Abstract
Background:
Walled-off necrosis (WON) is a sequela of acute necrotizing pancreatitis preferentially managed with lumen apposing metal stents (LAMS). Adverse events including buried stent syndrome and bleeding have been associated with a longer duration of LAMS placement.
Objectives:
We attempt to examine our outcomes of a standardized approach to LAMS dwell time and hypothesize that LAMS removal based on imaging characteristics and patient symptoms improves outcomes.
Design:
From November 2015 to May 2022, a prospectively maintained database on patients with symptomatic WON undergoing endoscopic drainage with LAMS was retrospectively reviewed and analyzed.
Methods:
Patient characteristics, procedure details, and outcomes were recorded. Imaging was performed at 1, 3, and 6 weeks after LAMS placement and 1 week after each necrosectomy. Imaging findings and patient symptoms were used to determine the need for repeat necrosectomy. The timing of LAMS removal was guided by adequate endoscopic necrosectomy and resolution of the patient’s symptoms. Subgroups were identified based on the duration of LAMS placement (less than 4 weeks and more than 4 weeks). Independent t-tests (continuous variables) and Fisher’s exact tests (categorical outcomes) were used to analyze the two groups.
Results:
In all, 104 patients underwent endoscopic necrosectomy during the study period. Of the two subgroups identified based on LAMS dwell time, 70 patients had a LAMS duration greater than 4 weeks and 34 patients had a LAMS duration less than 4 weeks. Collections with >50% necrosis were more commonly seen in patients with longer LAMS dwell time (<4 weeks (12%) vs >4 weeks (33%), p = 0.031). The median number of necrosectomies was fewer in the early LAMS removal group compared to the late LAMS removal cohort (p = 0.03). Clinical outcomes including technical success, clinical success, delayed adverse events, and 6-month mortality were similar in both groups.
Conclusion:
A patient-specific customized approach to endoscopic drainage of WON improves outcomes. Patients with extensive necrosis may require a longer LAMS dwell time to achieve adequate debridement and clinical resolution. An increased risk of bleeding or adverse events related to prolonged duration of LAMS placement was not observed in our study. Future larger prospective studies are needed to confirm these conclusions.
Introduction
Walled-off necrosis (WON) is a sequela of necrotizing pancreatitis that develops once necrotic pancreatic and peripancreatic tissue forms a well-circumscribed capsule. These collections, which are defined by the revised Atlanta classification, typically take 4 weeks to form. Patients with these collections are often asymptomatic though once they develop symptoms including abdominal pain, fever, signs of gastric outlet obstruction, including nausea, vomiting, and early satiety as well as biliary obstruction, drainage is indicated. 1 Further complications of these collections include the development of fistulae necessitating surgical intervention, erosion into adjacent vessels forming pseudoaneurysms, and most commonly infection requiring hospitalization for intravenous resuscitation and antibiotics. 2 Despite innovations in management techniques including Endoscopic Ultrasound-guided drainage with lumen apposing metal stents (LAMS), this condition is associated with high morbidity and mortality; thus, a multidisciplinary protocolized management approach is essential to improve outcomes. 3
EUS-guided drainage of pancreatic fluid collections has supplanted surgical and percutaneous drainage as the preferred technique. LAMS are widely noted to improve clinical outcomes of the management of WON due to their large stent diameter, ease of deployment, and anti-migratory design. 3 The strategy of managing these collections with LAMS, while proven effective, is highly variable among endoscopists and dependent on the availability of a multidisciplinary team, institutional protocols, and training techniques.
