaging studies undergoing manual review. Data on anticoagulant/antiplatelet use, procedure-related details, and pertinent clinical outcomes was collected. Data was stored in a secured database in accordance with our local institutional review board policies (IRB number: 00077330). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (see supplemental material). 16
Outcomes
The primary outcome was clinical success which we defined as resolution of abdominal pain and fever with normalization of white blood cell count by 96 h post procedure without recurrence of cholecystitis.
Secondary outcomes included technical success, number of gallbladder interventions (including index procedure), procedure duration, post-procedure length of stay, and adverse events.
Technical success was defined by appropriate positioning of the LAMS or cholecystostomy tube in the gallbladder confirmed by imaging. Adverse events were defined and the severity classified according to the American Society of Gastrointestinal Endoscopy lexicon. 17
Statistical analysis
Propensity score matching based on age, gender, and CCI was completed using a greedy-matching algorithm with no replacement in a 1:1 ratio of EUS-GBD and PT-GBD patients. Post matching, a standardized mean difference (SMD) of less than 0.10 was used to determine if a balanced covariate distribution was achieved between the two groups. For age, the SMD was 0.05, while the SMD was 0.06 for gender and 0.09 for CCI. In the matched dataset, categorical variables were compared using a Fisher’s exact test and continuous variables were assessed using independent t tests. The Kaplan–Meier method was used to estimate survival; the log-rank test of the Chi-square approximation assessed statistical significance in comparing survival and follow-up length between groups. For all analyses, a p value of <0.05 was assumed to be statistically significant. All the statistical analyses were conducted using SAS (version 9.4; SAS Institute, Cary, NC, USA).
Procedure detail
Endoscopic ultrasound-guided transmural gallbladder drainage
All the EUS-GBD procedures were performed under general anesthesia. All the patients received intraoperative intravenous antibiotics. A linear echoendoscope (GF-UTC 180; Olympus Co Ltd, Tokyo, Japan) was used to identify a site in the gastric antrum or duodenal bulb with an avascular path to the gallbladder. An electrocautery-enhanced LAMS (10 mm × 10 mm, Axios; Boston Scientific, Marlborough, MA, USA) was then advanced into the gallbladder using a direct puncture technique. Deployment of the distal flange was performed with EUS guidance and deployment of the proximal flange was completed with direct endoscopic visualization (Figure 1).
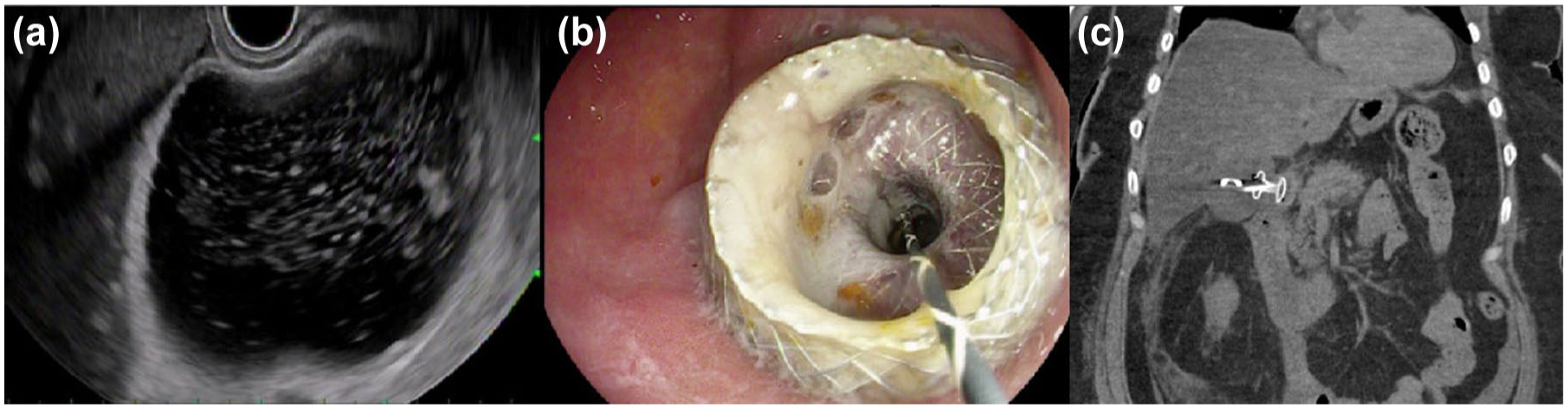
EUS-guided gallbladder drainage. (a) Endoscopic ultrasound image of a distended gallbladder with sludge. (b) Endoscopic image following transduodenal LAMS placement in the gallbladder with pus draining from the cholecystoduodenostomy tract. (c) CT scan showing LAMS and double pigtail stent in appropriate position.