The success of LAMS has not come without controversy. The duration of LAMS has been correlated with an increased risk of bleeding and buried stent syndrome.4,5 In a randomized controlled trial by Bang et al., 6 after 3 weeks, there was a precipitous increase in stent-related bleeding prompting an early protocol amendment and formal recommendation for LAMS retrieval within 4 weeks. This proposal has been adopted by many endoscopists and formally recommended by the European Society of Gastrointestinal Endoscopy (ESGE). While the increased risk of bleeding with a longer duration of LAMS is concerning, we hypothesize that tailoring the duration of LAMS to collection characteristics including the degree of necrotic pancreatic tissue and the presence of disconnected duct syndrome (DDS) improves clinical outcomes. 7
Methods
Patients
All consecutive patients with symptomatic WON managed with endoscopic necrosectomy from November 2015 to May 2022 were retrospectively reviewed and analyzed. IRB approval (IRB00035936) and informed consent was obtained. Prior to each procedure, the case was reviewed and discussed by a multidisciplinary team which consisted of abdominal radiologists, a hepatobiliary surgeon, and advanced endoscopists (Figure 1). The percentage of necrosis was calculated by an experienced radiologist in conjunction with the multidisciplinary team. The axial, coronal, and sagittal views of cross-sectional imaging were analyzed. The head, body, and tail of the pancreas were viewed independently with identification of the areas of necrosis following which a percentage was calculated based on the total volume of glandular involvement. Patients were divided into two categories: less than or greater than 50% necrosis. Inclusion criteria included patients with symptomatic WON who underwent drainage with LAMS followed by direct endoscopic necrosectomy (DEN) and had a clinic follow-up duration of greater than 6 months from the date of the index procedure. Exclusion criteria included patients less than 18 years of age, patients with concern for malignancy, postoperative fluid collections, thrombocytopenia with a platelet count of less than 50,000, and an international normalized ratio greater than 1.5. All patients included in the study were diagnosed with WON following confirmation with cross-sectional imaging and direct endoscopic visualization.

Magnetic resonance imaging (axial T2 Haste) of large walled-off necrosis (yellow asterisk) with necrotic material (red arrow).
Procedural technique
The WON was identified during endoscopy using a therapeutic linear echoendoscope (Olympus, Tokyo, Japan). A 15 mm × 10 mm LAMS (Boston Scientific, Marlborough, MA, USA) was used in all patients for endoscopic drainage (Figure 2). Following stent deployment, the lumen of the LAMS was dilated to 15 mm using a balloon. DEN was performed with rat tooth forceps, snares, or baskets (Figure 3). All collections underwent lavage with 100 cc of 0.3% hydrogen peroxide after the procedure. No double pigtail stents or nasocystic drains were placed in the collection following LAMS placement. Interval imaging was obtained at 1, 3, and 6 weeks following the index procedure and 1 week after each DEN. Imaging findings were correlated with patient symptoms and repeat necrosectomy was planned accordingly. The timing of LAMS removal was determined based on adequate debridement and collection size of less than 2 cm on imaging. Subgroups were identified based on the duration of LAMS placement (less than 4 weeks and more than 4 weeks).
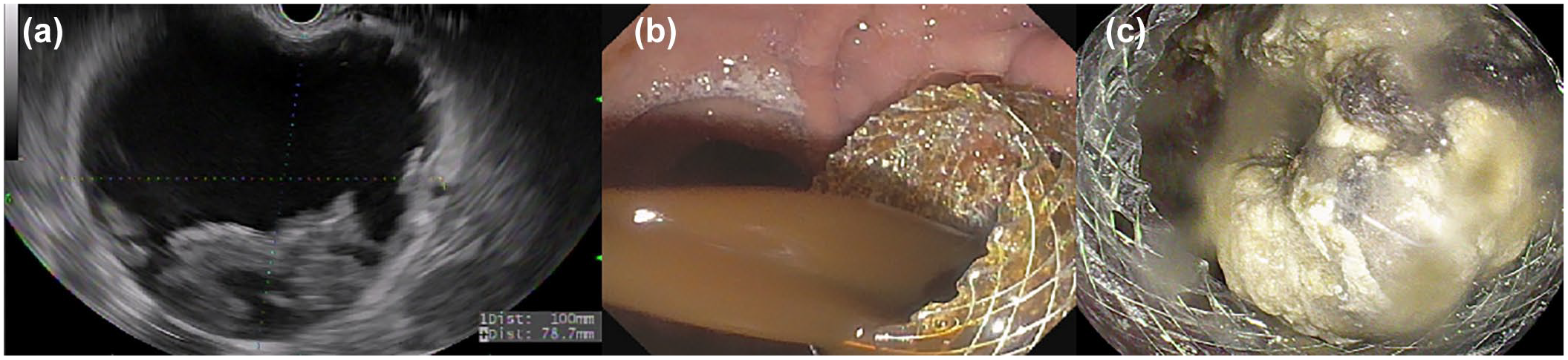
Endoscopic ultrasound-guided drainage of WON. (a) Endoscopic ultrasound image of a WON. (b) Endoscopic imaging following lumen apposing metal stent placement with fluid draining in the stomach. (c) Endoscopic imaging with necrotic material inside the cyst cavity.