If the gallbladder was contracted on initial endosonographic evaluation, then it was distended prior to inserting the LAMS. A 22-gauge fine needle aspiration (FNA) needle (EchoTip Ultra; Cook Medical, Winston-Salem, NC, USA) was used to puncture the gallbladder. FNA was performed with aspiration of bile confirming needle position. Dilute contrast was then injected to distend the gallbladder lumen in order to facilitate LAMS deployment.
Following LAMS deployment, a 0.025 inch in diameter and 450 cm in length VisiGlide 2 wire (Olympus, Tokyo, Japan) was placed into the gallbladder. Wire and fluoroscopy-guided balloon dilation of the LAMS was then performed up to the stent diameter with a CRE balloon (Boston Scientific, Marlborough, MA, USA). A 7 Fr by 4 cm DPT stent was placed through the LAMS and into the gallbladder.
All the patients were kept nil per oral overnight and maintained on intravenous hydration and antibiotics. The next day, patients started a clear liquid diet which was advanced as tolerated. For patients with life expectancy <6 months, the LAMS was left in place indefinitely. For the remaining patients, esophagogastroduodenoscopy with cholecystoscopy was completed 4 weeks after the index procedure for removal of gallstones. The LAMS was removed at the time of repeat endoscopy to decrease the risk of complications including delayed bleeding, perforation, and stent occlusion (resulting from degradation of stent covering and tissue hyperplasia). A 7 Fr × 4 cm DPT stent was replaced into the gallbladder via the cholecystoenteric tract for long-term drainage.
Percutaneous transhepatic gallbladder drainage
All the procedures were completed with local anesthesia using 1% lidocaine with or without moderate sedation (midazolam and fentanyl). A 22-gauge needle was used to puncture the gallbladder via a transhepatic approach under ultrasound guidance. If a transhepatic route was not feasible, a transperitoneal path was utilized. After gallbladder puncture, a wire was placed into the gallbladder under fluoroscopic guidance. Tract dilation was subsequently performed followed by placement of an 8 or 10 Fr cholecystostomy tube. Cholecystogram was used to confirm appropriate positioning of the tube.
Results
A total of 249 patients with cholecystitis underwent drainage procedures during the study period. EUS-GBD was performed for 57 patients while 192 patients underwent PT-GBD. Using propensity score matching in a 1:1 ratio, 57 PT-GBD patients were matched with the 57 EUS-GBD patients. The SMD for each variable used during matching is shown in Table 1(a) and (b) for both pre- and post-matched patients. A SMD of less than 0.10 was used to confirm a balanced covariate distribution between the two cohorts.
(a) Variable distribution prior to propensity score matching and (b) variable distribution after propensity score matching.
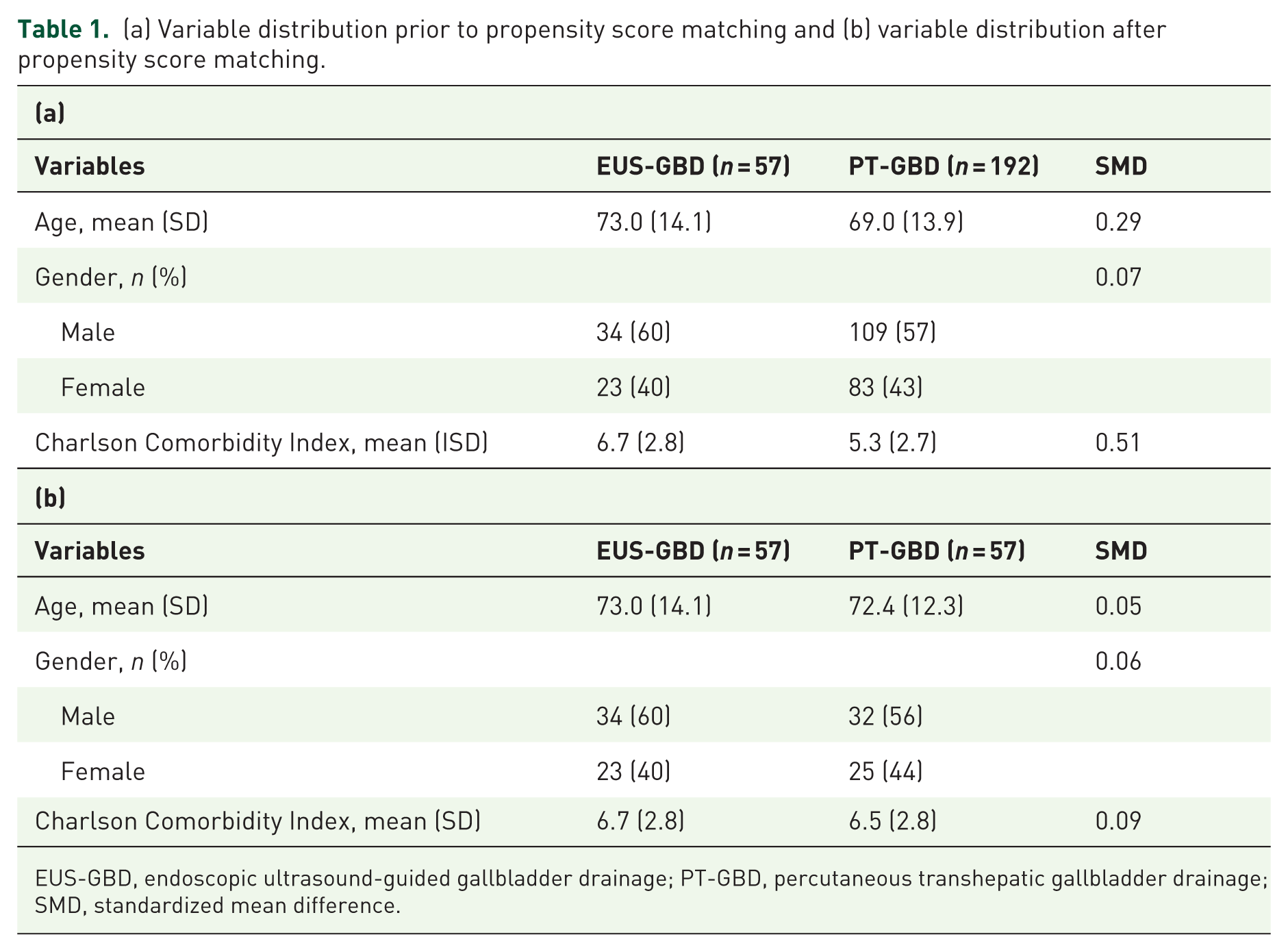
EUS-GBD, endoscopic ultrasound-guided gallbladder drainage; PT-GBD, percutaneous transhepatic gallbladder drainage; SMD, standardized mean difference.
In the post-matched patients, AC of Grade I (mild) or Grade II (moderate) severity per the Tokyo guidelines was seen in 52 patients in the EUS-GBD groups and 50 patients in the PT-GBD group (91% EUS-GBD vs 88% PT-GBD, p = 0.76). 18 Calculous cholecystitis was the most common indication for both EUS-GBD and PT-GBD, comprising 70% of the patients in each cohort. Malignant cholecystitis (defined as AC due to cystic duct obstruction from the tumor or after placement of self-expanding metal stents in patients with pancreatico-biliary malignancies) was more frequent in the EUS-GBD cohort (30% EUS-GBD vs 16% PT-GBD) while acalculous cholecystitis was more common in the PT-GBD group (0% EUS-GBD vs 14% PT-GBD; p = 0.0028). There was no significant difference in antiplatelet or anticoagulant use between the two groups. Ascites was present in a similar proportion of patients for each group (35% EUS-GBD vs 25% PT-GBD; p = 0.31). Additionally, the mean prognostic nutritional index did not differ significantly between the EUS-GBD and PT-GBD cohorts (34.6 vs 37.2; p = 0.11). Table 2 demonstrates notable pre-procedure laboratory values for each cohort.