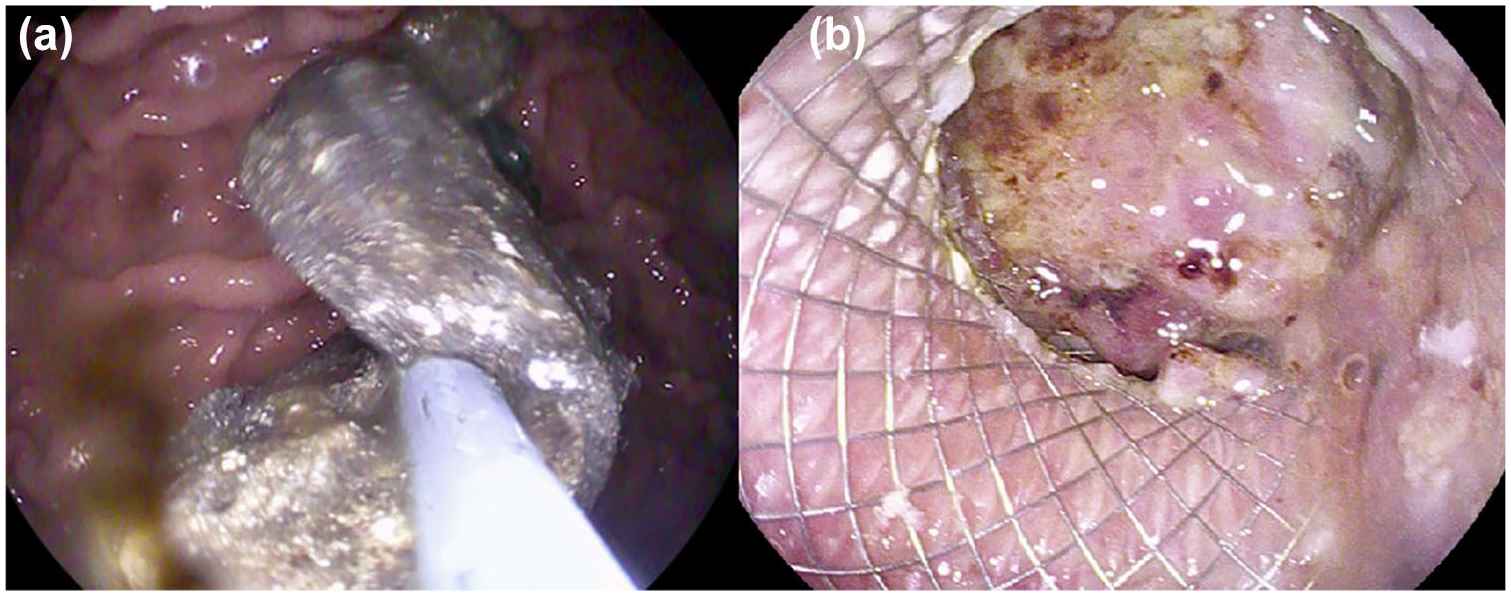
DEN for walled-off necrosis. (a) Endoscopic imaging with removal of necrotic material from the cyst cavity. (b) Endoscopic imaging with healthy-appearing cyst wall following DEN.