Procedure indication and patient-related variables following propensity score matching.

EUS-GBD, endoscopic ultrasound-guided gallbladder drainage; INR, International Normalized Ratio; IQR, interquartile range; PT-GBD, percutaneous transhepatic gallbladder drainage.
Technical success was similar for EUS-GBD patients compared to PT-GBD patients (96% vs 98%, p > 0.99). There were two technical failures in the EUS-GBD cohort, both related to stent misdeployment. One patient was managed with the same session transpapillary gallbladder drainage. The second patient underwent successful transgastric EUS-GBD after a transduodenal stent misdeployed. In the PT-GBD cohort, the lone technical failure ultimately required CT-guided percutaneous drainage the following day.
The remainder of the clinical outcomes are outlined in Table 3. Clinical success was noted in 93% of EUS-GBD patients and 80% of PT-GBD patients (p = 0.093). EUS-GBD patients required fewer gallbladder-related procedures (median 2 vs 3; p < 0.0001) compared to PT-GBD. However, there was no significant difference in procedure duration, post-procedure hospital length of stay, 30-day mortality, and follow-up duration between the two groups. The median survival was 573 days for the EUS-GBD group and 452 days for PT-GBD group (p = 0.77; Figure 2).
Comparison of clinical outcomes for patients undergoing EUS-GBD and PT-GBD.

EUS-GBD, endoscopic ultrasound-guided gallbladder drainage; IQR, interquartile range; PT-GBD, percutaneous transhepatic gallbladder drainage.

Kaplan–Meier curve comparing survival in patients undergoing EUS-guided gallbladder drainage versus percutaneous cholecystostomy (p = 0.77).
EUS-GBD was associated with a significantly lower adverse event rate (16% EUS-GBD vs 44% PT-GBD; p = 0.0020) when compared to PT-GBD. A comparison of adverse events is illustrated in Table 4. The adverse events seen in EUS-GBD group included post-procedure LAMS migration (n = 5), bleeding requiring blood transfusion (n = 2), abdominal pain requiring hospitalization (n = 1), and cholecystocolonic fistula (n = 1) treated with stent removal and fistula closure with clips. None of the five patients with LAMS migration into the gallbladder lumen experienced any symptoms. This was identified on follow-up endoscopy when patients presented for LAMS removal. The DPT that was placed coaxially at the time of initial endoscopy was noted to be in appropriate position. After performing cholecystoenteric tract dilation with a balloon, the LAMS was removed without complications. The DPT was left in place for long-term drainage.
Adverse events after EUS-GBD and PT-GBD.

EUS-GBD, endoscopic ultrasound-guided gallbladder drainage; PT-GBD, percutaneous transhepatic gallbladder drainage.
In the PT-GBD group, adverse events included drain dislodgement (n = 21), peri-cholecystic abscess (n = 4), and Systemic Inflammatory Response Syndrome (SIRS) (n = 1) requiring ICU admission, biloma (n = 3), cellulitis (n = 2), drain occlusion (n = 1), bleeding requiring blood transfusion (n = 1), and cholecystocolonic fistula (n = 1). Notably, six PT-GBD patients experienced multiple adverse events while no EUS-GBD patients experienced more than one. Serious adverse events were noted in 5 PT-GBD patients compared to zero in the EUS-GBD group, although this finding did not reach statistical significance (p = 0.057).
Discussion
Laparoscopic cholecystectomy remains the standard of care treatment for cholecystitis. In poor surgical candidates, PT-GBD has traditionally been the treatment modality of choice. 4 EUS-GBD is a method of endoscopic gallbladder drainage that enables patients to avoid a percutaneous drain and the associated negative impact on quality of life. 1 Utilizing propensity score matching, our study retrospectively compared EUS-GBD and PT-GBD in patients with cholecystitis deemed high risk for surgery.
In our study, EUS-GBD demonstrated similar high rates of technical and clinical success when compared to PT-GBD. These results closely align with those reported in early retrospective comparative studies.19–21 However, in these studies, the type of stent used for EUS-GBD was not uniform as patients with electrocautery-enhanced LAMS, non-cautery LAMS, and plastic stents were included. More recently, the DRAC1 trial randomized 80 patients with AC to EUS-GBD with electrocautery-enhanced LAMS or PT-GBD. In that study, Teoh et al. showed similar high rates of technical success (97.4% EUS-GBD vs 100% PT-GBD) and clinical success (92.3% EUS-GBD vs 92.5% PT-GBD). The summation of these studies strongly suggests that both EUS-GBD and PT-GBD are highly effective at treating AC.