Outcomes
Technical success was defined as successful endoscopic deployment of LAMS within the WON followed by complete debridement of all necrotic material. All patients were seen in the clinic following discharge at 1, 3, and 6 months or sooner if they developed recurrent symptoms. Clinical success was determined based on repeat imaging showing a residual collection size of less than 2 cm and improvement of patients’ symptoms at 6-month follow-up. The duration of follow-up was determined based on the date of the index procedure until the last clinic encounter. Procedure-related adverse events (bleeding, stent migration, buried stent syndrome, and stent occlusion) and their severity were determined by the American Society for Gastrointestinal Endoscopy Lexicon criteria. 8 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 9
Data analysis
Patient data were summarized using descriptive statistics including median and interquartile range. Continuous variables were analyzed using independent t-tests and categorical variables were analyzed using Fisher’s exact test. Statistical analysis system (version 9.4; SAS Institute Inc., Cary, NC, USA) was used for all analyses.
Results
During the study period from November 2015 to May 2022, 104 patients diagnosed with WON underwent DEN. Of these patients, 34 had their LAMS removed within 4 weeks of placement, while 70 had an LAMS duration exceeding 4 weeks.
The median age of the cohort was 58 years (interquartile range (IQR): 44, 64), with a median BMI of 29 kg/m² (IQR 24, 32). There were 37 female patients (35.6%). The etiologies of pancreatitis included alcohol (n = 32), gallstones (n = 40), hypertriglyceridemia (n = 4), idiopathic (n = 23), medication-induced (n = 3), and iatrogenic (n = 2). The median area of the collection was 94 cm² (IQR 54, 149) with 27 patients (25.9%) having greater than 50% necrosis (Figure 4). The median ASA score was 3 (IQR 3, 3) and concomitant infection was present in 46 patients (44.2%). Hemorrhage was noted in 16 patients (15.4%). These patients underwent a CT angiography and a pseudoaneurysm was identified in two patients which was managed with angioembolization prior to drainage.
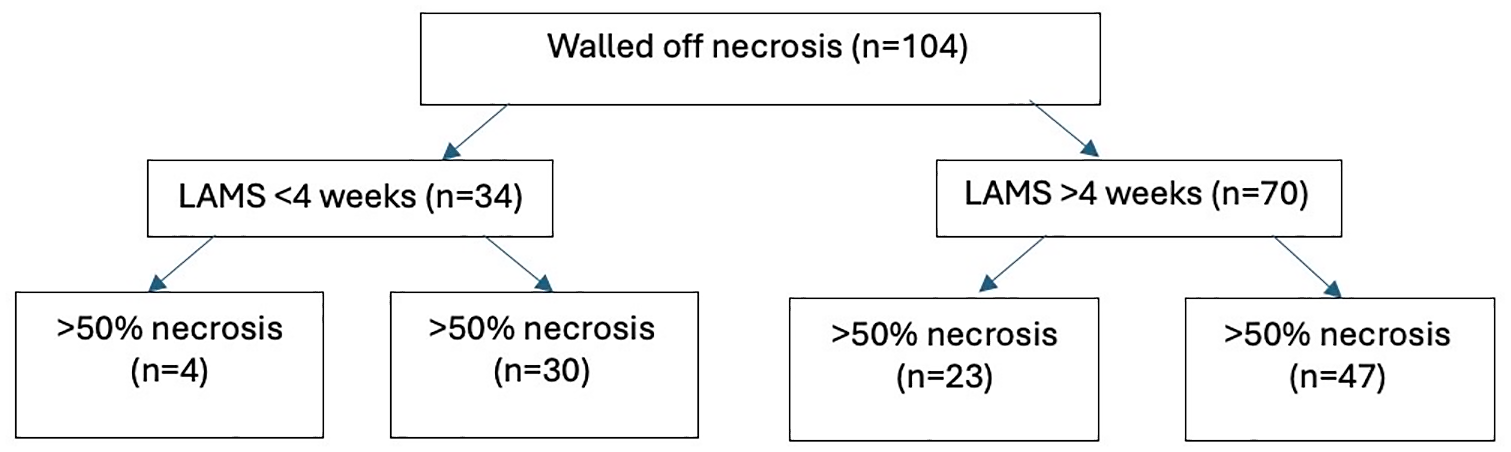
Flow diagram of patients with walled-off necrosis managed endoscopically.