When compared to PT-GBD, we report that EUS-GBD was associated with fewer and less severe adverse events. More specifically, 16% of EUS-GBD patients and 44% of PT-GBD patients experienced at least one adverse event (p = 0.0020). Furthermore, there were no serious adverse events in the EUS-GBD group while serious adverse events occurred in 5 PT-GBD patients, including one fatality. The most commonly observed adverse event was drain dislodgement (n = 21). This finding likely explains why patients undergoing PT-GBD underwent significantly more total procedures compared to EUS-GBD patients. Our findings closely align with those reported in a recent systematic review and meta-analysis. Boregowda et al. 22 noted adverse events in 14.6% of patients (n = 477) undergoing EUS-GBD and 30% of patients (n = 698) undergoing PT-GBD.
Despite EUS-GBD first being described in 2007, more widespread use was delayed by limitations related to stent design with plastic stents and fully covered self-expandable metal stents (fcSEMS). The fixed, narrow diameters of plastic stents led to stent clogging and bile leaks.5,23,24 In contrast, fcSEMS had fewer issues with stent occlusion due to their wider diameter. Unfortunately, the tubular nature of fcSEMS did not adequately maintain apposition of the two non-adherent lumens and thus led to issues with stent migration.5,23–25 EUS-GBD with a LAMS was first described in 2012. 7 The unique shape of LAMS features bilateral terminal flanges that anchor the two lumens together. Additionally, unlike plastic stents and fcSEMS, LAMS deployment is accomplished with a single device, obviating the need for device exchanges over a wire. Furthermore, the larger diameter of LAMS permits further therapies such as cholecystoscopy with electrohydraulic lithotripsy and stone extraction. 5
Looking forward, the role of EUS-GBD with LAMS in treating cholecystitis may expand in the future. Teoh et al. 26 retrospectively compared EUS-GBD and laparoscopic cholecystectomy using propensity score matching. There was no difference in technical success, clinical success, 30 day adverse event rates, and mortality between the two groups. Reintervention rates and rates of recurrent biliary events were also similar. It should be noted that this was a fairly small study with 30 patients in each cohort. Furthermore, the included EUS-GBD patients were deemed poor surgical candidates while the laparoscopic cholecystectomy patients fit for surgery. In consequence, the results of the comparison of two heterogeneous cohorts must be interpreted cautiously.
Our study has several notable strengths. First, it is a large direct comparative study between EUS-GBD and PT-GBD. Second, all EUS-GBDs were completed at a single institution with standardized procedural and post-procedural protocols. Last, propensity score matching was performed to mitigate some of the inherent risk of selection bias in retrospective studies.
In contrast, there are some limitations to our study. First, the study was conducted retrospectively and despite propensity score matching, the risk of selection bias persists. Second, the median follow-up duration for EUS-GBD was around 1 year. Our study did not adequately address long-term safety and efficacy of the chronic indwelling transmural DPTs left in the gallbladder indefinitely following LAMS removal. Last, all the EUS-GBD procedures were performed by high-volume therapeutic endoscopists with robust experience with EUS-guided transmural LAMS placement for a variety of indications. Thus, these results may not be applicable to lower volume centers.
Conclusion
EUS-GBD provides comparable effectiveness to percutaneous drainage for managing AC in patients who are poor surgical candidates, with the additional advantages of fewer complications, a better safety profile, and a need for fewer interventions. In high-volume centers, this technique should be preferred over PT-GBD in such patients.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251375376 – Supplemental material for EUS-guided gallbladder drainage versus percutaneous gallbladder drainage in high-risk patients with acute cholecystitis: a propensity score-matched analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848251375376 for EUS-guided gallbladder drainage versus percutaneous gallbladder drainage in high-risk patients with acute cholecystitis: a propensity score-matched analysis by Rishi Pawa, Nicholas Koutlas, Alexa Cecil, Robert Dorrell, Gregory Russell and Swati Pawa in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