The patient and collection characteristics of both cohorts (LAMS duration <4 weeks vs >4 weeks) are analyzed and summarized in Table 1. Demographic characteristics including age, gender, etiology, Charlson comorbidity index, ASA score, and pre-procedure albumin were similar in both groups. Severe pancreatitis, as defined by the revised Atlanta classification, was observed in 11 patients (32%) in the early LAMS removal group and 21 patients (30%) in the late LAMS removal group (p = 0.69). The median collection size was 105.3 cm2 in the less than 4-week group and 88.5 cm2 in the more than 4-week cohort (p = 0.51). Among these characteristics, only the presence of >50% necrosis was statistically significant between the two groups (<4 weeks: 12% vs >4 weeks: 33%, p = 0.031). The prevalence of infection, hemorrhage, paracolic gutter extension, and multi-organ failure among the two groups were comparable.
Patient demographics and collection characteristics prior to endoscopic drainage.

IQR, interquartile range; LAMS, lumen apposing metal stents.
Technical success was achieved in 100% of patients in both groups. Clinical success was observed in 29 patients (85%) with LAMS removal within 4 weeks and 66 patients (94%) with removal after 4 weeks (p = 0.15). The median number of necrosectomies was 1 (IQR: 1, 2) in the early LAMS removal cohort compared to 2 (IQR: 1, 3.75) in the late LAMS removal cohort (p = 0.03). The prevalence of DDS was significantly higher in the early LAMS removal group (p < 0.01; Table 2).
Clinical outcomes after endoscopic intervention.

IQR, interquartile range; LAMS, lumen apposing metal stents.
Early adverse events (<48 h) were observed in two patients, both of whom developed systemic inflammatory response syndrome after endoscopic necrosectomy, requiring intensive care unit admission. The incidence of delayed adverse events (>48 h) was similar between the two groups (p = 0.75, Table 2). A total of 12 delayed adverse events occurred across the study cohort, with 3 (9%) in the early LAMS removal group and 9 (13%) in the late LAMS removal group. Delayed adverse events included bleeding (n = 5), stent migration (n = 1), stent occlusion (n = 5), and buried stent syndrome (n = 1). Among the five patients who experienced bleeding (<4 weeks: n = 2 vs >4 weeks: n = 3), four were found to have pseudoaneurysms and underwent angioembolization. In one patient, bleeding was secondary to cyst wall trauma from the LAMS, exacerbated by anticoagulation, and was managed conservatively. All bleeding events occurred within 3 weeks of LAMS placement in both groups. Stent migration (on day 60, n = 1) and buried stent syndrome (on day 32, n = 1) were reported in the late LAMS removal group, necessitating endoscopic intervention. Stent occlusion occurred in one patient in the early LAMS removal group (on day 9) and four patients in the late LAMS removal group (on days 30, 35, 36, and 50). All of these patients required endoscopy for stent clearance.
At the 6-month follow-up, there were five deaths in the early LAMS removal group and seven deaths in the late LAMS removal group (p = 0.52). Of the deaths, five were related to clinical failure and seven were secondary to unrelated causes.
Discussion
Lumen-apposing metal stents have revolutionized the management of WON. Despite the emerging consensus on the utility and benefits of LAMS compared to plastic stents, data regarding the optimal timing for LAMS removal remain limited and inconsistent across institutions. Bang et al. 6 reported an increased risk of bleeding if LAMS were not removed within 3 weeks of placement, prompting similar guidelines from the ESGE.8,9 In our study, which included 104 patients with WON, we found no significant differences in technical success, clinical success, or adverse events between the cohorts with LAMS removed in less than 4 weeks versus those removed after 4 weeks. In our study, the timing of LAMS removal was guided by successful endoscopic necrosectomy and a collection size of less than 2 cm on imaging. Our findings demonstrated a high technical and clinical success rate, consistent with existing literature.2,10 –13 In the group with LAMS removal within 4 weeks, only four patients exhibited more than 50% necrosis. By contrast, those with LAMS in place for more than 4 weeks had a higher proportion of patients with greater than 50% necrosis, necessitating additional necrosectomies and ultimately longer LAMS dwell time. Our results suggest that LAMS removal should be individualized based on a patient’s imaging findings and collection resolution, rather than adhering to a standardized timeline.
Despite the longer duration of LAMS placement, the rate of bleeding-related adverse events in our study was comparable between the two cohorts. The overall risk of bleeding in patients undergoing LAMS placement is low. Garcia-Alonso et al. reported a cumulative bleeding risk of 6.9% over 12 months of LAMS placement. In addition, the literature indicates there are other potential sources of bleeding beyond the stent itself. 13 Abdallah et al. 14 found that 39 out of 607 patients diagnosed with necrotizing pancreatitis developed pseudoaneurysms, with only 17 (46.3%) occurring after LAMS placement. Although Bang et al. 6 suggested early LAMS removal due to increased incidence of delayed bleeding after 3 weeks, all adverse events related to bleeding in our study occurred within the first 3 weeks after the procedure, suggesting that stent dwell time should not guide device removal.
It is crucial to recognize that early LAMS removal may jeopardize clinical success. Dhillon et al. 15 followed a strict protocol, removing LAMS 3 weeks after placement, yet only achieved an 80% (33/41) clinical success rate. In another study by Willems et al., 12 108 patients with pancreatic fluid collections underwent drainage with LAMS with a median stent duration of 48 days. The authors concluded that increased clinical failure was associated with early LAMS removal (<4 weeks) and that this did not prevent adverse events. As noted in our study, patients with a larger percentage of necrosis often required multiple endoscopic necrosectomies, leading to delayed LAMS removal. Without this extended stent dwell time, achieving clinical success and resolution of WON would have been unlikely.
Our study has several notable strengths. All LAMS procedures were conducted at a single institution and followed a strict protocol. The study cohort was relatively large compared to existing literature, with all patients undergoing LAMS placement specifically for WON. However, there are limitations to consider. First, the retrospective design introduces a risk of selection bias. Next, we used contrast-enhanced CT rather than MRI for initial assessment and follow-up for patients with WON. While MRI is better than CT for assessment of fluid contents and evaluating feasibility of endoscopic drainage, the logistical constraints associated with MRI including higher cost and complexity of the procedure as well as lack of easy accessibility made it less preferable over CT in our study protocol. In addition, all LAMS procedures were performed by high-volume endoscopists at a large tertiary referral center, which may limit the generalizability of our findings to lower-volume institutions.
In conclusion, a one-size-fits-all approach to the drainage of WON is inadequate. Clinical resolution in patients with a higher degree of necrosis may necessitate a longer duration of LAMS placement to facilitate endoscopic necrosectomy. Our study did not find an increased risk of bleeding or delayed adverse events associated with stent dwell time. Larger prospective studies are needed to validate these findings.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251320739 – Supplemental material for Standardized approach to removal of lumen apposing metal stents following endoscopic necrosectomy: one size does not fit all
Supplemental material, sj-docx-1-tag-10.1177_17562848251320739 for Standardized approach to removal of lumen apposing metal stents following endoscopic necrosectomy: one size does not fit all by Robert Dorrell, Alexa Cecil, Swati Pawa, Gregory Russell and Rishi Pawa in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
